Abstract
Recurrent airway obstruction (RAO) in the horse is a disease characterized by reversible bronchoconstriction and by mucus and neutrophil accumulation in the airways. It has been hypothesized that in horses with RAO, remodeling changes occur that are similar to those described in humans with asthma. Although collagen fibrils are present surrounding normal airways, they are a prominent feature of airway remodeling in human asthma with evidence of enhanced collagen III and I fibril deposition. An immunolabeling method was developed to identify collagen I and III in equine lung and to describe the collagen fiber type and distribution within the walls of the noncartilagenous bronchioles. The health status of 14 horses was characterized by clinical respiratory exam, bronchoalveolar lavage cytology, and pulmonary function tests. Following postmortem examination and histological assessment, horses were divided into RAO-affected (n = 4) and nonaffected (n = 10) groups. Eight sections per horse from all lung regions were evaluated histologically. Results of the study showed that collagens I and III were present in the lamina propria and adventitial area of the noncartilaginous bronchioles. There was clear staining differentiation between collagen I or III, airway smooth muscle, and the airway epithelium. Collagen I and III were present in the lamina propria and adventitial areas of the noncartilaginous bronchioles of horses, and there was no significant difference in the relative amount of collagen I and III between this group of RAO-affected and nonaffected horses.
Collagen provides an important structural component to the lung parenchyma, with greater than 95% of lung collagen consisting of type I and III fibers. 2,20 As a constituent of the extracellular matrix, collagen imparts structural support and compartmentalization of tissue and serves a barrier function. 13 Airway remodeling in asthmatics is defined by both structural and functional changes. In humans, goblet cell and mucous gland hypertrophy, collagen deposition and subepithelial fibrosis, elastin deposition, neovascularization, and smooth muscle hyperplasia and/or hypertrophy characterize airway remodeling. 5,8 –10,15,16,33 Increased collagen deposition is one of the proposed mechanisms for bronchial thickening in airway remodeling. However, there is little information regarding type, amount, and distribution of collagen in the equine lung. In a previous collagen quantification study, a core section from the caudal lung region from 7 horses aged 2 to 5 years was examined using histochemical detection of collagen with Sirius red stain. 19 The distribution, relative abundance, and thickness of collagen fibrils in the parenchyma and small airways were semiquantified. Findings indicated that in clinically normal, racing Thoroughbred horses, airway collagen in the caudal lung region correlated to radiographic and histological changes compatible with exercise-induced pulmonary hemorrhage. 19 However, such assessment did not determine regional differences in lung collagen, changes occurring with age, or changes in horses with airway diseases other than exercise-induced pulmonary hemorrhage.
It has been suggested that asthma in humans and recurrent airway obstruction (RAO) in horses have similar pathophysiological characteristics. 11,28 Therefore, horses with RAO might also develop airway remodeling akin to human asthmatics. RAO is considered to be a disease of the noncartilaginous bronchioles 17,30 ; however, there has been only a limited assessment of pulmonary remodeling in horses with RAO. 14
Immunohistochemical methods for quantifying collagen are likely more sensitive than histochemical stains and may delineate the type of collagen present, even if degraded. 22,29 Identification of collagen I and III has aided in the understanding of equine ocular disease. 7 Hence, this study was undertaken to develop an immunohistochemical method to identify collagen in the equine lung, to describe the type and location of collagen in the equine noncartilaginous bronchiole, and to determine whether horses histologically classified as RAO have increased lung collagen compared with horses without RAO.
Methods
Animals, Respiratory Function Testing, and Tissue Preparation
This study used 14 horses that were donated for research purposes. Eleven horses were donated for reasons other than respiratory disease (Table 1). Horse Nos, 2, 3 and 4 were donated with a long-term history of RAO that required medical treatment to manage the clinical signs of disease. All procedures were performed in compliance with Animal Care Committee guidelines at the University of Guelph. Respiratory function of each horse was assessed with a respiratory score, 28 analysis of bronchoalveolar lavage fluid (BALF), 24 and pulmonary function testing. 12 The respiratory score was based on a cumulative value for specific indices of the respiratory exam, including respiratory rate and effort (nasal flaring and abdominal lift) and quality of lung sounds. Pulmonary function testing evaluated the mechanics of ventilation including dynamic lung compliance (Cdyn) and lung resistance (RL) as well as the change in transpulmonary pressure (▵Ppl). Histamine inhalation challenge described the reactivity of the airway to increasing doses of a nebulized irritant, resulting in altered pulmonary mechanics. Subsequent to the bronchoalveolar lavage, horses were euthanized and heart and lungs were removed. A 3 × 3 × 3 cm cube of lung tissue was dissected from each of the 4 quadrants of the right and left lungs, including the cranial, dorsal, ventral, and caudal regions. Each sample was obtained from locations 10 cm medial to the exterior lung margin. For each horse, a total of 8 tissue blocks were fixed by immersion in 10% neutral buffered formalin for 72 hours before routine paraffin embedding and sectioning.
