Abstract
A panel of 36 sera has been assembled from experimental cattle that had been infected by inoculation or contact exposure with 4 serotypes of foot-and-mouth disease virus (FMDV) with or without prior vaccination. Virus replication and persistence had been characterized in all of the animals. The proportion of the sera scored positive by 5 tests for antibodies to the nonstructural proteins of FMDV varied, suggesting that the panel can discriminate between the sensitivity with which such tests are able to identify infected cattle. Use of this panel will help in assessment of new tests and quality control of existing methods.
Foot-and-mouth disease (FMD) affects domestic and wild cloven-hoofed animals in parts of Asia, Africa, the Middle East, and South America. It is highly contagious and economically important. The FMD virus (FMDV) belongs to the genus Aphthovirus and the family Picornaviridae, and it includes 7 serotypes. The disease has an acute course but African buffalo, cattle, sheep, and goats may become persistently infected (carriers) by virus replicating subclinically in the oropharynx. 17
Serology is used to help diagnose FMD and to certify freedom from infection. This is complicated by the administration of FMD vaccines, which elicit antibodies that may cross-react in tests designed to detect evidence of previous infection. 5 FMD vaccines can prevent illness and reduce viral replication and excretion. 1 However, FMD vaccination does not necessarily prevent FMDV infection and some degree of virus replication; such animals can become viral carriers and may be considered a threat to other animals. Consequently, a constraint to the use of vaccination is the need for a reliable method to demonstrate that vaccinated animals have not been subclinically infected with FMDV and are not still carrying the virus. 6,16
Tests for antibodies against the nonstructural proteins (NSPs) of FMDV have certain advantages over conventional FMD serology targeted at antibodies to structural proteins of the virus such as the virus neutralization test (VNT), liquid-phase blocking enzyme-linked immunosorbent assay (ELISA) (LPBE) and solid-phase competition ELISA (SPCE). For NSP antibody tests, different antigens are not needed for each of the 7 FMDV serotypes, and the assays are specific for infection and not vaccination, provided that vaccines are used from which most of the NSPs have been removed. 5 To validate and compare tests for NSP antibodies, sera are needed from various categories of vaccinated and infected stock. It has proved difficult to assemble large panels of such sera, but results obtained so far have indicated that there is variability in the performance of different NSP serologic tests, especially regarding their ability to detect subclinical infection in vaccinated and subsequently exposed animals. 2,13 We have developed a panel of cattle sera that should be useful for evaluating the relative sensitivity of new NSP tests and for batch control of existing assays.
The NSP panel consists of 36 bovine sera collected in bulk amounts at the termination of 6 vaccine/challenge experiments conducted at the Pirbright laboratory in the past 4 years. Experiments were designed to evaluate vaccine-induced protection against challenge 21 days later by contact with infected animals or direct inoculation with FMDV. Vaccines used were oil adjuvanted and complied with the Office international des épizooties (OIE) guidelines for freedom from NSPs Each experiment included unvaccinated cattle as controls, all of which contracted FMD. The available volumes of sera average 390 ml (90 to 1,000 ml) and are stored frozen at −20°C.
The first experiments involved serotype O (UV, UY, and VH series; Table 1) and vaccination with either a full or a 10-times dose of O Manisa vaccine (Cox et al. 7,8 ; Parida et al. 12,13 ). Challenge was affected by 5 days of contact with donor cattle that had been infected with O UKG 34/2001 by tongue inoculation. All 40 of the vaccinated cattle were protected from the disease. However, replication of virus was detected in most of the vaccinated cattle, and 12 animals were scored as persistently infected based on the results of virus isolation and real-time reverse-transciption polymerase chain reaction (RT-PCR).
Sera representing serotypes Asia 1, A, and SAT 2 were derived from European Pharmacopoeia–compliant vaccine potency tests. 10 Each test used 17 cattle (VE, UZ, and VL series; Table 1) with animals divided into 4 groups and housed in separate boxes. In the first group 2 animals were kept as nonvaccinated controls. Groups 2 through 4 consisted of 5 animals, each vaccinated with 1/16, 1/4, and full doses of vaccine, respectively, by the intramuscular route. All animals were challenged by intradermolingual injection with a 10,000 ID50 dose of virulent homologous virus.
