Abstract
This report describes the signalment, clinical findings, gross appearance, histological and immunohistochemical characteristics, and behavior of 39 cases of canine synovial myxoma. Large-breed middle-aged dogs—especially, Doberman Pinschers and Labrador Retrievers—were most commonly affected. The stifle and digit were the most common sites. Grossly, the tumors were composed of gelatinous nodules that often filled the joint cavity and exuded viscous fluid on cut section. In 12 cases (31%), radiographic bony lysis or grossly invasive growth was noted clinically. Histologically, the nodules were sparsely cellular and composed of stellate to spindle cells suspended in an abundant myxomatous matrix. By immunohistochemistry, the cells were positive for vimentin, heat shock protein 25, and cadherin 11 and negative for cytokeratin and S100 protein; some cells (20–40%) were positive for CD18. Affected dogs had long survival times (average, 2.5 years), even with incomplete excision of the tumor. Three cases had local recurrence, but none metastasized or directly resulted in death. Canine synovial myxoma is a histologically distinctive tumor with a good prognosis.
The synovial membrane is composed of multiple cell types that can be differentiated by immunohistochemistry or electron microscopy but not by light microscopy. 3,15 The cells are loosely arranged in a membrane 1 to 3 cell layers thick with no tight junctions, desmosomes, or basement membrane. 2 Synoviocytes have been classically divided into 2 cell types: type A, phagocytic macrophage/dendritic cells; and type B, fibroblast-like synoviocytes. Type A synoviocytes are of hematopoietic origin, and they express CD18. Type B synoviocytes produce synovial fluid and express vimentin, cadherin 11, and heat shock protein 25 (HSP25). 13,15 Type C synoviocytes have recently been described as transitional or stem cell–like with properties of type A and B cells. 15
The 3 most common tumors of the canine synovium are histiocytic sarcoma, synovial cell sarcoma, and synovial myxoma. 5 All can cause lameness with bony lysis and proliferation on both sides of the joint. Histiocytic sarcoma is an aggressive malignancy of dendritic cell origin that metastasizes widely; it can be differentiated from synovial cell sarcoma by CD18 expression. Synovial cell sarcoma is a locally aggressive tumor that less commonly metastasizes and often contains a small proportion of cells that express cytokeratin. Synovial myxoma is a benign but sometimes infiltrative tumor that consists of myxomatous nodules. 5
Eleven cases of synovial myxoma have been reported in dogs. 4 –6,10 Ten of those cases exhibited benign behavior with long survival times and no metastasis; 1 case was an incidental finding at necropsy. 4 –6,10 Five of the 11 cases involved Doberman Pinschers. 4,5,10 The purpose of this report is to describe the signalment, clinical findings, gross appearance, histological and immunohistochemical characteristics, and behavior of 39 cases of canine synovial myxoma. Six cases were included in a previous report, 5 but follow-up information is included here.
Materials and Methods
Cases were selected by histologic appearance and synovial location from the biopsy services of the University of Pennsylvania School of Veterinary Medicine (26 cases, 1986–2000), University of Tennessee College of Veterinary Medicine (5 cases, 2004–2009), University of Georgia College of Veterinary Medicine (5 cases, 2004–2007), University of Wisconsin School of Veterinary Medicine (2 cases, 1997–1999), and Antech Diagnostics (1 case, 2007). When available, medical records were reviewed for breed, sex, age, weight, tumor site, duration of clinical signs, treatment, and outcome.
Immunohistochemistry was performed on 11 synovial myxomas for cytokeratin (AE1/AE3, Dako, Carpinteria, CA), vimentin (V9 Biogenex, San Ramon, CA), CD18 (Ca16.3c10, Peter Moore, University of California, Davis), S100 protein (Dako), HSP25 (Stressgen Bioreagents, Ann Arbor, MI), cadherin 11 (Invitrogen, Camarillo, CA), and proliferating cell nuclear antigen (PCNA) (PC10 Biogenex, San Ramon, CA) on formalin-fixed, paraffin-embedded 5-μm sections using an Autostainer Immunostaining System (Dako). Antigen retrieval was performed for cytokeratin and CD18 (using proteinase K [Dako] at room temperature for 5 minutes), vimentin (using heat [95°C] for 25 minutes), and cadherin 11 and HSP25 (using heat [95°C] for 40 minutes). Antigen retrieval was not used for S100 protein or PCNA. Positive control tissues for cadherin 11 and HSP25 were mouse urinary bladder and canine synovium, respectively. A horseradish peroxidase–labeled polymer system (Envision+, Dako), with diaminobenzidine (DAB, Dako) as chromogen, and Harris hematoxylin counterstain were applied to all slides.