Horse Characteristics
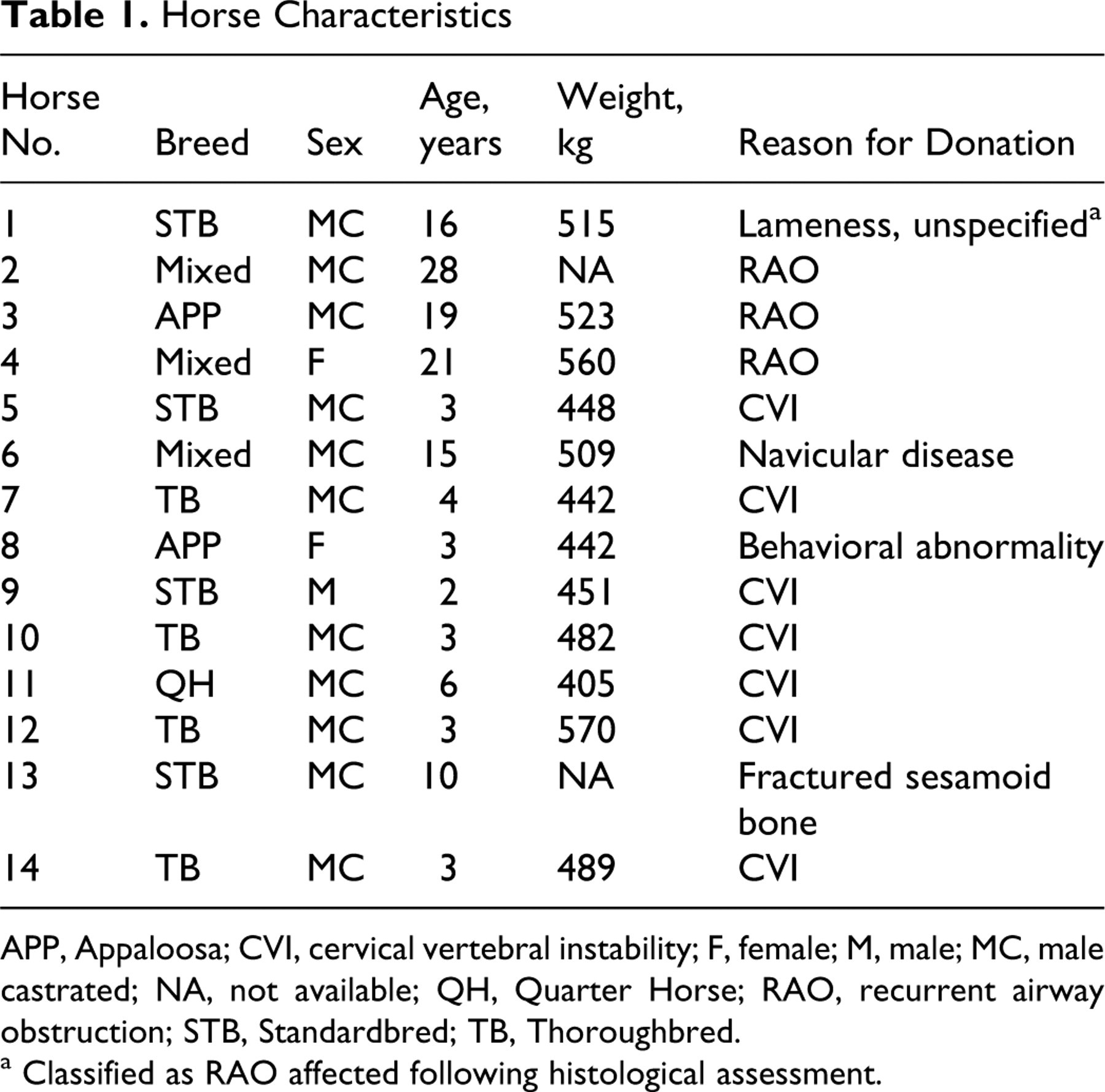
APP, Appaloosa; CVI, cervical vertebral instability; F, female; M, male; MC, male castrated; NA, not available; QH, Quarter Horse; RAO, recurrent airway obstruction; STB, Standardbred; TB, Thoroughbred.
a Classified as RAO affected following histological assessment.
