Abstract
Diabetes mellitus (DM) is a group of chronic metabolic diseases characterized by persistent fasting hyperglycemia, and it can be of either polygenic or monogenic origin. Animal models have played an important role in elucidating the pathophysiology of the polygenic Type 1 and type 2 DM forms; however, useful animal models of the monogenic forms do not exist. The authors describe 4 cases of naturally occurring DM in vervet monkeys (Chlorocebus aethiops sabaeus), 1 of which has clinicopathologic findings consistent with type 2 DM, including persistent hyperglycemia, hypertriglyceridemia, islet amyloidosis, and reduced islet insulin immunostaining. In contrast, the 3 remaining animals have clinicopathologic similarities to a monogenic form of the disease, including a lack of islet amyloidosis and hypertriglyceridemia, as well as normal islet insulin immunostaining. In addition, pedigree analysis conducted on one of these animals is consistent with either an autosomal dominant or mitochondrial inheritance pattern, which supports a monogenic form of DM. The authors thus hypothesize that a naturally occurring monogenic form of diabetes may occur in vervet monkeys, making them a potential animal model for future studies.
Keywords
Diabetes mellitus (DM) is a group of chronic metabolic diseases characterized by persistent fasting hyperglycemia owing to defects in insulin production, secretion, or action in target tissues. Numerous subtypes of DM have been identified including type 1, type 2, maturity-onset diabetes of the young (MODY), and mitochondrial diabetes. Animal models have played an important role in elucidating the pathophysiology of type 1 and type 2 DM; however, useful animal models of MODY and mitochondrial diabetes do not exist.
Type 1 DM is a polygenic autoimmune disease in which pancreatic beta cells are targeted and destroyed. 13 It presents more commonly in children and adolescents than adults; however, adult-onset type 1 DM does occur. Treatment generally consists of exogenous insulin administration, although pancreatic islet cell transplantation is being investigated experimentally. 4,14 Spontaneous type 1 DM occurs at a low frequency in macaques. 18 Other useful animal models of type 1 DM include the nonobese diabetic mouse and streptozotocin-treated animals. 1,5,15,16,18 Conversely, type 2 DM more commonly develops in adults, but prevalence in children is increasing as the incidence of childhood obesity increases. This form of DM accounts for more than 90% of overall diabetes prevalence, with US and worldwide incidence increasing at epidemic rates. 20 This is a complex polygenic form of the disease in which persistent insulin resistance in target tissues (eg, liver, muscle, fat) results in a compensatory increase in pancreatic insulin production in attempts to maintain euglycemia. Insulin secretion is accompanied by coproduction of islet amyloid polypeptide, also known as amylin. Amylin has been shown to induce beta cell apoptosis, and the accumulation of islet amyloid with concurrent islet cell loss is a hallmark of type 2 DM. 6 Macaques are a valuable animal model of type 2 DM because of the close phylogenetic relationship to humans, the similar reproductive and endocrine physiology to humans, and the potential to develop spontaneous obesity and insulin resistance with similar pancreatic pathology. 12,17 –19
MODY is a heterogeneous group of disorders of monogenic etiology that comprises about 5% of the clinical cases of diabetes in the United States and the world. 3 According to traditional clinical nomenclature, there are at least 6 types of MODY, each associated with mutations in different autosomal genes and inherited in an autosomal dominant pattern: HNF4A (hepatic nuclear factor 4 alpha; MODY 1), GCK (glucokinase; MODY 2), HNF1A (MODY 3), IPF1 (insulin promoter factor 1; MODY 4), HNF1B (MODY 5), and NEUROD1 (neurogenic differentiation 1; MODY 6). 11 Of these, all except GCK (MODY 2) are transcription factors that regulate expression of insulin and other proteins involved in glucose metabolism and transport within the pancreatic beta cell. 3 The result of these mutations is defective glucose sensing, resulting in impaired insulin secretion in the face of normal or near-normal insulin production and islet morphology. Even so, the phenotypic expression of these related conditions is quite diverse, and authors have suggested that the term MODY as a clinical diagnosis be replaced with a more suitable description based on genetic subgroup. 10 These subgroups consist of neonatal DM, mild hyperglycemia caused by mutations in GCK, DM associated with mutations in transcription factors (eg, HNF1A, HNF1B, HNF4A, and NEUROD1), and mitochondrial DM. In addition, and in contrast to type 2 DM, body weight of MODY-affected individuals is not associated with disease development and pathogenesis. In human cases of MODY, body weights distribute as they do in the general population, including both normal-weight and overweight individuals.
