Abstract
Tularemia, caused by Francisella tularensis, is a sporadic zoonotic disease with the potential to be an agent of biowarfare or bioterrorism. We describe here the gross, histologic, immunohistochemical, and ultrastructural findings in a group of 5 African green monkeys (AGMs) that received an average inhaled dose of 729 colony-forming units of F. tularensis and died or were euthanatized between days 7 and 11 post infection. Clinical changes were evident by 48 hours post infection, and key physiologic abnormalities included increases in body temperature, heart rate, peak cardiac pressure, and mean blood pressure. Prominent gross changes in all cases included numerous pinpoint to 1-cm, well-demarcated, necrotic foci present consistently in the lungs, mediastinal lymph nodes, and spleen but also seen in the heart, mediastinum, diaphragm, liver, urinary bladder, urethra, and mesentery. The lungs, mediastinal lymph nodes, and spleen were most severely affected, with as much as 50% of the tissue replaced by necrotic foci. Histologic changes in all tissues consisted of welldelineated foci of necrosis and neutrophilic and histiocytic inflammation, with varying amounts of hemorrhage, edema, fibrin, and vasculitis. Some lesions were immature pyogranulomas. Strong immunoreactivity was identified primarily within macrophages. Ultrastructurally, bacteria were present within cytoplasmic vacuoles of alveolar macrophages, many of which were degenerate. In summary, AGMs infected with F. tularensis by aerosol develop lethal multisystemic disease that particularly targets the lungs and lymphoid tissues. Thus, AGMs should serve as a suitable and reliable animal model for further studies of tularemia.
Introduction
Francisella tularensis is a small pleomorphic Gram-negative coccobacillus and is the causative agent of tularemia, also known as “rabbit-fever.” Tularemia is a sporadic zoonotic disease in humans, and most cases in the USA are concentrated in south central and western states, primarily in rural areas of Arkansas, Missouri, and Oklahoma. 2 An enzootic life cycle of F. tularensis exists among wildlife, particularly involving rabbits, hares, and rodents. Humans may become infected through arthropod bites, through intact skin by handling infected animal carcasses, by ingesting contaminated food or water, or by inhaling contaminated aerosols. 7 F. tularensis is a highly virulent bacterium, with as few as 10 organisms constituting an infectious dose, and it can survive for long periods in the environment. 7,12 Most naturally acquired human cases can be successfully treated if it is diagnosed early and the patient is maintained on antibiotics for extended periods. 5 Tularemia has been placed on the Centers for Disease Control and Prevention list of Class A biothreat agents.
In natural cases, pneumonic tularemia is often a consequence of inhalation of bacteria. In a biowarfare or bioterrorism event involving F. tularensis, it is very likely that the bacteria would be disseminated via aerosolization, resulting in a high incidence of pneumonic tularemia. 7 Pneumonic tularemia presents some particular challenges relative to other forms (ulceroglandular, glandular, oculoglandular, oropharyngeal, typhoidal, and septic) of the disease. For instance, the antibiotic regimens most applicable for the treatment or postexposure prophylaxis of pneumonic tularemia, especially in a mass casualty event, are not completely certain. Also, vaccines that protect against ingestional or transdermal infection may not protect against high doses of inhaled bacteria. 10 Further, the correlates of immune protection against aerosolized F. tularensis have not been established with certainty. Therefore, appropriate animal models of aerosolized tularemia are required to develop the necessary medical countermeasures.
To date, there are only a few published reports of experiments with tularemia in nonhuman primates, in rhesus macaques (Macaca mulatta), describing the pathology of aerosolized tularemia. 1,6,8,14,15 There is also a single report describing intranasal infection of African green monkeys (AGMs, Chlorocebus aethiops) by F. tularensis. 1 To our knowledge, this is the first report describing the pathology of aerosolized tularemia in AGMs. It is essential to continue to develop and fully characterize inhalational tularemia in AGMs to establish their reliability in mimicking human disease for future studies with F. tularensis, as an alternative to using rhesus macaques, and to provide documentation that will allow regulatory agencies to accept tularemia studies using AGMs in place of rhesus macaques.
Materials and Methods
Five adult male AGMs (Nos. 1–5) were obtained from the United States Army Medical Research Institute of Infectious Diseases (USAMRIID) colony. The 5 cases described herein were part of a larger study consisting of 15 total adult AGMs that were all surgically implanted with a T27-F-1B telemetry system (Data Integrated Scientific Systems, Inc., D.I.S.S., Dexter, MI) and 4 to 6 weeks later were aerosol challenged with either Bacillus anthracis (anthrax), Yersinia pestis (plague), or F. tularensis (tularemia). The continuous monitoring implantable telemetry device displayed parameters using a 5-lead system and included continuous readings on temperature, heart rate, ventricular and aortic pressures, single-lead electrocardiogram, and intrathoracic pressure values.
The monkeys were exposed to F. tularensis spp. tularensis SCHU S4, Type A strain, reproduced at USAMRIID (FT4, third passage) in a class III biologic safety cabinet in a head-only chamber. 9 The cases received an average dose of 729 cfu (range 177–1,433 cfu) that was calculated based on minute-volume, and whole-body plethysmography was performed to determine minute-volume for each animal. 9 All exposed animals either died or were euthanatized between 7 and 11 days post exposure. The high- and low-dose animals were found dead on days 7 and 8, respectively, and no relationship between dose and time to death was evident in this small group (Table 1).
African green monkeys aerosolized with Francisella tularensis spp. tularensis SCHU S4, Type A strain.