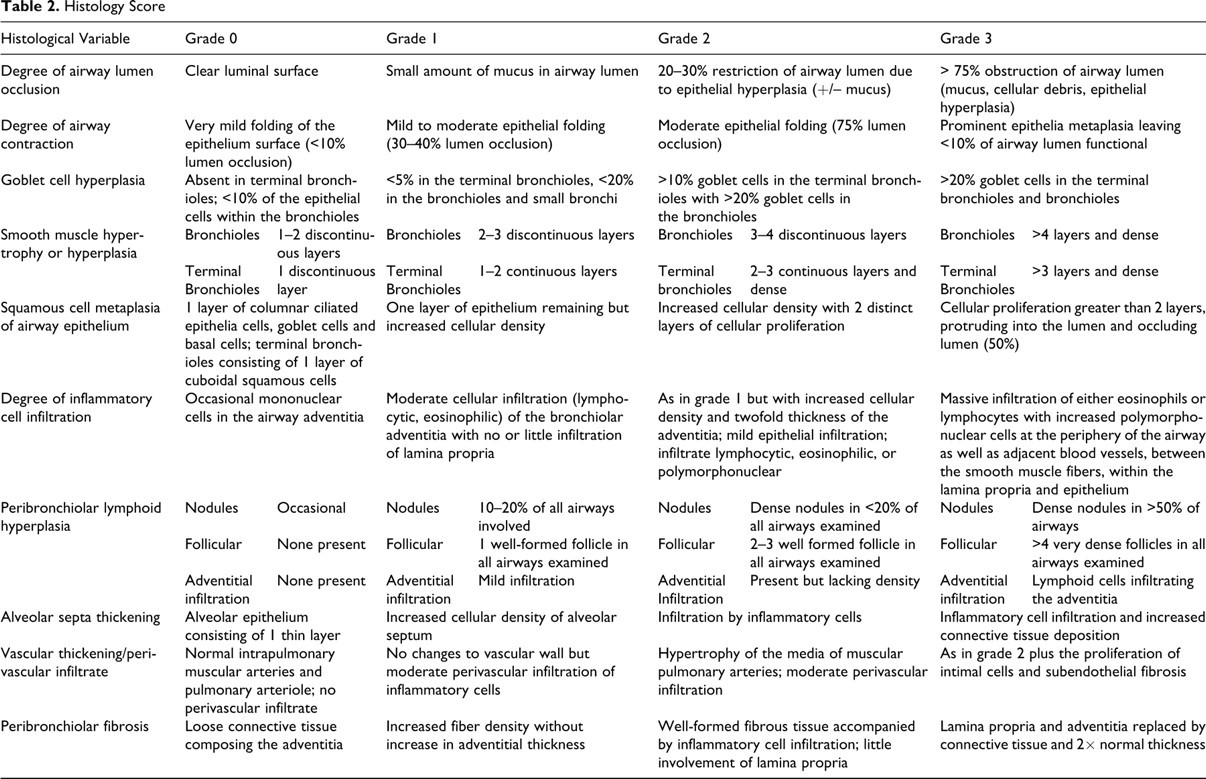
Histological assessment and scoring using 1 slide per block stained with hematoxylin and eosin were used to evaluate histological changes reflective of RAO, including the degree of mucus accumulation in the airway, goblet cell hyperplasia, inflammatory cell infiltration of the bronchiolar wall, smooth muscle hyperplasia, and inflammatory cell infiltration (Table 2 ). The maximum cumulative histologic score was 240. A histologic score greater than 80 was considered to be indicative of RAO. The histologic assessment was used as the sole measure to classify horses into RAO-affected and nonaffected designations.
