Abstract
Leishmaniasis is a zoonotic disease caused by intracellular Leishmania protozoa that are transmitted by sandflies. The disease occurs in 3 forms: cutaneous, mucocutaneous, and visceral. Cutaneous leishmaniasis has been reported in cats in Europe and South America and in 1 cat from Texas. Leishmania mexicana is endemic in Texas and has been reported to cause cutaneous lesions in humans. This article describes the pathology of 8 biopsy cases of feline cutaneous leishmaniasis presented to the Texas Veterinary Medical Diagnostic Laboratory over a 3.5-year period. The median age of the cats was 3 years; each was presented with nodular, ulcerative lesions on the pinnae and less commonly on the muzzle and periorbital skin. Histologically, the lesions were nodular to diffuse histiocytic dermatitis with numerous amastigotes (2–4 μm) within macrophages and occasionally within the interstitium. Organisms were often contained within round, clear, intracellular vacuoles. In areas of necrosis, organisms were also free within the interstitium. The overlying epidermis was hyperkeratotic, hyperplastic, and often ulcerated. The organisms were not argyrophilic (Gomori methenamine silver), reacted poorly with periodic acid–Schiff reagent, and were inconsistently basophilic with Giemsa. Although not readily visible histologically, kinetoplasts were evident in amastigotes in cytologic preparations. The lesions were similar to those described for cutaneous L. mexicana infection in humans. In 5 of the 8 cats, Leishmania mexicana DNA was amplified from paraffin-embedded tissue by polymerase chain reaction and sequenced.
Keywords
Leishmaniasis is caused by obligatory, intracellular, diphasic protozoa of the genus Leishmania that can infect humans and a wide variety of domestic mammals and wildlife. These parasites are usually transmitted by the bite of various species of female phlebotomine sandflies and occasionally by blood transfusion, sharing of needles, and congenital transmission. Numerous Leishmania species are geographically widespread, with the highest prevalence in parts of Latin America, southern Europe, and the Middle East. 2 Three forms of clinical leishmaniasis are recognized in humans: cutaneous, mucocutaneous, and visceral. Cutaneous leishmaniasis has been reported in cats in Europe and South America and in 1 cat from Texas. 4,5,12 Parasites isolated from cats in these regions include Leishmania braziliensis, L. amazonensis, and L. infantum chagasi in Brazil; L. infantum in southern France, Spain, Switzerland, and Italy; and L. mexicana in Texas. 4,5,21,23,26,27
Leishmaniasis is not widely recognized as an endemic zoonosis in the United States; however, L. mexicana is endemic in Texas and has been reported to cause cutaneous lesions in humans. 13 There is a single case report of a cat from Texas that developed progressive, coalescing, nonulcerated nodules on the face, ears, and muzzle. 3,4 In this study, we examined 8 domestic cats with autochthonous L. mexicana infection diagnosed by skin biopsy at the Texas Veterinary Medical Diagnostic Laboratory in College Station, Texas, between January 2004 and April 2008. In addition to histologic examination of biopsy specimens and evaluation of 1 cytologic preparation, all samples were evaluated using polymerase chain reaction (PCR) and sequencing to define the species of Leishmania.
Materials and Methods
Biopsy submission forms, completed by veterinary practitioners, were reviewed to collect signalment, history, and geographic location. The postal code for each referring veterinarian was used to plot the incidence of cutaneous leishmaniasis by county and year of diagnosis.
Tissue samples were received in 10% buffered formalin, embedded in paraffin, and stained with hematoxylin and eosin (HE). Additional sections were stained with Giemsa, periodic acid–Schiff (PAS), and Gomori methenamine silver (GMS). Cytologic samples, obtained from 1 cat (No. 2) by fine needle aspirate or impression of a surgical specimen, were stained with modified Wright’s stain.
