Abstract
Eyes and nuclei of the visual pathways in the brain were examined in 30 Rocky Mountain elk (Cervus elaphus nelsoni) representing 3 genotypes of the prion protein gene PRNP (codon 132: MM, ML, or LL). Tissues were examined for the presence of the abnormal isoform of the prion protein associated with chronic wasting disease (PrPCWD). Nuclei and axonal tracts from a single section of brain stem at the level of the dorsal motor nucleus of the vagus nerve were scored for intensity and distribution of PrPCWD immunoreactivity and degree of spongiform degeneration. This obex scoring ranged from 0 (elk with no PrPCWD in the brain stem) to 10 (representing elk in terminal stage of disease). PrPCWD was detected in the retina of 16 of 18 (89%) elk with an obex score of > 7. PrPCWD was not detected in the retina of the 3 chronic wasting disease–negative elk and 9 elk with an obex score of < 6. PrPCWD was found in the nuclei of the visual pathways in the brain before it was found in the retina. Within the retina, PrPCWD was first found in the inner plexiform layer, followed by the outer plexiform layer. Intracytoplasmic accumulation of PrPCWD was found in a few neurons in the ganglion cell layer in the PRNP 132ML elk but was a prominent feature in the PRNP 132LL elk. Small aggregates of PrPCWD were present on the inner surface of the outer limiting membrane in PRNP 132LL elk but not in PRNP 132MM or 132ML elk. This study demonstrates PrPCWD accumulation in nuclei of the visual pathways of the brain, followed by PrPCWD in the retina.
Chronic wasting disease (CWD), a transmissible spongiform encephalopathy, has been reported in captive and free-ranging cervids. 3,32,37,38 An abnormal isoform of the prion protein has been found in cervids with CWD (PrPCWD), and it can be detected with immunohistochemistry. 33 PrPCWD has been reported in the brain, lymphoid tissues, and other organs (including the retina) in mule deer and white-tailed deer with CWD. 9,16,27,32 Misfolded prion proteins have been associated with other transmissible spongiform encephalopathies and have been reported in retinas of domestic sheep inoculated with the scrapie agent 7,14 and in humans with sporadic and variant Creutzfeldt–Jakob disease. 10 This communication documents the occurrence of PrPCWD in the retina, optic nerve, and visual pathways of the brain in Rocky Mountain elk with natural occurring CWD. Disease progression in preclinical elk with natural exposure to CWD was estimated from the intensity and distribution of immunoreactivity and spongiform change in the brain stem at the level of the dorsal motor nucleus of the vagus nerve (obex score). Distribution of PrPCWD in the retina and optic pathways was a late-stage event, observed only in elk with relatively high obex scores. Minor variation in the distribution pattern of PrPCWD in the retina may be associated with a polymorphism at codon 132 of the PRNP gene, a site associated with incubation time in elk 8,23 and relative susceptibility and disease expression in humans (human codon 129) with sporadic and acquired transmissible spongiform encephalopathies. 19
Materials and Methods
Eyes and brain were collected postmortem from 27 Rocky Mountain elk diagnosed with CWD by immunohistochemistry assay of antemortem biopsy samples of rectal mucosal-associated lymphoid tissue 31,34 or postmortem assay of brain and lymphoid tissues. 33 Negative-control elk included 2 ranch-raised elk from a herd free of a history of CWD and 1 free-ranging elk with no detectable PrPCWD in brain or lymphoid tissues.
The open reading frame of the PRNP gene was sequenced in all the elk as described. 22 The previously described coding change at codon 132 was detected; elk were identified as homozygous for the wild-type allele encoding methionine (132MM), homozygous for the alternative allele encoding leucine (132LL), or heterozygous for the alleles (132ML).
Fourteen elk (47%) were free-ranging adult cows (eleven 132MM, three 132ML) originating from north-central Colorado and 11 elk (37%) originating from captive herds in northern Colorado. The captive group included 8 bulls (seven 132MM, one 132ML) and 3 cows (two 132MM, one 132ML). In addition, two 132LL elk experimentally fed a pooled brain homogenate from two 132ML CWD-positive elk were examined. 8,23 Both were euthanized at 60 months postinfection.