Esophagopharyngeal fluids were collected with a probang sampling cup to evaluate the status of infection of each of the NSP serum-panel animals and to see if the animals were viral carriers. The presence of FMDV in probang samples was scored by virus isolation and confirmatory ELISA and, except for the Asia 1 series, also by quantitative RT-PCR. 8 In the O Manisa/O UKG experiments, the exact time-point at which the contact-challenged animals became infected cannot be determined because of the nature of exposure; therefore, the term “days after challenge” rather than the term “days after infection” is used.
Serum antibodies to FMDV NSPs were measured by commercially available ELISAs and one in-house test (Istituto Zooprofilattico Sperimentale, Brescia [IZS]). 1,9 The commercial tests used were the Ceditest FMDV-NS, a , 15 the FMDV NSP ELISA, b , 18 the FMD virus 3ABC Svanovir, c and the CHEKIT-FMD-3ABC. d , 4 These tests are referred to hereafter as IZS-Brescia, Cedi, UBI, Svanova, and Bommeli tests, respectively. Each was used according to the manufacturer's recommendations, except that sera scoring inconclusively positive in the Bommeli test (n=5) were considered positive for the purpose of test comparisons.
The virus neutralization test (VNT), LPBE, and SPCE were as described 11 ; titrated sera was tested in VNT and LPBE, and sera were tested at a fixed dilution of 1 in 5 in SPCE with a 60% inhibition cutoff point. Each test used viruses or antigens of a serotype matched to that against which the animals had been vaccinated/infected with.
Heat treatment of serum for 2 hours at 56°C can reduce the likelihood of samples being contaminated with live FMDV and was evaluated at Institute for Animal Health, Pirbright Laboratory (IAH) for its effect on the Cedi, Bommeli, UBI, and Svanova tests. An additional 39 sera from FMD-naive cattle, neither vaccinated nor infected, were included in this trial. The 36 NSP panel sera were tested both at IAH and IZS Structural protein tests were only performed at IAH once on untreated sera and once on heat-treated sera. The commercial NSP tests were each repeated 4 times at IAH, twice with untreated sera and twice with heat-treated sera. Only heat-treated sera were supplied to IZS, and these were tested twice with each commercial test and 3 times with the in-house IZS test. At IAH, all serologic test results are the mean of sera tested in duplicate wells, whereas each serum was tested at IZS as a single replicate in one well.
The sera fell into 4 groups according to whether they were from cattle vaccinated before challenge exposure and whether or not these became FMDV carriers. Twenty were from carriers that had been vaccinated, and 7 were from carriers that had not been vaccinated before challenge. Four were from noncarriers that had been vaccinated and 5 were from noncarriers that had not been vaccinated before challenge. The timing of collection of the sera with respect to vaccination, challenge, and infection are shown in Table 1. The sera were collected when the animals were killed. The last day on which virus was recovered before this is given in the table, but it is a conservative estimate of virus replication and persistence because virus recovery from probang samples may be intermittent, and samples were not collected on every day. For carrier animals, there is a possibility that further recoveries would have been made had the animals been retained and sampled for longer.
Heat treatment had no significant effect on the results of tests for antibodies to structural proteins, and all NSP panel sera scored positive in these tests. With NSP tests, only Bommeli results were affected by heat treatment; it increased the percentage positivity of the results for both the NSP panel sera (7 sera changed from consistently negative to positive in one or more test replicates) and the 39 negative sera, 2 of which became false-positive (mean results for before and after treatment ratios for negative sera tested with Bommeli, Cedi, Svanova, and UBI tests were 3:9, 6:5, 13:12, and 4:4, respectively). For all other assay/serum combinations the results of testing untreated or heat-treated sera could be analyzed together for purposes of comparing test repeatability and reproducibility.