Ten biopsy samples from the 8 cats, which contained Leishmania organisms, were analyzed using PCR amplification and sequencing methods to identify the organisms at the species level. DNA was extracted from three 5-μm-thick sections of formalin-fixed, paraffin-embedded tissues using the Puregene Genomic DNA Purification Kit (Gentra Systems, Minneapolis, MN), following the xylene extraction method recommended by the manufacturer for paraffin-embedded tissue samples. A 603 base-pair portion of the ribosomal RNA gene was amplified using the previously published primer pair, R221 (sense) 5′-GGTTCCTTTCCTGATTTACG-3′ and R332 (anti-sense) 5′-GGCCGGTAAAGGCCGAATAG-3′. 11 Amplifications were done in 50-μL reactions containing 20 ng of DNA template, 25 μl of 2X GoTaq Green Master Mix (Promega, Madison, WI), and MgCl2 adjusted to final primer concentrations of 3 mM and 20 pM. After initial denaturation at 95°C for 2 minutes, the reactions were processed in a touchdown program for 24 cycles of denaturing for 45 seconds at 95°C, annealing for 45 seconds starting at 62°C and decreasing 1/2 degree each cycle to 50°C, and extension for 90 seconds at 72°C, followed by 15 additional cycles with a 50°C annealing temperature and completed with 10 minutes final extension at 72°C. A positive control of Leishmania infantum was obtained from a case of visceral leishmaniasis in a dog. Appropriately sized amplicons were extracted from low-melt agarose gels (Wizard SV Gel and PCR Clean-Up System, Promega, Madison, WI) and processed for sequencing using ABI Prism BigDye Terminator cycle sequencing reaction ready-kit (Promega, Madison, WI). Amplicons were sequenced in both directions by a fee-for-service laboratory. A consensus sequence was constructed for each sample using Sequencher software (Gene Codes Corp., Ann Arbor, MI). Sequences were compared among the samples and evaluated for homology with reported protozoal parasite sequences in the NCBI Genbank database, using a simple Basic Local Alignment Search Tool-Nucleotide (BLASTN) search. Sequences were submitted to Genbank to obtain accession numbers.
