Abstract
During postslaughter inspection of a 4-year-old male dromedary camel (Camelus dromedarius), numerous small nodules to large masses up to 4 cm in diameter were found on the serosal surfaces of forestomachs, large intestines, mesentery, liver, and spleen. Grossly, the masses were discrete, round, smooth, and white to gray that bulged from the serosal layer. Cut surfaces of the masses were discrete, round, white, and relatively homogeneous without any necrotic foci. Histopathologically, the masses were encapsulated and composed of a mixture of round and spindle-shaped cells in loose whorls of neoplastic cells with small elongated hyperchromatic wavy nuclei and a small amount of pale eosinophilic, poorly defined cytoplasm. Masson’s trichrome staining showed mild amounts of collagen fibers forming an irregular, loose stroma. In immunohistochemistry, immunoreactivity for the Schwann cell marker (S100) was diffusely positive in the neoplastic cells. The immunoreactivity for CK, c-kit, and CD34 were negative. Ultrastructural examination confirmed the tumor was entirely formed of neoplastic Schwann cells. On the basis of the histopathological, immunohistochemical, and ultrastructural findings, the tumors were diagnosed as multicentric fibromyxoid peripheral nerve sheath tumor (multicentric schwannoma). This tumor has not been previously recorded in camel worldwide.
Generally, peripheral nerve sheath tumors (PNSTs) occur as slow-growing tumors that can be located anywhere in the peripheral nervous system. They are derived from either Schwann cells or modified Schwann cells, fibroblasts, or perineurial cells.12,22 These tumors are relatively common in humans but occur infrequently in domestic animals. 23 In humans, PNSTs are subclassified as benign (BPNSTs) or malignant (MPNSTs) and include schwannomas (neurilemmomas) and neurofibromas, as well as perineurial cell tumors, nerve sheath myxomas (neurothekomas), and a few other entities.4,17,20,28 Terminology used for nerve origin neoplasms in animals is quite inconsistent and confusing, and different terms have been used by different pathologists to identify the same tumor.18,30 The World Health Organization classification of tumors of the nervous system and mesenchymal tumors of skin and soft tissues in domestic animals combines malignant and benign forms of neurofibroma and schwannoma under the title of PNST.9,11 Schwannomas and neurofibromas are the most common BPNSTs. Certain histopathologic patterns supported by immunohistochemistry and electron microscopy are often necessary in establishing the diagnosis of these tumors.4,18,20,28 In humans, schwannomas and neurofibromas are subclassified according to their growth patterns (schwannoma: localized or plexiform; neurofibroma: localized, plexiform, or diffuse) and histopathologic features (eg, classic, collagenous, cellular, and pigmented).18,28 Schwannomas of animals have been best recognized in the canine and bovine species and less commonly in the cat. 30 Neurofibromas have not been convincingly documented in animals, but more recently, localized, plexiform, diffuse, and other variants of this tumor have been reported in dogs, horses, and a chicken, and studies identified the existence of multiple subtypes of neurofibroma in animals similar to humans. 18
In animals, PNSTs are most often reported in dogs and cattle,23,30 infrequently in cats and horses,15,22 and rarely in other species, such as goats,16,26 pigs, 25 and birds. 14 Spontaneous cases were also reported in laboratory animals. 7
To date, there is no report about the occurrence of these tumors in the dromedary camel. Therefore, for the first time, this article describes gross, histopathological, immunohistochemical, and ultrastructural features of a rare PNST as a multicentric fibromyxoid PNST (schwannoma) in a dromedary camel (Camelus dromedarius).
