Abstract
Parenteral selenium (Se) and vitamin E (Vit E) were administered to all newborn kids at a Boer goat farm where there was previous high neonatal mortality assumed to be due to nutritional myopathy. All treated kids were affected by severe respiratory distress and died within 8 hours of Se/Vit E administration. Gross lesions included severe pulmonary edema, hydrothorax, and hydropericardium. The primary histopathologic finding was severe, acute, and monophasic myocardial contraction band necrosis. The diagnosis was accidental acute selenosis based on trace mineral analysis of the liver. This case highlights an important differential diagnosis in cases of acute myocardial contraction band necrosis and sudden death in goats and emphasizes the need for caution when administering parenteral Se/Vit E preparations.
History and Gross Findings
Three 1-day-old Boer goat kids (one singleton [No. 1] and 1 set of twins [No. 2 and No. 3]) were submitted for postmortem examination to Prairie Diagnostic Services Inc at the Western College of Veterinary Medicine in Saskatoon, Saskatchewan, Canada, with a history of severe respiratory signs rapidly progressing to death. Ataxia had also been observed. The herd consisted of 150 does, and the referring veterinarian reported high mortality on the farm in the previous year, with loss of approximately 50% of neonatal kids. The mortality was attributed to nutritional myopathy, based on areas of acute myocardial necrosis visualized microscopically and the location of the farm in a selenium-deficient area. However, the diagnosis of nutritional myopathy was not confirmed, as tissues were unavailable for selenium (Se) and vitamin E (Vit E) analysis. At the time of the current submission, all does were supplemented with selenium in the feed (rate unknown), and at birth, kids were injected intramuscularly with 0.5 ml of a commercial Se/Vit E preparation containing 3 mg/ml sodium selenite and 136 IU/ml dl-α-tocopherol acetate (Dystosel, Pfizer Canada Inc, London, Canada). In the current year, 4 kids had been born, and all 4 had died within 8 hours of parturition. Three carcasses were submitted for postmortem examination.
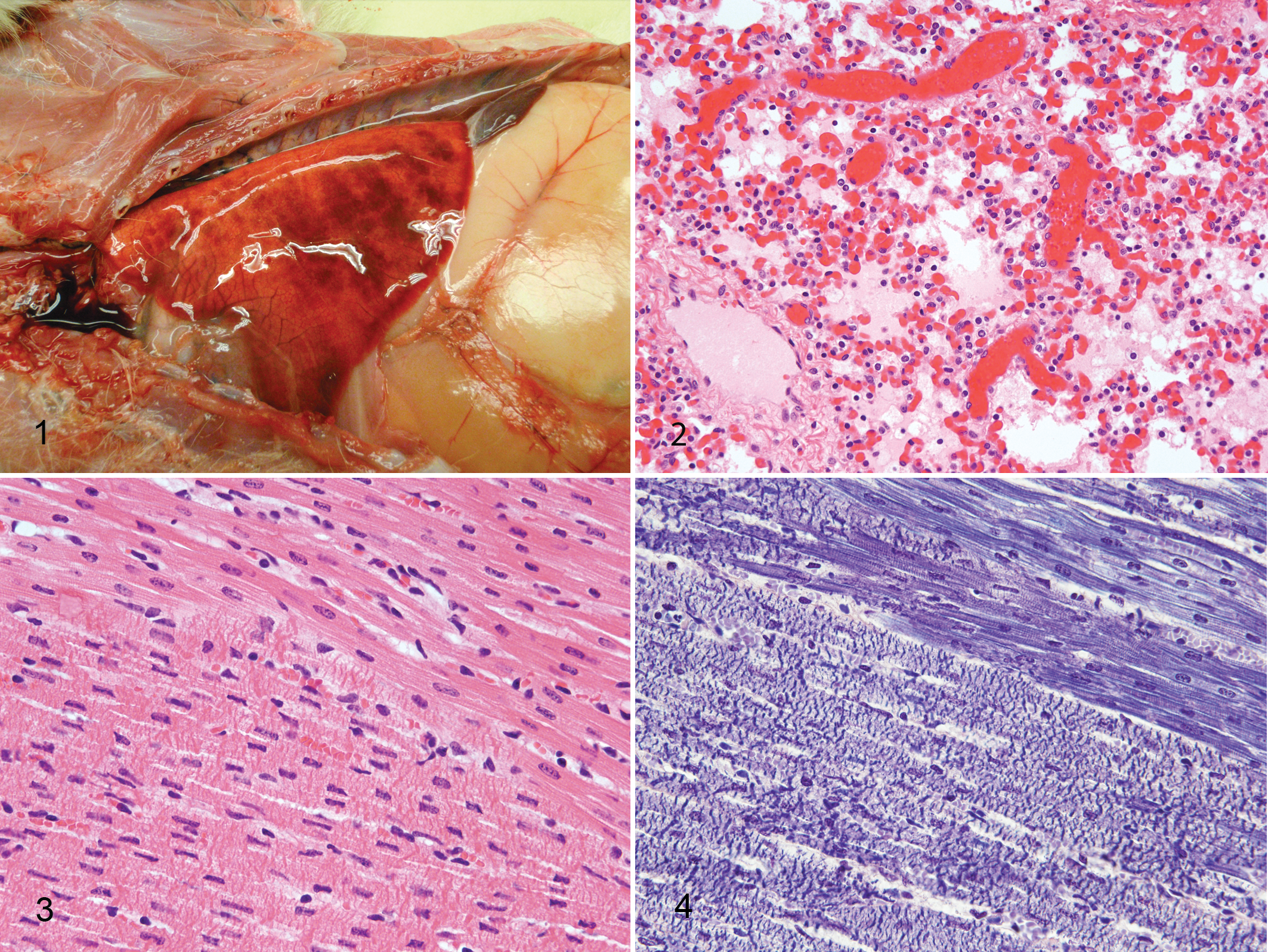
The carcasses weighed 3–4 kg each and were in good nutritional condition with no evidence of dehydration. The lungs of all 3 animals had severe diffuse edema evidenced by heavy, wet lungs that had failed to collapse; stable white froth in the major airways; and variably distended interlobular septa containing clear fluid (Fig. 1). Areas of red mottling were observed in the caudal lung lobes and were interpreted to be areas of hemorrhage (Fig. 1). Kids No. 2 and No. 3 had 40 ml of clear, straw-colored fluid within the thoracic cavity representing hydrothorax. Similar fluid was in the pericardial sac, which was minimally distended. The amount of fluid within the pericardium was not measured but was interpreted to be mildly increased. Diffuse hepatic congestion was observed in all 3 animals. Other macroscopic lesions that were inconsistently observed included serosal