Results
All cats were of unknown (mixed) breed and had nodular, variably ulcerative lesions on the pinnae and, less commonly, on the muzzle and periorbital skin. The median age of the cats was 3 years. Five were castrated males; 3 were female (2 spayed). The age, gender, geographic location, biopsy date, clinical outcome, and Genbank accession numbers are in Table 1 . Two biopsy accessions were received in the same year for 1 cat (No. 5).
Age, Sex, Texas County of Origin, Biopsy Date, Genbank Accession No., and Clinical Outcome in 8 Cats With Cutaneous Leishmaniasis
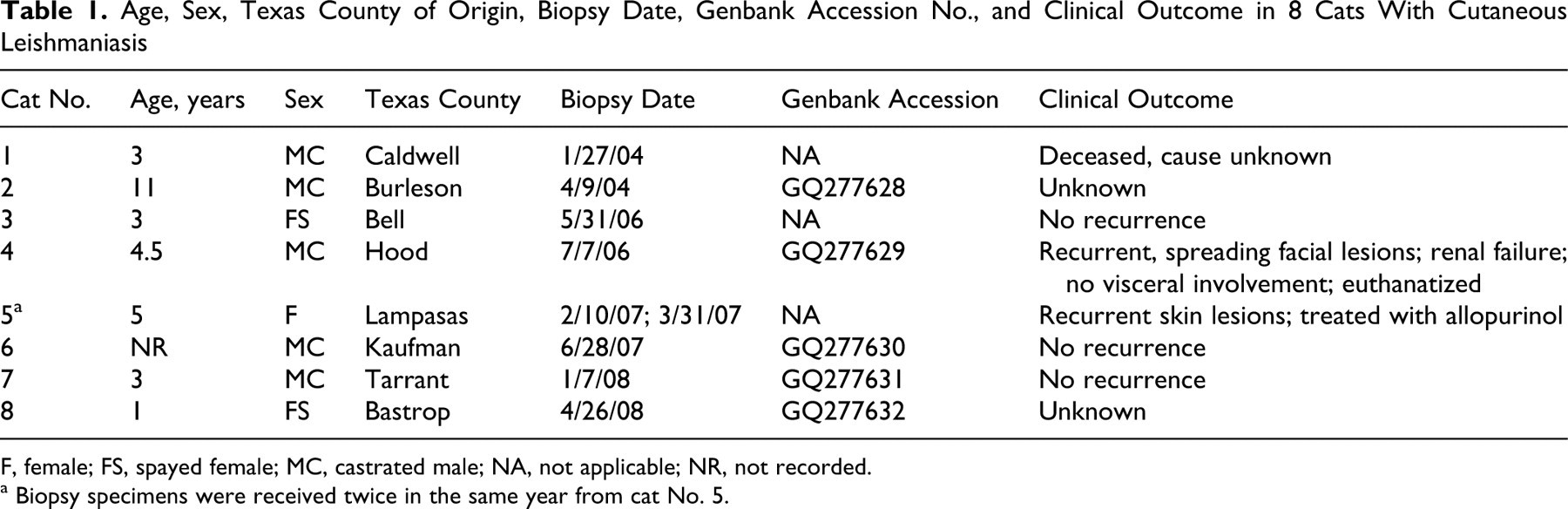
F, female; FS, spayed female; MC, castrated male; NA, not applicable; NR, not recorded.
a Biopsy specimens were received twice in the same year from cat No. 5.
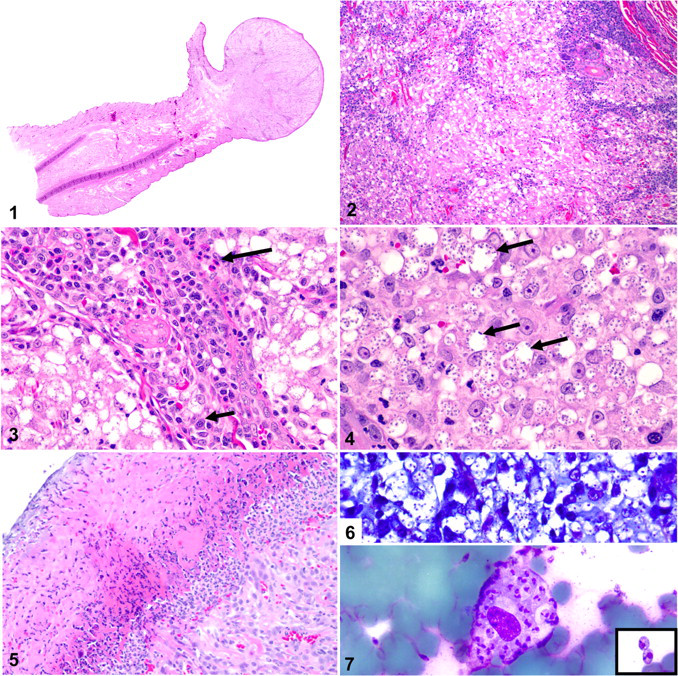
The veterinarians who submitted the biopsy specimens generally reported 1 or multiple nodules, with variably ulcerated, smooth, or scaled surfaces, on the pinnae and muzzle (Fig. 1). Histologically, the lesions consisted of nodular to diffuse accumulations of macrophages throughout the dermis with scattered neutrophils, lymphocytes, plasma cells, and mast cells (Fig. 2). There were accumulations of lymphocytes, plasma cells, and fewer eosinophils around blood vessels (Fig. 3 ). Numerous amastigotes (2–4 μm) were within macrophages and less commonly in the interstitium (Figs. 3, 4). Organisms were often contained within round, clear, intracellular parasitophorous vacuoles, many of which contained more than a dozen amastigotes at their periphery (Fig. 4). The overlying epidermis was hyperkeratotic (both orthokeratotic and parakeratotic), hyperplastic, and often ulcerated (Fig. 5). The protozoa were negative with GMS, reacted poorly with PAS, and stained well with Giemsa in only 5 of the 8 biopsy specimens (Fig. 6). Kinetoplasts were difficult to visualize in histologic preparations without oil immersion and were best evaluated cytologically (Fig. 7).
