Abstract
The aims of this study were to determine whether the appearance of stromal myofibroblasts and the expression of tenascin-C (Tn-C) correlate with the grade of malignancy in canine mammary tumors and to determine the main cellular source of Tn-C in these tumors. Single or double immunostaining using antibodies against α-smooth muscle actin (α-SMA) and Tn-C was performed on serial sections of normal canine mammary glands as well as those with lobular hyperplasia, simple adenoma, and simple carcinoma. Thirty-nine of 42 simple carcinomas (93%) exhibited stromal α-SMA–positive myofibroblasts and Tn-C expression. Only 6 of 11 cases of simple adenoma (55%) showed these changes, whereas no changes were observed in normal mammary gland tissue or cases of lobular hyperplasia. The distribution of stromal Tn-C correlated with the presence of myofibroblasts. However, Tn-C immunoreactivity was also occasionally observed in the basement membrane zone surrounding the myoepithelial layer in normal tissue, benign lesions, and tubulopapillary carcinomas. This pattern of staining was not related to the presence of myofibroblasts. The appearance of stromal myofibroblasts and expression of Tn-C were significantly correlated with higher histological grades of malignancy and vascular/lymphatic invasion in simple carcinomas. Stromal myofibroblasts appear to be a major cellular source of Tn-C and play an important role in the development of canine mammary tumors. The Tn-C expressed in the basement membrane zone of normal, hyperplastic, and neoplastic mammary tissue, which is likely produced by neighboring myoepithelial cells, may differ functionally from the Tn-C produced by myofibroblasts.
Keywords
It is accepted that the stromal microenvironment of the host contributes to carcinogenesis in various cancers. 15,19 Recent evidence suggests that the accumulation of genetic mutations in cancer cells and the interaction between cancer cells and the stromal microenvironment are important for cancer progression. Myofibroblasts, a unique group of smooth musclelike fibroblasts, have long been observed in the stroma of various cancers and are considered to play an important role in carcinogenesis, organogenesis, inflammation, wound healing, and fibrosis. 27 The myofibroblasts that reside in the stroma of cancerous tissue, which are often referred to as carcinoma-associated fibroblasts, promote the proliferation and invasion of carcinoma cells and induce angiogenesis by the secretion of several extracellular matrix molecules, proteases, and cytokines. 8,15,16,19 Although knowledge about stromal myofibroblasts within carcinomas has accumulated steadily in human medicine, little attention has been paid to their role in veterinary medicine, although myofibroblasts have been recognized in canine and feline mammary tumors. 6,17,18,31
Tenascin-C (Tn-C) is a huge extracellular matrix protein that consists of 6 identical subunits constructed from a variable number of repeated domains. 4,24,25 Tn-C is expressed transiently during organogenesis and is absent or greatly reduced in most adult tissues. However, it increases again in pathological conditions such as inflammation, wound healing, and fibrosis. 4 Tn-C also increases markedly in the stroma of neoplastic tumors. 4 In general, the intensity of Tn-C expression correlates with tumor malignancy, and the strongest expression indicates a poor prognosis. 4,25 Several studies have shown that Tn-C counteracts fibronectin cell adhesion signaling. 4,24,25 This leads to cell rounding and potentially stimulated cell proliferation. 25 Furthermore, it is possible that Tn-C plays a role in tumor angiogenesis and supports escape from tumor immunosurveillance. 25 These findings suggest that Tn-C is an important factor in the stromal microenvironment of tumor tissue.
It has been shown that Tn-C accumulates at the tumor invasion front of cancerous growth. Myofibroblasts also appear in cancerous tumor stroma, particularly at the invasion front. This correlation led to a hypothesis that myofibroblasts may be an important cellular source of Tn-C within the cancer stroma. 10 There are few reports concerning the relationship between myofibroblasts and Tn-C in the stroma of human endometrial cancer, 5 breast cancer, 38 prostate cancer, 32,35 and colon cancer 12 as well as in canine gastrointestinal tumors. 22 The expression of Tn-C has also been associated with myofibroblasts in Dupuytren’s disease. 3
Arai et al 1 reported that expression of Tn-C was observed in areas of proliferating myoepithelial cells and chondroid metaplasia in canine mixed mammary tumors. Nederbragt and coworkers 10,22,23,28 have studied the role of Tn-C in tumor progression for various canine neoplasms including mammary, gastrointestinal, and melanocytic tumors. These investigators' immunohistochemical data indicated that Tn-C was present in all mammary tissues examined, with increased expression in remodeling tissues and neoplastic lesions. 10 Thus, the distribution of Tn-C in canine mammary tumors is not necessarily similar to that reported in human breast tumors. 10
The aims of this study were to assess the relationship between the appearance of myofibroblasts and Tn-C expression in the tumor stroma of canine mammary carcinomas compared with the grade of tumor malignancy and to reveal the main cellular source of Tn-C.