Results of all NSP tests are summarized in Table 2. Twenty sera were from vaccinated, carrier cattle exposed to 4 different serotypes. Eight cattle (UV and UY) were exposed to the O serotype, 4 (UZ) to the A serotype, 5 (VE) to Asia1, and 3 (VL) to SAT 2. There were differences in the performance of the 5 NSP ELISAs on the samples that had been collected from O serotype experiments at 3.5 to 6 months after exposure of cattle by contact, whereas the samples collected from potency test cattle 1 or 2 months after infection by tongue inoculation gave similar results (Table 2), except for animals UZ 59 and UZ 62 from the A Iran 96 trial and animal VL 90 from the SAT 2 experiment. The Cedi and IZS tests detected the highest number, that is, 18 of 20 carriers (IZS results were scored as positive if ≥2 of 3 repeats were positive). A further serum from animal UV 19 was scored positive by the IZS ELISA once out of 3 repeats. The same animals were not missed by both tests. The Svanova and UBI tests scored 8 of the sera positive in all repetitions, increasing to 11 and 10, respectively, if the majority of positive test results are considered. In each of the tests, 1 and 2 additional sera were scored positive, respectively, in a minority of test repeats. The Bommeli test on untreated sera detected 10 carriers consistently (2 of them were in the inconclusive range), while UV 11 was also scored an inconclusive positive on 1 of 2 test repeats. After heat treatment, UV 9 and UV 11 became consistently positive (Table 2), and UV 13, UY 90, UZ 62, and VL 90 were sometimes positive.
Origins of NSP serum panel.
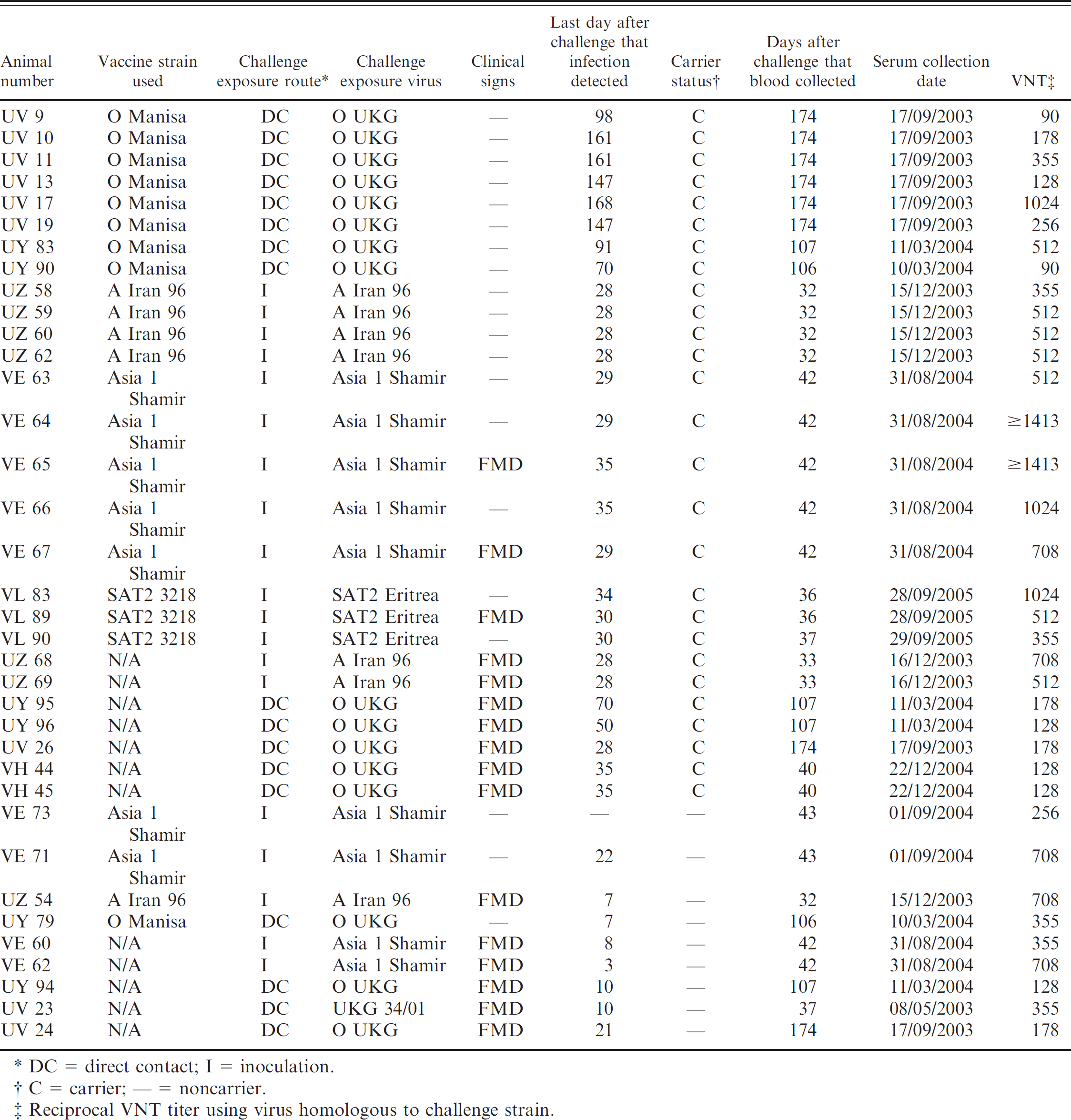
DC = direct contact; I = inoculation.