Complete necropsies were performed on each animal in a biosafety level 3 necropsy facility. A complete set of tissues were collected from each animal for histopathology and were immersion fixed in 10% neutral buffered formalin and held in biocontainment for 21 days. Tissues for histopathology underwent routine histologic processing, were embedded in paraffin, sectioned, and were stained with hematoxylin and eosin. Immunoelectron microscopy was performed on sections of lung from case No. 2, and immunohistochemistry was performed on all tissue sections of all cases using a mouse monoclonal antibody (1 : 1,200 dilution) that recognizes the lipopolysaccharide of F. tularensis (Meridian Life Science, Inc, Cincinnati, OH) and a commercial immunoperoxidase kit (EnVision System, Dako Corp., Carpinteria, CA). After pretreatment with proteinase K for 6 minutes at room temperature, primary and secondary antibodies were applied and the slides were incubated with substrate-chromagen solution according to the manufacturer's recommendations. Sections were counterstained with hematoxylin.
Select sections of lung from AGM case No. 2 were processed for transmission electron microscopy and immunoelectron microscopy from formalin-fixed tissue removed from a paraffin block. Briefly, the sections were dehydrated and placed in a 2 : 1 solution of 70% ethanol : London resin (LR) White acrylic resin followed by immersion in a solution of 1 part 70% ethanol and 2 parts LR White acrylic resin. The specimens were incubated in LR White for 48 hours, sectioned on a Leica Ultracut UCT ultramicrotome, and collected on carbon-stabilized formvar-coated nickel grids. Grids were analyzed in a JEOL 1011 transmission electron microscope at 80 KV. Additional staining for immunoelectron microscopy was performed by floating the grids (section side down) on drops of 1% bovine serum albumin in phosphate-buffered saline for 5 minutes, followed by blocking in 4% normal goat serum in phosphate-buffered saline for 20 minutes at room temperature. The grids were then rinsed by floating on buffer drops then floated on drops containing a 1 : 50 dilution of the primary antibody for 1 hour at room temperature. The grids were then rinsed as before and incubated on drops containing a 1 : 25 dilution of goat anti-mouse IgM conjugated to 10-nm gold spheres for 1 hour at room temperature followed by another buffer rinse. These were placed onto drops of 1% gluteraldehyde stained in uranyl acetate followed by lead citrate. The grids were rinsed in distilled water, air dried, and analyzed in a JEOL 1011 transmission electron microscope at 80 KV.
Results
The initial changes in clinical parameters were noted 48 to 72 hours after exposure. The earliest changes evident were physiologic parameters that included increased body temperature, increased heart rate, increased peak cardiac pressures, and increased mean blood pressures. Additional alterations occurred in systemic blood pressure, electrocardiogram parameters, and intrathoracic pressures. Detailed analysis of the clinical and telemetry data will be the subject of a subsequent report (B. Purcell et al., manuscript in preparation). Decreased activity and altered resting postures were the initial behavioral changes seen in the monkeys. These often lagged behind the measured deviations in telemetry by 1 or 2 days.
The main gross pathologic feature in all cases was the presence of necrotic foci in multiple tissues, including lung, spleen, tracheobronchial and mediastinal lymph nodes (Figs. 1–4), and liver. These lesions were single or multiple, discrete or coalescing, randomly distributed foci. The lesions were variably sized (pinpoint to 1 cm), well demarcated, oval or irregularly oval, pale, raised, and dome-shaped or slightly raised with flattened surfaces.
Lung; African green monkey No. 4. Within each lung lobe there are multiple and coalescing white raised lesions measuring 0.1 to 1 cm. The lobes are noncollapsing with rounded edges, and the lung parenchyma between the lesions is dark red, indicating marked congestion and edema. Bar = 1 cm.

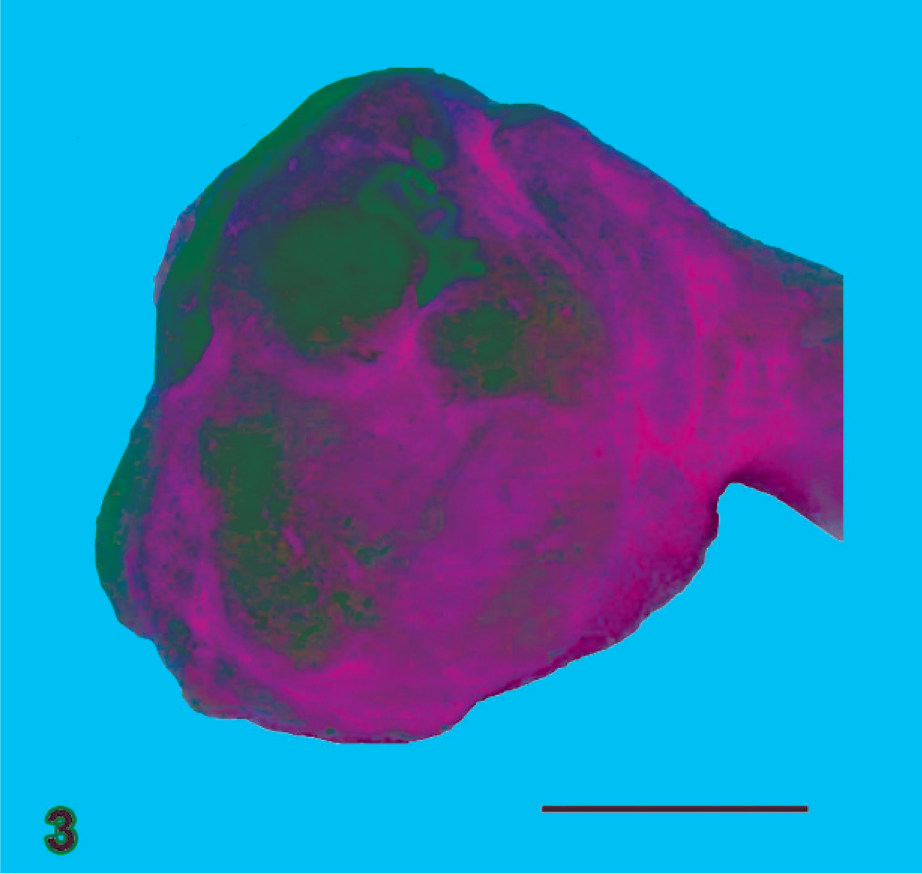
Lung; African green monkey No. 3. Lung with cut surface showing the extent of the lesions within the pulmonary parenchyma. Additionally, there is a milky and somewhat granular appearance to the pleural surface (pleuritis). Bar = 0.5 cm.