Mitochondrial diabetes, also called “maternally inherited diabetes and deafness,” is another monogenic form of the disease. In this form, mutations in mitochondrial DNA—most commonly, an A3243G mutation in the mitochondrial DNA-encoded tRNA (Leu, URR) gene—result in beta cell dysfunction and impaired glucose metabolism. 8 Clinically, this manifests as pronounced age-dependent deterioration of pancreatic function and insulin production. 8
Unfortunately, useful animal models for any of the monogenic forms are lacking. We describe 4 cases of naturally occurring DM in vervet monkeys (Chlorocebus aethiops sabaeus), 3 of which have clinicopathologic and inheritance similarities to a monogenic form of the disease. Thus, we hypothesize that a naturally occurring monogenic form of diabetes may occur in vervet monkeys, making them a potential animal model for future studies.
Materials and Methods
Animals
Four female vervet monkeys between 18 and 20 years of age presented with diabetes. All were part of the multigenerational pedigreed Vervet Research Colony (VRC), currently located at Wake Forest University Primate Center. The VRC was founded in 1975 as a facility jointly managed by the University of California, Los Angeles, and the Department of Veterans Affairs. The founder animals (28 males, 29 females) were wild caught from 1975 to 1983 in St. Kitts, West Indies, as described in McGuire 9 and Fairbanks et al. 2 The current population consists of approximately 530 descendants with 24 of the original matrilines now in their second to eighth generation. For clarity, the animals of interest—Nos. 978, 1005, 1988014, and 982—are referred to as Nos. 1, 2, 3, and 4, respectively. All animals were handled in accord with the Guide for the Care and Use of Laboratory Animals, and the Institutional Animal Care and Use Committees at Wake Forest University Health Sciences, UCLA, and the Department of Veterans Affairs Greater Los Angeles Healthcare System. All animals were fed Purina Lab Diet Monkey Diet 5038 (5% fiber, 13% fat). Animal Nos. 2 and 3 were fed Purina Lab Diet Fiber Balanced Monkey Diet 5052 (12% fiber, 15% fat) for the final 2 and 4 years of life, respectively. Animal No. 4 was fed a noncommercial experimental diet (8% fiber, 35% fat) on two separate occasions lasting 5 and 9 months each. All animals also received fresh fruits, vegetables, and water ad libitum.
Clinical Pathology
Animals were fasted overnight and sedated with ketamine hydrochloride (10 to 15 mg/kg intramuscular) (Ketaset, Fort Dodge Animal Health, Fort Dodge, IA) before examination and sample collection. Circulating glucose, insulin, and hemoglobin A1c (HbA1c) concentrations were determined as previously reported. 7 Briefly, glucose was measured in whole blood by glucometer (One Touch Ultra Glucometer, Lifescan, Inc, Milpitas, CA) or in plasma by the glucose oxidase method (Roche, Basel, Switzerland). The blood glucose reference range (95% confidence interval) for vervet monkeys (C aethiops sabaeus) was calculated on the basis of 157 measures from adult nonpregnant colony-born females. Samples were analyzed for insulin and glycation of HbA1c with a commercial ELISA kit (Mercodia, Uppsala, Sweden) and via high-performance liquid chromatography (Primus PDQ, Primus Diagnostics, Kansas City, MO), respectively. 7 Triglycerides were measured enzymatically. Body mass index (BMI) was calculated as follows: weight / crown–rump length 2 (kg/cm2). The reference range (95% confidence interval) for vervet monkeys (C aethiops sabaeus) was calculated on the basis of 157 measures from adult nonpregnant colony-born females. Intravenous glucose tolerance tests were conducted on 2 animals (Nos. 1 and 4) and a control animal as previously described: 17 After sedation with ketamine (15 mg/kg intramuscular, supplemented at 3 to 5 mg/kg as necessary) and collection of baseline blood samples via percutaneous femoral venipuncture, 50% dextrose (750 mg/kg) was infused via the saphenous vein. Blood samples were subsequently collected at 5, 10, 20, 30, and 60 minutes into EDTA-treated tubes and placed on ice. Samples were centrifuged, and plasma was stored at –80°C until analysis for glucose and insulin concentrations. Calculated parameters include area under the curve for glucose and insulin and glucose disappearance rate (Kg).
Histopathology and Immunohistochemistry
All animals underwent complete diagnostic necropsies with gross and histologic evaluations of all major organs, except No. 3, in which case only formalin-fixed pancreas was examined. Sections of formalin-fixed paraffin-embedded pancreata from each animal were cut at 5 μm and stained with hematoxylin and eosin (HE) and Congo red. For immunohistochemistry, 5-μm sections were incubated with guinea pig anti-insulin polyclonal antibody (prediluted; Biomeda, Foster City, CA) or rabbit anti-glucagon polyclonal antibody (prediluted; BioGenex, San Ramon, CA) for 75 minutes at 35°C. Negative controls were incubated with nonimmune sera, instead of primary antibody, for 75 minutes at 35°C. Primary antibodies were localized with appropriate biotinylated secondary antibodies, streptavidin–alkaline phosphatase (Biogenex) and Vector Red (Vector Labs, Burlingame, CA) substrate. Sections were counterstained with Mayer’s hematoxylin and examined by light microscopy by a board-certified veterinary pathologist (J.A.C.).