Three myxosarcomas from nonsynovial sites (subcutis of the digit, thorax, and abdomen), 2 synovial cell sarcomas, and 1 synovial histiocytic sarcoma were also subjected to immunohistochemistry for CD18, HSP25, and cadherin 11.
Results
The duration of clinical signs—typically, lameness with or without joint swelling—was recorded in 24 cases; the range was 1 week to 3 years, with a 6-month average. The initial clinical diagnosis was often degenerative joint disease. Common radiographic findings included soft tissue swelling, periarticular osteophytes, and in 8 cases (21%), well-circumscribed areas of lysis on both sides of the joint (Fig. 1). In 5 cases, the masses were noted to be grossly invasive; in 1 case (No. 12), bony lysis and gross invasiveness were noted before amputation.
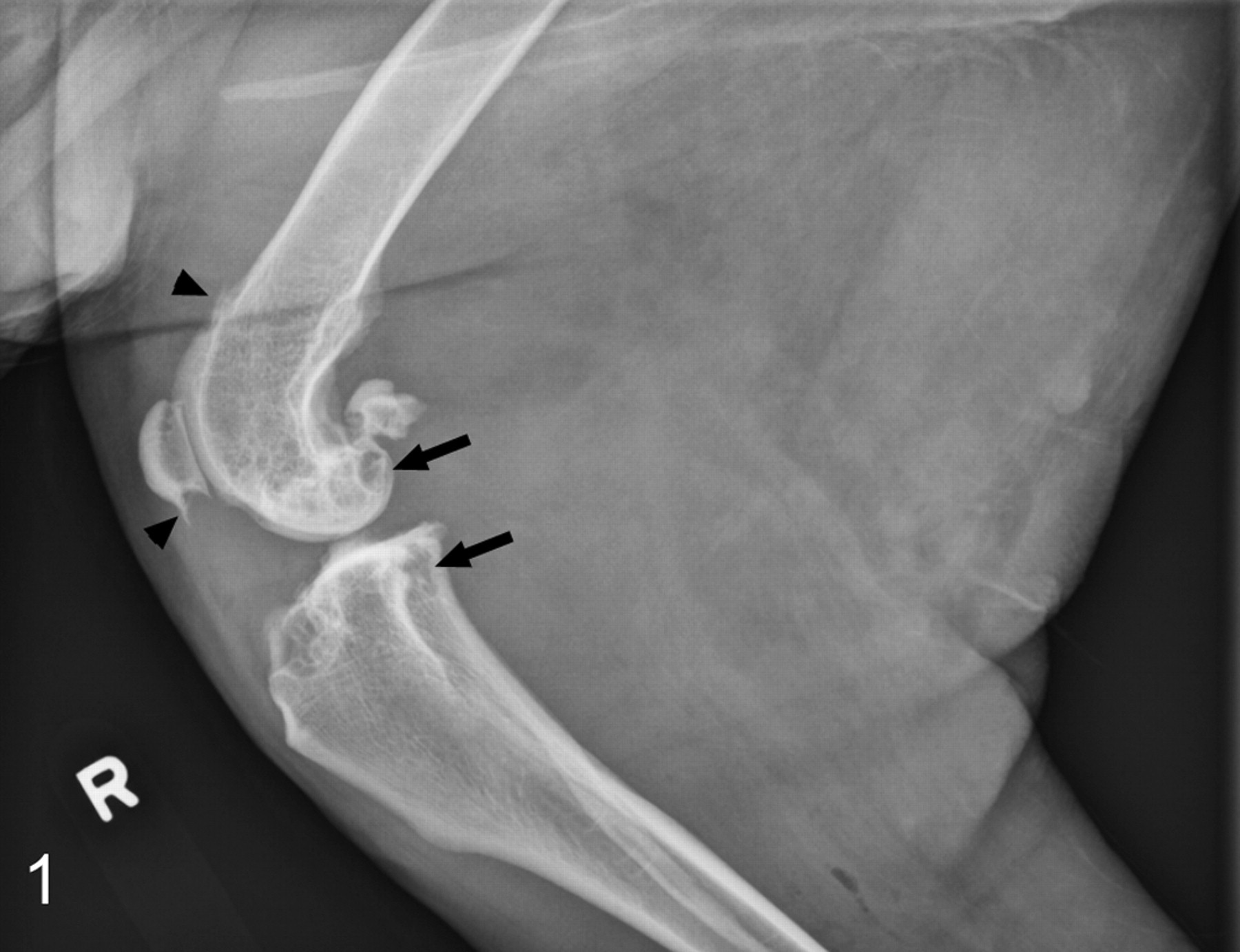
Stifle; synovial myxoma; dog, case No. 28. Radiograph showing well-circumscribed areas of lysis (arrows) and periarticular osteophytes (arrowheads) of the distal femur, proximal tibia, patella, and fabellae.