Tracheobronchial lymph node; African green monkey No. 2. Multiple irregularly oval raised lesions are evident within the lymph node. Bar = 0.3 cm.
Spleen; African green monkey No. 1. The spleen is showered with 0.1 to 0.2 cm diameter, raised, white lesions. Splenic borders are rounded, indicating marked congestion. Bar = 1 cm.
All lung lobes contained many generalized and randomly distributed lesions that were up to 1 cm, coalescing, white, and raised (both domed and flattened) that distorted the surface architecture and extended into the pulmonary parenchyma on cut surface. The normally smooth pleural surface of the lung appeared dry and granular at the site of the lesions (pleuritis). Lungs were noncollapsing and deeply red between single and coalesced lesions (edema and congestion and/or hemorrhage).
The spleen typically contained myriad small (0.1–0.5 cm), white, raised pinpoint lesions that in some areas were coalescing. The edges of the spleen were markedly rounded, indicating congestion. Gross liver lesions occurred in most animals as single or a few scattered foci. Similar lesions were also present, but less frequently, in the trachea, heart, mediastinum, diaphragm, mesentery, and epithelial surface of the urinary bladder and urethra.
Histologically, the lesions in multiple organs invariably consisted of well-delineated areas of parenchymal degeneration and necrosis with loss of architecture; large numbers of macrophages and viable and degenerate neutrophils; and varying amounts of hemorrhage, edema, fibrin, and vasculitis. Many lesions were immature pyogranulomas consisting of central necrosis with cellular debris and viable and degenerate neutrophils, surrounded by many macrophages with abundant microvacuolated cytoplasm. However, multinucleate giant cells, lymphocytes, and fibroblasts were not present. Histologic lesions were particularly prominent in the lungs and lymphoid tissues. Multifocally within the lung, the airways, alveolar septa, alveolar spaces, and pleura were replaced by cellular and karyorrhectic debris (necrosis), fibrin, hemorrhage, edema, many viable and degenerate neutrophils, and large numbers of macrophages with abundant cytoplasm (Fig. 5). An association of these lesions with small and medium airways (bronchopneumonia) was apparent in less affected areas (Fig. 6). However, as the lesions became more extensive, the association with the airway was obscured. Entrapped blood vessels were characterized by disrupted tunica intima and expansion of the tunica media and tunica adventitia with necrotic debris, degenerate neutrophils, macrophages, fibrin, edema, and hemorrhage (necrotizing vasculitis) (Fig. 7). Lesions were noted in the upper respiratory tract (tracheitis and laryngotracheitis) in 4 cases, consisting of degeneration, necrosis, and loss of surface epithelium and expansion of the subepithelium with eosinophilic cellular and karyorrhectic debris forming domed shaped lesions (Fig. 8). Lung parenchyma surrounding areas of necrosis was moderately to markedly congested, and alveolar spaces were often flooded with edema or hemorrhage. There were also increased numbers of alveolar macrophages that contained abundant foamy cytoplasm. Multifocally, there was marked pleuritis characterized by necrotizing and pyogranulomatous inflammation, edema, and occasionally hemorrhage. Similarly, the splenic architecture, including the white pulp, red pulp, and capsule, was disrupted, effaced, and replaced by necrosis and macrophages with abundant cytoplasm and few degenerate neutrophils (Fig. 9). There was diffuse moderate to marked lymphoid depletion of the white pulp. The less severely affected splenic red pulp was markedly congested. The mediastinal lymph nodes contained multiple necrotizing lesions that disrupted 50% or more of the normal architecture (Fig. 10). There was complete necrosis of some mediastinal lymph nodes, with only remnants of normal architecture remaining. Other mediastinal lymph nodes were draining inflammation from the lungs and surrounding tissues and contained large numbers of sinus and subcapsular macrophages, accompanied by neutrophils, edema, fibrin, and hemorrhage. Lymphoid depletion was a prominent feature of all mediastinal lymph nodes. In the liver, few to many randomly scattered foci of necrosis and inflammation disrupted the architecture of hepatic cords. In addition to these changes, tonsillitis, myocarditis, mediastinitis, cystitis, urethritis, enteritis, peritonitis, lymphadenitis, adrenalitis, and osteomyelitis were variably present. F. tularensis organisms were not identified on hematoxylin and eosin–stained sections.