hemorrhage of the bladder (No. 1), bilateral congestion of the renal medulla (No. 2), and congestion of the meningeal vessels (No. 2). Gross lesions were not identified in any other tissues including the heart. Tissue samples from each kid were fixed in 10% buffered formalin for 24 hours, processed routinely for histological examination, and stained with hematoxylin and eosin. Fresh samples of lung, spleen, and kidney were submitted for aerobic and anaerobic bacterial culture, and liver samples were submitted for trace mineral analysis.
Differential Diagnoses
Based on the clinical history and necropsy findings, the first consideration was for myocardial injury leading to severe pulmonary edema, hydrothorax, and hydropericardium due to either Se/Vit E deficiency or Se toxicity. Given the history of acute neonatal death, other conditions such as septicemia and toxemia were also considered.
Microscopic Findings
All major organ systems were examined microscopically, and the most significant lesions were restricted to the lungs and myocardium. Severe changes in the lungs were noted in all 3 kids and included acute, diffuse, and severe pulmonary congestion and protein poor edema, with small, multifocal areas of intra-alveolar hemorrhage (Fig. 2). In all 3 goats, there were multiple, locally extensive areas of acute myocardial degeneration and necrosis, which were restricted to the left ventricular wall and interventricular septum. Affected areas were characterized by prominent, thick, eosinophilic, transverse bands; loss of cross-striations; sarcoplasmic hypereosinophilia; and nuclear pyknosis (Fig. 3). The myocardial injury was monophasic, and there was no evidence of mineralization. Areas of necrosis contained small focal hemorrhages. Phosphotungstic acid-hematoxylin staining 2 highlighted the loss of cross-striations and the presence of contraction bands (Fig. 4). All 3 goats also had mild diffuse hepatic congestion. The reason for the relatively mild hepatic congestion in the face of heart failure is not definitively known but could be explained by the localization of the myocardial lesions to the left side of the heart. Other findings were hemorrhage on the serosal surface of the bladder (No. 1), bilateral renal medullary congestion (No. 2), and meningeal congestion (No. 2). Lesions were not observed in the spinal cord, brain, thyroid gland, adrenal gland, spleen, thymus, lymph nodes, skeletal muscle, tongue, diaphragm, gastrointestinal tract, or bone marrow.
Laboratory Findings
Aerobic and anaerobic bacterial culture of the lung, spleen, and kidney failed to isolate any significant pathogens. Quantification of trace elements in liver samples from kid No. 1 and kid No. 2 was performed by Inductively Coupled Plasma Mass Spectrometry (ICP-MS0; X series, Thermo Electron Corporation, Winsford, UK). Liver Se values were 8.92 parts per million (ppm) in kid No. 1 and 11.6 ppm in kid No. 2. Normal liver Se values in goats are 0.25–1.20 ppm wet weight. 10
Diagnosis
Acute myocardial degeneration and contraction band necrosis associated with Se toxicity.