Grossly, the synovial myxomas were composed of soft white translucent nodules and pockets of viscous fluid. The nodules often lined the entire inner surface of the joint capsule (Fig. 2) and exuded abundant viscous fluid on cut section. Histologically, the tumors were characterized by variably sized, sparsely cellular round nodules composed of stellate to spindle cells suspended in an abundant hypovascular myxoid matrix (Fig. 3). The nodules were separated by bands of collagen or by more densely cellular areas of the tumor. In these more densely cellular areas, the neoplastic cells were larger with oval nuclei and eosinophilic cytoplasm. Few foamy macrophages were scattered throughout the tumor. The intra-articular portions of the tumor were covered with synovium, which was often hyperplastic. There were occasional cystic spaces filled with fluid and lined by synovium or compressed mesenchymal cells. Within the tumor subjacent to the synovium, some cases had inflammatory cells—predominantly, lymphocytes and plasma cells with fewer macrophages and neutrophils. In some cases, the myxomatous nodules extended well beyond the joint capsule, infiltrating along fascial planes between muscles. Rarely, the myxomatous nodules were detected invading into bone (Fig. 4 ).
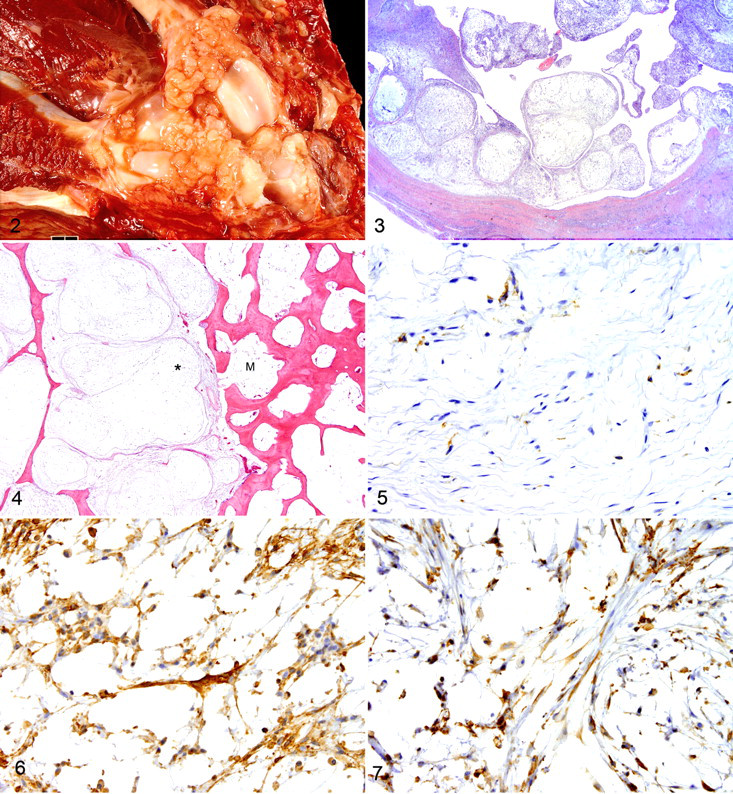
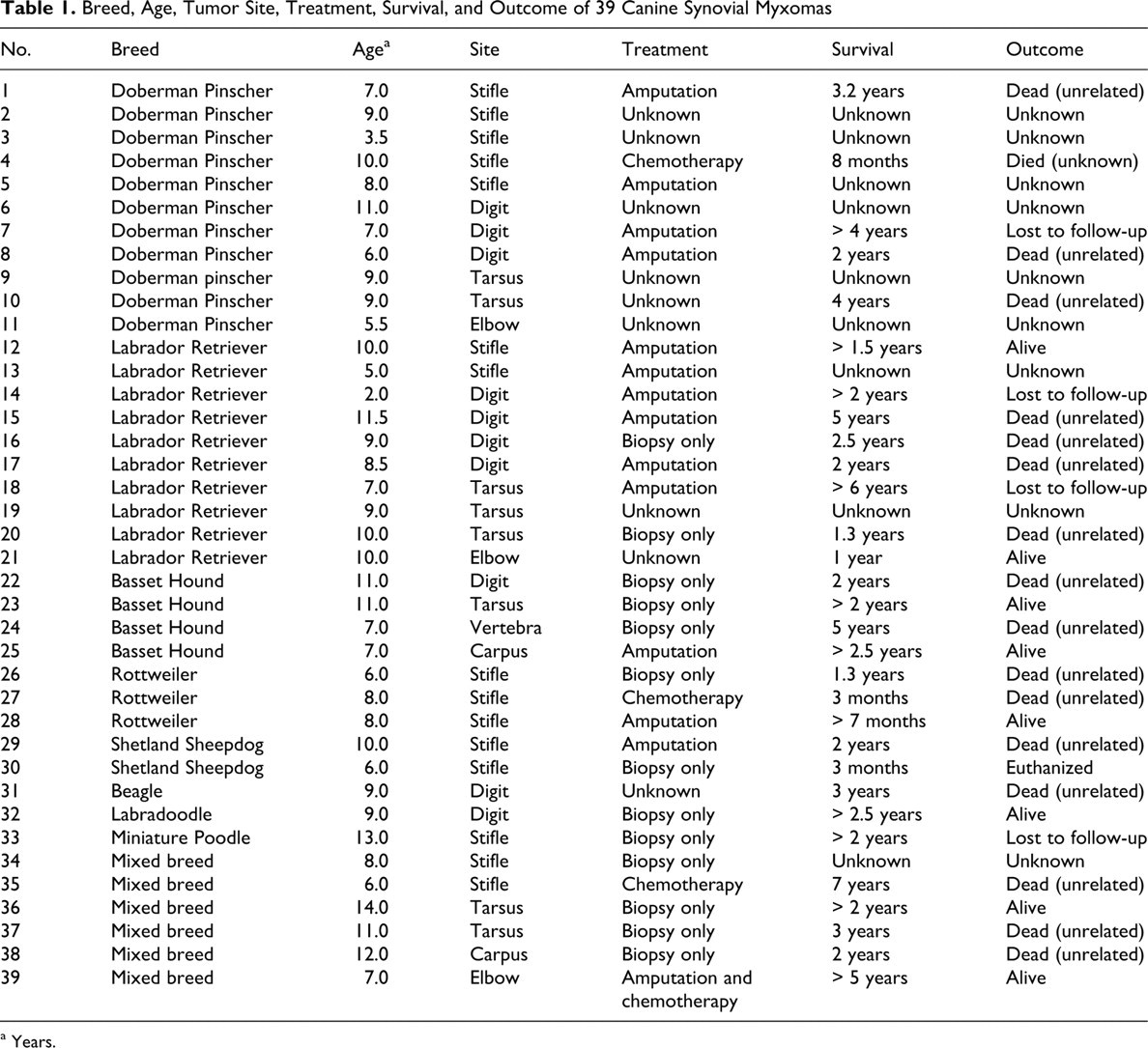