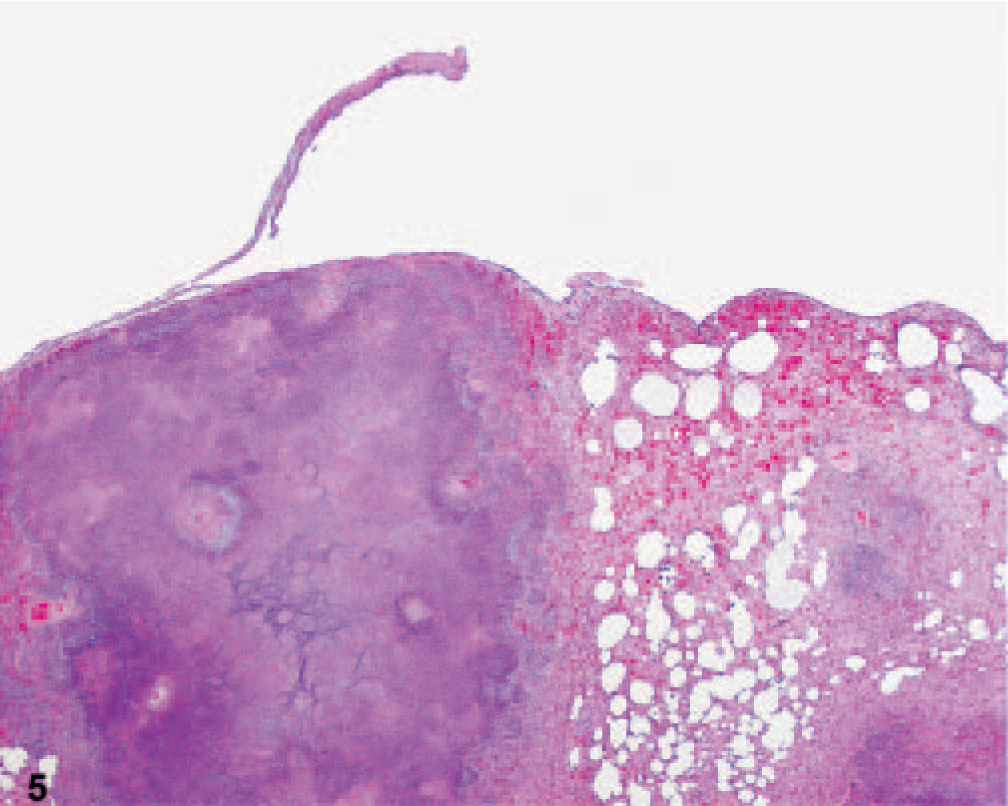
Lung; African green monkey No. 5. Multiple pulmonary necrotic lesions replace normal parenchma. The expanded pleura (pleuritis) has separated from the lung at the site of a large lesion. There are congestion, hemorrhage, and edema surrounding the lesions. HE.
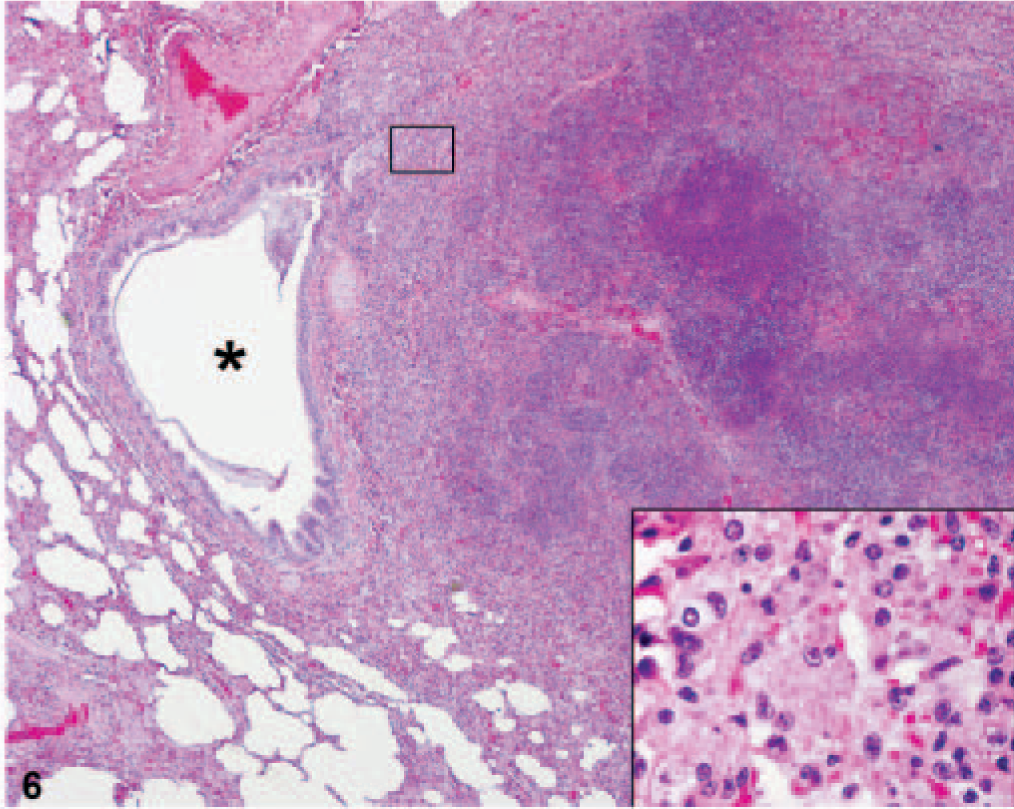
Lung; African green monkey No. 5. A pulmonary lesion associated with a medium-sized airway is composed of large numbers of macrophages with microvacuolated cytoplasm (insert) surrounding central necrosis. Note the deviation in the airway space (asterisk) due to the expanding lesion. HE.
Lung, blood vessels; African green monkey No. 1. Necrotizing vasculitis of blood vessels within a pulmonary lesion. Note the disrupted tunica intima. The tunica media and tunica adventitia are expanded by cellular and karyorrhectic debris, degenerate neutrophils, microvacuolated macrophages, fibrin, edema, and hemorrhage. Surrounding parenchyma is obscured by necrosis, inflammation, edema, and hemorrhage. HE.