Histology Score
Immunohistochemistry
For each tissue sample, four 5-μm-thick sections were placed on polylysine-charged slides (Surgipath, Richmond, IL). One slide was used for immunostaining for collagen type I and type III, and the remaining slides served as negative controls. Healthy bovine lung tissue served as a positive control. Following deparaffinization and rehydration, endogenous peroxidase activity was quenched with 0.3% hydrogen peroxide (LabChem Inc., Pittsburgh, PA) in methanol (Fisher Scientific, Fairlawn, NJ) for 5 minutes. Antigens were retrieved by incubation in 10% pepsin/0.05 M acetic acid solution (Fisher Scientific; LabChem) for 2 hours at room temperature. Nonspecific antigen binding was blocked with a non–serum protein–blocking agent (Dako Cytomation, Carpinteria, CA). Antibodies were polyclonal rabbit anti-bovine collagen type I (CL50121AP), type III (CL50321AP, both from Cedarlane Laboratories, Hornby, ON). The slides were incubated at 4°C with antibody (1:500) overnight for collagen I and III. All slides were maintained in a sealed humidified chamber. Control slides were incubated in nonimmune rabbit serum with the protein concentration adjusted to that of the undiluted anticollagen antibodies. The protein concentration was measured by refractometry. Bound antibody in sections was detected with a labeled polymer (Envison; Dako Cytomation, Carpinteria, CA) and diaminobenzidine (DAB) chromagen (Dako Cytomation, Carpinteria, CA). The chromagen was applied for 10 minutes. Slides were counterstained with Harris hematoxylin (Fisher, Kalamazoo, MI).
The specificity of the antibodies for the target epitopes was investigated with 2-color immunohistochemistry. A sequential method was developed that was similar to the previously described procedure except all incubations were carried out at room temperature. Anticollagen III antibody was applied at 1:500 for 1 hour, and slides were then incubated with labeled polymer and AEC reagent (3-amino-9-ethylcarbazole in N,N-dimethylformamide; Sigma, St. Louis, MO). Anticollagen I antibody was then applied at 1:500 for 1 hour, and bound antibody was visualized with anti-rabbit secondary antibody linked to alkaline-phosphate/BCIP/NCT (5-bromo-4-chloro-3-indolyl phosphate/nitroblue tetrazolium; Labvision, Fremont, CA).
Microscopic Assessment
Immunolabeled slides were randomized and masked for blinded examination at 200 × magnification. A semiquantitative scoring system proposed by Roche et al 26 was adapted and applied to assess the intensity of collagen staining at 4 specific locations of the noncartilaginous bronchiole. These locations included the connective tissue of the bronchiolar lamina propria and adventitia, airway smooth muscle, and airway epithelium. The semiquantitative score used by Roche et al 26 evaluated immunostaining for collagen in the airway at the region immediately beneath the epithelium, the thickened subepithelial band, the junction of the band with the submucosa, the vascular basement membrane, the basement membrane of the bronchial glands, smooth muscle, and the connective tissue of the submucosa. Similar to the scoring system described by Roche et al, 26 each component of the airway was assessed for the intensity of immunolabeling as 0 (no labeling), 1 (mild labeling), 2 (moderate labeling), and 3 (intense labeling). Airway smooth muscle and epithelium regions did not show any immunolabeling in any of the sections and so were not included in the final assessment. The immunohistochemical scores of the lamina propria and adventitia were added together from the 8 regions of the lung assessed per horse for a maximum immunohistochemical score of 48 (lamina propria [3] + adventitia [3] × 8 lung regions = 48). The regions of the pulmonary blood vessels and alveolar septa were examined but not scored.
Statistical Analysis
The age, BALF data, pulmonary function test data, and histopathology score were analyzed using a ProcT-test. The normality of the data was assessed using a Pro-univariate test for normality. The BALF data required a logit transformation. The respiratory score was assessed using a Mann-Whitney-Wilcoxon 2-tailed exact test for nonnormally distributed data. The presence of type I and III collagen within a group was compared using the Mann-Whitney-Wilcoxon 2-tailed exact test for nonparametric data, because the data were not normally distributed as determined by the Shapiro Wilks test. P values of ≤.05 were considered significant. All statistical analyses were performed with SAS 9.1.3 (SAS Institute, Cary, NC).