Methods
Pathologic Examination
Normal and proliferative mammary tissues from dogs were obtained from surgical or necropsy samples submitted to the Department of Veterinary Pathology, Nippon Veterinary and Life Science University. Tissues were fixed in 10% neutral buffered formalin and embedded in paraffin wax, and sections were cut at 4 μm and stained with hematoxylin and eosin. Normal (nonneoplastic) mammary gland (n = 11), lobular hyperplasia (n = 12), simple adenoma (n = 11), and simple carcinoma (n = 42: 20 tubulopapillary, 15 solid, 7 anaplastic) were diagnosed on the basis of the World Health Organization classification system. 21 Moreover, 42 cases of simple carcinoma were graded according to the Elston and Ellis criteria. 9,13,20 Briefly, each carcinoma was evaluated by quantification of anaplasia, tubule formation, mitotic activity, and nuclear pleomorphism of cancer cells and was assigned to grade I, II, or III. Complex and mixed mammary tumors were excluded from this study, because areas where myofibroblasts were admixed with myoepithelial cells made it difficult to determine which cells were predominantly involved in the production of Tn-C. The accessory lymph nodes excised with mammary carcinomas were routinely processed and examined microscopically to determine whether metastasis was present.
Immunohistochemistry
Serial sections were immunostained with a labeled streptavidin–biotin method using a mouse monoclonal antibody against Tn-C (4F10TT), α-SMA, vimentin, or p63 and a rabbit polyclonal antibody against S-100 protein. The monoclonal antibody 4F10TT recognizes an epitope within the EGF-like sequences that is common to all isoforms of Tn-C. The primary antibodies, their source, dilution, and the pretreatment used for immunohistochemical analysis are shown in Table 1 . After reaction with the primary antibodies, the sections were incubated with biotinylated goat antimouse IgG or antirabbit IgG antibodies (DAKO, Glostrup, Denmark), followed by peroxidase-conjugated streptavigin (DAKO). Finally, the reaction of each antigen was visualized by addition of diaminobenzidine tetrahydrochloride chromogen (DAB), and the slides were counterstained with hematoxylin. Sections from canine mixed mammary tumors that had been previously shown to express Tn-C 1 were used as positive controls. Negative controls were mammary tissue sections incubated with PBS instead of the primary antibodies.
Primary Antibodies Used in This Study

Double Immunostaining
Double immunostaining for α-SMA and Tn-C was performed on additional sections mounted on MAS-coated slides (Matsunami, Osaka, Japan). Endogenous peroxidase activity was blocked by incubation with 0.3% hydrogen peroxide in methanol for 30 minutes at room temperature (RT). For antigen retrieval of α-SMA, sections were heated using a microwave in 0.01 M citrate buffer (pH 6.0) for 10 minutes. After incubation for 30 minutes with Block Ace (DS Pharma Biomedical Co., Oosaka, Japan) to block background staining, the samples were treated overnight (4°C) with α-SMA primary antibody. After washing, they were incubated with EnVision+ System HRP Labeled Polymer Antimouse (DAKO) for 30 minutes at RT, followed by visualization in DAB. For blocking antibody cross-reactivity, samples were heated in a microwave oven in 0.01 M citrate buffer (pH 6.0) for 10 minutes. 14 For retrieval of Tn-C, the samples were treated with 0.1% trypsin at 37°C for 20 minutes, followed by incubation with Histofine Simple Stain AP (NICHIREI, Osaka, Japan) for 30 minutes at RT. The enzymatic activity of alkaline phosphatase was visualized with new fuchsin.
Assessment and Scoring of Myofibroblasts and Expression of Tn-C
Myofibroblasts were distinguished immunohistochemically from myoepithelial cells by positive staining for α-SMA and vimentin and negative staining for the myoepithelial markers p63 and S-100. 11 The appearance of myofibroblasts in mammary tissue was assessed using a semiquantitative evaluation proposed by Surowiak et al. 30 Briefly, samples were graded from (–) to (3+), with (–) indicating no stromal myofibroblasts, (1+) indicating scanty stromal myofibroblast presence (<10%), (2+) indicating focally abundant stromal myofibroblast growth (10%–30%), and (3+) indicating globally abundant stromal myofibroblasts (>30%). The extent of Tn-C expression in tumor stroma or the basement membrane zone was graded semiquantitatively from (–) to (3+), with (–) indicating no expression of Tn-C, (1+) indicating scanty Tn-C expression (<10%), (2+) indicating focal Tn-C expression (10%–30%), and (3+) indicating global Tn-C expression (>30%). The number of stromal myofibroblasts and the extent of Tn-C expression were evaluated under 400× magnification. The Mann-Whitney U-test was used to determine whether there was a statistically significant difference in the scoring of stromal myofibroblasts or Tn-C expression between samples and in comparison to the histopathological parameters (histological type and grade, intravasation, and metastasis to the neighboring lymph node in cases of carcinoma). All values of P < .05 were considered significant.