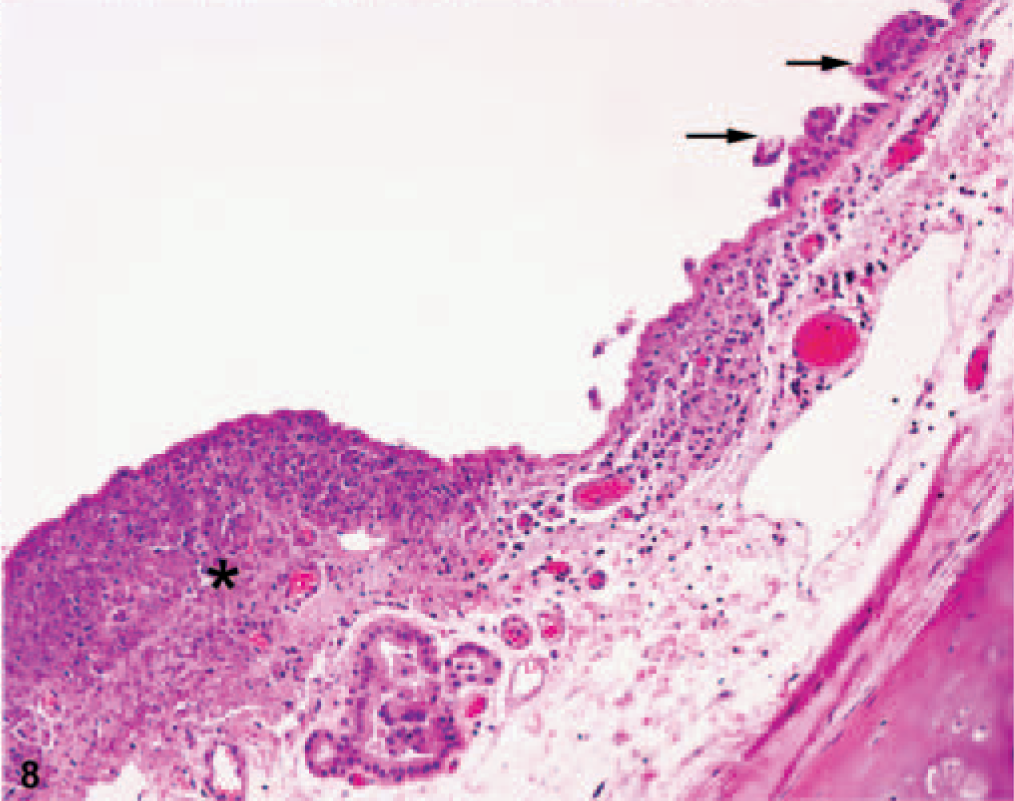
Trachea; African green monkey No. 1. Cellular and karyorrhectic debris expand the subepithelium to form a dome-shaped lesion (asterisk). There are degeneration and loss of respiratory epithelium (arrows). The subepithelium is also expanded by congestion, lymphatic ectasia, and increased clear space (edema). HE.
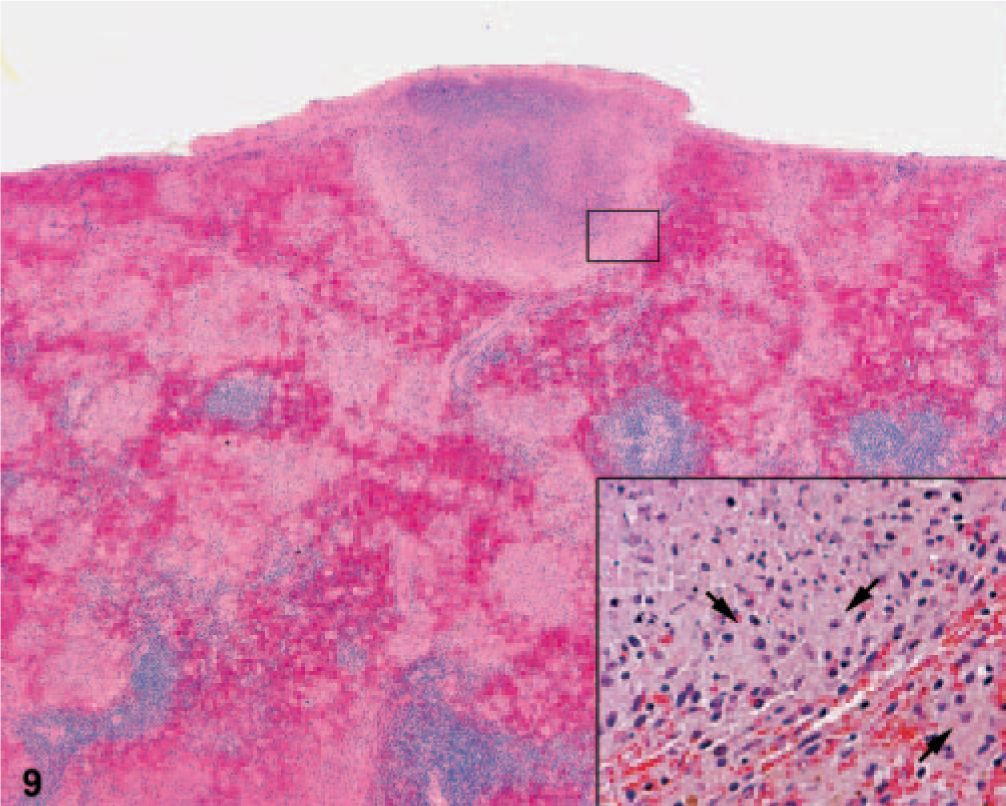
Spleen; African green monkey No. 4. A splenic lesion elevates the surface and capsule and is composed of central necrosis and a few degenerate neutrophils rimmed by macrophages (arrows, insert). Smaller pale lesions of the same are present throughout the parenchyma. There is diffuse paucity of white pulp, and the remaining red pulp is markedly congested. HE.