Results
Animals and Respiratory Function
Clinical scoring, respiratory function testing, and cytological analysis of BALF are presented in Table 3 . The RAO-affected horses were considered to be in a state of remission because they did not fulfill the definition of RAO exacerbation as described by the International Workshop of Equine Chronic Airway Disease. 25 No abnormalities were detected upon gross examination of the lungs. Histological assessment and application of the scoring system revealed 2 very distinct groups of horses within the study population (Table 4 ). Ten horses had histopathology scores ranging from 10 to 36 (median 20) and were designated non–RAO affected, whereas 4 horses had histopathology scores ranging from 90 to 117 (median 96.5) and were designated RAO affected. There was a statistically significant difference between the 2 groups (P = .001). The histological findings for non–RAO-affected horses included an absence of goblet cell hyperplasia and peribronchiolar lymphoid hyperplasia, rare inflammatory cell infiltration of the adventitia or lamina propria, and no occlusion of airways with mucus or inflammatory cells. Histologically, lung tissue from horses with RAO consistently demonstrated goblet cell hyperplasia in the noncartilaginous bronchioles, lymphocyte and neutrophil infiltration of the lamina propria and adventitia, peribronchiolar lymphoid hyperplasia consisting of prominent aggregates and occasionally airway smooth muscle hypertrophy or hyperplasia. Mucus and inflammatory cells, predominantly neutrophils, occluded many airways. The median age of RAO-affected horses was 20 years (range, 16–28 years) and of non–RAO-affected horses 3 years (range, 2–15 years) (P < .001). The median proportions of neutrophils in BALF from horses with and without RAO were 13.4% and 4.0% (P = .005), whereas the median respiratory scores were 7.0 and 1.0, respectively (P = .01). Among horses with RAO, the median values for ▵Ppl, Cdyn, and RL were 9.6, 0.7, and 0.9, respectively, which were significantly different from the values for the non–RAO-affected horses: 5.1, 1.6, and 0.5, respectively (P = .002, P = .03, and P = .02). Horses with RAO had markedly increased airway responsiveness to histamine with a median response at 0.5 mg/ml of nebulized histamine compared with 5.3 mg/ml (P = .02) in non–RAO-affected horses. Six of the nonaffected horses demonstrated airway responsiveness of less than 8.0 mg/ml, indicating airway reactivity.
Median (± Standard Deviation) Antemortem Diagnostic Findings
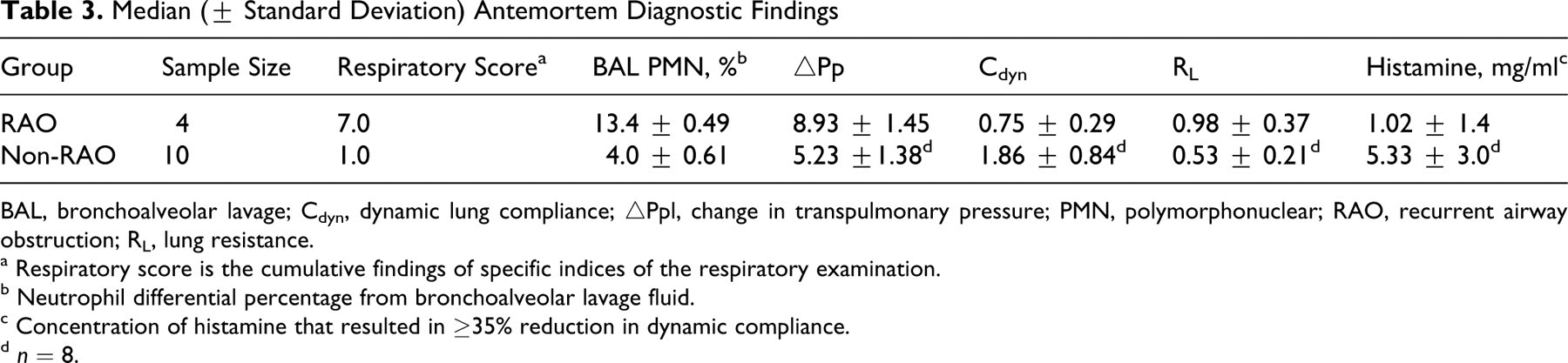
BAL, bronchoalveolar lavage; Cdyn, dynamic lung compliance; ▵Ppl, change in transpulmonary pressure; PMN, polymorphonuclear; RAO, recurrent airway obstruction; RL, lung resistance.
a Respiratory score is the cumulative findings of specific indices of the respiratory examination.
b Neutrophil differential percentage from bronchoalveolar lavage fluid.
c Concentration of histamine that resulted in ≥35% reduction in dynamic compliance.
d n = 8.