C = carrier; –- = noncarrier.
Reciprocal VNT titer using virus homologous to challenge strain.
Quantitative results of NSP ELISAs for heat-treated serum panel.∗
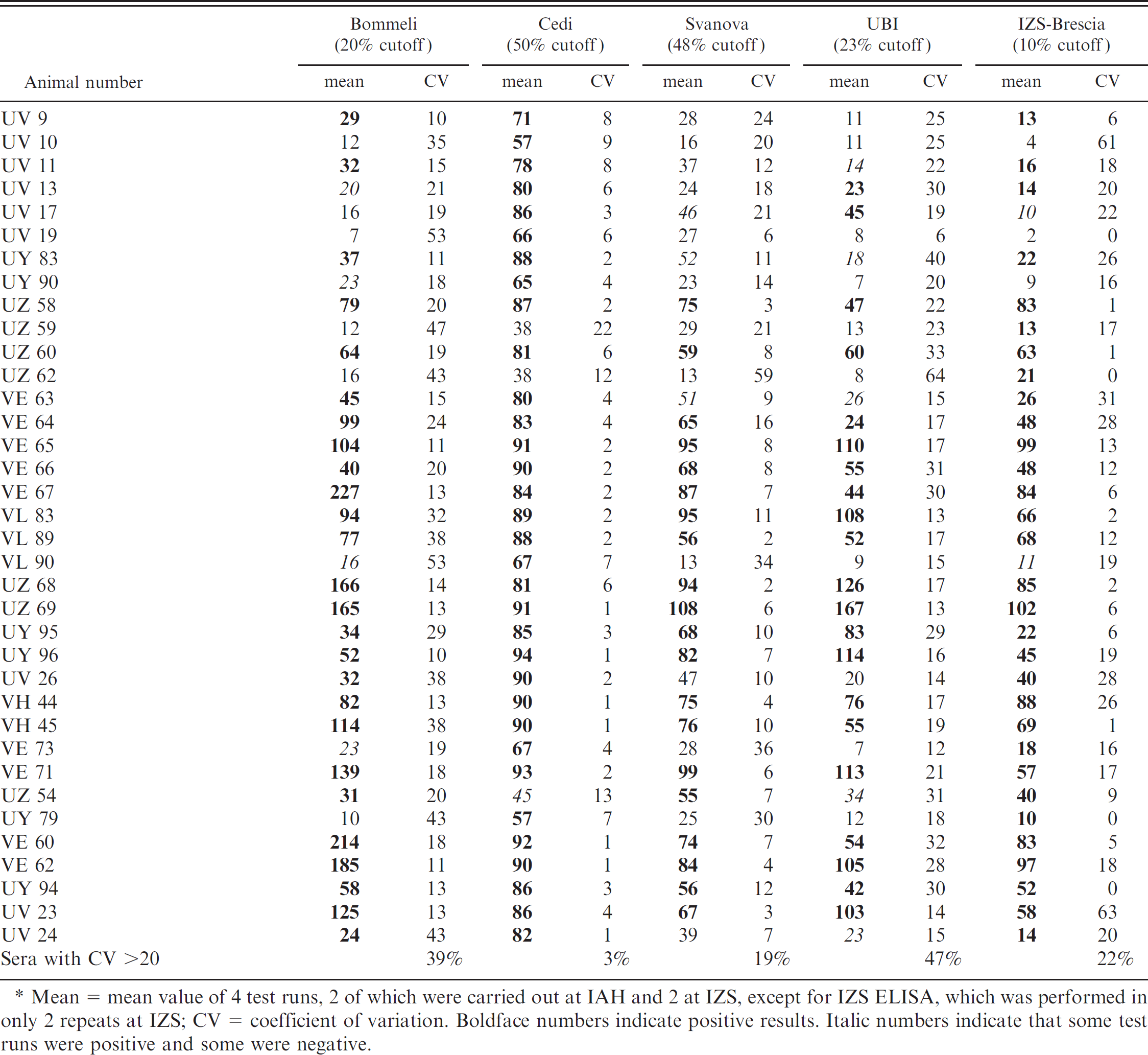
Mean = mean value of 4 test runs, 2 of which were carried out at IAH and 2 at IZS, except for IZS ELISA, which was performed in only 2 repeats at IZS; CV = coefficient of variation. Boldface numbers indicate positive results. Italic numbers indicate that some test runs were positive and some were negative.