Following euthanasia, eyes were collected within 1 to 5 minutes from each elk. The globe of each eye was opened via an incision through the sclera just posterior to the limbus, thus increasing exposure of the retina to fixative. Eyes were placed in Davidson’s fixative for 3 days. 13 Brains were collected within 4 hours of death from each elk, hemisectioned, placed in 10% neutral buffered formalin, and fixed for 1 week. The formalin was changed at least twice for each brain. Sections from each brain were cut at 5 μm and immunolabeled as previously described with anti–prion protein monoclonal antibody F99/97.6.1 33 (mAb99; Anti-Prion (99), Ventana Medical Systems, Inc, Tucson, AZ) and monoclonal antibody P4 (mAbP4). 36 The latter binds an epitope at residues 93–99, near the amino terminus of the processed prion protein; the former binds an epitope at residues 220–225, near the carboxyl terminus of the processed prion protein. 21 Hematoxylin and eosin–stained sections were examined from all sections for spongiform degeneration. 29
To confirm the CWD status, one section of obex was examined from each elk (ie, brain stem at the level where the fourth ventricle converges into the central canal of the spinal cord); this section contained the dorsal motor nucleus of the vagus nerve (DMNV), medial retropharyngeal lymph node, and palatine tonsil. In addition, the stage of the disease was estimated by scoring the degree of detectable positive chromogen immunoreactivity in the nuclei and white matter tracts, combined with the degree of spongiform degeneration in this section. The evaluated nuclei included the DMNV, area postrema, solitary nucleus, cuneate nucleus, nucleus of the spinal tract of the trigeminal nerve, nucleus ambiguus, reticular formation area/nucleus, olivary nuclei, medial raphe, and hypoglossal nucleus. 39 The obex was scored as a progressive increase in intensity of PrPCWD immunoreactivity and distribution, essentially as described, 30 with additional classification criteria based on the intensity of spongiform degeneration and PrPCWD in axonal tracts. An obex score of 0 was assigned to elk with no detectable PrPCWD in the brain stem. Scoring of samples with detectable PrPCWD in the obex ranged from 1, characterized by minimal immunoreactivity of the ventral aspects of the DMNV (Fig. 1), to 10, characterized by all the nuclei and axonal tracts containing a moderate to heavy PrPCWD immunoreactivity (Fig. 2). All nuclei had mild to severe spongiform degeneration. Intermediate stages were defined by an increase in immunoreactivity intensity and spongiform degeneration. A detailed description of this scoring system is in preparation.
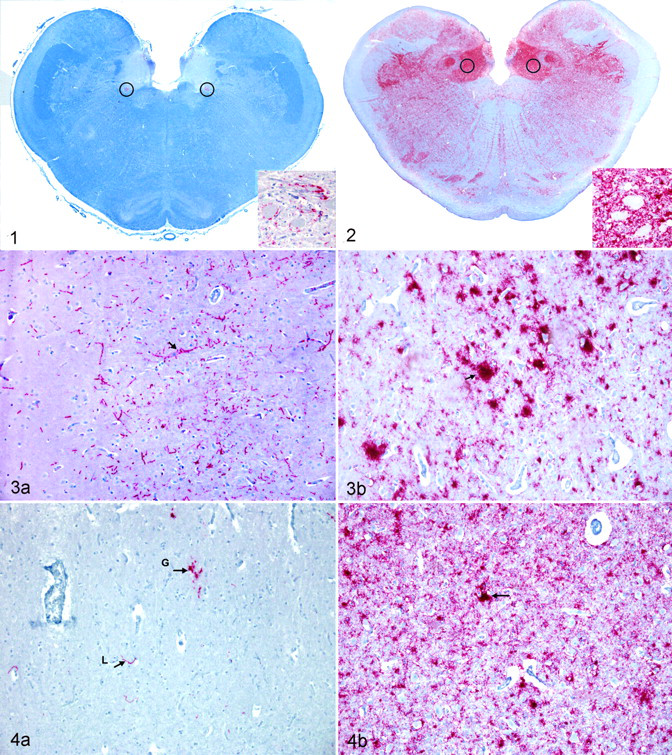
Eyes from each animal and brain sections of the proposed visual pathways were trimmed and embedded in paraffin blocks. The visual pathways within the brain of elk have not been described; therefore, the visual pathways described in humans were applied. The pathway is assumed to include, in order, retina, optic nerve, optic chiasm, optic tract, lateral geniculate nucleus, optic radiations, and visual cortex of the occipital lobe—all of which were sampled. Impulses leaving the lateral geniculate nucleus also travel to the superior colliculus (sampled) and midbrain tectum; these are primarily reflex. The oculomotor nucleus (sampled), even though not directly a part of the visual pathway, was examined because this nucleus controls multiple functions of the eye, including pupillary size. 1,2,26 Immunoreactivity intensity was scored as 0 (no labeling detected), 0.5 (trace amounts of chromogen in some areas of the region), 1 (light labeling within the region), 2 (moderate labeling in the region), and 3 (heavy chromogen deposits throughout the region). Elk were grouped by obex score, and the median value was calculated for each group at each anatomic site in the visual pathway.
