Abstract
To characterize the early events of foot-and-mouth disease virus (FMDV) infection in cattle subsequent to simulated natural exposure, 16 steers were aerosol inoculated with FMDV and euthanized at various times. Samples were collected from each steer antemortem (serum, nasal swabs, and oral swabs) and postmortem (up to 40 tissues per animal) and screened for FMDV by virus isolation and for FMDV RNA by real-time reverse transcription polymerase chain reaction. Tissues that tested positive for FMDV or viral RNA were examined by immunohistochemistry and multichannel immunofluorescence microscopy. In previremic steers, FMDV was most consistently localized to nasopharyngeal tissues, thereby indicating this region as the most important site of primary viral replication. The earliest site of microscopic localization of FMDV antigens was the lymphoid follicle-associated epithelium of the pharyngeal mucosa-associated lymphoid tissue of the nasopharynx at 6 hours postaerosolization. At early time points after aerosol inoculation, viral antigens colocalized with cytokeratin-positive pharyngeal epithelial cells; intraepithelial FMDV-negative, MHCII/CD11c-double-positive dendritic cells were present in close proximity to FMDV-positive cells. Onset of viremia coincided with marked increase of viral loads in pulmonary tissues and with substantial decrease of viral detection in nasopharyngeal tissues. These data indicate that subsequent to aerogenous exposure to FMDV, the temporally defined critical pathogenesis events involve (1) primary replication in epithelial cells of the pharyngeal mucosa-associated lymphoid tissue crypts and (2) subsequent widespread replication in pneumocytes in the lungs, which coincides with (3) the establishment of sustained viremia.
Foot-and-mouth disease (FMD) is a highly contagious picornaviral disease affecting domestic and wild cloven-hoofed animals. 2,13 Under natural transmission conditions, within and between herds of cattle, the etiologic agent, FMD virus (FMDV), is spread through inhalation of aerosolized virus, and under appropriate environmental conditions, virus-laden droplets may travel vast distances while maintaining infectivity. 1,13,22
Numerous researchers have experimentally investigated the pathogenesis of FMD in cattle; 1,4,6,7,14,15 however, a consensus still does not exist regarding many basic aspects of the early stages of infection. Most notably, the anatomic sites and cellular events involved in primary infection and the establishment of viremia are not well defined. Elucidation of these critical events and improved understanding of virus–host interactions have high probability to favorably affect the goal of improving efficacy of vaccines and biotherapeutic countermeasures to protect domestic livestock against FMDV.
Early evidence that the respiratory tract was the natural route of infection implicated the nasal cavity 14 or lungs 10 as the sites of primary replication. Yet, as investigation proceeded, opinion on this subject diverged into 2 camps favoring either the nasopharynx 2,7 or lungs 4,6 as the primary infection site. A unique set of experiments performed in the 1970s effectively isolated the contributory roles of the upper and lower respiratory tract by placement of indwelling tracheostomy tubes in cattle. 24 This work concluded that both the lungs and the pharynx could similarly serve as portals for the establishment of systemic infection. The only studies to microscopically localize FMDV in the early (previremic) stages of infection utilized in situ hybridization and concluded that lungs supported infection earlier than the pharynx. 4,6 In consideration of the disparate findings across FMD pathogenesis studies, it is necessary to remember that the various studies have utilized a broad range of serotypes and subtypes of FMDV, which may have substantial differences in virulence and tissue tropism. Additionally, heterogeneity of inoculation systems and differences in sensitivity and/or specificity of virus detection methods may account for some disparity among the published studies.
Recent reports from our laboratory have described a novel method for aerosol inoculation of cattle with FMDV and trimodal systems for detection of FMDV in bovine tissues during the early stages of infection. 3,18 In the current study, we utilized similar experimental systems to further characterize the distribution of FMDV in cattle during the previremic and early viremic phases of infection, with the overall conclusion that subsequent to aerogenous inoculation of cattle, infection initiates in the nasopharynx, it is promptly followed by pulmonary infection, and the onset of viremia is coincident with increased viral load in the lungs and decreased virus in the nasopharynx.
Materials and Methods
Experimental Animals, Virus, and Inoculation Systems
Sixteen 9- to 18-month-old Holstein steers weighing 400 to 500 kg were obtained from an experimental-livestock provider accredited by the Association for Assessment and Accreditation of Laboratory Animal Care (Thomas-Morris Inc, Reisterstown, MD). For all experiments, animals were individually housed in a biosafety level 3 animal facility from time of inoculation until time of euthanasia. Experiments were terminated by euthanasia via intravenous barbiturate overdose at predetermined end points at 0.1 hours post aerosol inoculation (hpa; steer No. 1), 3.0 hpa (steer Nos. 2, 3), 6.0 hpa (steer Nos. 4, 5), 12.0 hpa (steer Nos. 6, 7), 24.0 hpa (steer Nos. 8-13), 48.0 hpa (steer Nos. 14, 15), 240.0 hpa (steer No. 16).
Virus inoculum consisted of a clarified, macerate of tongue epithelium harvested from 2 steers experimentally infected with the FMDV strain O1-Manisa as previously described. 12,18 The inoculum was quantitated as BTID50 (bovine tongue infectious dose 50%). All steers were aerosol inoculated with 107 BTID50 of FMDV-O1-Manisa as previously described. 18 Briefly, each steer was sedated with xylazine and fitted with a commercially available aerosol delivery system (Aeromask-ES, Trudell Medical, London, Ontario, Canada), which was placed over the muzzle. The mask was attached to a jet nebulizer (Whisper Jet, Vital Signs Inc, Totowa, NJ), which was subsequently attached to an air compressor that generated 25 psi of pressure. Aerosolization proceeded until the complete inoculum was expelled from the nebulizer cup (10 to 15 minutes).
Sample Collection
Antemortem sampling consisted of collection of whole blood in serum separation tubes, oral swabs, and nasal swabs. All animals were sampled before inoculation to ensure FMDV-free status and at several time points throughout the duration of the experiment, which varied according to goals of the individual experiments. Swabs and serum tubes were transported from the animal room to the laboratory on ice and were immediately centrifuged for harvesting of serum, saliva, and nasal secretion. Samples were then stored at –70°C until time of processing.
