Abstract
Beta–gamma bridging (β-γ bridging) on serum protein electrophoresis is touted as being virtually pathognomonic for hepatic disease. However, the criteria for β-γ bridging are not defined, and few publications support a relationship between β-γ bridging and liver disease. The goal of this retrospective study was to evaluate the prevalence of hepatic pathology in animals with β-γ bridging. All serum protein electrophoretograms from clinical patients generated at the University of Georgia between 1994 and 2008 were evaluated for the presence of β-γ bridging, defined as (1) an albumin:globulin ratio below the reference interval; (2) indistinct separation between all β and γ globulin fractions or between the β2 and γ fractions, with a negative shoulder slope of < 5%; and (3) predominance of γ proteins versus β proteins. Of the 237 electrophoretograms examined, 25 (11 dogs, 11 cats, 3 horses) met the inclusion criteria for β-γ bridging. Patients were classified into disease categories on the basis of biochemical, cytologic, and/or histologic findings. Positive predictive values of β-γ bridging for hepatic and infectious diseases were determined with a one-sided exact binomial test. Of 25 animals, 8 had evidence for hepatic disease, whereas 9 had infectious diseases. As such, the positive predictive value of β-γ bridging for hepatic disease was 32.0%, with a 95% confidence interval of 15.0% to 53.5% (P < .001), whereas for infectious disease, the positive predictive value was 36.0%, with a similar confidence interval. Beta–gamma bridging is not pathognomonic for liver diseases and is as frequently found with infectious diseases.
Beta–gamma (β-γ) bridging, also known as β-γ linking 4 and β-γ fusion, 11 is an uncommon pattern observed on electrophoretograms following serum protein electrophoresis. Despite its frequent usage, the term β-γ bridging is actually poorly defined. The use of this terminology is commonly based on the lack of demarcation between the peaks of the β and γ globulins on an electrophoretogram, along with a decreased albumin:globulin ratio (A:G). 29 According to many human and veterinary medicine and diagnostic pathology textbooks published over the past 60 years, the pattern of β-γ bridging is considered “virtually pathognomonic” for chronic active hepatitis or hepatic cirrhosis. 5,14,15,18 Reports of this correlation date back to the early 1950s and are based on studies by Demeulenaere and Wieme, utilizing paper serum protein electrophoresis, 4 and Sunderman and Sunderman, utilizing sucrose gradient electrophoresis of sera. 28 The relationship between liver disease and this electrophoretic pattern is presumed to result from increased levels of various protein fractions, predominantly immunoglobulins A and M, resulting in poor separation between the β and γ fractions. 18,19 Despite the reported occurrence of β-γ bridging in other nonhepatic diseases—including lymphoma, mycobacteriosis, heartworm disease, leishmaniasis, and γ heavy chain disease—human medical sources suggest that the presence of a β-γ bridge on an electrophoretogram warrants a liver biopsy as the next appropriate diagnostic step. 2,8,12,18 The original studies of β-γ bridging in people have been widely accepted as being applicable to domestic species, but to our knowledge, it has not been evaluated in animals.
The positive predictive value (PPV) is defined as the probability that a positive test result indicates that the animal has the condition being evaluated. 26 Unlike test sensitivity, PPV varies with disease prevalence: When disease prevalence is high, a patient is more likely to have the disease, regardless of the test result; thus, false-negative results are more likely. For a test to be considered pathognomonic for a disease, the PPV should be 100%; all animals with the disease test positive, whereas all animals without the disease test negative. If β-γ bridging is virtually pathognomonic for hepatic pathology, the PPV should be high. The aim of this retrospective study was to determine the prevalence of hepatic pathology in animals with β-γ bridging, based on biochemical, cytologic, and histologic findings consistent with liver disease.
Materials and Methods
All mammalian serum protein electrophoresis profiles were evaluated as generated on hospitalized patients at the University of Georgia’s clinical pathology laboratory between September 1994 and July 2008. In all cases, protein electrophoresis was performed with cellulose acetate gels (Titan III Zip Zone Plates, Helena Laboratories, Beaumont, TX) stained with Ponceau S and sulfosalicylic acid (Helena Laboratories), and electrophoretograms were produced by a scanning densitometer utilizing ultraviolet light detection (Cliniscan 2, Helena Laboratories). All electrophoretograms were evaluated by two board-certified clinical pathologists for β-γ bridging, without prior knowledge of any medical information or previous interpretation. Electrophoretogram analyses were performed under the premise that all proteins to the cathode side of midline were considered to be β and γ globulins.