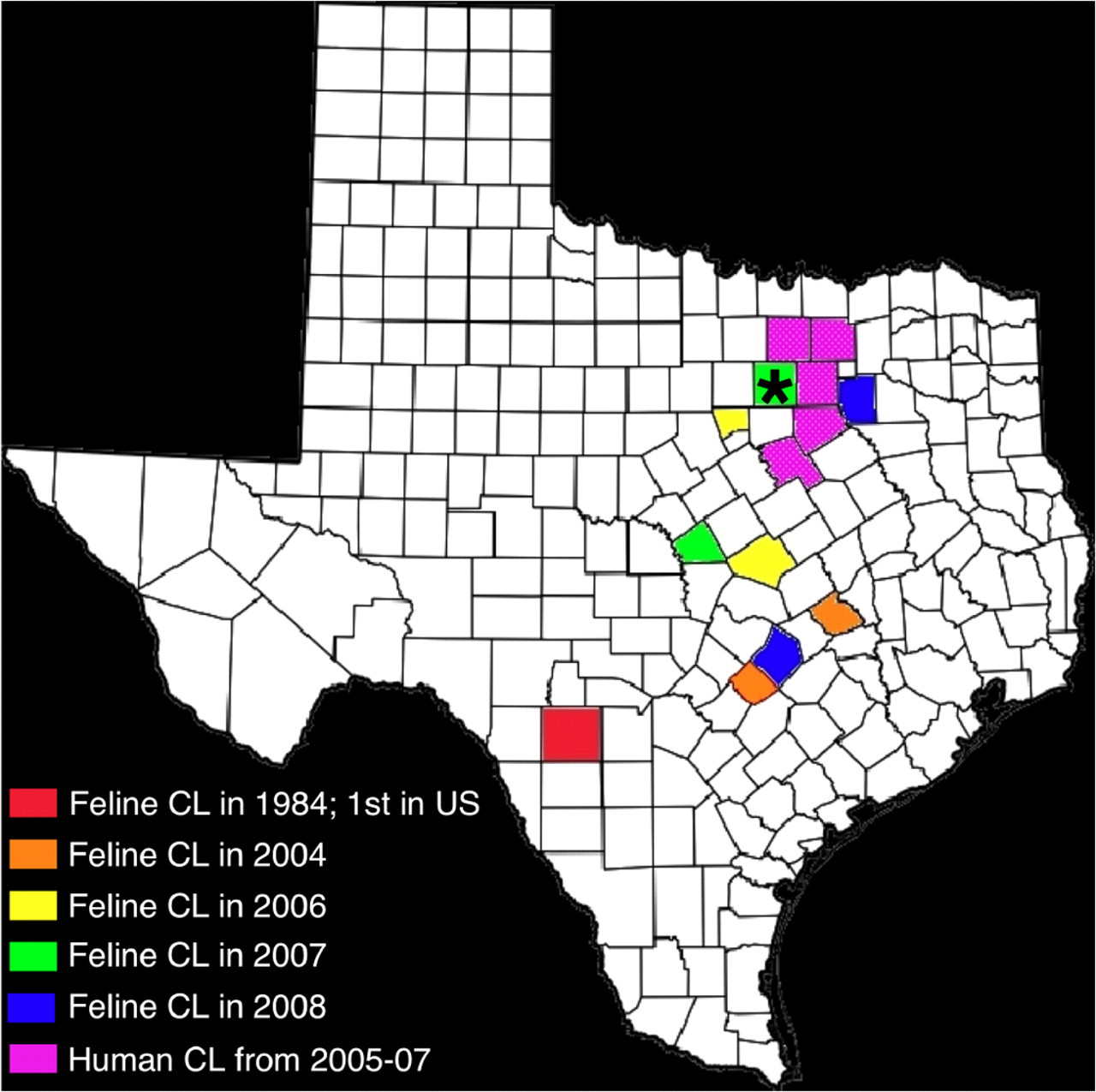
Incidence of cutaneous leishmaniasis (CL) in humans and cats in Texas by county. *Co-incidence of a feline and human case. The feline cases in this study occurred in the following counties: Burleson (2004, orange), Caldwell (2004, orange), Bell (2006, yellow), Hood (2006, yellow), Lampasas (2007, green), Tarrant (2007, green), Bastrop (2008, blue), and Kaufman (2008, blue). Uvalde County, site of the first reported case of feline CL (in 1984), is highlighted in red 4 . The counties highlighted in purple (Dallas, Collin, Ellis, Hill, Denton, and Tarrant) were the sites of 9 human cases in 2006–2008.28
Positive PCR and sequence results were obtained from biopsy samples from 5 of the 8 cats. Sequence lengths ranged from 491 to 563 bases in length; Genbank accession numbers are in Table 1. All sequences were identical to each other and aligned 100% (563/563 bases) with L. mexicana amazonensis (Genbank accession No. M80293.1).
Case outcome is summarized in Table 1. Two cats had recurrent cutaneous leishmaniasis: cat No. 4 was euthanatized for renal failure but did not have visceral involvement, and cat No. 5 was treated with allopurinol. In 3 cats (Nos. 3, 6, and 7), excisional biopsy was apparently curative, and lesions did not recur during the 2- to 4-year follow-up period. The case outcome was unknown for 3 cats (Nos. 1, 2, and 8).
The geographic distribution of the 8 cats is depicted in Fig. 8. The first reported case of feline cutaneous leishmaniasis, in Uvalde County, Texas, in 1984, 4 is also highlighted, as are 9 cases of autochthonous human cutaneous leishmaniasis, reported in north Texas between 2005 and 2007. 28
All previously reported cases of autochthonous human cutaneous leishmaniasis had been documented in the southern and central portions of the state. Uvalde County (red) is where the first case of feline cutaneous leishmaniasis was diagnosed in 1984. 3,4 The cats diagnosed in this study are highlighted by county and are color-coded according to the year of diagnosis to highlight the northern spread of the disease over time.