Results
Clinical Findings
Routine blood glucose screening of animals within the VRC between 2001 and 2008 revealed 23 vervets with persistent fasting hyperglycemia (> 100 mg/dl), 4 of which are featured here. Figure 1 shows the fasting blood glucose concentrations as determined over a 5-year period. Animal Nos. 1 and 2 were markedly hyperglycemic. Further workup of No. 1 revealed an elevated HbA1c concentration (16.2%) and elevated plasma glucose with reduced insulin in response to intravenous glucose tolerance testing (Fig. 2). Management with parenteral NPH insulin (Novolin 70/30, NovoNordisk, Bagsvaerd, Denmark) at a dose adapted from diabetic cynomolgus macaques was attempted on both animals. 17 However, both were found to be unusually sensitive to insulin therapy, as evidenced by rapid-rebounding hypoglycemia posttreatment, and both succumbed to posttreatment hypoglycemic complications within 4 months of initiation of insulin therapy. BMI of both animals was greater than normal throughout life; however, it fell to within or below the reference range during the final 3 to 5 years (Fig. 3). Triglyceride concentrations were within reference range for both animals (88 mg/dl and 28mg/dl, respectively). Animal No. 3 had moderate persistent fasting hyperglycemia (Fig. 1) and did not receive insulin therapy. BMI of this animal was greater than normal throughout life but fell to below normal during the final year (Fig. 3). Triglyceride concentrations were within reference range (75 mg/dl). Animal No. 4 had a mild to moderate persistent fasting hyperglycemia (Fig. 1) that did not require insulin therapy. Further workup of this animal revealed an elevated HbA1c concentration (6.4%) and persistently elevated plasma glucose and insulin in response to intravenous glucose tolerance testing (Fig. 2). BMI of this animal was greater than normal throughout life but fell to within or below normal during the final 2 years of life (Fig. 3). Triglyceride concentrations were markedly elevated (189 mg/dl).
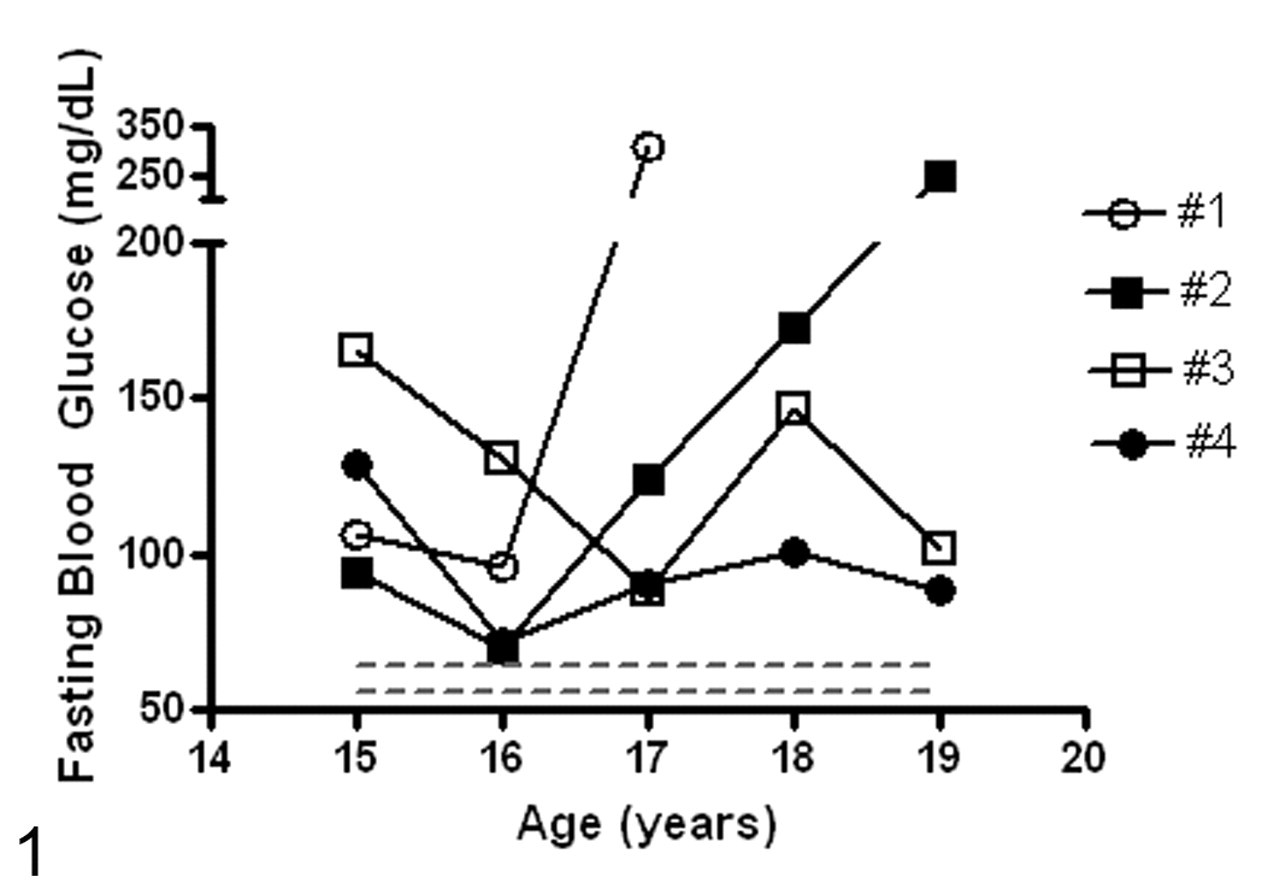
Fasting blood glucose concentrations over time in diabetic vervets (Nos. 1–4). Dotted lines represent the reference range (95% confidence interval) for nondiabetic vervets.
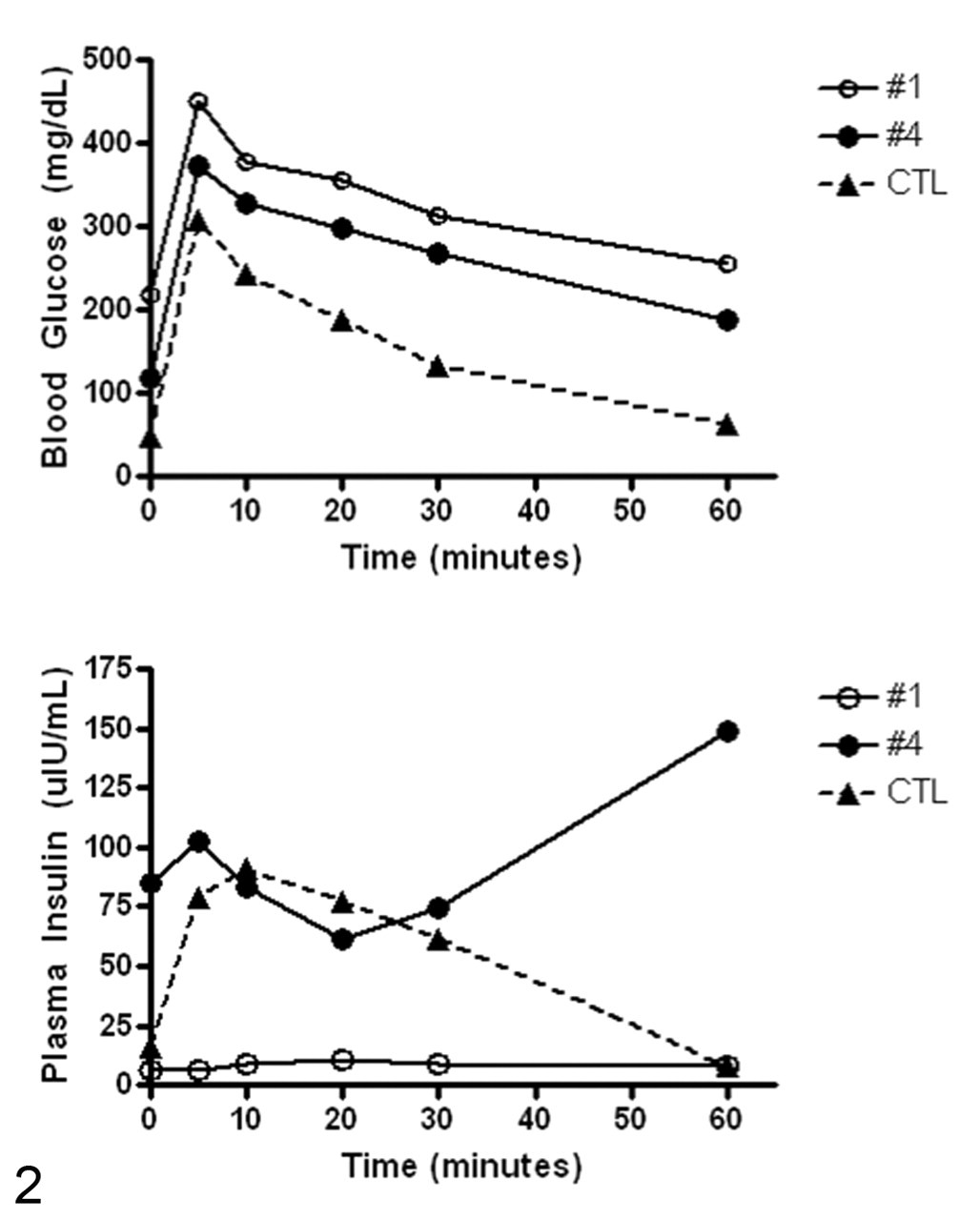
Glucose (top) and insulin (bottom) responses during intravenous glucose tolerance tests in diabetic (Nos. 1 and 4) and nondiabetic (control) vervets.