26
Based on the literature and a collective discussion among the pathologists, the following criteria were used to define a β-γ bridge (Fig. 1
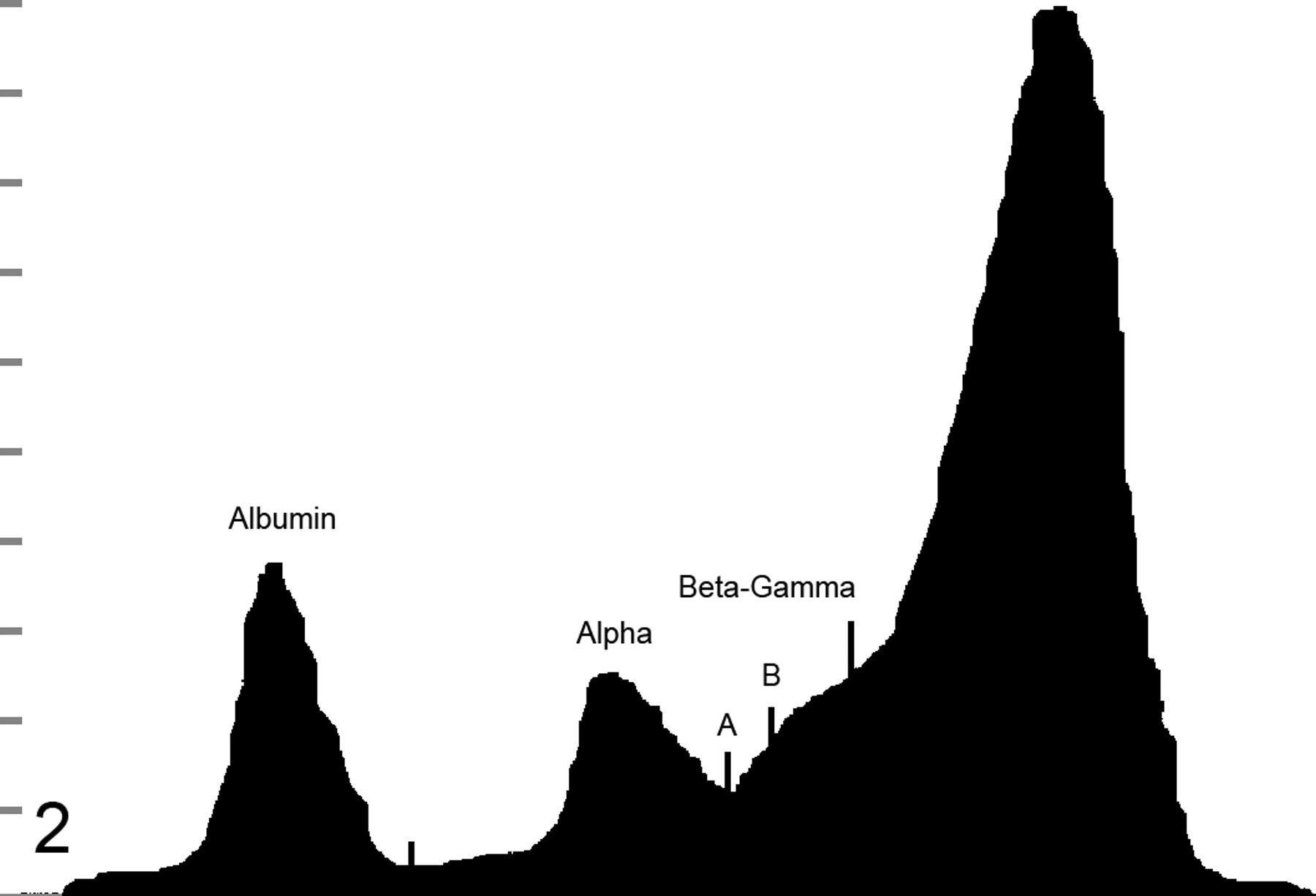
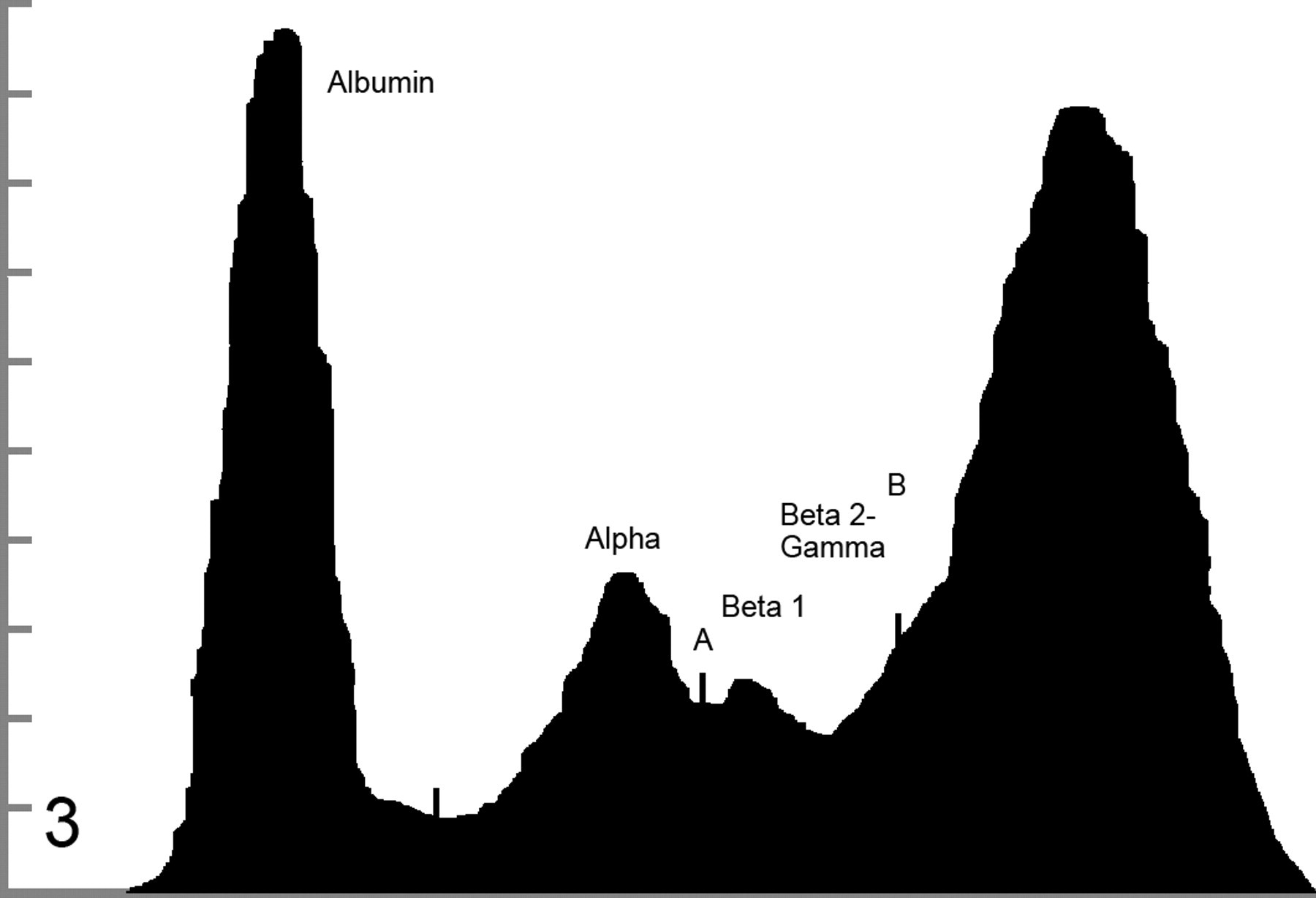
): The A:G ratio was ≤ 0.6 (the low end of the reference interval established at the University of Georgia’s College of Veterinary Medicine for all included species). There was a lack of obvious separation between all