Mean Histopathology and Immunohistochemistry Scores
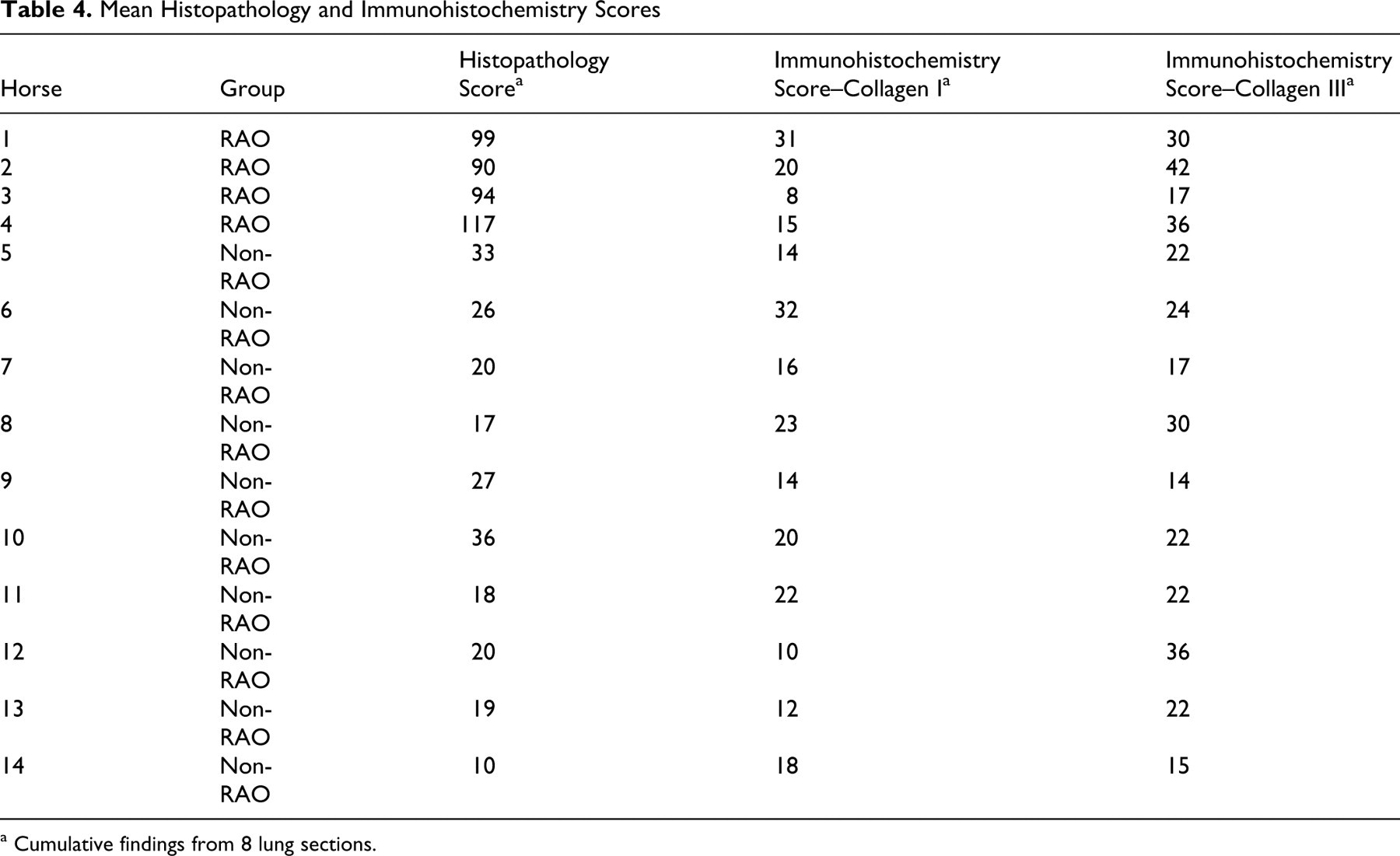
a Cumulative findings from 8 lung sections.
Immunohistochemistry
The immunohistochemical technique resulted in consistent immunolabeling of collagen I and III in positive control slides and equine lung tissue. There was minimal background staining with no evidence of positive immunolabeling in the negative control slides. There was no difference in the density of immunolabeling of collagen I (P = .13) or collagen III (P = .06) when comparing the adventitia and lamina propria regions (Figs. 1–4). No immunolabeling was noted in the airway smooth muscle, bronchiolar epithelium, or bronchiolar reticular basement membrane (RBM). Both the pulmonary venules and arterioles demonstrated intense immunolabeling for collagen I and III fibers (Fig. 5). There was occasional faint labeling of collagen I and III in the alveolar septa (Fig. 6). Double immunohistochemistry experiments confirmed lack of cross-reactivity between collagen I and III antibodies.