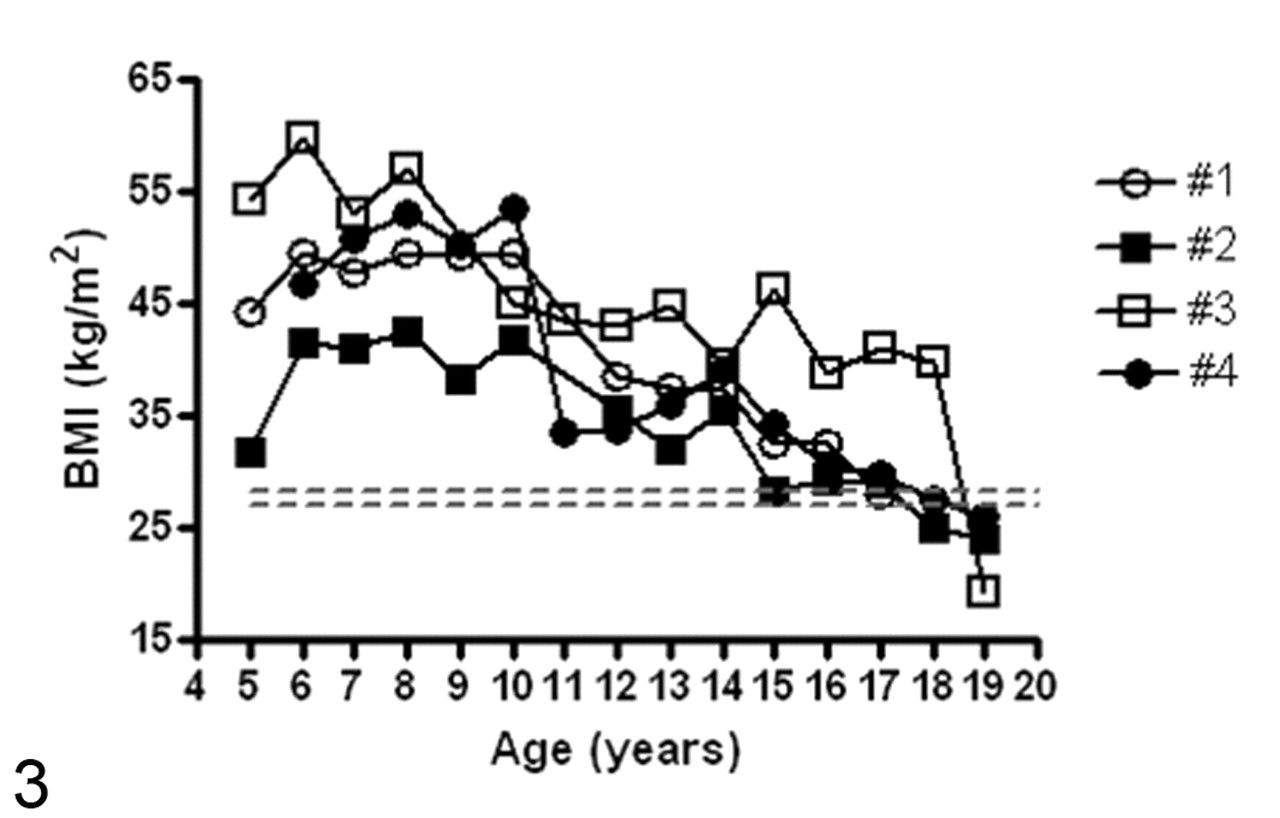
Body mass index of diabetic vervets calculated as follows: weight / crown–rump length2 (kg/cm2). Dotted lines represent the reference range (95% confidence interval) for vervet monkeys.