Necropsies were performed immediately subsequent to euthanasia at predetermined time points, and tissue specimens were collected from oral cavity, nasal cavity, soft palate, pharynx, larynx, trachea, lungs, lymph nodes, and skin (Table 1 ). Tissues collected from each animal varied depending on the goals of individual experiments. Detailed descriptions of tissue designations and collection strategies has been published. 18 For each anatomically defined specimen, two 30-mg tissue samples were aliquoted into separate screw-cap 1.5-ml tubes and frozen immediately in liquid nitrogen for transfer within 2 hours to a –70°C freezer in which they were stored until the time of processing. An adjacent specimen from each tissue was placed in a cryomold, embedded in Optimal Cutting Temperature Compound (Sakura Finetek, Torrance, CA), frozen on a bath of liquid nitrogen, and stored at –70°C for immunohistochemistry (IHC).
Tissue-Specific Distribution of FMDV in Steer Nos. 2-15 with Cumulative Percentage Positivity a
a HPA, hours post aerosol inoculation; rRT-PCR, real-time reverse transcription polymerase chain reaction; VI, virus isolation; R, rostral; C, caudal; V, ventral; LN, lymph node; NEG, rRT-PCR negative. Blank cells indicate not examined. Tabulated numerical data are log10 foot-and-mouth disease virus (FMDV) RNA genome copies per ml of serum or mg of tissue as determined by rRT-PCR. Negative tissues indicate that RNA quantity was below the detection threshold of 2.26 log10 FMDV genome copies per mg of tissue (corresponding to cycle threshold = 40.00). Entries in bold text were positive for FMDV by virus isolation; nonbold text indicates that virus isolation was negative.
b Additional tissues tested were negative for all previremic steers examined, as follows: dental pad, tongue (caudal), tongue (torus), hard palate (rostral), hard palate (caudal), nasal planum, turbinates (rostral), trachea (orad), trachea (mid).
c Previremia is defined as serum virus isolation negative at all times sampled, including the time of euthanasia. Percentage positivities for rRT-PCR and VI separately are defined as the number of positive results using one modality, divided by the number of steers for which a tissue type was examined by that modality. Percentage positivity for rRT-PCR or VI is defined as the number of times that a tissue was positive by either modality, divided by the number of steers for which that tissue type was examined by both modalities.
FMDV RNA Detection
Two samples of each tissue listed in Table 1 per animal were thawed and immediately macerated in a TissueLyser bead beater (Qiagen, Valencia, CA) as previously described. 18 After maceration, 50 μl of each sample was transferred to a 96-well plate (Thermo Scientific, Waltham, MA) containing 150 μl of lysis/binding solution. RNA was then extracted with Ambion’s MagMax-96 Viral RNA Isolation Kit (Ambion, Austin, TX) on a King Fisher-96 Magnetic Particle Processor (Thermo Scientific, Waltham, MA). RNA was eluted in a final volume of 25 μl. Once extracted, 2.5 μl of RNA was analyzed by real-time reverse transcription polymerase chain reaction (rRT-PCR) on the ABI 7000 system (Applied Biosystems, Austin, TX) as previously described. 8 Samples with cycle threshold values < 40 were considered positive. The remaining macerated tissue was clarified at 1,000 rpm for 2 minutes at 4°C, and the supernatant was cleared of bacterial contamination using centrifuge tube filters (Spin-X, Costar, Corning, NY). Clarified and cleared samples were stored at –70°C until virus isolation (VI) was performed.
To convert cycle threshold values generated by rRT-PCR from experimental samples to RNA genome copies per milligram, serial 10-fold dilutions of in vitro synthesized FMDV RNA of known RNA concentration were analyzed by the rRT-PCR protocol described above. The equation of the curve of RNA copy versus cycle threshold value, further adjusted for average mass of tissue samples and dilutions during processing, was used for subsequent conversions. The cycle threshold positivity cutoff of 40 corresponded to a detection threshold value of 2.26 log10 FMDV RNA copies per milligram (RNA/mg) of tissue. The rRT-PCR results reported in Table 1 are the higher RNA/mg value of the 2 samples processed per tissue per animal; the rRT-PCR results reported in Figure 1 are the mean log10 FMDV RNA copies per milliliter (RNA/ml) for all animals sampled at each time point.
Time course of foot-and-mouth disease virus RNA quantity (log10 genome copy number / ml) determined by real-time reverse transcription polymerase chain reaction in serum (red), nasal swab samples (green), and oral swab samples (blue) collected from steers aerosol-inoculated with foot-and-mouth disease virus O1-Manisa. Each data point represents the mean ± SE for all steers sampled at a given time (hours postaerosol inoculation). Lesion (vesicle) score was calculated by evaluating each foot and the head independently, with 2 points assigned for a small vesicle (≤ 1.0cm) and 4 points for a large vesicle. Viremia (*) was defined by positive findings on serum virus isolation. X-axis calibration not to scale.
FMDV Isolation
VI was performed separately on the duplicate samples of each tissue on LFBK cells as previously described. 18,25 Upon detection of cytopathic effect, FMDV positivity was confirmed by rRT-PCR on cell culture supernatants. Samples in which no cytopathic effect was observed were amplified through 3 blind passages and the supernatants were tested by rRT-PCR before they were deemed negative. Each VI result in Table 1 is reported positive if either or both duplicate samples per tissue were positive.
Immunohistochemical and Immunofluorescent Localization of FMDV Antigens
Microscopic localization of FMDV antigens was performed in cryosections as previously described. 3,18 Briefly, tissue sections were blocked for 2 hours at 20°C; primary antibodies were diluted in blocking buffer and applied to tissue sections for 18 hours at 4°C. For IHC, specific anti-FMDV immunoreactivity was detected with a micropolymer alkaline phosphatase kit (Biocare, Concord, CA). For multichannel immunofluorescent (MIF), detection was performed with goat, anti-rabbit, and isotype-specific anti-mouse secondary antibodies labeled with AlexaFluor dyes (AF 350, 488, 594, 647). Slides were examined with a wide-field, epifluorescent microscope, and images were captured with a cooled, monochromatic digital camera. Images of individual detection channels were adjusted for contrast and brightness and merged in commercially available software (Adobe Photoshop CS2). Mouse monoclonal anti-FMDV antibodies against viral structural proteins were 10GA4 and 12AF4; 23 anti–nonstructural protein antibodies were F19-6 and F19-51. 27 Antibodies used to label cell markers in MIF experiments were mouse monoclonal anti-pancytokeratin plus (Biocare No. CM162), anti-bovine cytokeratin (CK; Sigma No. C6909, Sigma-Aldrich, Inc, St. Louis, MO), anti-CD11c (VMRD No. BAQ153A, VMRD, Pullman, WA), anti-MHCII (VMRD No. CAT82A), and anti-vimentin (Dako No. M0725, Dako, Carpinteria, CA), and rabbit polyclonal anti–Von Willibrand factor (Dako No. A0082).