Results
Immunohistochemistry
Normal and hyperplastic mammary tissue
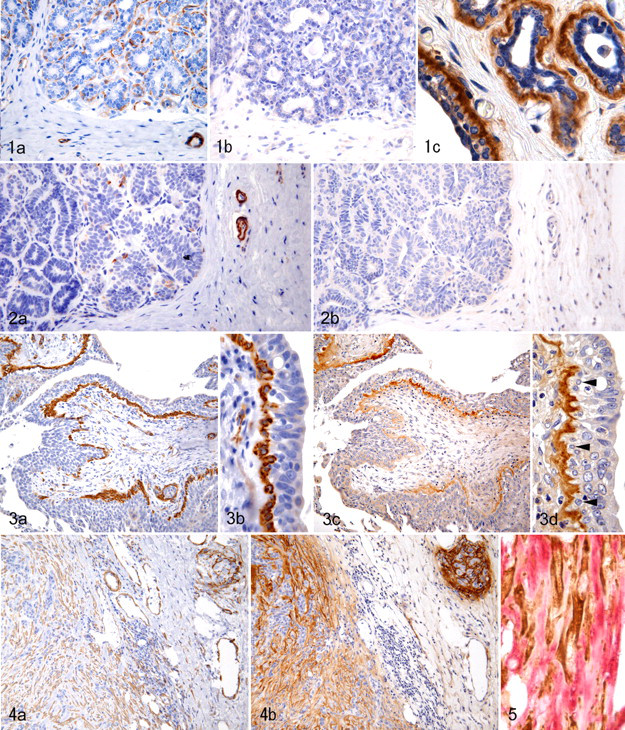
Stromal cells in histologically normal (nonneoplastic) mammary glands and mammary lobular hyperplasia were negative for α-SMA and Tn-C (Fig. 1a, b). However, a positive reaction for Tn-C was sometimes observed in the ductal basement membrane zone and, less frequently, in the acinar basement membrane zone (Fig. 1c). Myoepithelial cells were positive for both α-SMA and p63, but myoepithelial cells adjacent to the Tn-C–positive basement membrane zone seldom immunostained with anti-Tn-C antibody. Smooth muscle cells and adventitial cells of the blood vessels often expressed Tn-C.
Simple adenoma
Neither α-SMA–positive myofibroblasts nor Tn-C–positive extracellular matrixes were present among tumor cells in simple adenomas (Fig. 2a, b). However, in 6 of 11 adenomas (55%), stromal myofibroblasts were seen in focal areas of the fibrous capsule and in the stroma surrounding dilated neoplastic ductules, particularly the basal stroma of intraductal papillary growths. Expression of Tn-C in the tumor stroma was located in areas of myofibroblast proliferation when serial sections were immunolabeled for Tn-C or α-SMA. In 7 of 11 adenomas (64%), the basement membrane zone of ductal-like tumor cell proliferation was positive for Tn-C despite the absence of myofibroblasts in the surrounding stroma. In these tumors, the positive reaction was restricted to the basement membrane zone with overlying myoepithelial cells that were positive for α-SMA and p63.
Simple carcinoma
In 10 of 20 cases of tubulopapillary carcinoma (50%), myofibroblasts positive for α-SMA were either absent or either located focally in the neoplastic stroma. The distribution of α-SMA–positive myofibroblasts seemed to coincide with sites of Tn-C expression when serial sections were immunostained. In the other 50%, the connective tissue surrounding and within the neoplastic lobules, particularly at the tumor invasion front, contained a large number of myofibroblasts and abundant Tn-C–positive extracellular matrix. The myofibroblasts located at the invasion front showed stronger expression of α-SMA than other areas of myofibroblast growth. Similarly, Tn-C expression was more prominent at the invasion front than in the neoplastic lobules. In 12 of 20 tubulopapillary carcinomas (60%), a discontinuous myoepithelial monolayer accompanied by an underlying Tn-C–positive basement membrane zone was sometimes observed (Fig. 3).
Of 15 solid carcinomas, 10 cases (67%) showed focally or globally abundant myofibroblasts and exhibited focal or global Tn-C expression in the stroma (Fig. 4a, b). The remaining 5 cases (33%) contained only scanty myofibroblasts and no or scant positivity for Tn-C. Double immunostaining for α-SMA and Tn-C showed a similar distribution of positive staining for both antibodies. Individual α-SMA–positive myofibroblasts were closely surrounded by an extracellular matrix immunolabeled with Tn-C (Fig. 5). In 3 of 15 solid carcinomas (20%), a variable number of tumor cells showed positive cytoplasmic staining for both Tn-C and vimentin. Tumor cells in the remaining 12 cases were negative for Tn-C. Necrotic areas were often positive for Tn-C. In solid carcinomas, the myoepithelial layer with a Tn-C–labeled basement membrane zone was not observed.