Results
Clinical Status and PRNP Genotype of the Test Population
Of the 27 CWD-positive elk, 20 were nonclinical and 7 showed typical clinical signs of CWD at the time of euthanasia. Clinical signs included behavior changes, mild to moderate aggression when cornered, weight loss, excessive salivation, grinding of the teeth, and emaciation. PRNP genotypes in the 25 naturally infected elk included twenty 132MM elk (80%) and five 132ML elk (20%), a ratio similar to that observed in the natural population. 22,24 Elk homozygous for the 132L allele are relatively rare in the population; therefore, 2 experimentally infected animals described in previous studies 8,23 were added to this study.
Obex Scores of Preclinical and Clinically Affected Elk
The obex was scored in all 30 elk based on the distribution and intensity of immunoreactivity combined with the degree of spongiform degeneration. Neither PrPCWD detected by immunoreactivity nor spongiform degeneration was observed in the brain stem from the 3 negative control elk. Of the twenty 132MM CWD-positive elk, obex scores were as follows: one 1, two 2s, one 3, one 4, one 6, three 7s, three 8s, three 9s, and five 10s (Table 1 ). Of the five 132ML-positive elk, the obex scores were as follows, one of each: 4, 5, 6, 8, 10. One 132LL elk had an obex score of 8 and the other, a score of 9. Elk with obex scores of < 9 were clinically normal except for 2 elk, 1 with a score of 8 and the other, a score of 9; both these elk had pneumonia at the time of necropsy.