For each tissue screened by IHC, a duplicate negative-control serial section treated with a mouse monoclonal anti-VSV-Indiana antibody of similar concentration was prepared. Additional negative control tissue sections were prepared from a steer that received a virus-free aerosol inoculum and was euthanized 24 hours post aerosol inoculation (hpa). Immunohistochemical and MIF labeling were considered positive when there was an intense cell-associated signal within the experimental tissue, with the absence of such staining in the negative controls.
Results
Inoculum Detection Controls
For the purpose of determining if the FMDV screening techniques would detect residual inoculated virus, 1 aerosol-inoculated steer (No. 1) was euthanized immediately after inoculation (0.1 hpa) and was subjected to standard tissue collection and screening techniques as described for the other animals. All 28 tissues collected from this animal were VI negative, and only a single tissue, proximal cranial lung, was positive by rRT-PCR (2.56 RNA/mg). This information confirmed that the inoculum reached as far as the lungs and strongly affirmed that any rRT-PCR- and VI-positive findings at subsequent time points represent FMDV replicated de novo. Based on the positive rRT-PCR finding for proximal cranial lung from steer No. 1, the frozen Optimal Cutting Temperature Compound–embedded tissue specimen from this tissue was examined by IHC to similarly test for detection of inoculated virus. Forty serial sections were examined (alternating anti-FMDV capsid and anti-FMDV 3D [polymerase] primary antibodies) with no detection of FMDV antigens in any section, confirming that inoculum quantity was insufficient to be detected by IHC. Data generated from this animal are not included in subsequent analyses.
Antemortem Profiles of FMDV-Infected Cattle
Neither vesicles nor fever (rectal temperature ≥ 40°C) was observed in any previremic animals. The earliest detection of fever in any animal occurred at 60 hpa, in steer No. 16; this animal was febrile from 60 to 96 hpa, with maximum body temperature of 41°C detected at 60 hpa. The 3 steers euthanized as 48 hpa or later (viremic) all developed vesicles of 1 (n = 2) or 2 (n = 1) interdigital clefts at 48 hpa (Fig. 1). One steer (No. 15) had subtle blanching of the epithelium of the lingual torus at 48 hpa, which was subsequently determined histologically to be an early vesicle. The 1 steer that survived 240 hpa (No. 16) developed vesicles of tongue and all 4 interdigital clefts between 48 and 120 hpa. No other clinical signs of infection or gross lesions were detected in the animals that survived 0.1 to 48.0 hpa.
The aerosol inoculation system provided highly consistent patterns of viral shedding, viremia, and clinical signs across experimental animals; however, some interanimal variation was observed. Antemortem data are presented as cumulative average log10 FMDV genome copy numbers per milliliter (RNA/ml) for all animals at each time point (Fig. 1). For oral swab samples, the quantity of FMDV RNA (inoculum) progressively decreased from 0.1 to 8.0 hpa with de novo replicated virus first detected at 24 hpa and peaking at 72 hpa at 8.17 RNA/ml. Similarly, nasal swab samples showed gradual elimination of inoculum from 0.1 to 4.0 hpa with de novo replicated viral RNA beginning to increase at 6 hpa. However, unlike oral swabs, the viral RNA quantity never dropped below detection threshold before de novo replication was detected (Fig. 1). Also unlike oral swabs, the nasal shedding pattern showed 2 peaks, at 12 and 72 hpa (5.22 and 8.92 RNA/ml, respectively) with an intervening trough. Maximum nasal and oral FMDV RNA/ml occurred coincidently at 72 hpa with the nasal detection peak being slightly higher but decreasing more precipitously relative to salivary samples.
The earliest detection of FMDV RNA in sera occurred at 22 hpa in 1 steer. At 24 hpa, 6 of 9 steers had RNA-positive sera, and at 48 hpa the sera of all 3 steers sampled were FMDV rRT-PCR positive (Fig. 1). Detection of infectious FMDV in sera was delayed relative to RNA, with a single VI-positive steer (1 of 9) at 24 hpa and with the remaining 3 steers (3 of 3) becoming serum VI positive by 48 hpa; viremia was defined by the detection of infectious virus in serum. Overall, FMDV RNA was detected in sera from 22 to 120 hpa, whereas viremia was detected from only 24 to 72 hpa.
Tissue-Specific Distribution of FMDV and Viral RNA
Steers were euthanized at predetermined time points regardless of clinical progression of disease in individual animals. Sample collection schemes were predetermined and standardized with minor variation among individual animals, based on the expected stage of disease at the time of euthanasia (Table 1). A maximum of 40 anatomically distinct tissue specimens were collected per animal. Both steers euthanized at 3 hpa had more FMDV RNA–positive tissues in the nasopharyngeal/laryngeal (NP/L) sites (6 of 12 tissues; 50.0%) than in other anatomic regions (2 of 18 pulmonary tissues; 11.1%) (Table 1). At this time point, FMDV RNA quantities were generally low, and no tissues contained infectious virus. The 2 highest quantities of FMDV RNA were detected in caudal dorsal soft palate (2.92 RNA/mg) and caudal dorsal nasopharynx (2.95 RNA/mg) of steer No. 2.