Pathologic Findings
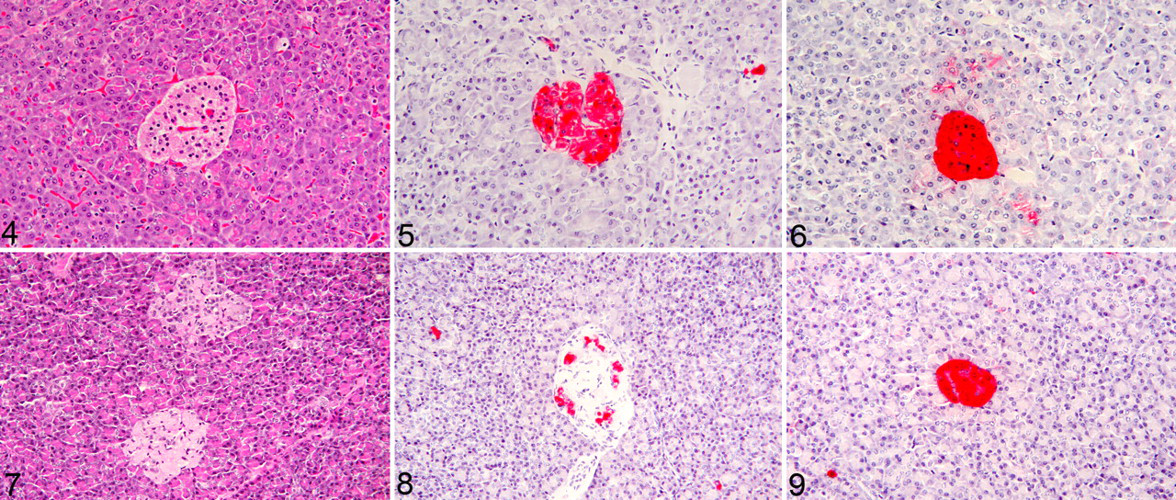
No gross lesions were noted in the pancreata or in any other organs involved in glucose metabolism (liver, fat, muscle, adrenal glands, pituitary gland). Pancreatic histology was normal in animal Nos. 1, 2, and 3 (Fig. 4), and immunohistochemistry for insulin (Fig. 5) and glucagon (Fig. 6) revealed diffuse moderate to intense cytoplasmic staining within pancreatic islet cells, interpreted as normal insulin and glucagon expression. In animal No. 4, multifocal mild to moderate segmental amyloidosis was seen on HE (Fig. 7 ) and confirmed with Congo red staining (not shown). In addition, there was multifocal mild to moderate cytoplasmic staining for insulin (Fig. 8) and diffuse intense cytoplasmic staining for glucagon (Fig. 9). Compared with that of animals Nos. 1–3, insulin staining was reduced in animal No. 4, and glucagon staining was equivalent.
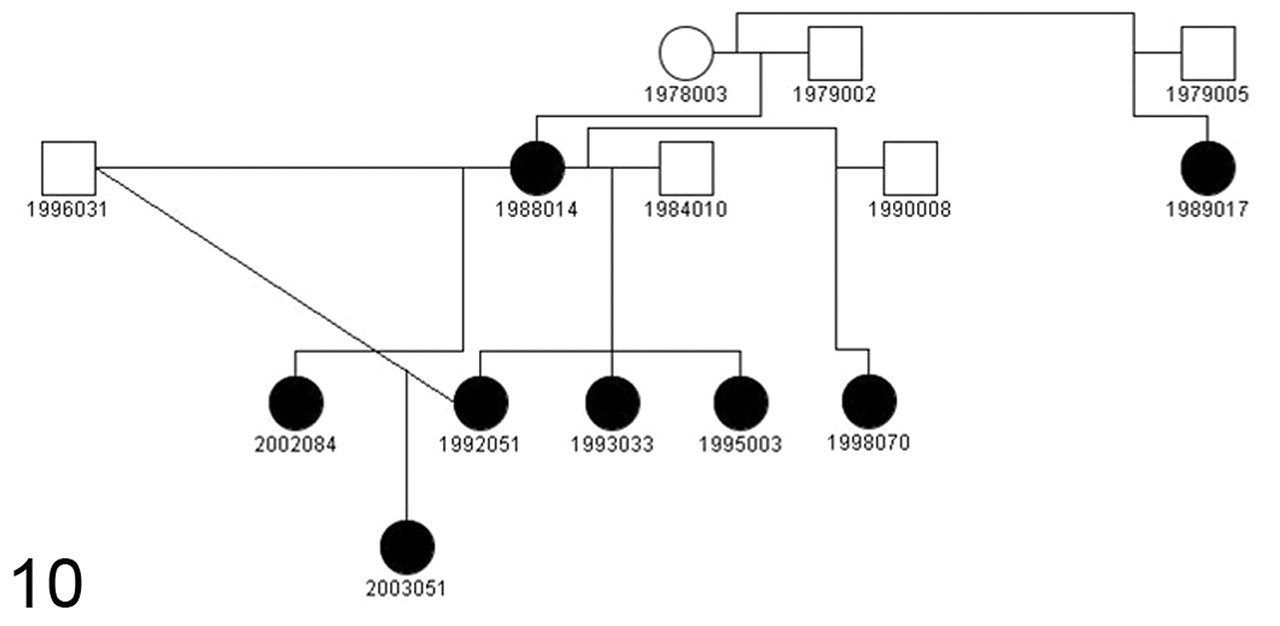
Solid symbols depict a familial pattern of increased blood glucose concentrations (shown in Table 1) of relatives of animal No. 3 (1988014). This animal had 5 offspring from 3 fathers, all of which had elevated fasting blood glucose concentrations. In addition, a grandchild (2003051) and a maternal half sibling (1989017) had elevated fasting blood glucose concentrations. These pedigree data are consistent with either an autosomal dominant or mitochondrial inheritance pattern. Circle = female; square = male; closed symbols = hyperglycemic; open symbols = normoglycemic.