Rocky Mountain Elk in Which Eyes Were Collected For Examination
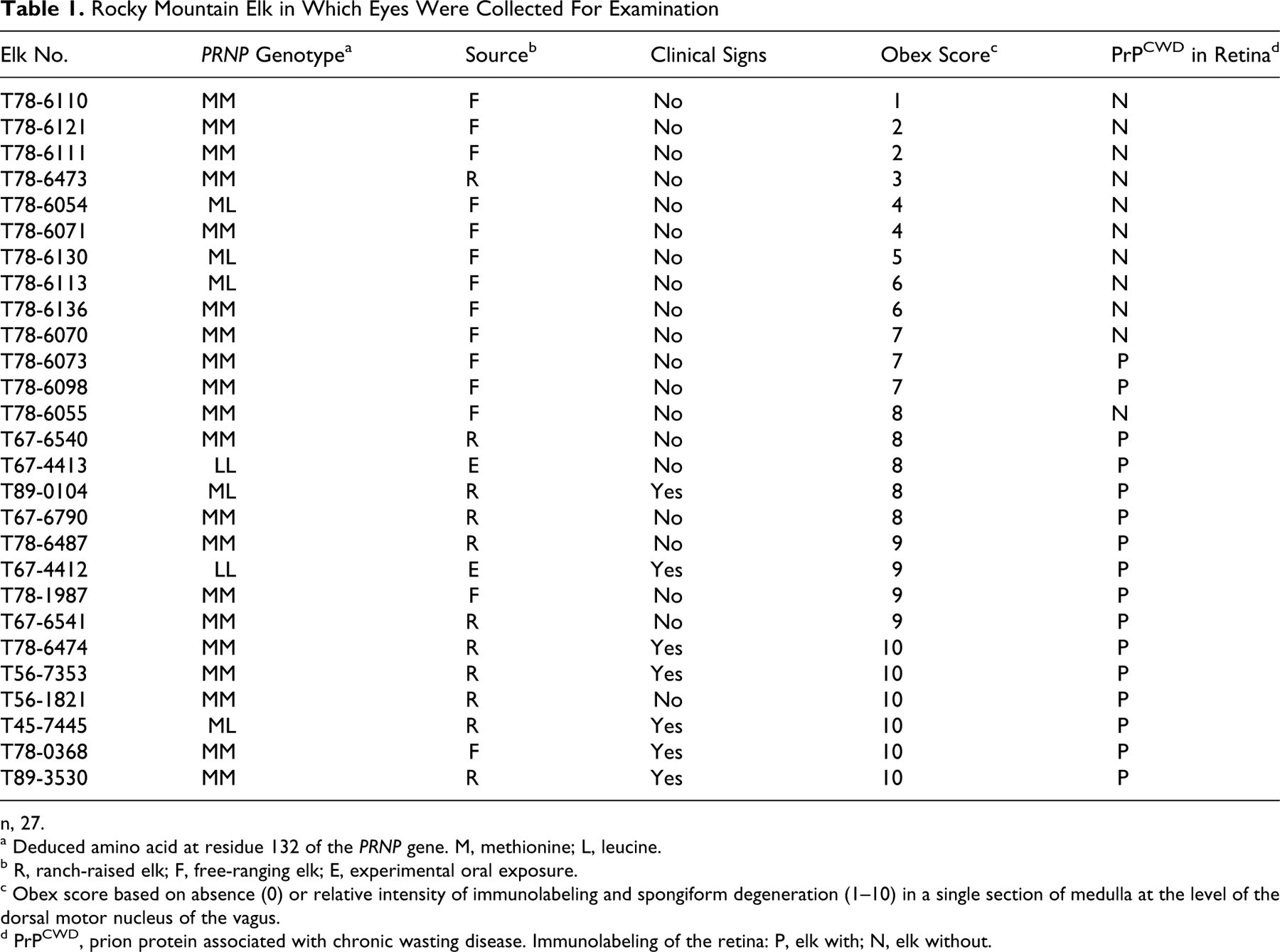
n, 27.
a Deduced amino acid at residue 132 of the PRNP gene. M, methionine; L, leucine.
b R, ranch-raised elk; F, free-ranging elk; E, experimental oral exposure.
c Obex score based on absence (0) or relative intensity of immunolabeling and spongiform degeneration (1–10) in a single section of medulla at the level of the dorsal motor nucleus of the vagus.
d PrPCWD, prion protein associated with chronic wasting disease. Immunolabeling of the retina: P, elk with; N, elk without.
PrPCWD in the Brain and Visual Pathway of Elk at Different Stages of CWD
Immunoreactivity of the visual pathway was scored at 9 neuroanatomic locations in samples from elk with obex scores ranging from 1 to 10. Table 2 shows the median scores for each location in relation to the obex score. The earliest sites of PrPCWD immunoreactivity were the visual cortex (1 of 2) and superior colliculus (2 of 2) in the brain of elk with an obex score of 2. Heaviest PrPCWD immunoreactivity was observed in the superior colliculus, oculomotor nucleus, visual cortex, lateral geniculate nucleus, and retina in elk with an obex score of 9 or 10. At each location, visual pathway scores were higher in elk with higher obex scores. PrPCWD immunoreactivity was detected in the retina of elk with obex scores of 7 or higher. The character and location of the PrPCWD immunoreactivity varied with obex score and genotype, as follows.