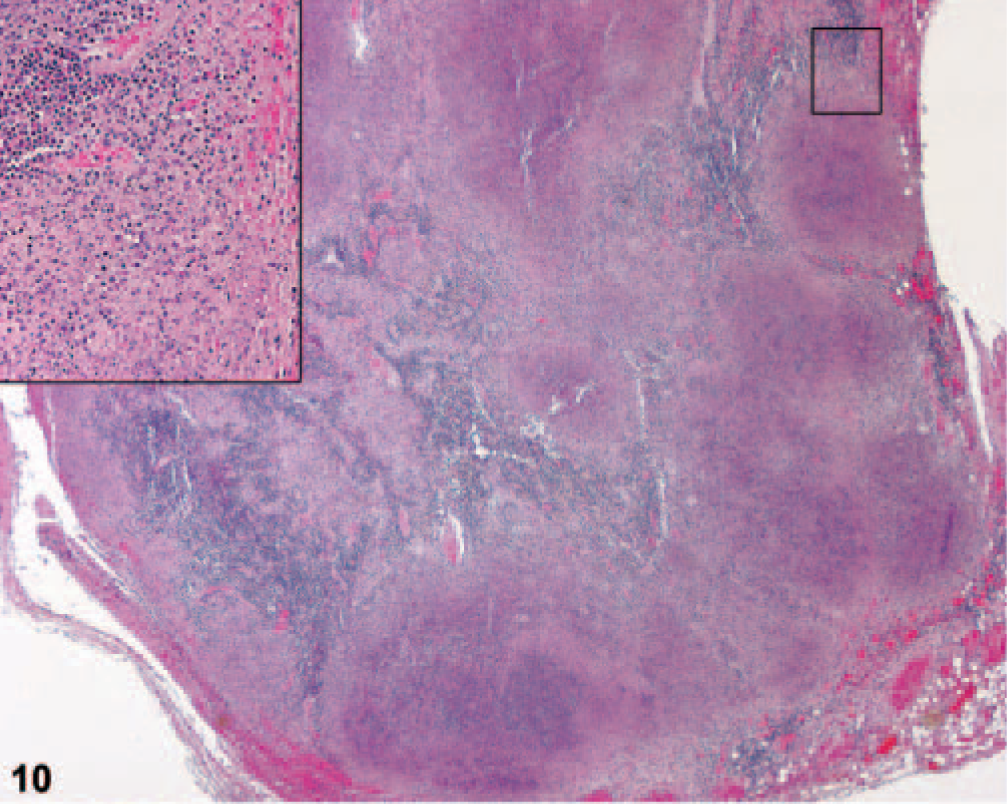
Lymph node, mediastinal; African green monkey No. 4. There are multiple and coalescing necrotic lesions replacing 80% of the normal architecture. There is marked lymphoid depletion. There is multifocal hemorrhage in the subcapsular sinus and surrounding mediastinum. HE.
Immunohistochemical labeling demonstrated strong F. tularensis immunoreactivity in the cytoplasm of multiple cell types but was especially prominent within macrophages of the tonsil; mandibular, mediastinal, mesenteric, axillary, and inguinal lymph nodes; and in alveolar macrophages. Strong F. tularensis immunoreactivity was also observed in the following cell types: pleural mesothelial cells; respiratory epithelial cells of the larynx, trachea, bronchi, and bronchioles (Fig. 11); epicardial cells; degenerating hepatocytes; reticuloendothelial cells and macrophages in the spleen; glomerular mesangial cells and/or endothelial cells; a variety of cell types in the bone marrow; cortical cells of the adrenal gland; the epithelium of the urinary bladder and urethra; and neutrophils and macrophages infiltrating multiple tissues (Fig. 12). In addition, granular or amorphous extracellular antigen was evident in areas of necrosis or pyogranulomatous inflammation in multiple tissues.
Trachea; African green monkey No. 1. There is strong tularemia immunoreactivity of the respiratory epithelium (insert) and amorphous extracellular antigen of the domed necrotic lesion. Immunohistochemistry, mouse monoclonal antibody to tularemia (#927), and hematoxylin counterstain.
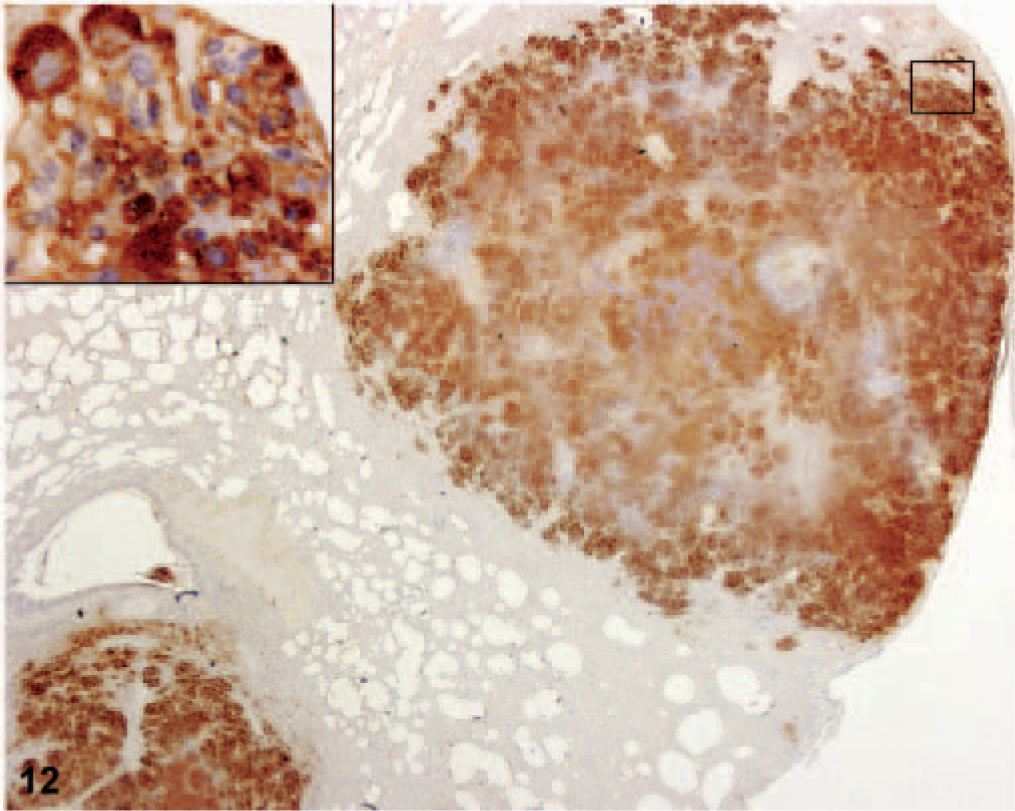
Lung; African green monkey No. 5. There is strong tularemia immunoreactivity of the macrophages and neutrophils (insert) and amorphous extracellular antigen of the necrotic lesions. Immunohistochemistry, mouse monoclonal antibody to tularemia (#927), and hematoxylin counterstain.