Discussion
L. mexicana infection is well documented in the medical literature and has spread as far north as the Dallas–Fort Worth area. 13,28 The clinical course of human cutaneous leishmaniasis, caused by L. major, L. mexicana, or L. braziliensis, is categorized as acute, diffuse, or chronic. 24 The strain of Leishmania often corresponds with a specific pattern of tropism, such as cutaneous, mucocutaneous junctions, or visceral. 16 L. mexicana has a strong tropism for the skin in both humans and cats, especially for exposed or sparsely haired parts of the body (e.g., face and ears) that are easily bitten by sandflies. 3,24 In humans, L. mexicana typically causes focal to locally extensive cutaneous lesions and less commonly results in the disseminated or diffuse form. 24 Grossly, lesions in humans often begin as a papule that becomes a shallow and expansile ulcer with raised edges, creating a “wet” lesion that heals within 6–18 months. Less commonly, the lesions are nodular and smooth-surfaced or hyperkeratotic and are classified as “dry” lesions. Microscopically, the lesions described in humans consist of dense granulomatous inflammation with numerous giant cells, fewer lymphocytes and plasma cells, and scant amastigotes. 14,16 Visceral organ involvement of L. mexicana is extremely rare in humans but has been documented in patients with concurrent HIV infection. 19,22
Feline cutaneous lesions of L. mexicana, L. amazonensis, L. infantum, and L. braziliensis are typically more nodular or diffuse, and superficial ulceration is less common than in human cases. 4,5,21,23,26,27 The histologic changes in cats are very similar to those reported in humans, but amastigotes are more numerous within parasitophorous vacuoles. Spontaneous resolution of cutaneous lesions caused by L. mexicana has been well documented in immunocompetent human patients; however, surgical cure without recurrence of lesions was reported in only 3 of the 8 cats in this study.