Sera from unvaccinated carriers came from 7 cattle; 5 that had been infected with O UKG FMDV by contact (UY, UV, and VH series) and 2 with A Iran 96 FMDV by injection (UZ series). These animals had served as unvaccinated controls, had developed FMD lesions on their tongue and feet, and had been scored as carriers by virologic tests. The 4 sera collected from cattle between 33 and 40 days after exposure to FMDV were scored as positive by all of the different NSP tests (Table 2). Of 3 sera collected from animals 3.5 to 6 months after exposure (UY 95, UV 26), 2 were scored less consistently. The UBI test missed UV 26 in all but one repetition, and it was inconsistently detected by the Svanova test; UY 95 was scored negative on one repetition by the Bommeli test.
Sera from vaccinated noncarriers were collected from 4 animals exposed to 3 FMDV serotypes (VE series, Asia1; UZ series, A; UY series, O). In these animals, virus or viral genome was detected in probang samples within the first 2 to 3 weeks after exposure to FMDV, but not thereafter. The various NSP tests performed differently with the sera from this group (Table 2). Two sera collected from cattle UY 79 and VE 73 at 3.5 and 1.5 months after contact exposure or direct inoculation were not scored positive by Bommeli, Svanova, and UBI tests, whereas both were scored positive in the Cedi and IZS tests. However, the serum from animal VE 73 became positive in the Bommeli test on 3 of 4 occasions after heat treatment. The other 2 sera came from cattle that had been directly inoculated with FMDV and were detected as positive in all tests, with the exception of serum from animal UZ 54, which was scored inconsistently positive in the Cedi test and was missed in one repeat of the UBI test.
Sera from unvaccinated noncarriers were derived from another group of 5 cattle used as unvaccinated controls in different experiments involving 2 serotypes (VE series, Asia1; UY and UV series, O) and both direct-contact challenge and tongue inoculation of FMD virus (Table 1). Four samples were scored positive in all the tests (Table 2), whereas the serum collected 6 months after infection from UV 24 was scored consistently positive only in the Cedi and IZS tests.
Repetitions of different tests revealed mostly unchanged result categories. This is summarized in Table 2, showing coefficient of variation between test results calculated as the standard deviation multiplied by 100 and divided by the mean. Cedi test results were qualitatively the most repeatable, despite 3 different kit batches having been used. Only one serum (from UZ 54) alternated between positive and negative both at IAH and IZS because of a borderline reaction. The next most repeatable were the Svanova and Brescia tests, whereas the UBI and Bommeli tests had the lowest repeatability and coefficient of variation.
NSP tests are useful for postoutbreak serologic surveillance to demonstrate freedom from FMDV whether or not vaccination has been used to help curtail outbreaks. Categories of reference sera needed to assess the performance of NSP serologic tests, therefore, include samples obtained from infected animals to evaluate sensitivity and from uninfected animals to evaluate specificity. Postoutbreak serosurveillance is normally delayed until at least a month after the last clinical case of FMD, and this period may be further extended after large outbreaks, as was the case after the 2001 outbreaks in the United Kingdom. Reference sera are therefore required from vaccinated and unvaccinated animals; in the case of infected animals, infection should have occurred one to several months before sample collection. Because a major aim of post-outbreak serosurveillance is to detect subclinical infection, especially virus carriers that cannot be recognized by clinical surveillance, samples from this category of animal should also be represented in a reference panel.