For anaplastic carcinomas, 3 of 7 cases (43%) had a large number of stromal myofibroblasts. However, the remaining 4 cases, consisting of extremely poorly differentiated cells lacking epithelial features, had no or sparse myofibroblast growth in the stroma. Stromal Tn-C immunoreactivity was observed focally or globally in all cases of anaplastic carcinoma.
The frequency of stromal myofibroblasts in simple carcinomas was significantly higher than in simple adenomas (Table 2 ). Among the 3 subtypes of carcinoma, the solid type had the highest appearance of stromal myofibroblasts.
Appearance of Myofibroblasts in Normal and Proliferative Mammary Tissues
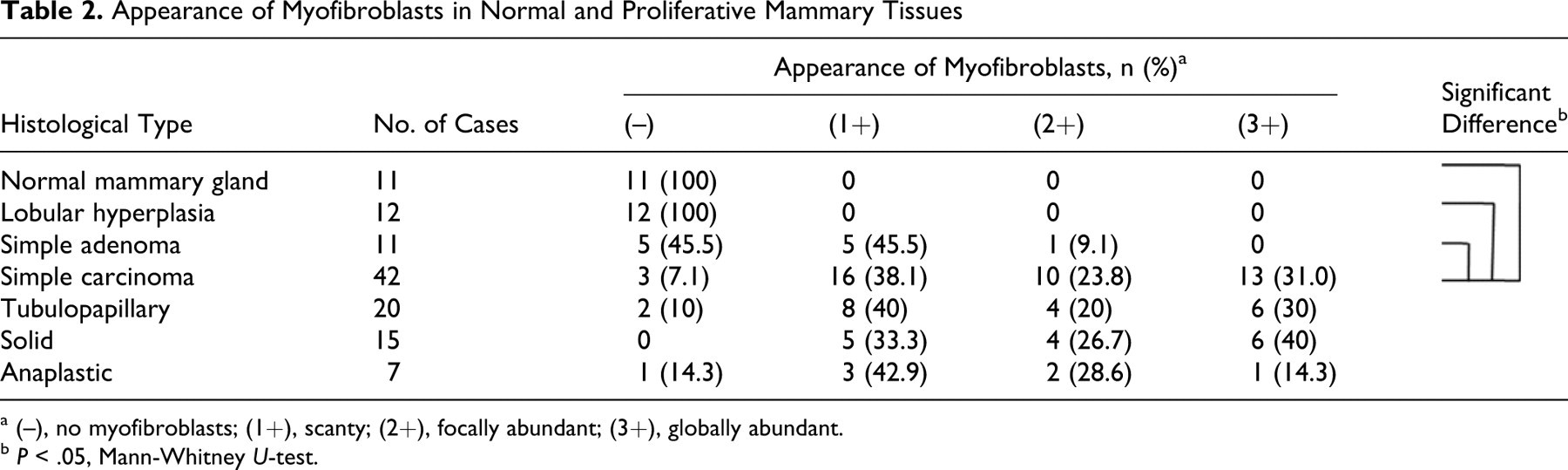
a (–), no myofibroblasts; (1+), scanty; (2+), focally abundant; (3+), globally abundant.
b P < .05, Mann-Whitney U-test.
In general, the expression of Tn-C in tumor stroma corresponded to the appearance of stromal myofibroblasts. Expression of Tn-C in the stroma was significantly greater in simple carcinomas than in simple adenomas (Table 3 ). In contrast, expression of Tn-C was significantly higher in the basement membrane zone in simple adenomas than in simple carcinomas.