From 6 to 12 hpa, within the NP/L sites there was greater quantity of tissues positive for infectious FMDV (14 of 24; 58.3%) and viral RNA (13 of 24; 54.2%) relative to all other anatomic regions; at 12 hpa, every NP/L tissue examined was positive by VI, rRT-PCR, or both. Within this period, rRT-PCR and VI positivity percentages for pulmonary specimens were also increasing but to a lesser extent than that of NP/L tissues. At the individual tissue-specific level, the greatest quantities of FMDV RNA between 6 and 12 hpa were detected in distal cranial lung (5.12 RNA/mg) and caudodorsal nasopharynx (4.9 RNA/mg) of steer No. 7 (12 hpa).
At 24 hpa, 97.2% of NP/L samples (35 of 36 at tissue level) were positive for either FMDV RNA or infectious virus, with 72.2% double positive (VI and rRT-PCR). By contrast, 61.1% of pulmonary specimens (22 of 36 at tissue level) were VI or rRT-PCR positive, with 36.1% double positivity. The highest quantity of FMDV RNA at 24 hpa (7.3 RNA/mg) was detected within the rostral dorsal soft palate of steer No. 9. The only positive findings (rRT-PCR or VI) from lymph nodes of previremic steers in the study were the hilar lymph node of steer No. 11 and the medial retropharyngeal lymph node of steer No. 12, which were VI positive at 24 hpa.
Both steers euthanized at 48 hpa were viremic at the time of euthanasia; as such, infectious FMDV and FMDV RNA detected in the tissues of these animals may have been present within and/or exterior to blood vessels in these tissues. Viral load was substantially decreased in the NP/L sites at 48 hpa relative to 24 hpa, with 66.6% positive by VI or rRT-PCR (8 of 12 tissues) and 33.3% double positive. By contrast, VI or rRT-PCR positivity of the lungs from 48-hpa steers was increased relative to 24 hpa, with 77.8% single positive (14 of 18 tissues) and 27.8% double positive. The greatest quantity of FMDV RNA detected in these animals was from the interdigital clefts of both steers (7.70 and 7.96 RNA/mg) and the lingual torus of steer No. 15 (6.19 RNA/mg), all of which had grossly detected vesicles. Among nonlesional tissues collected from the 48 hpa steers, the greatest quantity of FMDV RNA was obtained from the distal anterior lung of steer No. 15 (5.54 RNA/mg).
Given that the primary goal of these experiments was to characterize the involvement of various tissues as primary replication sites of FMDV, greater attention was focused on the animals that had VI-negative sera at the time of euthanasia (Table 1). For these animals (all except steer Nos. 13–16), tissue-specific cumulative positivity percentages (PPs) were calculated for each tissue for rRT-PCR, VI, and both. PP was defined as follows:
total positive results at tissue X by modality Y in previremic steers ∕ total specimens of tissue X examined by modality Y in previremic steers:
Thus, PP served as an indicator of the relative frequency of involvement of each tissue in previremic FMD in these animals. The PP index demonstrated that among previremic animals, the specimens most frequently positive for infectious FMDV or FMDV RNA were the tissues of the NP/L sites. Specifically, the only 100% PP value occurred for rRT-PCR or VI positivity in rostrodorsal nasopharynx. The next-highest PP values (81.8%) were achieved in only caudodorsal soft palate and caudodorsal nasopharynx. Overall, 15 of 18 PP values from NP/L sites were greater than all PPs from other tissues. PP values from lung and ventral soft palate indicate lesser consistency of FMDV positivity relative to NP/L sites. PP values from lymphoid tissues were uniformly low, with lingual tonsil having the highest overall PP in this tissue category.
Microscopic Localization of FMDV Antigens and Phenotypic Characterizations of Associated Cells
Inoculation to 12 hpa
Despite numerous attempts, FMDV antigens could not be microscopically (immunohistochemically) identified in any of the rRT-PCR-positive tissues from the steers euthanized at 3 hpa. The earliest time point at which viral antigens were localized in situ was at 6 hpa, within the rostrodorsal nasopharynx of both steers (Nos. 4 and 5) euthanized at this time point. Viral antigens were not localized in any other tissues from these animals despite screening of more than 100 additional IHC slides from pulmonary and pharyngeal sites that were rRT-PCR or VI positive. In both steers, FMDV capsid antigen was similarly localized to microscopic epithelial crypts of the follicle-associated epithelium (FAE) overlying pharyngeal mucosa–associated lymphoid tissue (PALT; Figs. 2, 3). Although the epithelial crypts were vaguely discernable by IHC-light microscopy (Fig. 2), the MIF examination of a serial section clearly demonstrated that FMDV in these regions was intraepithelial rather than within subjacent lymphoid tissue (Fig. 3A). Furthermore, simultaneous labeling with CD11c and MHCII indicated that the FAE and subjacent lymphoid regions had diffuse distribution of cells positive for both these antigens individually and as CD11c–MHCII double positives (presumptive dendritic cells [DCs]); Fig. 3B–3D). Despite the presence of DCs in close proximity to FMDV antigens, virus colocalized exclusively with CK in these tissues. Individual cells containing FMDV antigens were rarely identified in the subepithelial dome region immediately subjacent to antigen-positive crypts (not shown); however, phenotypic characterization of these cells with various markers was not achieved.
At 12 hpa, the distribution of viral antigens in the nasopharynx was similar to that observed at 6 hpa with the exception that there was slightly greater intimacy of association between FMDV antigens and the DC markers CD11c and MHCII (not shown). However, colocalization of FMDV antigens with DC markers was not observed. The lungs of both steers euthanized at 12 hpa (Nos. 6, 7) had regional anti-FMDV immunoreactivity (Figs. 4–7). Steer No. 7 had numerous FMDV capsid and nonstructural antigen–positive cells within the distal segment of the cranial lung lobe (Figs. 5–7). In this animal, there were 2 distinct morphological variants of immunopositive cells when viewed by light microscopy: a squamous cell type associated with alveolar septa (type I pneumocyte; Fig. 5, Insert A), and a polygonal cell type variably associated with alveolar septa or lumina (type II pneumocyte or alveolar macrophage; Fig. 5, Insert B). MIF labeling of this tissue indicated that both morphologic categories of FMDV-positive cells were additionally CK positive (Figs. 6A, 7), von Willebrand factor (vWF) negative (Fig. 7), vimentin negative (Fig. 6B), confirming that infected cells were of epithelial histogenesis. However, throughout the alveolar parenchyma, vWF- and vimentin-positive cells were ubiquitously interspersed with CK-positive pneumocytes (Figs. 6B, 7). The other steer euthanized at 12 hpa (No. 6) had antigenically and morphologically distinct pulmonary viral distribution. The distal segment of the middle lung lobe of this animal had a single, small focus of immunopositivity for FMDV 3D protein (Fig. 4), which was negative for capsid antigens (not shown). Unlike steer No. 7, FMDV-positive cells in this region were exclusively squamous.