Electron microscopy was performed on the lung of one AGM. Organisms were readily identified within membrane-bound vacuoles within the cytoplasm of alveolar macrophages adjacent to the nucleus. The bacteria varied in shape but were generally oval or elongate and measured 0.4 to 0.5 µm in width (Fig. 13). Bacteria contained a thin cell wall, a pale central cytoplasm, and a darker rim of cytoplasm near the cell wall (Figs. 13, 14). An outer membrane was present in some organisms and appeared as an irregular or wavy membrane loosely surrounding the organism (Fig. 13). Many cells containing internal bacilli were seen in various stages of degeneration characterized by swollen irregular mitochondria with faded or absent internal cristae, cytoplasmic vacuoles, enlarged membrane-bound vacuoles with indistinct borders (with or without bacteria), disrupted smooth and rough endoplasmic reticulum, disrupted Golgi apparatus, and disrupted cell membranes. Because severe degeneration and necrosis often hindered recognition of these small bacteria, we used immunoelectron microscopy to distinguish organisms. The identification of F. tularensis was made with confidence when the bacterial cell wall contained black grains of gold particles that were evenly distributed over the entire bacterium (Fig. 14).
Lung; African green monkey No. 2. Ultrastructural view of an alveolar macrophage within an alveolus. The cell has several cytoplasmic vacuoles (arrow) surrounding an irregular cell nucleus with peripheralized heterochromatin. Bacteria measuring 0.4 to 0.5 µm diameter are present in most vacuoles. Some cytoplasmic vacuoles are losing structural integrity, and some bacteria are irregularly shaped. Most bacteria have darker peripheralized cytoplasm, and one bacterium has an irregular enveloping membrane (inset). Transmission electron microscopy. Bar = 2 µm.
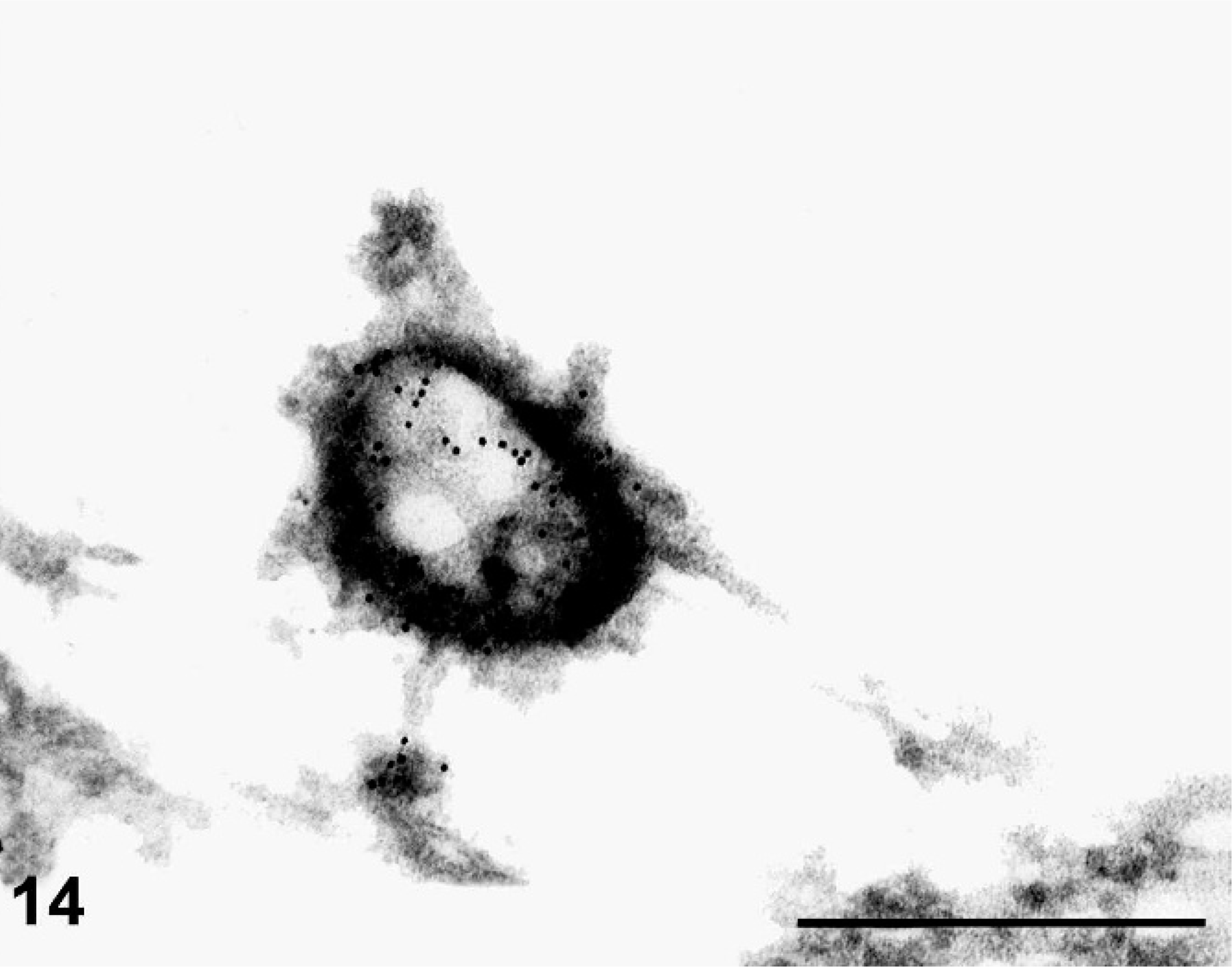
Lung; African green monkey No. 2. Immunoelectron micrograph of tularemia. Note the black grains of gold particles attached to the organism's wall. The central light and peripheral dark cytoplasm of the bacteria is clearly represented. Immunoelectron microscopy, mouse monoclonal antibody to tularemia (#927) 1 : 200, gold 1 : 25. Bar = 500 nm.