β and γ globulin fractions (Fig. 2; type 1 β-γ bridge) or between the β2 and γ fractions (Fig. 3
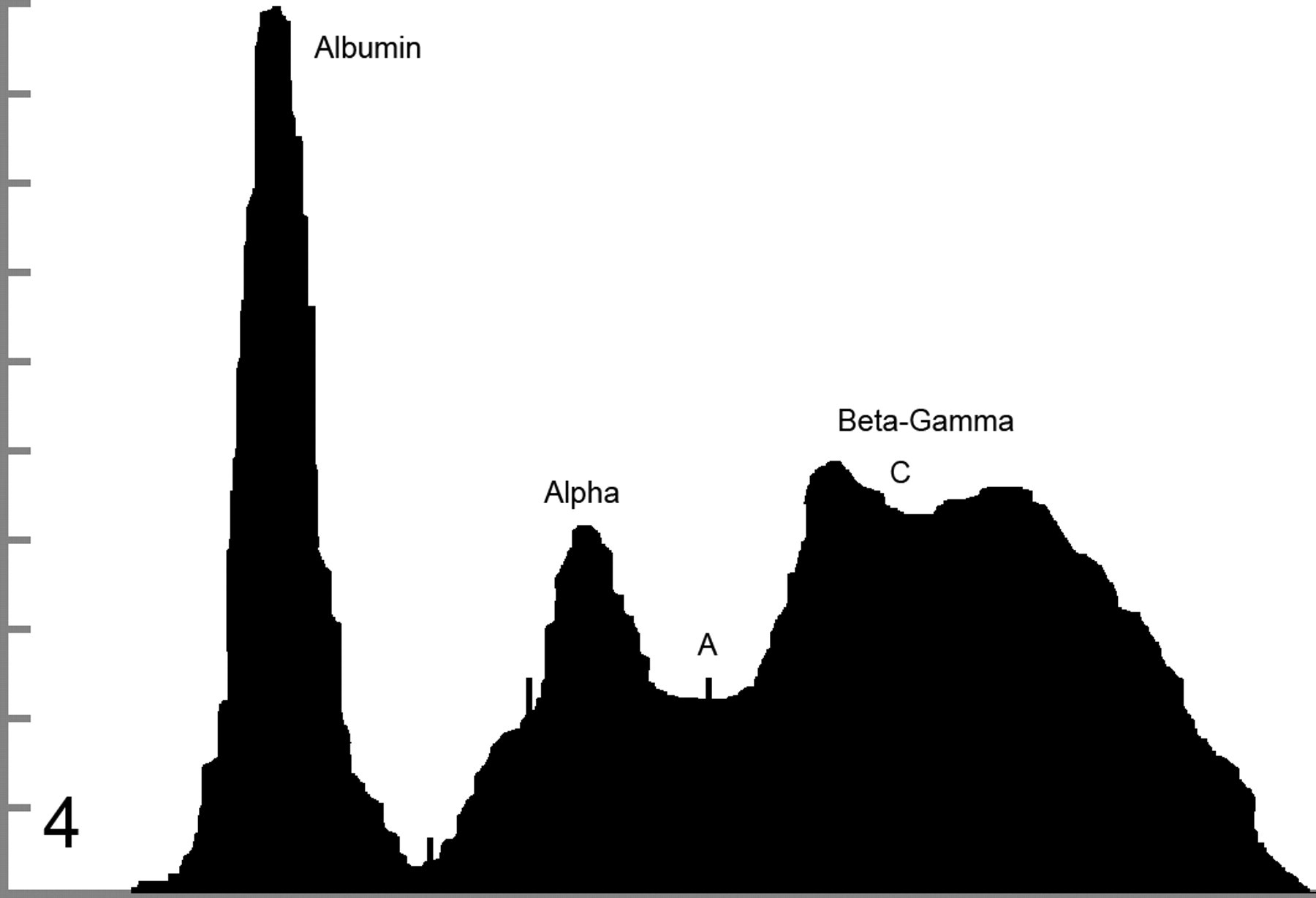
; type 2 β-γ bridge). If any visible trough on the β-γ bridge was present, it was less than 5% of the height of the β peak, based on the sensitivity of our densitometer. In these cases, a distinct separation between the β and γ regions could not be definitively identified (Fig. 4
; type 3 β-γ bridge).