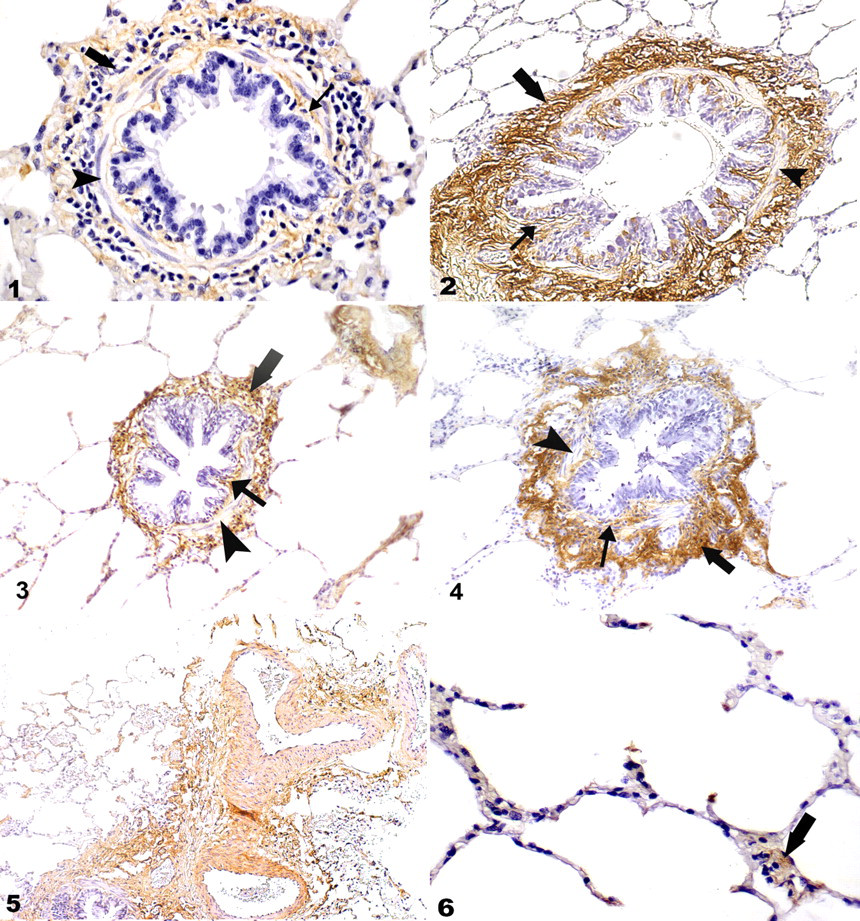
There was no significant difference in the intensity of immunolabeling for collagen III between horses with and without RAO (P = .2), nor was there a significant difference in the intensity of immunolabeling for collagen I between the groups (P = .2) (Table 4). Collagen immunolabeling was homogenous among all lung regions.
Discussion
The immunohistochemical labeling technique used in this study localized collagen I and III in the equine lung. Immunolabeling imparts specificity to the recognition of collagen fiber types, which is lacking with the use of routine collagen-specific histochemical stains. 22,29 The type and location of collagen I and III surrounding the noncartilaginous bronchioles in horses affected with RAO were compared with non–RAO-affected animals.
Total lung collagen as well as the abundance of collagen surrounding pulmonary structures has been described in humans, particularly as it relates to collagens I and III. 18,21,27,29 In humans, the fibrillar collagens, including collagen I and III, are important structural components of connective tissue. 13,29 In humans, the presence of collagen I and III has been described in the lamina propria regions surrounding the large and small airways, 10,25,26 interlobular septa, 21 and alveolar septa. 18,21 Collagen I and III were demonstrated in the lamina propria and adventitial regions surrounding the noncartilaginous bronchioles in all tissue sections of the horses examined, which is in agreement with reports in the human literature. 4,10,26 There was no difference in the quantitative distribution of collagen I or III within the components of the small airway wall, suggesting a uniform distribution of collagen fibers throughout the lamina propria and adventitia of noncartilaginous bronchioles. Concurrent with the human literature, there was an absence of collagen type I and III fibers in the airway epithelium and airway smooth muscle.
The histological assessment and cumulative score evaluate specific histopathologic variables associated with RAO. Descriptive grading systems have been used by other authors 23,34 to classify horses into groups based upon the severity of lung lesions without the use of antemortem diagnostic evaluation. Other investigators have reported a strong relationship between the clinical diagnosis and histological changes in horses with RAO. 17 In this population of horses, there was a very clear distinction in scores between RAO-affected and nonaffected individuals with no overlap. Therefore, the histological assessment was used as the sole means to diagnose RAO in this study.