Expression of Tenascin-C in Normal and Proliferative Mammary Tissues
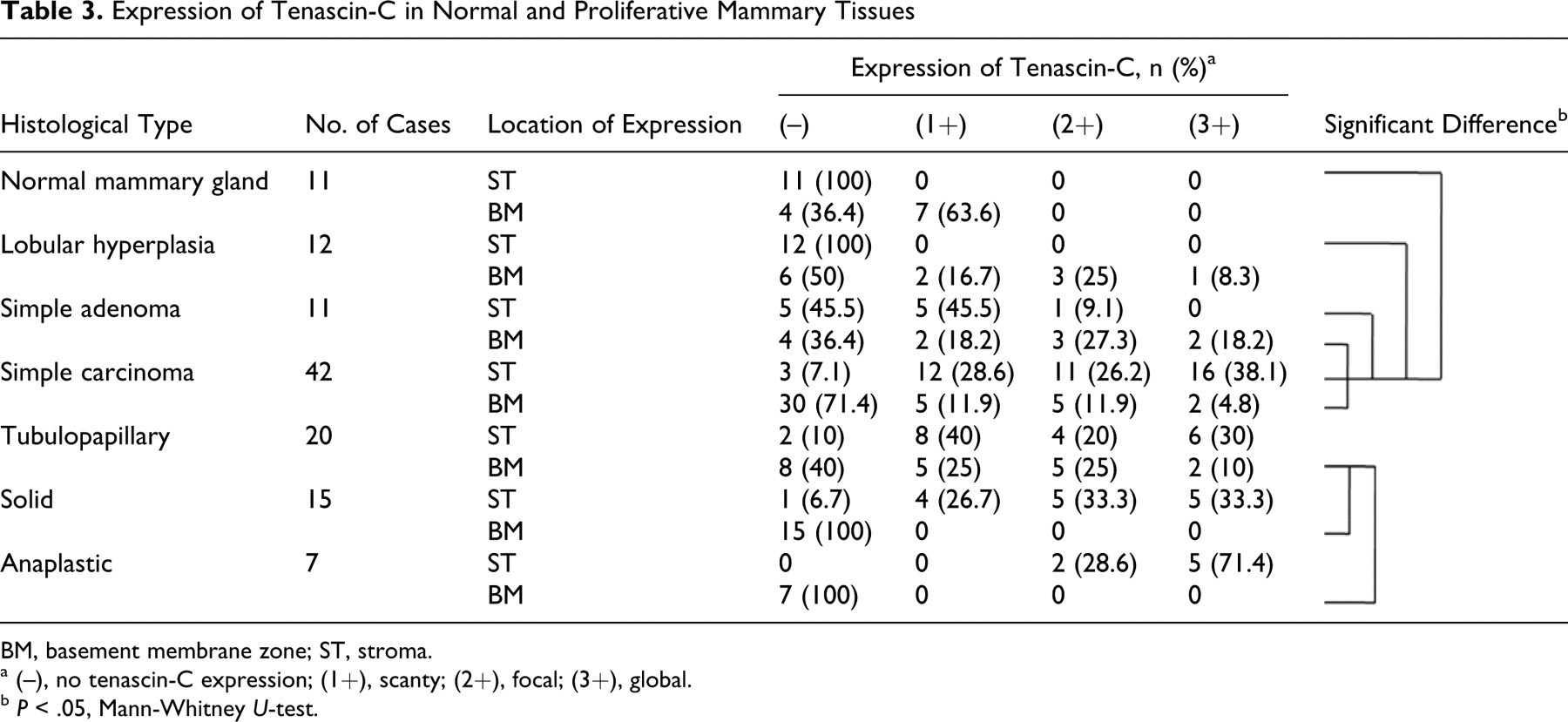
BM, basement membrane zone; ST, stroma.
a (–), no tenascin-C expression; (1+), scanty; (2+), focal; (3+), global.
b P < .05, Mann-Whitney U-test.
Relationship Between the Appearance of Myofibroblasts and Histopathological Parameters in Simple Carcinomas
Histologically, 42 cases of simple carcinoma were graded as follows: grade I, 11 cases (all tubulopapillary); grade II, 12 cases (6 tubulopapillary, 3 solid, 3 anaplastic); and grade III, 19 cases (3 tubulopapillary, 12 solid, 4 anaplastic). Seventeen of 42 cases (40.5%) had vascular/lymphatic invasion and 8 cases (19.0%) showed lymph node metastasis.
In simple carcinomas, there were significantly more myofibroblasts at grade III than at grade I (Table 4 ). Also, myofibroblasts appeared significantly more frequently in carcinomas with stromal intravasation and/or metastasis than in those without (Table 4).