10
Decision tree illustrating classification process for beta–gamma (β-γ) bridging. Solid arrows indicate yes decisions. Open arrows indicate no decisions. A:G, albumin:globulin. Type 1 beta–gamma bridge: lack of distinction between all beta and gamma components. A, midpoint; B, shoulder. Albumin, 2.2 g/dl; alpha, 2.1 g/dl; beta–gamma, 11.0 g/dl. Type 2 beta–gamma bridge: lack of distinction between beta 2 and gamma fractions. A, midpoint; B, shoulder. Albumin, 2.43 g/dl; alpha, 1.13 g/dl; beta–gamma, 5.44 g/dl. Type 3 beta–gamma bridge: trough less than 5% of the height of the beta fraction. A, midpoint; C, trough. Albumin, 1.84 g/dl; alpha, 1.42 g/dl; beta–gamma, 4.14 g/dl. Based on the area under the curve, the highest concentration of proteins is found in the quartile most closely associated with the cathode, indicating a γ predominance rather than a β predominance.
For the cases that met the initial inclusion criteria, medical records were examined to determine biochemical abnormalities and/or cytologic or histologic evidence for liver disease, along with a final diagnosis. The animals that met the inclusion criteria and had adequate follow-up data were then categorized as having hepatic or nonhepatic disease. For inclusion as a case of liver disease, animals had to possess either cytologic or histologic evidence for hepatic pathology, or they had to exhibit at least one of the major, or two of the minor, biochemical indicators of hepatic damage or dysfunction, as outlined in Table 1
. Cutoff values for biochemical parameters were generally based on twofold changes in magnitude above or below the upper and lower ends of the reference interval, respectively. Patients not demonstrating microscopic or biochemical evidence for liver involvement were classified as having nonliver disease and were further subclassified as having infectious, inflammatory, or other etiologies, based on the final diagnosis. If a final diagnosis was not reached, patients were classified as simply having hepatic or nonhepatic disease, with no further subclassification. Potential associations between all protein and biochemical parameters were visually assessed on scatter plots using Cartesian coordinates, as generated with spreadsheet software (Microsoft Office Excel 2007). Then, linear regression analysis was performed to evaluate any potentially significant associations; PPV and a 95% confidence interval were determined with a one-sided exact binomial test (SAS 9.1). For this analysis, we defined virtually pathognomonic as an expected value of 95%.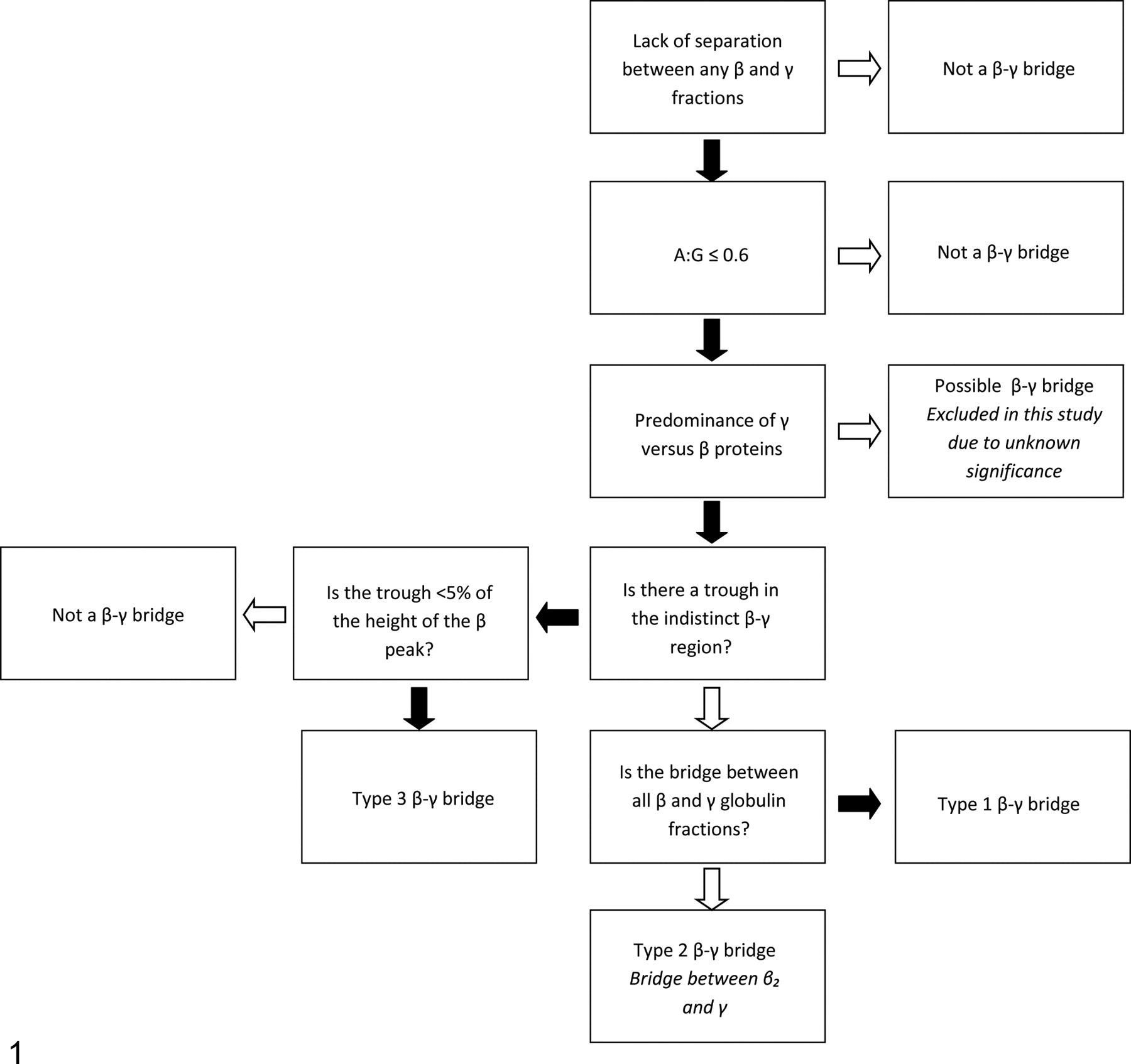