Median PrPCWD Immunoreactivity Intensity Scores of Rocky Mountain Elk With Disease Progression as Estimated From Obex Scores (Number of Elk Affected)
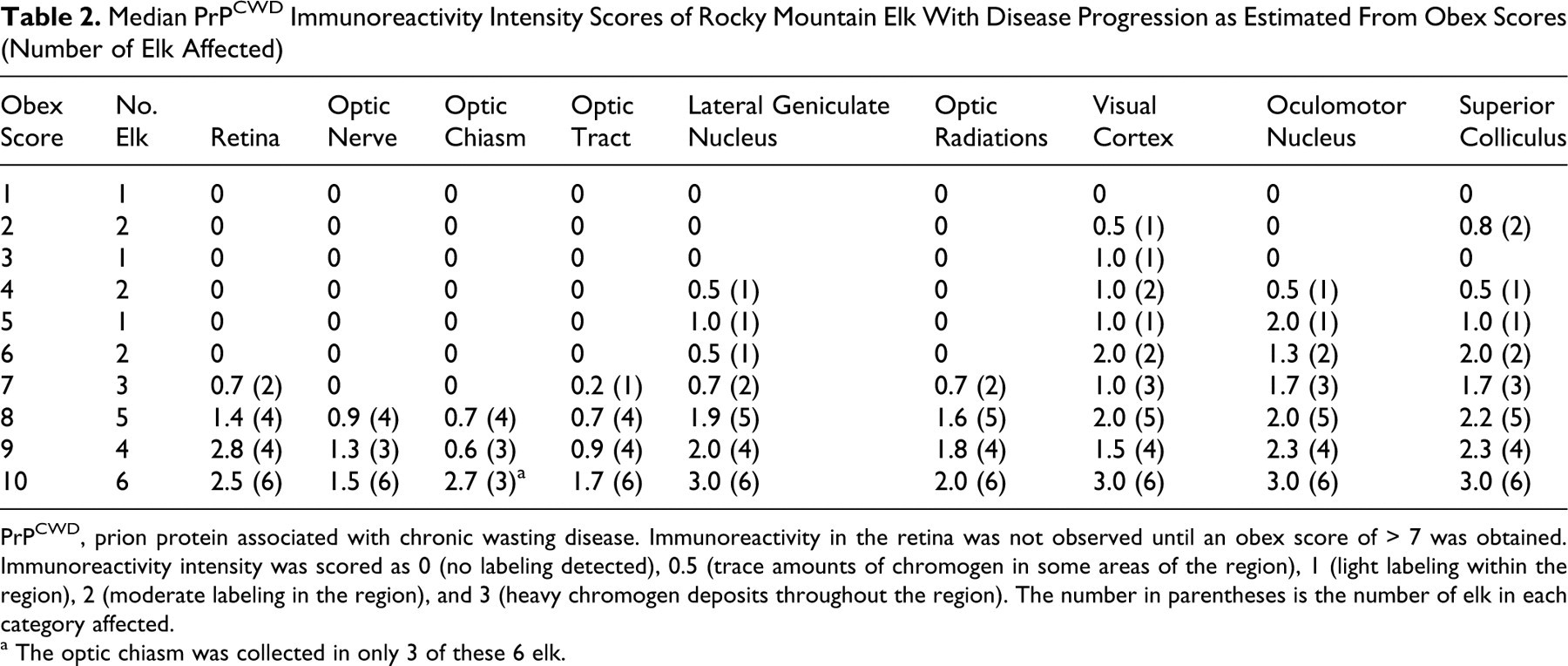
PrPCWD, prion protein associated with chronic wasting disease. Immunoreactivity in the retina was not observed until an obex score of > 7 was obtained. Immunoreactivity intensity was scored as 0 (no labeling detected), 0.5 (trace amounts of chromogen in some areas of the region), 1 (light labeling within the region), 2 (moderate labeling in the region), and 3 (heavy chromogen deposits throughout the region). The number in parentheses is the number of elk in each category affected.
a The optic chiasm was collected in only 3 of these 6 elk.
Obex score = 0
Neither histological lesions nor PrPCWD immunoreactivity was detected in obex, brain, lymphoid tissues, or sections of the visual pathway including the eye of the 3 control elk.