During postslaughter inspection of a 4-year-old male dromedary camel (C dromedarius) in Yazd Province, Iran, slaughterhouse, numerous small nodules to large masses up to 4 cm in diameter were found on the serosal surfaces of forestomachs, large intestines, mesentery, liver, and spleen. Selected tissues of the nodules and masses were processed for light microscopic, electron microscopic, and immunohistochemical evaluation. Samples of tumoral tissues were fixed in 10% neutral buffered formalin, processed routinely, and embedded in paraffin wax. Sections (5 μm) were stained by hematoxylin and eosin and Masson’s trichrome. Consecutive sections to those used for the histopathological examination were subjected to immunohistochemistry with primary monoclonal antibodies specific for S-100 protein (S1/61/69), pan-cytokeratin (CK), c-kit (CD117), and CD34 (Novocastra Laboratories, Newcastle, UK). Immunolabeling was detected with an avidin-biotin conjugate procedure (Novocastra Laboratories) with positive internal and external controls.
For the ultrastructural study, tissue samples were fixed in 4% glutaraldehyde solution (pH 7.3), washed in buffer phosphate, and then postfixed in 2% osmium tetroxide. After further washing, the tissues were progressively dehydrated in graded ethanol solutions, cleared in propylene oxide, and embedded in agar resin. At least 3 semithin sections from each sample stained with toluidine blue and the areas of interest were selected by light microscope. Ultrathin sections were contrasted with lead citrate and uranyl acetate and were examined by using a Philips CM-10 transmission electron microscope.