Biochemical Indicators of Hepatic Damage or Dysfunction Used to Support a Diagnosis of Liver Disease in Patients With Beta–Gamma Bridging a
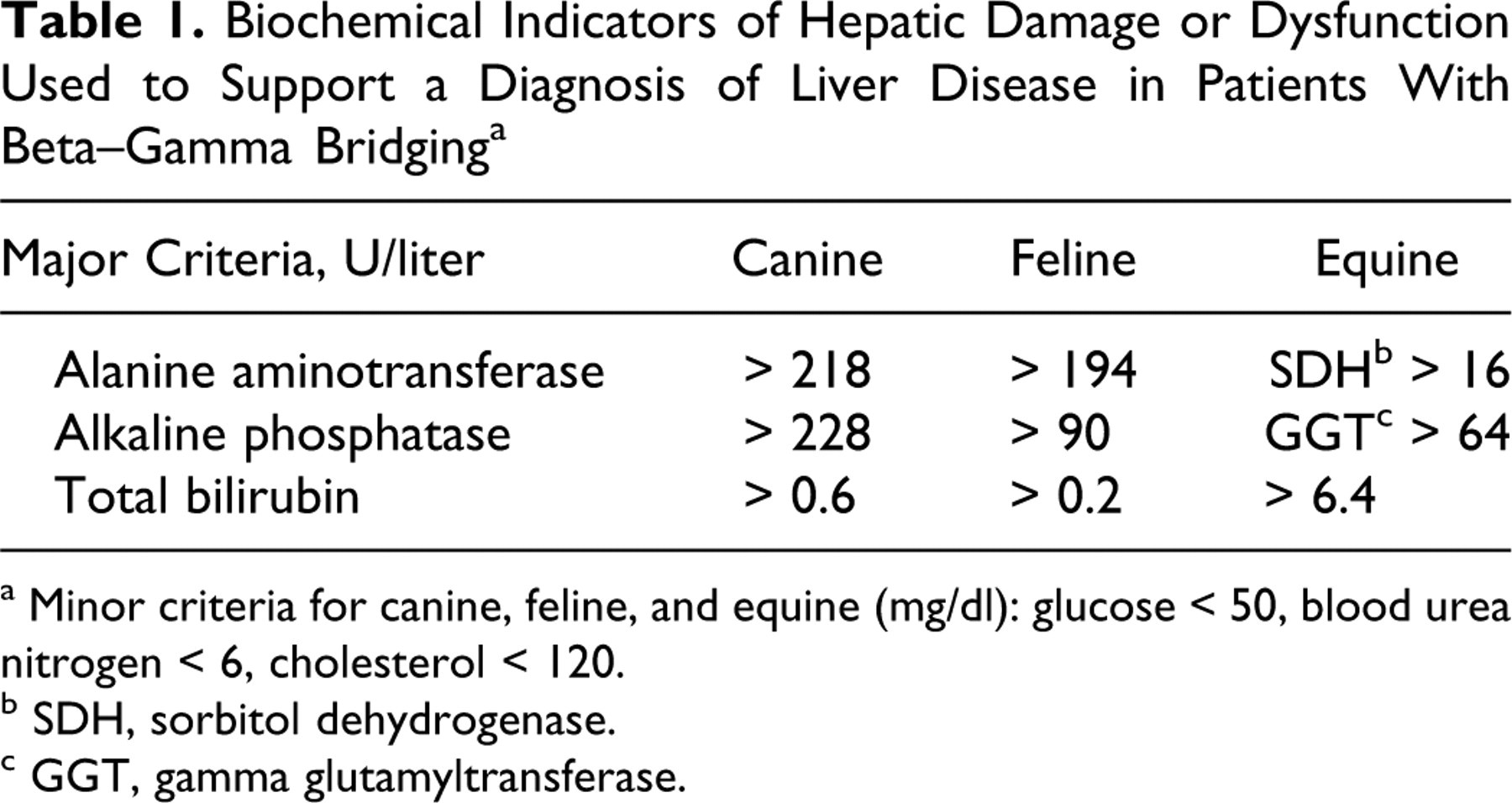
a Minor criteria for canine, feline, and equine (mg/dl): glucose < 50, blood urea nitrogen < 6, cholesterol < 120.
b SDH, sorbitol dehydrogenase.
c GGT, gamma glutamyltransferase.
Results
In summary, 237 electrophoretograms were performed between September 1994 and July 2008. Based on the defined criteria, 36 cases were initially identified as having a β-γ bridge. Of these initial 36 cases, only 25 met all the inclusion criteria. Table 2 presents the results for these 25 cases. Eleven dogs (ages, 1–11 years), 11 cats (ages, 7 months–14 years), and 3 horses (ages, 2 months–16 years) were included. Eight of these animals (32%) had microscopic or biochemical evidence for liver disease: Case Nos. 1 and 2 had intrahepatic neoplasia; case No. 3 had hepatic lipidosis; case Nos. 4 and 5 had infectious diseases directly involving the liver (leishmaniasis and bacterial septicemia); and case Nos. 6–8 had an undetermined disease process involving the liver. Case No. 6 was a dog with dramatically increased liver enzyme activities and a provisional clinical diagnosis of cholangiohepatitis. Unfortunately, this patient was discharged without further diagnostics or a final diagnosis. The second animal with unclassified liver disease (case No. 7) was a cat with increased alanine aminotransferase and a palpably enlarged liver. The cat had a provisional diagnosis of feline infectious peritonitis (FIP). This animal was euthanized without a postmortem examination. The third animal with unspecified liver disease (case No. 8) was a horse with extensive nodular hepatic lesions. A liver biopsy revealed multifocal inflammatory foci and fibrosis, which were interpreted as “potentially consistent with chronic active hepatitis.” Hairy vetch toxicosis was a primary consideration but never confirmed.
