Abstract
The plant piquillín (Condalia microphylla) grows in arid regions of Argentina and is the cause of mal del piquillín in cattle. The salient histologic features of this leukomyelopathy are vacuolation of white matter and axonal degeneration in the spinal cord. The histologic lesions can be experimentally reproduced in cattle and pigs by feeding milled bark of the plant, but naturally occurring piquillín toxicosis has not been reported previously in pigs. The authors report an outbreak of progressive ataxia on an Argentine hog farm, where partially consumed piquillín plants were found in the pens of affected pigs. Histologic lesions included vacuolation of white matter, edema, and mild axonal degeneration in lumbosacral segments of the cord. The diagnosis of mal del piquillín was based on the history, clinical signs, histologic changes, and elimination of other potential causes of leukomyelopathy. No new outbreaks developed after elimination of piquillín from the hog lots. Results of this investigation indicate that C microphylla toxicosis can affect pigs under natural conditions and should be included in the differential diagnosis for porcine ataxia and leukomyelopathy in regions where the plant grows.
Mal del piquillín was reported in cattle after consumption of Condalia microphylla (piquillín), 1 a drought-tolerant, spiny shrub of the family Rhamnaceae. Piquillín grows throughout arid regions of Argentina, from the northwest to the Península of Valdez in Patagonia. The plant is 0.5 to 2.0 m tall, with spicate branches 2–6 cm long and sessile leaves with elliptical to obovate stipules. 3 Although its fruit was traditionally consumed by native human populations, the plant is not considered suitable forage for livestock. Nevertheless, animals eat it under conditions of overcrowding, boredom, or limited access to food during dry seasons or drought.
Piquillín toxicosis is a cause of ataxia and leukomyelopathy. 1 The disease develops when cattle ingest new growth following rain in the dry season, when forage is scanty. 1 The clinical signs are exclusively neurologic. Affected animals have flexure and restricted mobility of 1 or both hind limbs. The forelimbs are subsequently involved in many cases; severely affected cattle become recumbent and die. 1 At necropsy, no gross lesions are found. Histologic lesions include vacuolation of white matter in the spinal cord with dilation of myelin sheaths and axonal degeneration, especially in lateral and ventral funiculi of thoracolumbar segments. 1
Mal del piquillín has been experimentally reproduced in calves, pigs, and guinea pigs. 4 Clinical signs began in the hind limbs and progressed cranially with continued ingestion of the plant. 4 As in the naturally occurring toxicosis, gross lesions were not found. Histologic lesions were qualitatively similar to those in naturally occurring cases but more widespread, involving all funiculi in all segments of the spinal cord. 4
The toxic principle of C microphylla has not been defined, although some chemical properties have been described, 5 and experimental studies have confirmed the presence of a toxic component in the bark of the shrub. 4 The toxic dose is unknown, but 7 days' feeding of 25 g per day caused disease in a 90-kg calf. Four grams per day of milled bark did not induce disease in 10-kg pigs; it was necessary to triple the dose and feed the bark for 11 days to produce clinical signs. 4
Mal del piquillín is considered a bovine disease, and there are no reports of natural toxicosis in other animals. The aim of this study was to describe an outbreak of ataxia with leukomyelopathy in pigs associated with consumption of C microphylla.
Case History
On a hog operation in La Pampa, Argentina, 112 of 320 feeder pigs developed motor dysfunction over a 30-day period. The farm had 2,247 pigs under a semi-intensive production system. Affected animals were 3 to 6 months of age and confined to dry lots in which many piquillín plants were found. A few other shrubs were present, such as Schinus fasciculatus (molle) and Licyum chilense (llaollín), along with xerophyte grasses. Housing consisted of rudimentary buildings. The ration was based on maize and cooked slaughterhouse offal. Drinking water was derived from a well and pumped through troughs for continuous access. Many of the piquillín plants had been partially uprooted; the stems and roots had been chewed (Fig. 1), and tooth-marked fragments of bark were strewn around the shrubs.
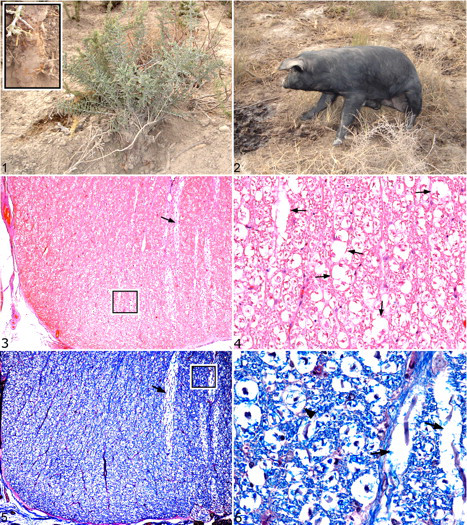
Piquillín (Condalia microphylla) shrub in a hog lot. Inset: Detail of a shrub with chewed stems and roots.
The clinical course started with mild motor alterations in 1 hind limb with progression to bilateral hind limb flexure and paresis over the course of 7 to 10 days. Despite altered locomotion and ataxia, all pigs were alert and able to eat and drink. An affected 6-month-old crossbred castrated male pig with clinical signs was examined. The animal was in ventral recumbency or in a sitting position (Fig. 2) and fell when forced to walk. Both hind limbs were paretic with proprioceptive deficits but responsive to painful stimuli. The pig was humanely euthanized and submitted to the laboratory for necropsy.