Discussion
This case illustrates the macroscopic and microscopic pathology of acute Se toxicity in Boer goat kids. Selenium is a metalloid that is an essential trace element but is toxic in excess.5,12 In animals, there is a fairly narrow range between Se deficiency and Se toxicosis. Diets containing <0.1 ppm result in deficiency, whereas those with concentrations >2.2 ppm are potentially toxic. 16 Two forms of Se toxicosis, acute and chronic, are described according to duration, level of exposure, and clinical signs.5,11,12 Signs of acute poisoning include restlessness, anorexia, respiratory distress, salivation, watery diarrhea, fever, tachycardia, abnormal posture and gait, prostration, and death after a short illness. Chronic poisoning is manifested by emaciation, rough hair coat, alopecia, lack of vitality, stiffness, lameness, and hoof abnormalities.11,12 Chronic forms of selenosis were previously called “alkali disease” and “blind staggers,” but these terms are now little used, as they are misleading and were based on misunderstandings of the cause. 11
In certain regions of the world, Se responsive disease and unthriftiness constitute important losses of production in livestock; therefore, use of Se/Vit E preparations for prevention of nutritional myopathy is common practice.9,16 The widespread use of inorganic Se salts therapeutically and prophylactically in combination with the narrow margin of safety has increased the risk of accidental poisoning. 4 Acute toxicity is almost always the result of oversupplementation or overdosing with parenteral preparations. 12 In lambs, the LD50 for intramuscularly administered sodium selenite has been reported to range from 0.45 to 1 mg of Se/kg of body weight.17 In the case reported here, the kids received between 0.38 and 0.5 mg Se/kg body weight, and all developed clinical signs and died within 8 hours of receiving the injection.
Experimental3,6,14,16 and accidental4,7-9 acute Se poisoning has been described in sheep, and accidental poisoning has been described in cattle. 13 Accidental cases are often associated with heavy losses. Literature review revealed only a single report of Se toxicosis in goats under experimental conditions. 1 To the authors' knowledge, this is the first report of accidental acute Se toxicosis in this species. In all 3 kids examined, the most significant lesion was acute myocardial degeneration and necrosis, and previous studies implicate the heart as the major target organ in selenium intoxication.6,14,16 A single report of experimental Se poisoning in goats 1 describes congestion and hemorrhage in the heart but not the myocardial contraction band necrosis observed in all 3 kids reported here. Previous studies suggest that the heart has an affinity for Se and accumulates a higher concentration than other tissues, 14 and myocardial damage has been positively correlated with Se concentration in the heart muscle. 16 Myocardial selenium concentrations could not be assessed in this case because samples were not available for analysis.
Severe pulmonary edema, hydrothorax, and hydropericardium were the most significant gross lesions in these kids and are believed to be manifestations of heart failure. However, Tiwary et al 16 suggested that in sheep, other mechanisms including alveolar vasculitis may contribute to pulmonary edema. In the current cases, there was no light-microscopic evidence of vasculitis. In addition, protein-poor pulmonary edema and congestion were considered to be more consistent with acute heart failure than with endothelial damage, as the latter is usually associated with protein-rich edema. Other lesions that have been inconsistently reported in acute selenosis in sheep include acute tubular nephrosis,6,8,9,14 atrophy of lymphoid tissue,6,14 and hepatocellular degeneration,6,14 but these features were not observed in the current case. Swine are the only species to consistently develop neurological signs as a result of Se toxicity, and the signs are attributed to poliomyelomalacia. 12 Interestingly, ataxia has been reported as a clinical sign in lambs 13 suffering from selenium toxicosis, and 1 report suggested that the hindlimb paresis in goats may be a manifestation of poliomyelomalacia, 1 although the spinal cord was not evaluated microscopically. In the cases described here, lesions were not observed in the spinal cord, and the ataxia observed clinically was attributed to generalized weakness and shock.
The specific mechanisms of Se toxicity have not been fully elucidated, but recent evidence suggests that oxidative stress contributes significantly.12,16 Certain chemical forms of Se react with tissue thiols to produce reactive oxygen species, which is exemplified by the reaction of selenite with reduced glutathione in vitro to produce superoxide anion and elemental Se. 15 Also, Vit E concentration in the liver of severely affected lambs given sodium selenite was significantly reduced, indicating increased oxidative stress. 16 The link between Se toxicity and oxidative injury is somewhat counterintuitive, as Se is primarily considered a powerful antioxidant because of its role in the glutathione peroxidase system. Given the potential for a common mechanism of cell injury in both Se deficiency and Se toxicity, it is not surprising that the primary lesion in these 2 diseases is myocardial necrosis. Therefore, in cases of acute myocardial necrosis, it is imperative that tissue Se levels are quantified before a diagnosis of Se deficiency is rendered.
In conclusion, we report the first case of accidental acute selenosis in goats that manifested as acute myocardial necrosis leading to severe pulmonary edema, hydrothorax, and hydropericardium. This report emphasizes the toxic potential of parenteral Se therapy and highlights an important differential diagnosis that needs to be considered in sudden death of goat kids. This case also illustrates the overlapping mechanisms and histopathological features of Se toxicosis and Se/Vit E deficiency and emphasizes the need for tissue Se quantification to confirm the diagnosis.
Footnotes
The author(s) declared no potential conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) received no financial support for the research and/or authorship of this article.