A number of studies have described the relative sensitivity and/or specificity of NSP tests. 2,3,13,15 The performance of the 4 commercial assays and the single in-house assay in the current study mirrors those reported in the aforementioned publications, and the implications of this for the application of postoutbreak serosurveillance has been reported separately. 14 NSP antibody development correlates to the extent of virus replication; therefore, prior vaccination, which may reduce virus replication, may also reduce the sensitivity of NSP tests for detecting infection. 13,15 Cattle that became infected by contact with infected donors rather than by needle inoculation were in general harder to detect by NSP serology as were sub-clinically infected animals. As noted by Parida et al. 13 there is a tendency for antibody detection to wane with increasing time after infection, although this effect was not seen when using the Cedi and IZS tests.
Despite a number of comparative studies on NSP test performance, a general difficulty has been obtaining sufficient samples representative of the different species that may be infected with FMDV, of the different categories of status with respect to vaccination and infection of the donor animals, and of the different serotypes of FMDV. Furthermore, the results of studies in different laboratories using different test sera are not readily comparable. Because new NSP tests are expected to be developed, and there is an ongoing need to quality control the available assays, it would be useful to have stocks of a panel of reference sera able to discriminate between tests with differing sensitivity of NSP antibody detection. This study reports on the establishment of such a panel of 36 well-characterized reference sera and has demonstrated the utility of the panel in discriminating between the performances of 4 commercially available NSP tests and one in-house test. The reactivity of the panel sera with serologic tests for antibodies to FMDV structural proteins has also been described, as well as the effect on different tests of heat inactivating the sera. The panel is not designed to help evaluate test specificity because this requires large numbers of samples from naive animals or else a subset of such sera known to give false-positive results. The panel also lacks representative sera of 3 FMDV serotypes, but as further animal experiments take place these gaps may be filled. Field sera have not been included in the panel because of the difficulty of obtaining sufficient volumes from animals of adequately known status. However, studies using field sera have suggested a similar relative performance of the 4 commercial NSP tests evaluated here. 2,3 Nevertheless, because of the many factors that will affect the proportion of NSP seropositive animals after an outbreak, as well as the strength of the serologic reactions induced in these animals, it cannot be expected that the diagnostic sensitivity of a test will remain constant nor be fully reflected by the test's performance with a single NSP serum panel. It is intended that the panel be made available to FMD reference laboratories to evaluate new tests and to quality control new batches of existing tests, especially when large diagnostic reserves are being established as part of contingency plans for dealing with future outbreaks. As a reference panel, the sera are not primarily intended for use in routine proficiency testing or to provide in-test controls. Future use of the panel may also enable further studies of the repeatability of the tests and their performance in different laboratories to be assessed. However, the present reproducibility study in 2 laboratories has shown that similar results can be obtained using all the 4 commercial assays and with different assay batches. Where there was some lack of qualitative repeatability and reproducibility, this was mainly seen with sera having borderline positive results or in the case of the Bommeli test, where heat treatment affected the outcome. Because heat treatment also increased the Bommeli test reactivity of negative sera, there is probably no overall improvement in sensitivity or specificity; rather a new cutoff would be required for testing this type of sera.
Acknowledgements. This work was supported by grants from the U.K. Department for Food and Rural Affairs, United Kingdom (SE2918 and SE1122) and from the European Commission (FMDJmprocon project of the EU 6th Framework Programme, SSPE-CT-2003–503603).
Footnotes
a.
Ceditest FMDV-NS, Cedi Diagnostics B.V. Lelystad, The Netherlands.
b.
UBI FMDV NS ELISA (CATTLE), United Biomedical Inc, Hauppauge, NY.
c.
Svanovir FMDV 3ABC-Ab ELISA, Svanova Biotech AB, Uppsala, Sweden.
d.
CHEKIT-FMD-3ABC bo-ov, Bommeli Diagnostics, Liebefeld-Bern, Switzerland.