Case Information for 25 Animals With Beta–Gamma Bridging: Type, Disease Category, Basis for Classification, and Final Diagnosis
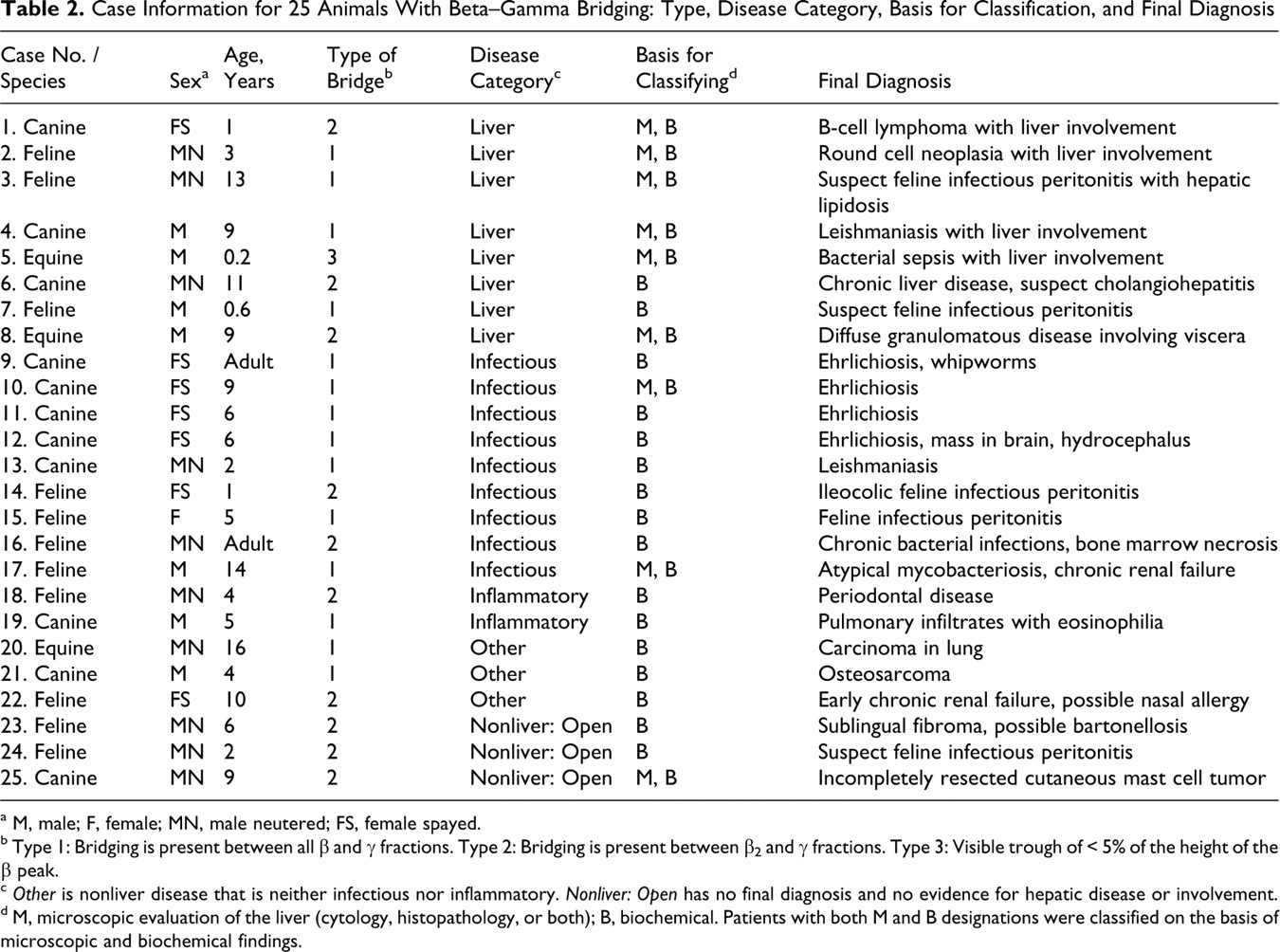
a M, male; F, female; MN, male neutered; FS, female spayed.
b Type 1: Bridging is present between all β and γ fractions. Type 2: Bridging is present between β2 and γ fractions. Type 3: Visible trough of < 5% of the height of the β peak.
c Other is nonliver disease that is neither infectious nor inflammatory. Nonliver: Open has no final diagnosis and no evidence for hepatic disease or involvement.
d M, microscopic evaluation of the liver (cytology, histopathology, or both); B, biochemical. Patients with both M and B designations were classified on the basis of microscopic and biochemical findings.