Table 1 lists the breed, age, tumor site, treatment, length of survival, and outcome of each dog. Doberman Pinscher and Labrador Retriever were the most commonly affected breeds (28% and 26%, respectively). Other breeds with more than 1 case included Basset Hound (10%), Rottweiler (8%), and Shetland Sheepdog (5%). The average age was 8.5 years; the range was 2 to 14 years. The sex distribution was 64% female (21 of 25 spayed) and 36% male (8 of 14 castrated). The weight was recorded for 25 dogs, an average of 31.7 kg; 84% of the dogs were more than 20 kg. The range was 5.0 kg (Miniature Poodle) to 68.2 kg (Rottweiler). The stifle was the most commonly affected site (38%), followed by the digit (26%), tarsus (20%), elbow (8%), carpus (5%), and vertebra (3%).
Breed, Age, Tumor Site, Treatment, Survival, and Outcome of 39 Canine Synovial Myxomas
a Years.
The treatment was recorded for 30 cases. Thirteen (43%) were only biopsied; the entirety of excision was often unknown, but in most cases the submitted tissue consisted entirely of tumor nodules. Surgical margins were commented upon in 10 cases, with neoplastic cells extending to the margins in 7 of those. Four cases of digital tumor were treated with digit amputation. Nine cases were treated with whole leg amputation: 6 stifle tumors and 1 each at the tarsus, carpus, and digit. Three cases were treated with chemotherapy (piroxicam, doxorubicin, adriamycin), and one case was treated with amputation and chemotherapy (adriamycin and cytoxan).