Grossly, the nodules and masses were often discrete, round, smooth, and white to gray that bulged from the capsular surface or serosal layer of affected parts of the alimentary tract, including forestomachs (Fig. 1), large intestines (Fig. 2), mesentery (Fig. 3), liver (Fig. 4), and spleen. Cut surfaces of the masses contained discrete, round, white to gray, and relatively homogeneous tumors without any necrotic or liquefied foci in the centers (Fig. 4). The masses in the liver were about 1–3 cm, but masses on the serosal surfaces of intestines and mesentery were somewhat smaller. No significant gross lesions were observed in the other body tissues or organs.
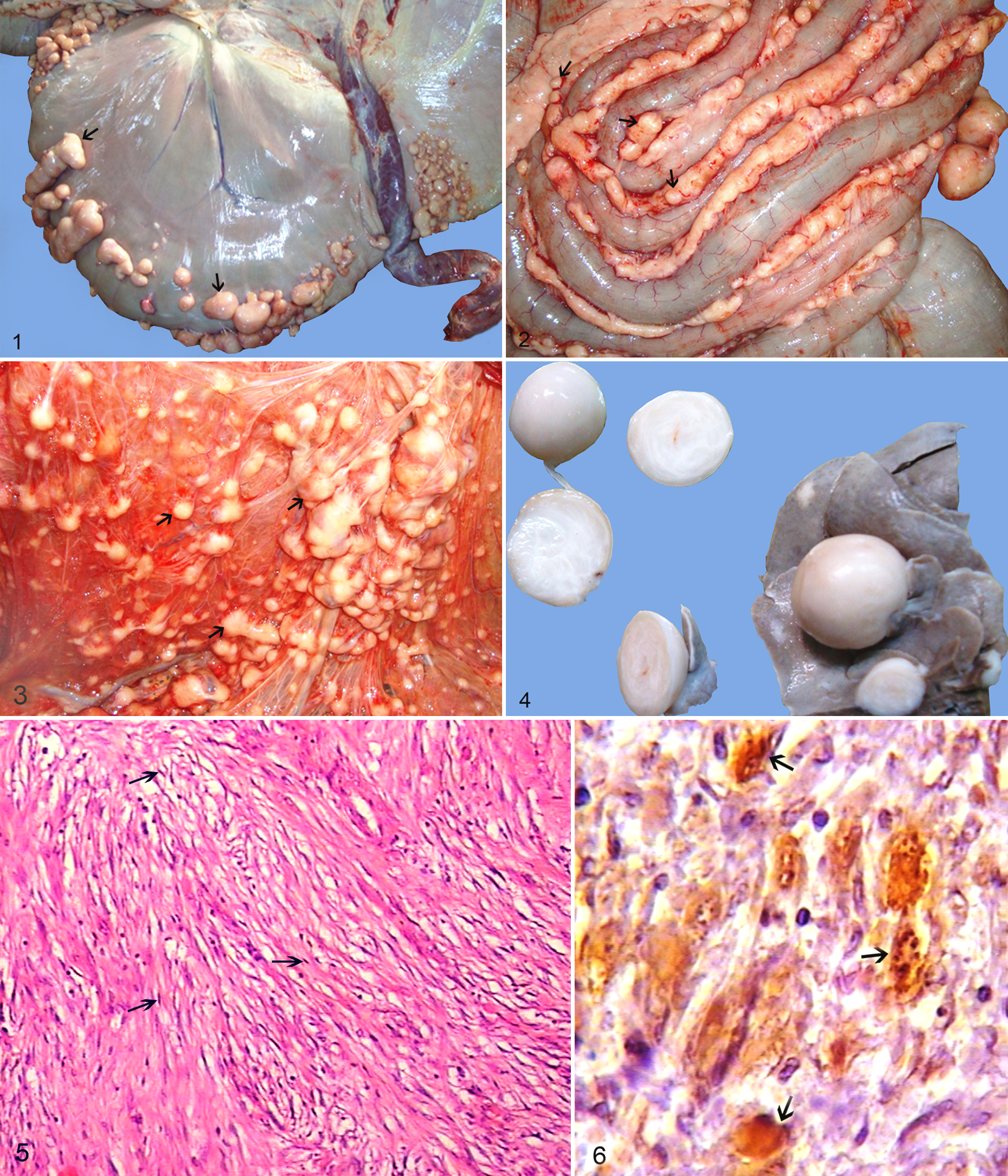
Histopathologic examination of the masses revealed a multicentric neoplastic process with a common cell type and growth pattern. They were encapsulated and composed of loose whorls of elongated spindle cells with indistinct cell borders. The tumor had low to moderate cellularity, including a mixture of round and spindle-shaped cells in a loose cellular arrangement. The cells were embedded in a loose fibromyxoid matrix. Antoni type B appearance was seen as a loose arrangement of sheets and whorls of neoplastic cells with small elongated hyperchromatic wavy nuclei and a small amount of pale eosinophilic, poorly defined cytoplasm (Fig. 5). The mitotic rate was very low, with less than 1 mitotic figure per ten 40× fields. There were no nerve fibers within the tumor tissue. Staining with Masson’s