The remaining 17 animals (68%) with a β-γ bridge on electrophoresis did not have microscopic or biochemical evidence of liver disease. Of these animals without liver involvement, 9 (53%) had infectious diseases that did not result in clinical hepatic disease, based on biochemical and microscopic findings. Of the 9 animals with infectious diseases but not liver disease, 4 dogs (case Nos. 9–12) had ehrlichiosis; 1 dog (case No. 13) had leishmaniasis; 2 cats (case Nos. 14 and 15) had FIP; and 2 cats (case Nos. 16 and 17) had bacterial septicemia. Of the 17 animals without cytologic, histologic, or biochemical evidence for liver disease, 2 (12%) had inflammatory diseases not associated with a proven infectious etiology: Case No. 18 was a cat with severe periodontal disease; case No. 19 was a dog diagnosed with pulmonary infiltrates with eosinophilia. Of the 17 animals, 3 (18%) had diseases classified as other (neither hepatic nor inflammatory/infectious disease): 2 (12%; case Nos. 20 and 21) were diagnosed with nonhepatic neoplasia, and 1 (6%; case No. 22) was a cat diagnosed with chronic renal failure. The remaining 3 patients did not have definitive diagnoses but lacked cytologic, histologic, or biochemical evidence for hepatic disease: 2 (12%; case Nos. 23 and 24) were cats suspected of having infectious diseases (bartonellosis and FIP, respectively), but neither was confirmed; the third animal (case No. 25) was a dog with an incompletely resected cutaneous mast cell tumor.
Based on visual inspection of scatter plots, a positive correlation appeared between total protein concentration and increases in the γ and β-γ fractions of liver and nonliver patients. This relationship was confirmed with linear regression analysis (P < .001). Linear regression analysis did not reveal correlations between any other biochemical parameter and the total serum protein concentration or protein fraction concentration. The PPV of β-γ bridging for hepatic disease was calculated to be 32% (P < .001), with a 95% confidence interval of 15.0% to 53.5%. The PPV of β-γ bridging for infectious disease not specifically associated with the liver was determined to be 36.0%, with a 95% confidence interval of 18.0% to 57.5% (P < .001).
Discussion
Based on the results of this retrospective study and despite the propagation of this information in the veterinary literature, β-γ bridging can occur in the absence of overt liver disease. In the present study, only 8 of 25 animals (32%) had hepatic disease, and only 1 of these was suspected to have chronic active hepatitis. None of the animals had clinical, biochemical, or microscopic evidence for hepatic cirrhosis.
For this analysis, we defined virtually pathognomonic as an expected PPV of 95% (ie, 95% of animals that have β-γ bridging truly have hepatic disease). In the study population of this report, the 95% confidence intervals for a PPV for hepatic disease ranged from 15.0% to 53.5%. Although some animals with liver disease certainly have β-γ bridging on electrophoretograms, these results indicate that most animals with bridging can be expected to have other diseases that do not affect the liver. Animals with confirmed liver disease and animals with infectious disease not involving the liver had similar PPVs and confidence intervals.
The lack of clearly defined universal criteria makes identification of β-γ bridging difficult, with marked variation among pathologists about how to define this phenomenon. At the time the analyses were performed, 23 of 237 (9.7%) were reported as having β-γ bridges. Only 10 of these met our narrow inclusion criteria. Others included in this report were not classified as β-γ bridges at the time of initial interpretation. For the purpose of this study, we utilized the most restrictive definition for β-γ bridging. During evaluation of electrophoretograms, two other potential manifestations of β-γ bridging appeared, which were not included in our study. Electrophoretograms in one excluded group met the first two inclusion criteria (decreased A:G ratio and indistinct separation between β and γ fractions) and were classified as a potential fourth type of β-γ bridge (Fig. 5 ). However, in this small group of cases (6 of 237), the predominance of β-γ proteins is within the third quartile, indicating β rather than γ protein predominance. These cases were excluded owing to uncertainty of the significance of this pattern. Electrophoretograms in a second excluded group (3 of 237) met the first and third inclusion criteria (decreased A:G ratio and predominance of γ proteins) but exhibited a trough > 5% of the height of the β peak. Because these may simply represent an overlap between distinct β and γ fractions, this group was not included. Unfortunately, the majority of these animals were lost to follow-up analysis.