Obex score = 1
One 132MM elk had an obex score of 1. This elk had no detectable PrPCWD immunoreactivity in the visual pathways of the brain or retina. Spongiform degeneration was not found in any of the nuclei of the visual pathway. Histological lesions were not detected in any of the layers of the retina.
Obex score = 2
Two 132MM elk had an obex score of 2. PrPCWD immunoreactivity was detected in the visual cortex of 1 elk and in the superior colliculus in both elk. The pattern of PrPCWD immunoreactivity was considered to be trace and primarily linear. Evidence of spongiform degeneration was not found in any of the nuclei of the visual pathway. Histological lesions or PrPCWD immunoreactivity was not detected in any of the layers of the retina.
Obex score = 3
One 132MM elk had an obex score of 3. PrPCWD immunoreactivity was detected in the visual cortex in this elk (Fig. 3A). The pattern of PrPCWD immunoreactivity was considered to be mild and primarily linear. No evidence of spongiform degeneration was found in any of the nuclei of the visual pathway. Histological lesions or PrPCWD immunoreactivity was not detected in any of the layers of the retina in this elk.
Obex score = 4
Two elk (one 132MM and one 132ML) had an obex score of 4. PrPCWD immunoreactivity was detected in the lateral geniculate nucleus, the oculomotor nucleus, superior colliculus, and visual cortex in the 132MM elk but only in the visual cortex in the 132ML elk. The pattern of PrPCWD immunoreactivity was primarily linear, but coarse granules of chromogen were found in the visual cortex and lateral geniculate nucleus in the 132MM elk and in the superior colliculus in both elk. Spongiform degeneration was confined to the DMNV in both elk and not found in nuclei of the visual pathway. Histological lesions or PrPCWD immunoreactivity was not detected in any of the layers of the retina in these 2 elk.
Obex score = 5
One elk (132ML) had an obex score of 5. PrPCWD immunoreactivity was detected in the lateral geniculate nucleus, superior colliculus, visual cortex, and oculomotor nucleus in this elk. Both linear and granular patterns were found in this stage, but the linear pattern was more prominent. Spongiform degeneration was confined to the obex and not found in nuclei of the visual pathway. Histological lesions or PrPCWD immunoreactivity was not detected in any of the layers of the retina in this elk.
Obex score = 6
Two elk (one 132MM and one 132ML) had an obex score of 6. PrPCWD immunoreactivity was detected in the superior colliculus (Fig. 4A), visual cortex, and oculomotor nucleus in the 132ML elk. PrPCWD immunoreactivity was detected in the lateral geniculate nucleus, superior colliculus, visual cortex, and oculomotor nucleus in the 132MM elk. Both linear and granular patterns were found in these elk, but the linear pattern was more prominent. Spongiform degeneration was confined to the obex and not found in nuclei of the visual pathway. PrPCWD immunoreactivity or histological lesions were not detected in any layer of retina in these 2 elk.
Obex score = 7
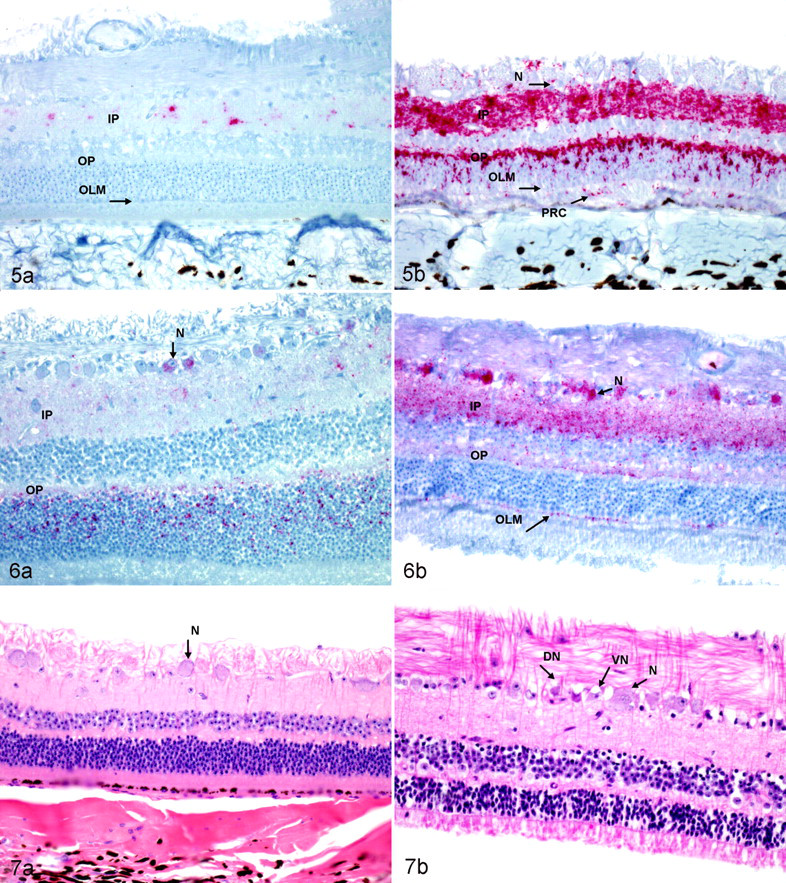