trichrome showed small amounts of collagen fibers forming an irregular, loose stroma with scanty dense bundles in the stroma. In immunohistochemistry, immunoreactivity for the Schwann cell marker (S100) was diffusely positive in the neoplastic cells (Fig. 6). The immunosignal ranged from variable degrees of moderate to intense intracytoplasmic staining. The results of immunohistochemistry for CK, c-kit, and CD34 were negative.
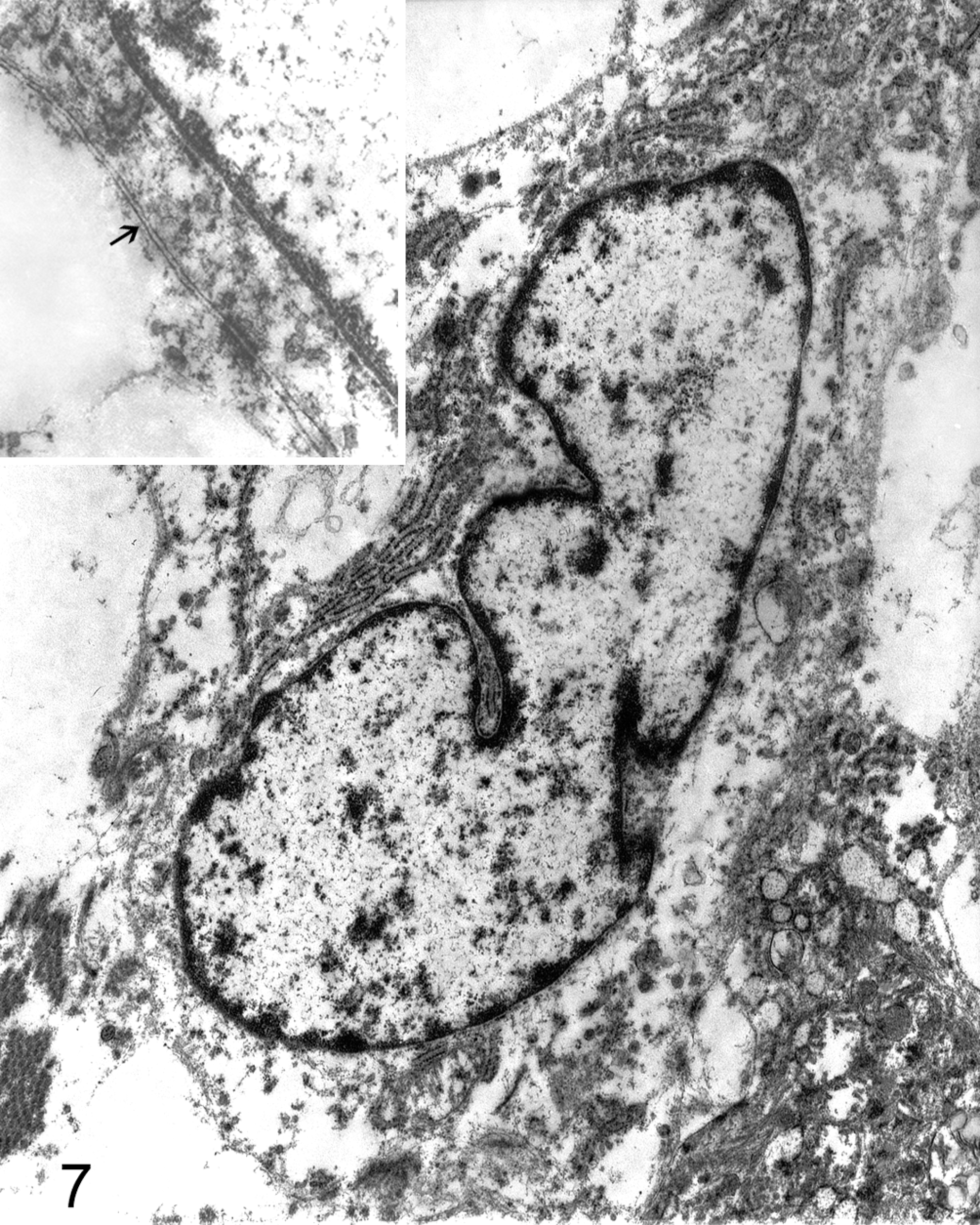
Ultrastructural examination confirmed the tumor was entirely formed of neoplastic Schwann cells. Neoplastic Schwann cells had elongated nuclei and double cytoplasmic membrane and were identified by their continuous basal lamina and branched cytoplasmic processes. Nuclear membrane was generally smooth with occasional marked indentation. The chromatin pattern was essentially diffuse with patches of heterochromatin more prominent adjacent to the nuclear membrane (Fig. 7).
On the basis of the gross, histopathological, immunohistochemical, and ultrastructural findings, the tumors were diagnosed as multicentric fibromyxoid peripheral nerve sheath tumor (multicentric schwannoma).
Although camelid neoplasia has been rarely reported in the literature, it is presumed that camelids are susceptible to all the various tumor types that affect domestic ungulates. 5 Vertebral osteoma, 2 mammary and pulmonary carcinoma, 1 multicentric t-cell lymphoma, 19 renal cell carcinoma, 27 bronchoalveolar adenocarcinoma, 6 and recently peripheral primitive neuroectodermal tumor 29 have been reported in the dromedary camel.