This retrospective study was limited by the difficulty of obtaining complete signalment, history, and treatment information. To improve our understanding of feline infection with Leishmania mexicana, more information, such as biochemical and hematologic data, infection status for feline leukemia and feline immunodeficiency viruses, and indoor/outdoor habits, in addition to imaging or other diagnostic techniques to detect visceral involvement, should be collected. Visceral infection was ruled out by the clinician in 1 cat from our study and was not identified by necropsy in the first reported feline case. 3
No effective treatment protocols for L. mexicana have been established in cats. One cat in this study was treated with allopurinol; however, the skin lesions have continued to recur. Numerous treatments for cutaneous lesions of L. mexicana have been reported in humans, but many are not completely effective, have toxic side effects, or are not available in the United States. 17
The seroprevalence of L. infantum in cats in Europe and the Middle East has been reported. 1,6,18,25 These studies have endeavored to explore the possible role of cats as a domestic reservoir for the disease. L. infantum causes lesions of both visceral and cutaneous leishmaniasis, in contrast to L. mexicana, which has very strong cutaneous tropism. Seroprevalence studies are a more effective diagnostic and epidemiologic method for visceral leishmaniasis than for cutaneous leishmaniasis with L. mexicana infection, for which PCR amplification and sequencing remain the gold standard. 8,20
Even in enzootic areas, clinical leishmaniasis is uncommon in the dog and rare in the cat. 7 The clinical differential diagnosis for nodular lesions on the face of a cat includes lymphoma, mast cell tumor, squamous cell carcinoma, feline sarcoid, pemphigus foliaceus, and dermatophytosis. The histologic diagnosis of cutaneous leishmaniasis is challenging without cytology to identify kinetoplasts in amastigotes or PCR for confirmation. The histologic differential diagnosis includes infection with Histoplasma capsulatum, Trypanosoma cruzi, Toxoplasma gondii, Neospora caninum, Cryptococcus neoformans, Blastomyces dermatitidis, and Sporothrix schenckii. 9 The differential staining patterns of these organisms may be helpful in a typical diagnostic setting; however, PCR is needed for definitive diagnosis because of the weak or variable histochemical reactions. Giemsa reportedly stains the cytoplasm of Leishmania organisms blue and the kinetoplasts purple; however, only 5 of the 8 cats were Giemsa-positive in this study, and lack of Giemsa-staining of Leishmania organisms has been reported. 13 The lack of silver impregnation with GMS should rule out S. schenckii and H. capsulatum as possible causes.
The results of this study indicate that cutaneous leishmaniasis is becoming more widespread in Texas; however, the cause of this is unclear. The burrowing wood rat, Neotoma micropus, is a mammalian reservoir for L. mexicana, and the northern spread of disease within the state may be related to expansion of its range. 10,15 Changes in vector populations could also be involved. All species of Leishmania in humans in Texas are reportable according to the Texas Department of State Health Services. Leishmaniasis is not reportable at the national level; however, if the disease continues to spread geographically, additional states may begin monitoring human and veterinary cases. The role of cats as a potential reservoir as a primary, secondary, or accidental host in the life cycle of Leishmania is still unknown. This study confirms the incidence of Leishmania mexicana infection in Texas cats and emphasizes the need to include leishmaniasis in the differential diagnosis for cutaneous lesions in cats in the United States.
Footnotes
Acknowledgements
We thank Sarah Jones (TAMU) for histology support. We also gratefully acknowledge John Roths (TAMU) for his help with image preparation.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