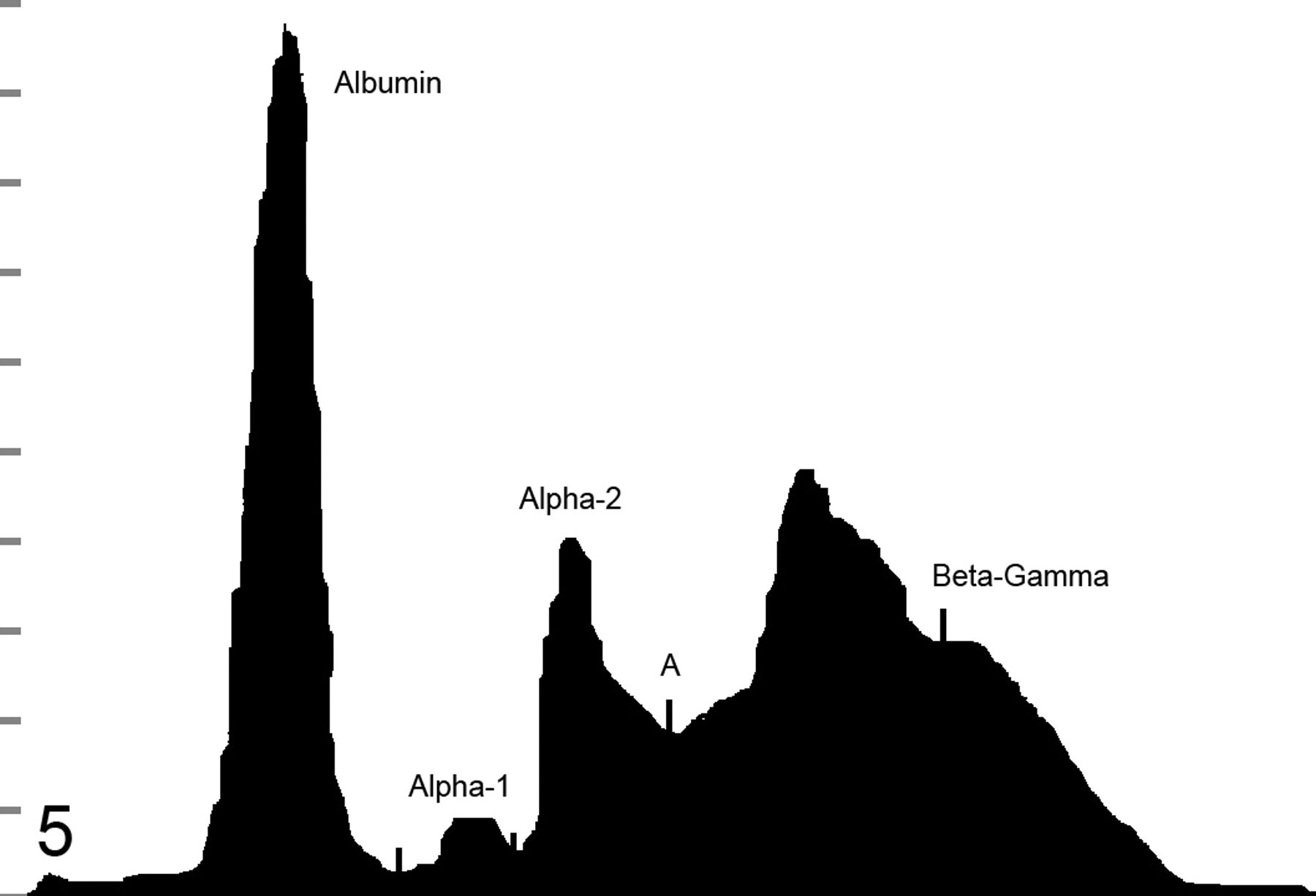
Increased proteins in the beta fraction as opposed to the gamma fraction. A, midpoint. Albumin, 2.5 g/dl; alpha 1, 0.2 g/dl; alpha 2, 1.3 g/dl; beta, 2.9 g/dl; gamma, 1.3 g/dl.
Few published studies have evaluated the strength of the correlation between β-γ bridging and hepatic disease. However, there have been many attempts to determine the nature of the substance that links the β and γ regions on the protein electrophoretograms of human patients with known hepatic disease—particularly, cirrhosis. 6,21,22,27,30 Although often referred to as substance X or substance T, the substances migrating between these two fractions are, according to most investigators' hypotheses, simply high levels of immunoglobulins. Some have theorized that these immunoglobulins are synthesized because of an autoimmune reaction occurring upon hepatocyte damage, resulting in exposure of hepatocyte nuclei and mitochondria that were previously immunologically protected. Others have suggested that there are excess immunoglobulins migrating within this fraction because of increased antigenic stimulation from enteric-derived proteins that are not adequately removed by the Kupffer cells of the diseased liver or that bypass the liver via acquired hepatic shunts. 21,23 Still others have proposed that increased globulins are actually produced locally with liver disease by proliferative reticuloendothelial cells lining hepatic sinusoids. 3,7,20,27 Different investigators have suggested that β-γ bridging occurs secondary to increased production of β and γ globulins, in an attempt to compensate for the hypoalbuminemia caused by decreased hepatic synthesis and/or loss of albumin in ascitic fluid. 9 Finally, some have theorized that liver disease—particularly, cholangiohepatitis—incites an acute inflammatory response, resulting in the increased production of fibrinogen, which is actually the substance responsible for bridging the β and γ regions. 3,7,20 It is possible that any or all of these proposed mechanisms may be correct in specific instances. Regardless, the nature of the elusive migrating substance (or substances) does not need to be clarified for the identification of a β-γ bridge and any associated disease. It is likely that proteomic analysis could help resolve the identity of this substance.
The moment of transition of this proposed association between β-γ bridging and hepatic disease from the human into the veterinary literature is unknown. However, Johnson and Wicks 13 compared the serum protein electrophoretic profiles of various mammalian species and humans, found marked species variation, and concluded that such a comparison (ie, between human and other mammalian protein profiles) is inappropriate for diagnostic purposes. Although arguments for the presence of increased levels of immunoglobulins seen with numerous diseases are reasonable explanations for β-γ bridging, the presence of other substances cannot be excluded. This is important to remember in interpreting the electrophoretogram of nonmammalian species in which plasma rather than serum is often utilized to evaluate fibrinogen levels. Fibrinogen migrates in the late β-globulin region and can bridge the area between the β and γ regions, particularly when increased levels of fibrinogen are present, making identification and interpretation difficult. 16,24 For this reason, all electrophoretograms from nonmammalian species were automatically excluded. Free hemoglobin–haptoglobin complexes resulting from hemolysis can occasionally migrate in the β-γ region and even within the α2 region, often resulting in further diagnostic confusion. 1,17,25 To our knowledge, none of the samples in this study exhibited gross hemolysis. Additional electrophoretic studies using immunoelectrophoresis and capillary band electrophoresis may prove useful in further defining the migrating protein fractions to correctly determine the underlying pathologic process.
This study has shown, in contrast to information in the veterinary literature, that β-γ bridging does not have a strong predictive value for hepatic disease in dogs, cats, or horses. Based on the results of this study, the finding of β-γ bridging on an electrophoretogram should not be considered pathognomonic for hepatic disease in dogs, cats, or horses.
Footnotes
Acknowledgements
We thank Shay Bush, Jeff Duke, Kelly Gibson, and Laura Amlung for their assistance in obtaining medical information and Deborah Keys for her assistance with statistical analysis. We also thank Tom Drapela, Lari Cowgill, Jennifer Towe, and Drs Holly Brown and Julie Webb for assistance in preparing the article.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