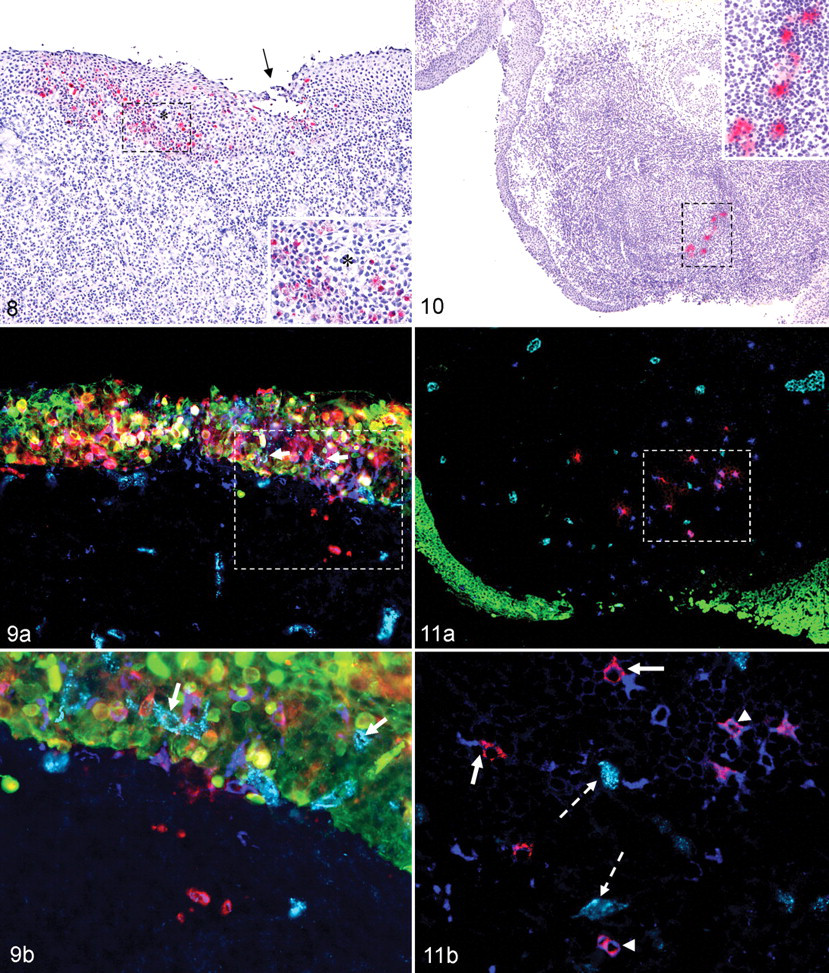
Twenty-four hpa
At 24 hpa, nasopharyngeal tissues had some of the same qualities described at 6 and 12 hpa but with variations. Viral immunopositivity was similarly localized to FAE of PALT regions but was less associated with crypt regions. Rather, FAE overlying large, expansive, cryptless regions were more commonly affected (Figs. 8–12). In such areas there were substantially greater quantities of FMDV-positive cells within the epithelium (Figs. 8–9) and the superficial and deep subepithelium (Figs. 9–12) relative to earlier time points. These cells were often positive for FMDV structural and nonstructural proteins (Fig. 12). Within subepithelial lymphoid regions, small quantities of FMDV structural and nonstructural protein–positive cells had morphologic and phenotypic (CK–, MHCII+, CD11c+) characteristics of DCs (Figs. 9–12). Additionally, concurrent labeling with anti-vWF antibody indicated that within lymphoid follicles, FMDV-positive DCs were occasionally present within 50 μm of capillary endothelia, but direct interaction between DCs and vascular cells was not observed in these areas. Microvesiculation (Figs. 8, 9) and erosion (Figs. 8–11A) of nasopharyngeal FAE were rarely observed; in such regions, capillary endothelia (vWF-positive cells) were present within the disrupted epithelium immediately adjacent to FMDV-positive epithelial cells and DCs (Figs. 8, 9). Intraepithelial vWF-positive cells were not observed in regions lacking erosion, suggesting that this was a response to viral disruption of epithelial integrity.