Postsurgical survival and outcome data were available for 31 cases. Eight dogs were still alive 7 months to 5 years after diagnosis. Four dogs were lost to follow-up after surviving 2 to 4 years. Nineteen dogs died or were euthanized an average of 2.5 years after diagnosis (range, 0 to 7 years). In only 1 case was death attributed to the tumor; case No. 30 was an obese (28.2 kg) Shetland Sheepdog with a synovial myxoma of the stifle for which euthanasia was elected over amputation. There was no evidence of metastasis in any case; however, 3 cases had local recurrence, 2 of which had neoplastic cells at the surgical margins of the initial biopsy specimen and 1 of which had grossly invasive behavior. Amputation achieved clean margins in 2 of the recurrent cases; 1 dog died of unrelated causes.
The original histologic diagnosis was available in 36 cases. Thirty-one (86%) were originally diagnosed as being malignant: 21 as myxosarcoma, 3 as synovial cell sarcoma (1 mucinous variant and 1 myxoid variant), 2 as lipomyxosarcoma, 2 as liposarcoma (myxoid variant), and 1 each of myxofibrosarcoma, myxoid sarcoma, and sarcoma. Four were diagnosed as synovial myxoma and 1 as synovial myxomatous change.
Eleven synovial myxoma cases had tissue available for immunohistochemistry. All 11 were uniformly positive for vimentin and negative for cytokeratin and S100 protein. In all cases, 20 to 40% of the cells were positive for CD18; these were round to stellate and morphologically indistinguishable from the CD18-negative cells (Fig. 5). The CD18-positive cells were more common in the cellular stroma than within the myxomatous nodules. The overlying synovial membrane was also CD18-positive in all cases. None of the nonsynovial myxomatous tumors had CD18-positive cells.
Immunohistochemically, the synovial myxoma cells, including their long cell processes, expressed cadherin 11 (Fig. 6), as did cells of the nonsynovial myxomatous tumors (subcutaneous myxosarcomas). Similarly, neoplastic cells of both the synovial myxomas and the subcutaneous myxosarcomas were positive for HSP25 (Fig. 7 ). The nonmyxomatous synovial tumors (synovial cell sarcoma and synovial histiocytic sarcoma) also expressed cadherin 11 and HSP25.
Nine of the 11 (82%) synovial myxomas had less than 1% of cells expressing PCNA. Case Nos. 13 and 39 had 5 to 10% cells with nuclear PCNA staining. Mitotic figures were rare to absent in all cases.
Discussion
Canine synovial myxoma was first described by Pool in 1990 as a morphologically unique lesion in 3 dogs; in those cases, the tumor filled the joint space and extended along fascial planes but did not invade bone or metastasize. Synovectomy and amputation were apparently curative. 11 The cases described in this report are similar in appearance and behavior to those described by Pool and others. 4,6,10 However, 8 dogs reported here had radiographic or histologic evidence of bony invasion. Bony invasion is not typical of a benign tumor, but we have elected to use the original name for this tumor based on its benign histologic appearance, slow growth, and overall good prognosis. In this respect, it can be compared to another invasive benign tumor, infiltrative lipoma, which requires amputation in some cases. Although malignant transformation of a benign myxoma into an invasive myxosarcoma was considered, there are no differences in cell morphology, mitotic rate, or PCNA immunohistochemistry between the invasive and noninvasive tumors.
Bony invasion makes this tumor impossible to differentiate radiographically from other common synovial tumors, such as synovial cell sarcoma and synovial histiocytic sarcoma. The addition of synovial myxoma to the differential diagnosis broadens the range of prognosis for these tumors from very good (synovial myxoma) to very poor (synovial histiocytic sarcoma), making a histologic diagnosis essential before treatment. Immunohistochemistry can be used to differentiate synovial cell sarcoma (cytokeratin positive, CD18 negative) from synovial histiocytic sarcoma (cytokeratin negative, CD18 positive). 5 The unique microscopic morphology of synovial myxoma is more useful than immunohistochemistry to diagnose this tumor.