Necropsy Examination
The only gross lesion was rupture of the round ligament of the left femur. Samples from liver, kidney, spleen, lung, brain (olfactory, frontal, parietal, and occipital lobes; thalamus; midbrain; pons; cerebellum; and obex), and spinal cord (cervical, thoracic, lumbar, and sacral segments) were fixed in 10% neutral buffered formalin and routinely processed for histologic examination. Four-micrometer paraffin sections of each organ were stained with hematoxylin and eosin. Sections of brain and spinal cord were also stained with Luxol fast blue with periodic acid–Schiff counterstain. 6
Histologic Findings
Histologic lesions were limited to the spinal cord (Figs. 3–6) and consisted of mild vacuolation of the white matter, mainly in lumbosacral segments. Ventral funiculi were especially involved, but dorsal and lateral funiculi were also affected. Luxol fast blue staining (Figs. 5, 6) accentuated the dilation and focal disruption of myelin sheaths. Vacuolation was also apparent in lumbar and sacral ventral spinal nerve roots, whereas dorsal nerve roots were unremarkable. A few axons appeared swollen or fragmented; many myelin sheaths appeared empty. Only a few microscopic hemorrhages were noted in spinal gray matter.
Discussion
A variety of infectious agents, toxins, and nutritional deficiencies produce neurologic disease in pigs. Because most infectious agents are expected to result in inflammation of the brain or spinal cord, 9 the degenerative nature of the lesions in this pig suggested a noninfectious cause. The clinical signs and histologic lesions resembled those reported in natural and experimental piquillín toxicosis. 1,4 These findings, with the known exposure to C microphylla shrubs, prompted the diagnosis of piquillín toxicosis. The ruptured round ligament of the femur was considered a consequence of the neurologic disorder, rather than a primary lesion.
Because the histologic lesions in the central nervous system produced by piquillín poisoning are not specific, other possible causes of leukomyelopathy were considered. Diplodia toxicity, produced by Stenocarpella (formerly Diplodia) maydis, was ruled out because it typically produces lesions in the cerebrum or cerebellum. 9 Furthermore, S maydis was not detected macroscopically in the ration. Cyanide poisoning was ruled out because consistent changes in lungs and heart were not found. Additionally, chronic cyanide ingestion 9 was unlikely because affected pigs had no access to cyanogenic feed. Consumption of Stypandra spp causes neurologic disease, with myelin vacuolation and blindness in mammals and birds in Australia and other countries. 9 It was ruled out in this outbreak because the affected pigs had no evidence of blindness and because plants of the Stypandra genus have not been identified, to our knowledge, in Argentina. In the present outbreak, there was no record of use of the phenolic antiseptic hexachlorophene, which may cause disease in young animals, 9 or other chemicals, such as insecticides that could result in delayed organophosphate poisoning. Metabolic disorders, such as hepatic or renal encephalopathy, 9 were considered unlikely because no hepatic or renal lesions were found.
The following entities or agents were also eliminated as a potential cause for the ataxia and leukomyelopathy, on the basis of history or lack of relevant lesions: water deprivation or salt poisoning, 9 pigweed (Amaranthus retroflexus) poisoning, 2 mycotoxins (eg, aflatoxin or ochratoxin), 9,11 selenium toxicosis, 9 copper or iron toxicosis, 2 delayed swayback (neonatal copper deficiency) or thiamine deficiency, 9 vitamin A toxicosis or deficiency, 12 or vitamin E deficiency. 8 Fixation artifacts and postmortem changes were ruled out because, although ultrastructural studies were not performed (only formalin-fixed tissues were available), other samples of brain and spinal cord that were processed in parallel did not have these changes or other evidence of artifact or autolysis.
The lesions in this pig were most severe in ventral funiculi of lumbosacral segments of the spinal cord, as in bovine mal del piquillín 1 and thus did not exactly match those described in experimental piquillín toxicosis, in which white matter vacuolation involved all funiculi of all segments of the spinal cord. 4 This discrepancy could reflect differences in the dose, age of the pigs, or stage of the disease. In the experimental toxicosis, pigs were fed the milled bark ration until altered sensorium (a late clinical sign) merited euthanasia. In contrast, the pig in this report was euthanized just a few days after the onset of neurologic signs.
The pathogenesis of piquillín poisoning is poorly understood. The histologic changes suggest that the toxin could damage myelin with secondary axonal degeneration. Despite the changes in the spinal cord, no brain lesions were found in this case or in experimentally poisoned animals. It has been suggested that the histologic lesions could be sequelae to biochemical alterations; 1,4 this hypothesis could explain the absence of changes in the brain and the mild histologic lesions in the cord in the presence of severe neurologic signs.
There is no specific antidote for piquillín toxicosis; however, mildly affected animals may recover after C microphylla consumption is halted. 1 In this outbreak, the farmer was advised to move affected pigs to a pen free of piquillín, to remove the plant from the premises, and to provide adequate feed and water, especially during the dry season and periods of drought, to minimize the risk of C microphylla consumption. No new cases of toxicosis occurred after the farmer instituted these management changes; however, a few pigs that had been in hog lots with piquillín developed ataxia and died.
In summary, pigs in semi-intensive production operations are susceptible to piquillín toxicosis. Although C microphylla may be confined to Argentina, other species of the genus Condalia are distributed throughout South and North America and may have toxic potential. 7,10,13 Consequently, piquillín toxicosis should be included in the differential diagnosis for neurologic diseases of domestic animals.
Footnotes
Acknowledgements
We thank C. Moreno, G. Francinelli, and M. C. Tagle for histologic support; R. Fortunato for plant identification; and N. Aznar, C. Rossetti, G. Rodriguez, and M. Fernandez Miyakawa for manuscript review.
The authors declared no potential conflicts of interests with respect to the authorship and/or publication of this article.
The present work was partially supported by Instituto Nacional de Tecnología Agropecuaria, PNI 2044.