Pedigree Analysis
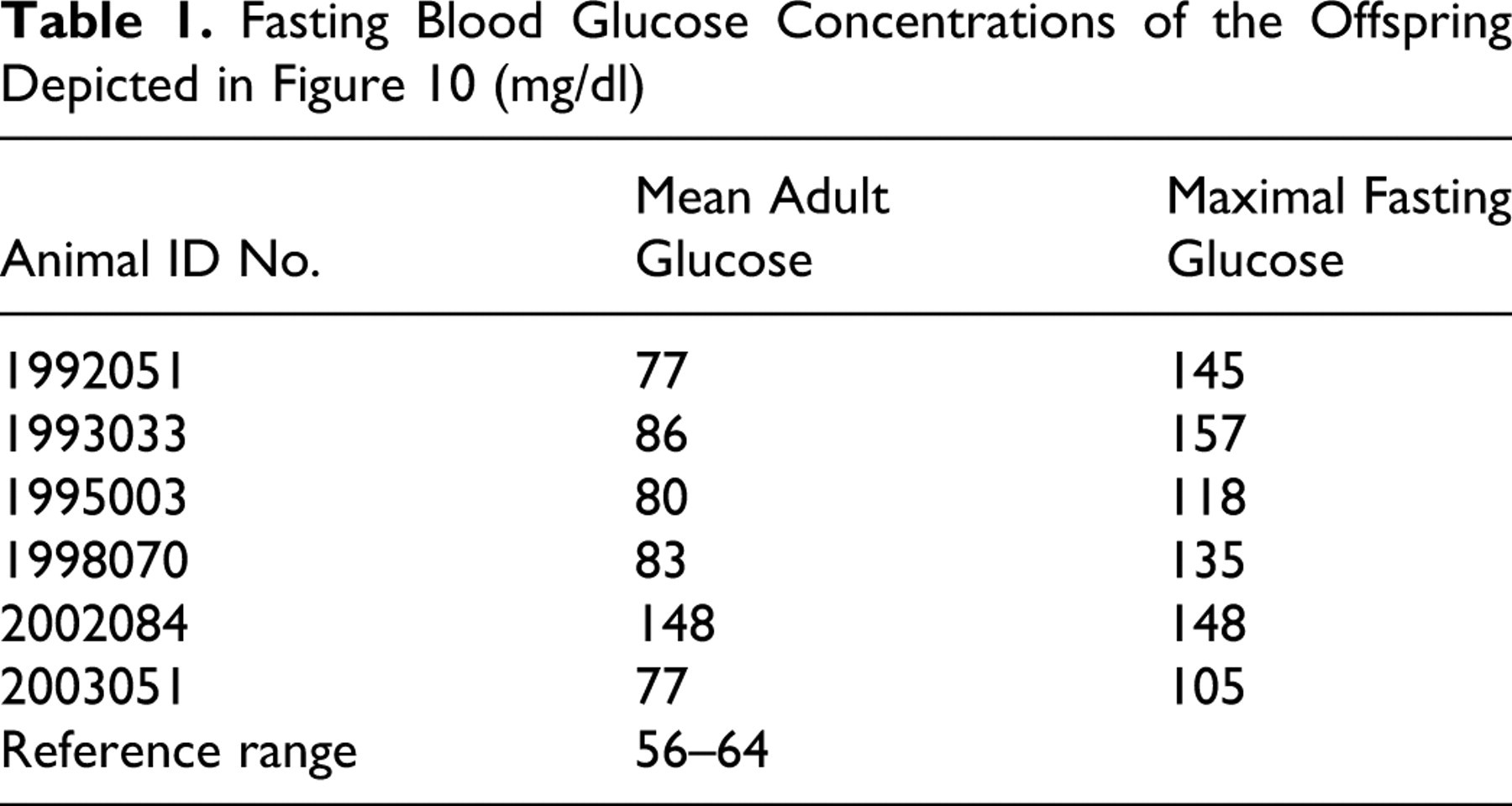
Given that the monogenic forms of DM in humans are passed as autosomal dominant traits, we evaluated blood glucose concentrations of relatives of animal No. 3 (1988014). This animal had 5 offspring from 3 fathers, all of which had elevated fasting blood glucose concentrations (Fig. 10), as did a grandchild (2003051) and a maternal half sibling (1989017). Table 1 shows mean adult fasting blood glucose concentrations and maximum fasting blood glucose concentrations of the offspring. These pedigree data are consistent with either an autosomal dominant or mitochondrial inheritance pattern.