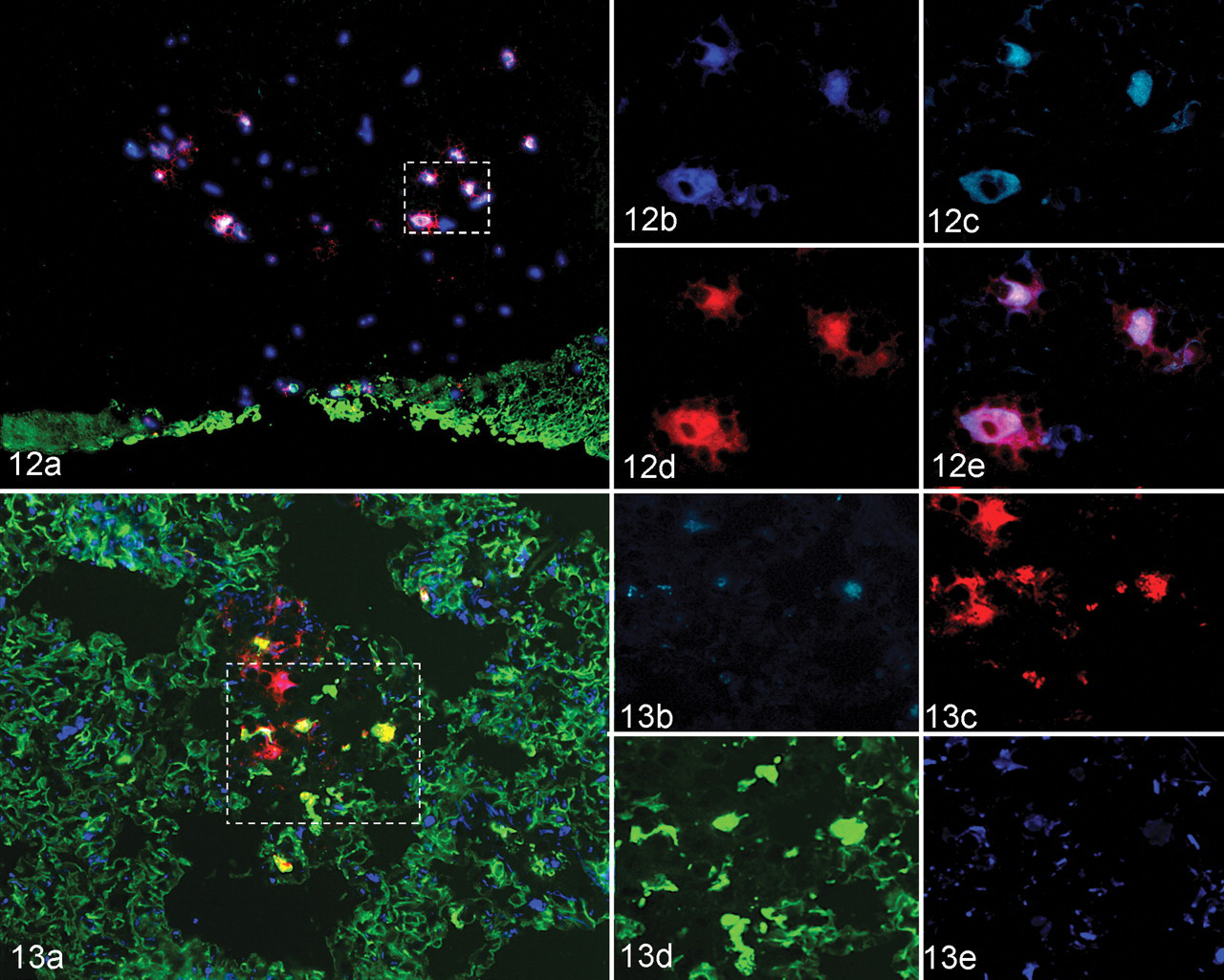
Compared to those at 12 hpa, the lungs at 24 hpa had more foci of immunopositive cells with a morphologic trend toward a greater quantity of polygonal versus squamous epithelial cell positivity (Fig. 13). Additionally, the CK/FMDV double-positive polygonal cells were often free within alveolar lumina with deterioration of surrounding CK architecture.
Forty-eight hpa
At 48 hpa there was a marked decrease of detection of FMDV immunopositive cells in the nasopharynx. Individual, and small clusters of, antigen-positive cells were present within the strata basale and spinosum of NP/L epithelia and lamina propria with no apparent predilection for FAE regions. A single cluster of FMDV-positive cells within the epiglottal epithelium of steer No. 14 was suggestive of a microvesicle. This steer was the only animal in which FMDV antigens were identified within the palatine tonsil (not shown).
In contrast to the lesser quantity of FMDV antigens localized to the upper respiratory tract (relative to steer euthanized at 6–24 hpa), at 48 hpa there was substantially increased quantity of structural and nonstructural FMDV antigens in pulmonary samples with multifocal, coalescing distribution (Figs. 14, 15). Several tissues had regionally extensive fields spanning up to 2.0 mm of FMDV antigen-positive cells within alveolar parenchyma. Within these regions, alveolar septa were expanded by infiltrates of mixed leukocytes and proteinaceous material (fibrin). Cells containing FMDV antigens were round to polygonal with eccentric nuclei and were more frequently free within alveolar lumina or loosely associated with alveolar septa when examined by IHC-light microscopy (Fig. 14). However, when examined by MIF, these cells were nearly exclusively strongly CK positive (Fig. 15). In these regions there were well-demarcated transitions from the normal CK latticework pattern in regions lacking FMDV positivity to disorganized CK clumping and dissociation of cells from septa in virus-positive regions. The pattern of MIF labelings also revealed that a subpopulation of FMDV-positive cells had somewhat greater association with alveolar septa than that detectable by light microscopy. Labelings for vimentin and vWF suggested that theses markers were present in a more normal architecture relative to the deterioration of the CK (pneumocytes) pattern. FMDV/CK double-positive cells were often directly adjacent to vWF-positive capillary endothelia (Fig. 15B). Overall, the pattern had strong similarities to the acantholytic degeneration seen in FMD vesicles.
Discussion
In considering the early pathogenesis of FMD in any species, 2 critical issues have substantial translational relevance to the development of vaccines and biotherapeutics. Identifying the primary site (or sites) of infection is foremost because blocking these sites is the only method of achieving complete sterile protection. Identifying the mechanism of the establishment of viremia is secondary but still substantial. In most cases, blocking (or impairing) this process would substantially decrease shedding, transmission, and severity of clinical signs. Previous investigations of the early pathogenesis of FMD in cattle have independently implicated the nasopharynx 7,15 and the lungs 4,6,10 as sites of primary infection. A single study indicated that upper or lower respiratory tract can similarly serve as sites of primary infection and portals for systemic generalization of FMD. 24 Recent reviews have suggested that subsequent to natural aerosol exposure, FMDV replicates in the pharynx and establishes viremia by draining through the lymphatic system, but after experimental aerosol inoculation, viremia is established directly through the lungs. 1,2 However, the primary experimental basis for these claims is not entirely clear. Another pathogenesis study implicated the nasal mucosa as the site of primary infection. 14 Overall, the published literature does not provide enough information to allow a clear interpretation of the critical virus–host interactions associated with early FMDV infection of cattle.
The current study provides a more thorough description of the early events of experimental FMD in cattle than that previously published. Additionally, the use of a controlled aerosol inoculation system has allowed a consistently repeatable method of exposure to virus while preserving the natural route of infection; high-throughput techniques have facilitated screening of numerous tissues per animal and has thus provided a highly detailed mapping of FMDV distribution.