Discussion
This report describes the gross, histopathologic, and ultrastructural changes in 5 AGMs with lethal inhalational tularemia. The monkeys in this study exhibited a number of similarities to human tularemia, in particular with regard to the pneumonic form of disease. The key pathologic features of inhalational tularemia in these monkeys were numerous and widespread necrotizing pyogranulomatous lesions that especially targeted the lungs and lymphoid tissues. Bacteria were present in many cell types but were most readily present in alveolar macrophages, as well as macrophages in other tissues. Ultrastructural features included the presence of bacteria within cytoplasmic vacuoles that were located adjacent to the nucleus (Francisella-containing vacuoles), 4 as well as degenerative changes in infected cells. In addition, all 5 AGMs in our study had significant pleuritis associated with the lung lesions, representing another feature in common with the human counterpart, and one also shared by the rhesus macaque model. 7,8 Likewise, our finding that most lesions were characterized by necrosis combined with neutrophilic and histiocytic inflammation progressing to form immature pyogranulomas is consistent with histologic findings in most forms of human tularemia, except the typhoidal form.
Two differences between the disease in AGMs and that in humans were noted. We did not observe granulomas associated with epithelioid macrophages and multinucleated giant cells in the target organs of AGMs. Possibly, the monkeys in this study succumbed to disease before such lesions had sufficient time to fully develop. Another exception is that the kidney is a reported target of human tularemia, yet none of our cases displayed gross or histologic changes in the kidney. We did observe, however, that most kidneys had positive immunohistochemistry labeling (for bacterial antigen) within glomerular mesangial and/or endothelial cells. The importance of positive staining in the absence of pathologic changes in the tissue is unclear. One report did describe histologic changes in the renal glomeruli of rhesus macaques. 8 It is uncertain if the renal lesions provide a significant contribution to the pathogenesis in human tularemia in the face of the significant bacterial burden and lesions in the pulmonary and lymphoid systems, as well as other key organs.
Our finding that F. tularensis was found in abundance in macrophages in the lungs, lymphoid tissues, and other tissues is a feature of the disease in AGMs with potentially critical significance to understanding the human condition. The ability of F. tularensis to infect macrophages, evade the immune system by preventing phagolysosome fusion, and then replicate in these cells is considered a key aspect of its pathogenesis. 3 Infected macrophages also appear to be important in the pathogenesis of tularemia in that they secrete both proinflammatory and anti-inflammatory cytokines, 13 although a full understanding of the host and pathogen factors that operate within infected macrophages is lacking. Nonetheless, the AGM may provide a useful experimental model for investigating the nature of host-pathogen interactions in macrophages, as well as other aspects involved in the pathogenesis of tularemia.
There are limited numbers of documented reports of experimental aerosolized tularemia in rhesus macaques, mostly dating back to the 1960s. These reports describe acute bronchiolitis progressing to bronchopneumonia, lymphadenitis, splenitis, and hepatitis with neutrophilic and histiocytic inflammation with intrahistiocytic bacteria. 6,8,14,15 Such features are similar to those seen in the AGMs of our study. Rhesus macaques have become increasingly expensive and limited in supply in recent years, which limits their usefulness as a model of tularemia. The most recent report of tularemia in nonhuman primates documents an epizootic of tularemia in a group of cynomolgus macaques (Macaca fascicularis) that contracted oropharyngeal tularemia by ingesting contaminated food and water. The tularemic lesions were present in the oral mucosa, tongue, lungs, liver, spleen, and lymph nodes consistent with ingestional human tularemia. 11 To date, there have been no published studies characterizing the pathology of aerosolized tularemia in AGMs. Therefore, the AGM model of inhalational tularemia we describe appears to share important features of the human condition and thus should provide a useful nonhuman primate model for future research. It is essential to continue to characterize the clinical and pathologic changes that occur in animal models of tularemia, as well as the molecular mechanisms involved in this disease. Future experimental studies to develop early preventive regimens, early diagnostic procedures (identifying target organs for sampling), and clinical algorithms to optimize treatment efficacy will depend on reliable animal models that consistently mimic human disease. Likewise, relevant animal models will also be necessary to investigate avenues of altering pathogen and host factors, to include novel antibiotic therapies, immunomodulators, or inflammatory inhibitors. In that light, the AGMs reported here displayed consistent gross and histologic lesions after exposure to aerosolized F. tularensis with important parallels to human tularemia. This species of nonhuman primate may serve as a suitable and reliable animal model for further studies with F. tularensis.
Footnotes
Acknowledgements
We gratefully acknowledge P. Fogle, J. Brubaker, N. Davis, G. Krietz, K. Kuehl, and C. Mech for excellent necropsy, histology, immunohistochemistry, and electron microscopy technical assistance; Dr. L. Asher for transmission electron microscopy and immunoelectron microscopy; and Larry Ostby for visual assistance. All work with F. tularensis was performed in a biosafety level-3 laboratory at USAMRIID. Research was conducted in compliance with the Animal Welfare Act and other federal statutes and regulations relating to animals and experiments involving animals and adheres to principles stated in the Guide for the Care and Use of Laboratory Animals, National Research Council, 1996 (27a). The facility where this research was conducted is fully accredited by the Association for Assessment and Accreditation of Laboratory Animal Care International. The research described herein was sponsored by the Military Infectious Disease Research Program, U.S. Army Medical and Material Command, DHS/NBACC no.: N836-007, USAMRIID Project Plan no. 92401 and NIAID Project Plan No. Y1-AI-6139-01, USAMRIID # A120-D.3, MRMC 05-0005. Opinions, interpretations, conclusions, and recommendations are ours and are not necessarily endorsed by the U.S. Army or the Department of Defense.