The mean age of the affected and nonaffected animals was significantly different. In healthy, nonsmoking humans, advancing age has not been associated with an increase in absolute wall area of bronchioles when compared with younger individuals, 1 suggesting that collagen deposition does not increase with age, although the relationship between age and amount of pulmonary collagen has not been described in horses. If age-related increases in pulmonary collagen deposition did occur, they would likely err on the side of the RAO-affected horses, because they are the older group. However, because no difference between the nonaffected horses and the limited number of RAO-affected horses was observed in this study, this limitation does not present a problem for data interpretation. Further studies using a larger, age-matched cohort may provide a more definitive answer to this quandary.
An environmental challenge was not performed in this study because 3 of the 4 horses with RAO had long-term histories of RAO that required therapeutic intervention when the horses were exposed to hay. At the time of the examination, the BALF and resting pulmonary function test data were not suggestive of RAO exacerbation 24 ; therefore, the horses were considered to be in a state of disease remission. The remaining horses had no history of respiratory disease. All of the RAO-affected horses were very reactive to histamine inhalation, which is indicative of the severity of the disease process.
Three of the 4 horses with RAO were donated with a long-term history of disease that necessitated therapeutic intervention. The influence of inhaled or systemic corticosteroids on collagen remodeling was not addressed in this study; however, others have suggested that the use of inhaled corticosteroids in patients with mild to moderate asthma does not modulate collagen deposition in the peripheral airways. 5 Therefore, we speculate that the past use of corticosteroid in these individuals would not likely have influenced collagen deposition in this group of horses.
Three unanticipated confines of this study should be noted: the low number of RAO-affected horses, the possible impact of clinically normal horses with airway hyperreactivity, and the inability to reliably identify the bronchiole RBM. The RAO status did not significantly affect the relative quantity of collagen I or III detected surrounding the noncartilaginous bronchioles. This was an unexpected finding because the concept of airway remodeling in RAO had been suggested by other authors, 14 perhaps based on the presumed similarities of RAO with human asthma. However, in a human study, alteration in collagen deposition within the lamina propria of asthmatics was not a consistent finding. 31 The inability to detect a difference between groups may be a reflection of the low number of RAO-affected horses (n = 4) examined in this study, because it is possible that enhanced collagen deposition as a component of airway remodeling is an inconsistent feature of RAO. Some of the horses classified as nonaffected in this study demonstrated airway hyperreactivity, and it is possible that these horses were affected by inflammatory airway disease (IAD). There was no evidence of an increased respiratory effort, which is considered characteristic of horses during RAO exacerbation but not IAD. 11 Horses with IAD have a history of poor athletic performance, exercise intolerance, nonseptic airway inflammation, and/or evidence of lower airway dysfunction including airway obstruction, airway hyperresponsiveness, or impaired gas exchange. 11 Histological changes associated with IAD have not been identified, and in particular it is not known whether horses with IAD have airway remodeling. If this were the case, then bronchiolar collagen deposition in IAD-affected horses may have confounded our findings. Recently it was reported that horses with exercise-induced pulmonary hemorrhage (EIPH) have alterations in collagen distribution as well as increases in the amount of collagen within the interlobular septa, intralobular veins, and subpleural lung regions. 32 Although none of the horses in the present study had a reported history of EIPH, it is plausible that previous episodes of EIPH contributed to collagen deposition in some of the nonaffected horses, thereby influencing the collagen scores and confounding the comparison between the 2 groups.
In studies of human asthmatics, collagen deposition as a consequence of airway remodeling is often assessed using the thickness of the bronchiole and bronchi RBM using routine histochemical stains. 3,6,10,26,33 However, there appears to be a large variation across studies with respect to bronchiole and bronchi RBM thickness in asthmatic and normal individuals, 31 which reduces the reliability of the assessment. The bronchiole RBM was not assessed in this study because of the inability to adequately and reliably identify the membrane structure using light microscopy alone. Therefore, it remains possible that deposition of collagen in this specific region could have been underestimated.
Using an immunohistochemical technique, this study has demonstrated the type and homogenous distribution of collagen within the noncartilaginous bronchioles of the horse. The RAO-affected horses in this study did not have measurably different amounts of collagen I or III in their airways compared with clinically normal horses, suggesting that collagen may not play an important role in equine airway remodeling or that enhanced collagen deposition is not a consistent feature of airway remodeling in horses with RAO.
Footnotes
Acknowledgements
We are grateful to Dr. Leslie Huber, Barb Jefferson, and Susan Lapos for technical assistance and Gabrielle Montheith for statistical analysis.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
Funding for this study was provided by Equine Guelph and Ontario Ministry of Agriculture, Food and Rural Affairs.