Three elk (132MM) had an obex score of 7. PrPCWD immunoreactivity was detected in the optic tracts of 1 elk; in the retina, lateral geniculate nucleus, and optic radiations of 2; and in the visual cortex, oculomotor nucleus, and superior colliculus of all 3. The patterns of PrPCWD immunoreactivity were predominately granular, but areas of linear staining were still found. An increased number of plaques of PrPCWD immunoreactivity were found in all visual pathways in the brain. In most areas, these plaques appeared to be surrounding and filling the cytoplasm of astrocytes. Mild spongiform degeneration was found within ocular motor nucleus and superior colliculus. PrPCWD immunoreactivity was detected in the inner plexiform layer of the retina in one (Fig. 5A) and in the inner and outer plexiform layer in the other. The pattern of PrPCWD immunoreactivity in the retina of the 132MM elk was characterized by light red granular chromogen staining that was first detected in the inner plexiform layer.
Obex score = 8
Five elk (three 132MM, one 132ML, and one 132LL) had an obex score of 8. PrPCWD immunoreactivity was detected in the retina of two 132MM elk and the 132ML and 132LL elk. PrPCWD immunoreactivity was detected in the optic nerve, optic chiasm, and optic tracts in two 132MM elk. PrPCWD immunoreactivity was detected in the lateral geniculate nucleus, optic radiations, visual cortex, oculomotor nucleus, and superior colliculus in all 5 elk. The pattern of immunoreactivity in these elk was of the coarse granular pattern with an increased number of plaques. Linear staining was minimal. Mild to moderate spongiform degeneration was found in the nuclei of the visual pathways within the brain. PrPCWD immunoreactivity was detected in multiple layers of the retina in all 5 elk. All layers had some degree of PrPCWD immunoreactivity, except the outer limiting membrane and the pigmented epithelium. Layers of the retina with the most intense PrPCWD immunoreactivity were the inner and outer plexiform layers. Intracytoplasmic PrPCWD immunoreactivity within neurons of the ganglion cell layer was present in the 132ML elk but was a prominent feature in the 132LL elk (Fig. 6A, 6B). Intracytoplasmic PrPCWD immunoreactivity within neurons of the ganglion cell layer was not present in the 132MM elk. Small aggregates of PrPCWD immunoreactivity were present on the inner surface of the outer limiting membrane in 132LL elk but not in 132MM or 132ML elk.
Obex score = 9–10
Four (three 132MM and one 132LL) elk had an obex score of 9, and 6 had an obex score of 10 (five 132MM and one 132ML). PrPCWD immunoreactivity was found in all the neuroanatomical visual pathways in the brain (Figs. 3B, 4B). The patterns of PrPCWD immunoreactivity were similar to those described in elk with an obex score of 8. Linear staining was rare. Mild to severe spongiform degeneration was found in the nuclei of the visual pathways in all these elk. PrPCWD immunoreactivity was found in all layers of the retina except the outer limiting membrane and the pigmented epithelial layers in the 132MM elk. Small aggregates of PrPCWD immunoreactivity were present on the inner surface of the outer limiting membrane in 132LL elk but not in the 132MM elk (Fig. 5B). A few ganglia of the retina showed evidence of central chromatolysis, vacuolation, and necrosis (Fig. 7A, 7B) but with slightly more frequency in the elk with a score of 10. The overall degree of spongiform degeneration of the retina was minimal in all genotypes.