In veterinary medicine, the generic diagnosis of BPNST is often used because clear diagnostic criteria for the distinction between neurofibroma and schwannoma in animals have not been established, and all BPNSTs are believed to have a similar prognosis.8,11
The tumors in this study were diagnosed as multicentric schwannoma in a dromedary camel based on its morphopathological features together with the absence of intratumoral nerve fibers, the diffuse pattern of S100 (the Schwann cell marker) immunostaining of the neoplastic Schwann cells, CD34-negative and also CK, c-kit–negative stromal cells. Also, the diagnosis was supported by the ultrastructural findings, which confirmed the uniform presence of Schwann cells. Several immunostains can be of utility in assisting with the differential diagnosis of schwannoma and neurofibroma. In addition to S100, CD-34 is a useful stain for the differential diagnosis, since neurofibromas typically demonstrate a significant subpopulation of CD34-positive stromal cells, unlike most schwannomas. CD34 and c-kit negativity rules out gastrointestinal stromal tumors.
In animals and humans, PNSTs occur in both cranial and spinal nerves of the PNS. In humans, neurofibroma and schwannoma are well-characterized, distinct entities within the category of BPNSTs. 21 The schwannoma is characterized by having variable proportions of 2 morphologic features, known as Antoni A and B areas. Antoni A areas are highly cellular and consist of monomorphic spindle-shaped Schwann cells with nuclear palisading (Verocay bodies) present in a collagenous stroma. Antoni B areas are also composed of Schwann cells, but their nuclei appear to be suspended in a copious myxoid, often microcytic matrix. 30 Also, schwannomas do not typically contain nerve fibers and immunoreactivity because S100 protein is strong and diffuse. Schwannomas should be differentiated from neurofibromas, which consist of a mixture of Schwann cells, perineurial cells, and fibroblasts. The common microscopic features of the neurofibromas include elongated spindle cells with poorly defined pale eosinophilic, tapering wavy or buckled nuclei, and numerous small nerve fibers. Within the tumor, nerve fibers are present and S100 immunoreactivity is restricted to a subpopulation of the tumor cells (Schwann cells) but not the remaining tumor cells.4,13,17,20,28 The presence of mast cells in the tumor is also reported. 30
Schwannomas occur most frequently in cattle, in which they are usually benign and often multiple. 3 A schwannoma was reported in the cecum of a cow. 24 In cattle, the tumor occurs most commonly in mature animals, although the lesion has also been reported in young calves and involves the cranial eight nerve, brachial plexus, and intercostal nerves. In addition, autonomic nerves of the liver, heart, mediastinum, and thorax can be affected. 30 Previously, in adult cattle, development of multiple PNSTs with a predilection for the autonomic nervous system has been referred to as neurofibromatosis, although they were microscopically similar to human schwannomas. 23 BPNST occur very rarely in horses but have been described in the periorbital region, extradurally in the cranium, in the small intestine, in the skin, and in the spinal cord. Also, multiple subserosal and intramuscular neurofibromas and schwannomas in association with hyperplasia of the myenteric plexus have been reported. 10 BPNSTs were reported in a young pony with concentric laminations of very loosely arranged Schwann cells embedded in an intervening myxomatous stroma (Antoni type B appearance). 22 An MPNST originating from a thoracic spinal nerve with metastasis to the liver, lungs, spleen, and vertebral canal has been described in a goat. 26 In the present case, Antoni type B appearance of the tumor tissue is consistent with schwannoma. Our report confirms the value of immunohistochemistry and electron microscopy in association with histopathology in differentiating schwannoma from neurofibroma. Also, this report presents the first case of multicentric PNST (multicentric schwannoma) in a camel.
Footnotes
Acknowledgements
The authors are grateful to Mr L. Shirvani and Mr G. Yousefi from the Department of Pathology; Mr A. Safavi from Electron Microscopy section, School of Veterinary Medicine, Shiraz University; and Dr Y. Daneshbod (A1) and Mrs K. Kalani from the Daneshbod Pathology Laboratory, Shiraz, Iran, for technical assistance.
The author(s) declared no conflicts of interest with respect to the authorship and/or publication of this article.
The author(s) received no financial support for the research and/or authorship of this article.