Correlation of Appearance of Myofibroblasts With Histopathological Parameters in Simple Carcinomas
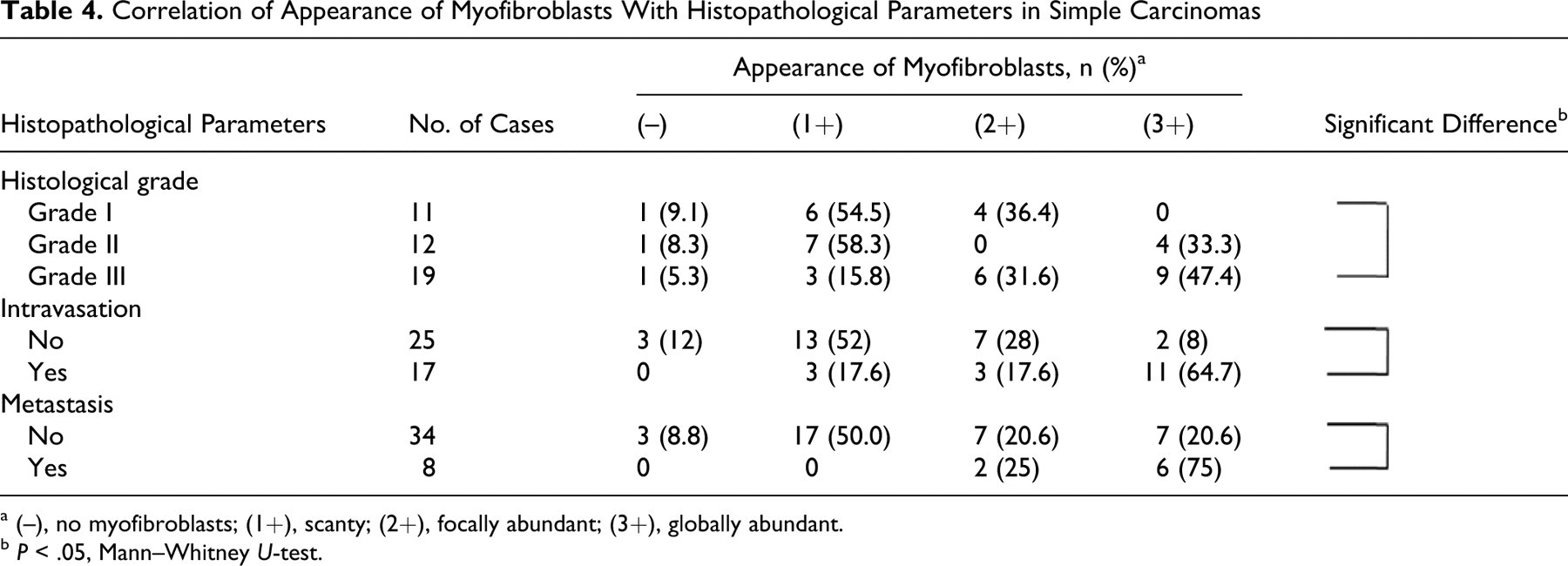
a (–), no myofibroblasts; (1+), scanty; (2+), focally abundant; (3+), globally abundant.
b P < .05, Mann–Whitney U-test.
Relationship Between Expression of Tn-C and Histopathological Parameters in Simple Carcinomas
Regarding the grading of simple carcinomas, the expression of Tn-C in the stroma was significantly higher in grade III than in grade I tumors. Stromal expression of Tn-C in carcinomas with intravasation was more global than in those with no intravasation (Table 5 ). Although there was a trend toward stronger expression of Tn-C in carcinomas with metastasis than in nonmetastatic carcinomas, the difference was not statistically significant.
Correlation of Expression of Tenascin-C With Histopathological Parameters of Simple Carcinomas
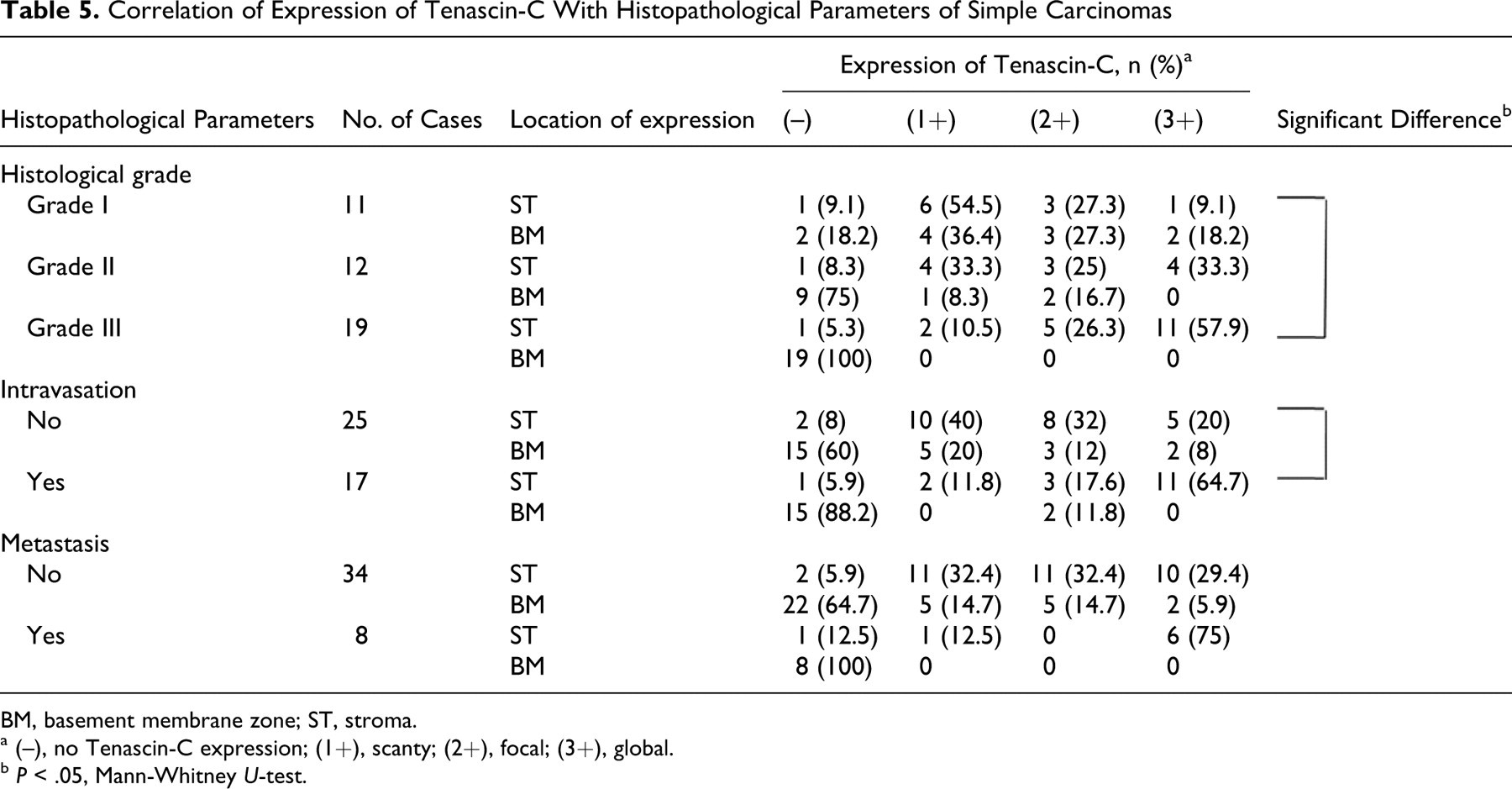
BM, basement membrane zone; ST, stroma.
a (–), no Tenascin-C expression; (1+), scanty; (2+), focal; (3+), global.
b P < .05, Mann-Whitney U-test.
Discussion
This study focused on the appearance and distribution of stromal myofibroblasts in samples of neoplastic and nonneoplastic canine mammary tissue. No myofibroblasts were observed in normal or hyperplastic mammary tissue. Myofibroblasts were rarely detected in the marginal fibrous tissue compressed by proliferating tumor cells in simple adenomas but were observed frequently in the interlobular stroma of simple carcinomas. Stromal myofibroblasts were most frequently found within the tumor invasion front of carcinomas, and myofibroblasts in this area also showed stronger expression of α-SMA. The appearance of stromal myofibroblasts was significantly related to the grade of malignancy, intravasation, and metastasis of carcinomas. Histological grading of mammary carcinomas using the Elston and Ellis method 9 is associated with prognosis, especially in cases of simple carcinoma in dogs. 13 Also, it has been confirmed that intravasation and lymph node metastasis are good criteria for indicating malignant behavior in canine mammary carcinomas. 