Fasting Blood Glucose Concentrations of the Offspring Depicted in Figure 10 (mg/dl)
Discussion
Clinical diagnosis of DM is not difficult, as blood glucose concentrations are routinely measured, and persistent fasting hyperglycemia is easily identified. However, differentiating the different types of DM can be challenging. As described here, the clinicopathologic presentation of animal No. 4 was similar to cynomolgus macaques with type 2 DM where persistent hyperglycemia, hypertriglyceridemia, islet amyloidosis, and reduced insulin immunostaining are common features. 17 This is in contrast to animal Nos. 1, 2, and 3, all of whom presented in a manner more closely resembling a monogenic form of the disease. With the monogenic forms, islet amyloidosis and hypertriglyceridemia are not pathologic features, and neither was found in any of these 3 cases. Likewise, insulin production as assessed immunohistochemically appeared to be within normal limits, although more definitive studies of long-term insulin production are needed to better characterize this finding. In addition, with the monogenic forms of DM, insulin sensitivity in target tissues is maintained, and at least 2 animals presented here were found to be unusually sensitive to exogenous insulin therapy, as evidenced by repeated rapid-rebounding hypoglycemia posttreatment and, eventually, death subsequent to posttreatment hypoglycemia. Thus, from a clinical perspective these findings indicate that it is important to determine the type of DM in monkeys before initiating therapy. In human medicine, MODY is generally considered easier to manage than the polygenic type 1 and type 2 forms of DM; oral hypoglycemic agents are generally sufficient, although parenteral insulin therapy is sometimes required. 3 Also important to note is that although the animals in our cases were adults at the time of diagnosis, the monogenic forms of DM can present clinically in adults and adolescents. 3
Also supporting our hypothesis that a monogenic form of diabetes may be present, pedigree analysis of animal No. 3 revealed that all 5 of this animal’s offspring were hyperglycemic. MODY and mitochondrial diabetes are highly heritable conditions; therefore, the possibility exists that a monogenic form of DM occurs naturally in vervet monkeys, making them a potential animal model of the human condition. The hallmark of mitochondrial inheritance is that all offspring of affected mothers are affected, whereas no offspring from affected fathers are affected because fathers contribute little to no mitochondria to their offspring. Interesting to note is that in mitochondrial diabetes, gradual aging-associated beta cell dysfunction and reduced insulin production are the main mechanisms by which glucose intolerance develops. 8 This is in contrast to the MODY forms where islet insulin production is not affected. In our presumed monogenic origin cases, insulin production as assessed immunohistochemically appeared to be within normal limits, suggesting that of the different monogenic types, one of the MODY forms is more likely. However, in humans, definitive diagnosis of MODY is made via genetic testing of target genes—including HNF4A (MODY 1), GCK (MODY 2), HNF1A (MODY 3), IPF1 (MODY 4), HNF1B (MODY 5), and NEUROD1 (MODY 6). 3 Similar genetic screening is not currently available for vervet monkeys; however, development of a suitable assay and further characterization of this potential animal model are being explored.
Footnotes
Acknowledgements
This work was funded in part by the Wake Forest University School of Medicine Venture Fund, the Skorich Diabetes Research Fund, the Monty Blackmon Diabetes Research Fund (J.D.W.), T32 RR07009 from the National Institutes of Health / National Center for Research Resources (S.B.G.), and an Animal and Biological Materials Resources grant from the National Center for Research Resources (P40RR019963: original principal investigator, Lynn Fairbanks, University of California, Los Angeles; current principal investigator, Jay Kaplan, Wake Forest). We would also like to thank Drs Nelson Friemer (University of California, Los Angeles) and Jay Kaplan (Wake Forest University School of Medicine) for their input and collaboration and Abdoulaye Diallo for technical support.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