Screening of tissues and swab specimens by rRT-PCR and VI indicated that there are tissue-specific temporal trends regarding distribution of FMDV and viral RNA subsequent to aerosol inoculation. The period of 3 to 12 hpa was generally dominated by amplification of virus in the nasopharyngeal/laryngeal (NP/L) tissues, with a trend toward progressively higher levels of viral RNA and greater prevalence of VI positivity over time. Similarly, within the period of 4 to 12 hours, FMDV RNA detection from nasal swabs was increasing toward the first of 2 peaks. The temporal coincidence of these trends suggests that the RNA in nasal swabs in this period originated from FMDV replication in the NP/L tissues. At 24 hpa, FMDV and viral RNA were increasingly abundant in the pulmonary tissues, with minimal change in the NP/L sites indicating pan–respiratory distribution of virus associated with numerous foci of replication in both upper and lower respiratory tract. However, at 48 hpa, detection of FMDV by both screening tests was decreasing in NP/L tissues, with further increase in the lungs. This coincided with the onset of viremia and the approach to the second peak in nasal swab RNA detection. Interpretation of the 2 peaks of the nasal swab FMDV RNA detection curve in the context of the tissue-specific rRT-PCR data suggests that there are distinct pharyngeal and pulmonary phases of FMDV release into the respiratory tract. Overall, the tissue-specific prevalence values for all previremic animals collectively indicates NP/L sites as the most important sites of primary FMDV infection.
The rRT-PCR and VI data indicate that subsequent to aerosol inoculation, virus inoculum was distributed throughout the entire respiratory tract; yet, immunolocalization of FMDV antigens indicated that primary infection was limited to specific regions of the nasopharynx and, somewhat later, the lungs. At early time points, tissues that were positive for FMDV by VI or rRT-PCR but IHC–MIF negative were interpreted as having virus or viral RNA on their superficial surfaces but not active infection and replication. Thus, VI and rRT-PCR served as screening tests for infection at the tissue and cellular level (high sensitivity), whereas IHC–MIF functioned as a confirmatory procedures (high specificity). In the present study, the overall success of this screening and confirmation approach provided adequate efficacy of FMDV localization.
The similar localization of FMDV antigens to the lymphoid FAE of PALT crypts of the rostrodorsal nasopharynx in both steers euthanized at 6 hpa demonstrated the importance of this region in the establishment of primary infection. The colocalization of FMDV antigens with CK and lack of colocalization with MHCII or CD11c in these tissues demonstrated that the first cells infected with FMDV are of epithelial origin. This is the first microscopic documentation of localization of FMDV to the respiratory tract of any aerogenously infected animal earlier than 24 hpa. Two previous works have demonstrated nasopharyngeal intraepithelial FMDV RNA in cattle by in situ hybridization at 5 days after contact exposure; 20,26 however, at this later phase of infection, it is impossible to separate primary aerogenous from secondary hematogenous infection. The exclusive infection of PALT FAE cells in the context of pan–respiratory exposure to virus suggests that there are specific, intrinsic qualities of these cells that make them highly susceptible to infection. The caudal segment of the dorsal soft palate is anatomically aligned with the rostral dorsal nasopharynx, and these 2 tissues were determined to be largely similar morphologically, phenotypically, and with regard to FMD pathogenesis within this study. The laryngeal tissues (epiglottis and larynx) are morphologically similar to the nasopharyngeal sites but with less mucosa-associated lymphoid tissue, which might explain the slightly decreased detection of virus in the former.
Although the nature of the susceptibility of the NP/L tissues remains elusive, possibilities include cell-specific expression of virus-specific receptors (αV integrins or other) and expression of other presently unidentified cellular factors by the susceptible cells or by other cells in the immediate microenvironment. Although αVβ6 and αVβ3 expression has been characterized in these tissues from uninfected cattle, 16,17 simultaneous localization with FMDV has not been described. The nearly diffuse epithelial distribution of αVβ6 described in one study 17 combined with the multifocal distribution of primary FMDV infection (current work) suggests that the expression of integrins does not solely dictate cellular susceptibility to infection.
The FAE is an extremely active epithelium, and candidate cells that might affect the microenvironment include intraepithelial DCs, lymphocytes, and natural killer cells. Similarly, M cells in these regions may directly facilitate infection by providing nonspecific portals for entry of virus into the host via pinocytosis. Morphologic and phenotypic characterization of bovine PALT has indicated that such cells are present in the FAE to varying extents in FMDV-infected cattle and in the resting (naïve) state (J.A., unpublished data). Additionally, physical characteristics of the FAE crypts may influence susceptibility to infection; it is possible that pooling of secretions in these regions allows increased time for adsorption of virions and thus increases efficiency of infection in a highly localized manner. Current efforts in our laboratory are directed toward investigating these mechanisms.
Determining the role of the lungs in early infection was less straightforward. Although viral RNA was detectable in pulmonary tissues at 3 hpa and infectious virus at 6 hpa (similar to NP/L sites), the prevalence of VI- and rRT-PCR-positive samples was low at these time points compared to that in the NP/L sites; additionally, viral antigens could not be detected in lungs until 12 hpa (ie, 6 hours later than the nasopharynx). At least 2 hypotheses are supported by this contrast in the data describing processes in the NP/L sites and lung. It is possible that the viral RNA and infectious virus detected in the lungs before 12 hpa represent foci that were in the process of establishing primary infection independent of infection of the nasopharynx but with a relative lag (eclipse) period. Alternatively, it is possible that primary infection of the NP/L sites serves as a requisite phase in the establishment of pulmonary infection presumably by dose amplification of inoculated virus. In this scenario, the nasopharyngeally amplified virus would serve as an endogenously generated secondary aerosol delivered to the lungs via normal respiration. It is also possible that under conditions of natural exposure (ie, direct or indirect contact), both infection scenarios are relevant with contributory roles determined by viral dose, aerosol droplet size, and various aspects of virus host interactions.