20 These results suggest that there is a relationship between the occurrence of stromal myofibroblasts and poor prognosis in canine mammary tumors. The findings are similar to results reported for invasive ductal carcinomas of the human breast. 29,30,37 Yazhou et al 37 demonstrated that a comparison between human breast carcinomas with and without stromal myofibroblasts revealed significant differences in histological grade and lymph node metastasis. Surowiak et al 29,30 reported that human cases with a larger number of myofibroblasts in breast carcinoma tissue manifested a higher histological grade, which was accompanied by lowered overall survival and relapse-free survival.
Our results suggest that severely infiltrative carcinomas could be grouped into 2 types, scirrhous carcinomas and anaplastic carcinomas. The first group consists of carcinomas with reactive stroma containing abundant myofibroblasts showing histological features of scirrhous carcinoma, which is known to be highly invasive. Tumor stroma plays an important role as a foothold for the organization of epithelial structures. These carcinoma cells are thought to induce excessive development of early stroma by secretion of various cytokines. The newly formed stroma then contributes to growth and infiltration of the carcinoma. 15,16,19,27 In contrast, some anaplastic carcinomas, consisting of pleomorphic epithelial cells with sarcomatous features, had a relatively small amount of reactive stroma. These anaplastic carcinoma cells may gain the ability to grow and infiltrate without a foothold, similar to inflammatory cells. With the exception of anaplastic carcinoma cases, abundant stromal myofibroblasts in canine mammary carcinomas were generally associated with aggressive tumor characteristics.