The cell of origin of synovial myxoma is still unknown. Given the abundant viscous synovial-like fluid produced by the neoplasm, we expected the cells to express markers of type B synoviocytes (cadherin 11 and HSP25), not type A synoviocytes (CD18). Normal type B synoviocytes produce hyaluronan, a large linear glycosaminoglycan that is a major component of synovial fluid. 13 However, a significant minority (20–40%) of the cells expressed CD18, suggesting a dendritic cell origin, and although cadherin 11 and HSP25 expression was widespread, it was not specific to synovial-origin neoplastic mesenchymal cells in this study. It is possible that synovial myxomas arise from the recently described transitional or stem cell type C synoviocyte. 15
Cadherins are membrane proteins involved in cell–cell interactions. Unlike most cadherins, cadherin 11 is predominantly expressed by mesenchymal cells. It is important in embryonic limb and joint development as well as organization and homeostasis of the adult synovial membrane. 14 Cadherin 11 is expressed by type B synoviocytes, but not type A synoviocytes, in normal mouse synovium. 1 It is also expressed in bone, placenta, brain, lung, and heart, but its distribution in other tissues is largely unknown. 14 In this study, most synovial myxoma cells were positive for cadherin 11; however, the neoplastic cells of the nonsynovial myxomatous tumors (ie, synovial cell sarcoma and synovial histiocytic sarcoma) were also positive. Therefore, cadherin 11 cannot be used to differentiate synovial myxomas from other myxomatous tumors or other synovial tumors. This widespread expression of cadherin 11 in mesenchymal tumors may represent upregulation associated with neoplastic transformation, as has been seen in invasive tumors in humans. 16
Like most heat shock proteins, HSP25 protects cells against various insults, such as hyperthermia, oxidative stress, and inflammatory cytokines. However, HSP25 is also involved in cell differentiation under normal conditions and has been used as a specific marker of type B synoviocytes in dogs and mice. 7,15 In this study, all tumor cells (synovial myxoma, nonsynovial myxosarcoma, synovial cell sarcoma, and histiocytic sarcoma) were positive for HSP25. Therefore, like cadherin 11, HSP25 cannot be used to differentiate synovial myxomas from other myxomatous tumors or other synovial tumors.
The canine synovial myxoma shares some features with a tumor described in humans, the juxta-articular myxoma, a poorly circumscribed myxoid mass that most commonly affects the knee (88%). Humans with this tumor typically have a long duration of clinical signs (up to 18 years) that suggest degenerative joint disease; 8 however, the lesion has been confirmed to be neoplastic by clonal chromosome changes. 9,12 Human and canine myxomas both consist of stellate to spindle cells within an abundant hypovascular myxoid matrix. Mitotic figures are rare or absent. None of the human tumors have been reported to metastasize, but approximately one-third recur locally. As with the canine synovial myxoma, many are misdiagnosed as a sarcoma. 8 However, the human juxta-articular myxoma differs from its canine counterpart in several ways. In humans, men are predisposed (72%), whereas approximately two-thirds of the affected dogs in this report were female. The subgross morphology of the tumors is also somewhat different. Although the human tumor has a diffuse pattern with only rare lobules, the canine tumor is distinctly nodular. Perhaps the most important difference is that the human tumor is located in the periarticular subcutis and only rarely abuts the synovium. 8 All the reported canine tumors were within joints, although some extended through the joint capsule and into the surrounding tissues.
Although we do not know the breed distribution of the biopsy sample population from the multiple institutions contributing to this report, we can assume that they roughly reflect national breed popularity. The Labrador Retriever is the most popular breed in the United States and has been for many years; however, this does not account for the fact that 26% of synovial myxomas were from that breed. If breed popularity were the determining factor, then the second- and third-most-popular breeds in the United States, Yorkshire Terrier and German Shepherd, would be represented in this report. Doberman Pinschers, only the 20th-most-popular breed over the past 5 years, made up 28% of the cases in this report, strongly indicating a breed predisposition. Four of the synovial myxomas were in Basset Hounds, which may represent a breed predisposition, given that the average popularity ranking for that breed over the past 5 years was 30th (see http://akc.org/reg/dogreg_stats.cfm).
In summary, this is a morphologically unique synovial tumor with a good prognosis. Most of the cases in this report had been diagnosed as being malignant, and some had been treated with chemotherapy. Accurate diagnosis will help ensure that less aggressive therapy, such as synovectomy or amputation, is used in these cases.
Footnotes
Acknowledgement
We thank Dr Danielle Reel for contributing case No. 21.
The authors declared that they had no conflicts of interests with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