Regardless of the contributory roles of lungs versus nasopharynx in the early pathogenesis of FMD in cattle, it is clear that the pulmonary epithelia are permissive to FMDV replication. Our trimodal identification of FMDV in distal lung segments of both steers euthanized at 12 hpa (previremic) indicates the consistent role of the lungs in the early progression of FMD. This is consistent with previous works that localized FMDV RNA to alveolar septa in previremic FMD in aerosol inoculated cattle. 4,6 In the present study, the morphologic localization of FMDV to alveolar septa combined with exclusive colocalization with CK in these regions suggests that pneumocytes are the most relevant cells supporting viral replication in the lungs. The morphologic variation of FMDV-positive cells (squamous versus polygonal) seen in the lungs of steer No. 7 likely represents a temporal and pathologic continuum with the squamous variant (type I pneumocyte) representative of early cellular infection that progresses to infection of cells with polygonal morphology (type II pneumocytes). Thus, the single, small (type I pneumocyte morphology) focus of anti-FMDV immunopositivity in middle lung lobe of steer No. 6 (Fig. 4) is interpreted as an earlier stage relative to cranial lung specimen of steer No. 7 (Figs. 5–7). This concept is further supported by the increasing abundance of FMDV-immunopositive type II pnuemocytes at 24 and 48 hpa and by “conventional wisdom” on the subject of responses to insults by pulmonary epithelia. 9 At 12 hpa, colocalization of FMDV antigens and CK was rarely identified within terminal bronchiolar epithelia, indicating that these cells are also susceptible to infection but with apparently lesser efficiency compared to pneumocytes.
The question of how viremia is established in FMD has substantial implications for ongoing and future development of FMD prophylaxis. It has been suggested that in bovine FMD, the mechanism of the establishment of viremia is that virus drains from the pharynx to the regional lymph nodes from which viremia is established. 1,2 In the present study, the lack of detection of FMDV or viral RNA in lymph nodes draining the respiratory tract and oral cavity in the previremic and early viremic phases of FMD suggests that this is not the route of establishment of viremia within the current work’s experimental conditions. A specific region’s ability to serve as a portal for establishment of viremia depends on local presence of virus in immediate proximity to blood vessels and on a competent mechanism of movement of virions from the extravascular to intravascular space. The transfer mechanism is a complex subject that was not addressed by the current study but likely involves several molecular and cellular recognition events. However, in the current work, the requirement for intimate association of FMDV and blood vessels was shown to be met in previremic animals in tissues of the nasopharynx and lungs.
As early as 12 hpa, FMDV-positive type I pneumocytes were present within alveolar septa that contained abundant vWF- and vimentin-positive cells (ie, capillary endothelia). This suggests that in the lungs, FMDV would have a high likelihood of gaining access to the vasculature. However, at 24 hpa, FMDV was also present in close proximity to vWF-positive (endothelial) cells within the PALT epithelium and lamina propria. The intraepithelial capillaries in regions of erosion may represent a neovascular response to ongoing FMDV-induced epithelial injury. Additionally, small caliber intraepithelial blood vessels have been demonstrated within the normal reticular epithelium (FAE) of the bovine palatine tonsil, which has many morphologic and phenotypic similarities to PALT. 19 The possibility that viremia may be established in the PALT is supported by the presence of these vessels associated with FMDV-positive epithelial cells and DCs. Overall, these findings are consistent with the earlier work demonstrating that the lungs or upper respiratory tract may independently serve as portals for systemic dissemination. 24
An additional, salient consideration regarding the site of establishment of viremia is that in the period in which viremia was established (24–48 hpa), viral load in the NP/L sites was decreasing by all detection methods, whereas in the lungs, virus was replicating to high genome copy numbers in cells that are in direct association with capillary endothelia. At the time of establishment of viremia, pulmonary quantities of FMDV, viral RNA, and structural and nonstructural antigens were all increasing. Although these data do not conclusively establish a single responsible anatomic site for the establishment of FMDV viremia in cattle, interaction between processes in upper and lower respiratory tract regions are likely, and current efforts in our laboratory are directed toward defining the relative contributory roles of these regions.
The FMDV-associated degenerative process observed in the lungs at 48 hpa has many microscopic similarities to the classic acantholytic degeneration described for vesicles caused by FMDV and other pathologic processes; 11,28 however, macroscopic lesions were not detected in the lungs. As is seen with keratinocytes in FMDV vesicles, the pneumocytes swell, lose association with adjacent structures, and dissociate into an expanding cavitary space. 21,28 However, unlike a classic vesicle in which the morphologic characteristics of acantholysis are well defined and recognizable, this pulmonary acantholytic-like degeneration could easily be mistaken for a histiocytic alveolitis. The morphology of the acantholytic pneumocytes is similar to that of alveolar macrophages, and it is only the CK positivity of these cells that confirms their epithelial histogenesis. An additional consideration on this subject is the relationship that this pulmonary process may have to the maintenance of high-titer viremia during acute FMD. Although it has been suggested that sustained FMDV viremia is maintained by viral replication in lesional and/or nonlesional skin, 1,5 this hypothesis has never been thoroughly elucidated. Several factors support the notion that the lungs may be important amplifiers of FMDV and may be responsible for maintaining high titer viremia, including (1) the relatively high FMDV RNA/mg and large quantities of FMDV structural and nonstructural antigens detected in the lungs of viremic cattle in this study, (2) the overall mass of the lungs, and (3) the extensive vascularity of lungs.
In conclusion, the data presented herein support a model for early FMDV infection of aerosol-inoculated cattle that includes the following key events: (1) primary replication in epithelial cells of the PALT crypts and (2) subsequent widespread replication in pneumocytes in the lung, which coincides with (3) the establishment of sustained viremia. Current work in our laboratory is directed toward similar investigation of additional serotypes and strains of FMDV and further elucidation of FMDV–host interactions in the early pathogenesis of FMD.
Footnotes
Acknowledgements
This research was funded by Agricultural Research Service–Current Research Information System project No. 1940-32000-052-00D. We wish to acknowledge Meghan Tucker for superb histotechnological support and Elizabeth Bishop, Ethan Hartwig, and George Smoliga for crucial assistance in collecting and processing samples. We also thank the animal resource unit personnel at the Plum Island Animal Disease Center for their patience and assistance in caring for and collecting samples from experimental animals. We thank Dr Manuel Borca and Dr Fawzi Mohammed for thoughtful review of the article.
The authors declared no potential conflicts of interests with respect to the authorship and/or publication of this article.
The authors are all salaried employees of the US federal government. The research described and the authorship of the manuscript was all conducted within the duties of the authors' job posts. There was no additional support.