The patterns of Tn-C immunolabeling in canine mammary tissues were assessed, which revealed 2 different localization patterns of Tn-C expression in nonneoplastic and neoplastic canine mammary tissues: (1) Tn-C expressed in reactive stroma and (2) Tn-C expressed in the basement membrane zone. Stromal Tn-C immunoreactivity was not detected in normal and hyperplastic mammary glands or in simple adenomas without stromal myofibroblasts. It was found in simple adenomas with stromal myofibroblasts and in simple carcinomas, particularly at the tumor invasion front. The areas of stromal Tn-C expression coincided almost exactly with the distribution of myofibroblasts. Yoshida et al 38 described in their review that myofibroblasts are candidates for Tn-C–producing cells in human breast cancers. Faustino et al 10 also suggested that myofibroblasts are an important cellular source of stromal Tn-C in canine mammary tumors. However, they did not immunohistochemically demonstrate the colocalization of Tn-C with myofibroblasts. The results of our current study agree with the concept that myofibroblasts produce excessive amounts of stromal Tn-C in canine mammary tumors, although fibroblasts, smooth muscle cells and the adventitial cells of blood vessels also produce it to some degree. De Wever et al 7 indicated that myofibroblasts isolated from human colon cancer tissue stimulated invasion of colon cancer cells in vitro and showed that Tn-C was a convergent proinvasive agent secreted by myofibroblasts. Furthermore, it has been demonstrated that Tn-C involves various mechanisms of tumor cell proliferation, including angiogenesis and inhibition of immunosurveillance. 24,25 Findings in our current study suggest that Tn-C is hyperproduced by myofibroblasts within the reactive stroma of canine mammary tumors and plays an important role in cancer progression, similar to findings presented in humans.
Tn-C immunoreactivity was often observed in the basement membrane zone surrounding ductal structures in normal mammary glands, simple adenomas, and low-grade tubulopapillary carcinomas but not in high-grade carcinomas. The Tn-C immunoreactivity in the basement membrane zone did not correspond to the localization of myofibroblasts. It is generally accepted that mesenchymal cells are the primary cellular source of extracellular matrix proteins, including Tn-C, 38 but epithelial cells in the basal layer of the skin also produce these proteins. 2 In this study, the Tn-C–positive basement membrane zone was always adjacent to myoepithelial cells showing expression of both α-SMA and p63. Arai et al 1 reported that proliferating myoepithelial cells in canine mixed mammary tumors produce Tn-C. It appears that myoepithelial cells of the basement membrane zone also produce Tn-C in normal canine mammary tissues. Other extracellular matrix proteins such as laminin and type IV collagen have been found in both the basement membrane and areas of proliferating myoepithelial cells in canine mammary tumors, and it has been suggested that myoepithelial cells are the main source of these proteins. 26 However, myoepithelial cells adjacent to the Tn-C–positive basement membrane zone were seldom positive for Tn-C. A possible explanation may be similar to that observed in the morphogenesis of chick feathers, where Tn-C messenger RNA is detected in the overlying epithelia by in situ hybridization, but by immunohistochemistry, the epithelia is Tn-C–negative whereas the basement membrane zone shows Tn-C–positive staining. 34
A high accumulation of Tn-C appears to be a sign of poor prognosis in human breast cancer. 4,24,25 In canine mammary tumors, however, even simple adenomas often express prominent Tn-C. 10 Therefore, Faustino et al 10 concluded that Tn-C cannot be used as a marker of malignancy in canine mammary tumors. The present study revealed that the Tn-C in the basement membrane zone in most simple adenomas and low-grade tubulopapillary carcinomas was likely to be produced by myoepithelial cells rather than by stromal myofibroblasts. It has been reported that resting myoepithelial cells are present in simple adenoma and carcinoma. 6,11 Furthermore, this study demonstrated that the Tn-C expressed in the stroma of carcinomas significantly correlates with histological parameters associated with a poor prognosis, whereas the Tn-C in the basement membrane zone does not. The Tn-C expressed in the basement membrane zone is thought to have different splice variants from that expressed in reactive stroma. Tn-C has a number of splice variants generated by alternative splicing of fibronectin-like type III repeats. 33 Tsunoda et al 33 performed an immunohistochemical study on human breast tissue using a monoclonal antibody (4F10TT) against an epitope within the EGF-like sequence that is common to all forms of Tn-C and a monoclonal antibody (4C8MS) against the fibronectin-like type III B domain. In normal human mammary glands, all the Tn-C variants that were immunolabeled with 4F10TT were located in the basement membrane zone surrounding ducts, but the large Tn-C variants labeled by 4C8MS were not. In contrast, both the antibodies showed strong binding to the cancer stroma. 33 Thus, in canine mammary tumors, the Tn-C in the basement membrane zone seems to have a different function from the stromal Tn-C that is directly associated with tumor progression. Tn-C immunoreactivity in normal mammary glands is confined mainly to the basement membrane zone surrounding ducts, known as a site in which mammary stem/progenitor cells reside. 36 Colocalization of Tn-C with stem cell niches in some organs has been described. 24 Further studies will be required to determine the roles played by Tn-C expressed in the basement membrane zone in relation to mammary stem/progenitor cells and mammary cancer stem cells.
Footnotes
Acknowledgement
We thank Mr. Rei Nakahira for photographic support.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
This work was supported in part by the Academic Frontier Project for Private Universities: matching fund subsidy (2005–2009) from the Ministry of Education, Culture, Sports, Science and Technology of Japan (MEXT).