Epitope Mapping With Amino Terminal Antibody P4 and Carboxyl Terminal Antibody 99
The intensity and location of PrPCWD immunoreactivity observed with mAbP4 in the brain and retinal sections were similar to those of the brain and retinal sections in the 132MM and 132ML elk stained with mAb99. Immunoreactivity with mAbP4 was identical in character and pattern to that observed with mAb99 except in one location in the 132LL elk. The accumulation of red chromogen granules on the inner surface of the outer limiting membrane as seen in the 132LL elk immunostained with mAb99 was not observed with mAbP4.
Lack of PrPCWD Immunoreactivity in Other Structures in the Eye
PrPCWD immunoreactivity or histological lesions were not found in the lens, ciliary body, iris, cornea, or sclera in any of the 3 genotypes of the 27 CWD-positive elk.
Discussion
The degree of PrPCWD immunoreactivity and spongiform encephalopathy is thought to correlate with the duration of disease. Researchers have suggested that for mule deer and white-tailed deer, the more detectable PrPCWD and severity of spongiform degeneration in neural tissues, the longer the animal has been incubating the disease 6,16,35 ; however, this relationship has not yet been delineated in elk. Neither the pathogenesis nor the dissemination of the agent through the host is well defined in Rocky Mountain elk. In this study, we examined the presumed progression of PrPCWD dissemination along the visual pathway. Detection of PrPCWD immunoreactivity in the retina has been reported in mule deer 35 and white-tailed deer 16 with CWD, ovine scrapie, 4,12 and bovine spongiform encephalopathy, 7 but the pathway and kinetics of prion protein accumulation are not reported. Data from this study demonstrated that detectable PrPCWD immunoreactivity first accumulated in the visual pathways of the brain, before the retina. Elk with an obex score of < 6 had no evidence of PrPCWD immunoreactivity in the optic nerve or retina but did have PrPCWD immunoreactivity in neural regions of the visual pathway in the brain. The first nuclear area where PrPCWD immunoreactivity was detected was in the superior colliculus, followed by the visual cortex (obex score of 2). Next PrPCWD immunoreactivity was detected in the lateral geniculate nucleus and oculomotor nucleus (obex scores of 3 to 6). PrPCWD immunoreactivity was first detected in the retina in elk with an obex score of 7. At this time, PrPCWD immunoreactivity was beginning to accumulate to detectable levels in the axonal tracts of the visual pathway (the optic radiations, optic tracts, optic chiasm, and optic nerve). Elk with an obex score of 7 usually had PrPCWD immunoreactivity in the optic nerve and retina but at much lower intensity as compared to that of the more advanced elk with an obex score of 9 or 10.
Elk with an obex score of < 5 were thought to be in relatively early stages of disease; elk with an obex score of 6 to 7 were thought to be in the midstage of disease; and elk with an obex grade of > 8 were in a more advanced stage. Clinical signs were primarily observed in elk with an obex score of 10; however, 2 elk (one with a score of 8 and the other, a score of 9) did show mild signs, but both had pneumonia at the time of necropsy. In addition to differences in distribution, the immunoreactivity characteristics of PrPCWD were different as the disease progressed. The pattern of PrPCWD immunoreactivity in the brain in early cases was mostly linear, apparently composed of small granular particles of PrPCWD immunoreactivity attached to the periphery of axons or dendrites. The pattern of PrPCWD immunoreactivity in later stages of disease was more granular, including plaque formation.
Retina had detectable PrPCWD immunoreactivity in elk with an obex score of > 7. Prion was first detected in the inner plexiform layer of the retina, then the inner and outer plexiform layers. The prion then spread throughout the remaining retinal layers, sparing the pigmented epithelial layer. In domestic sheep with scrapie, the inner plexiform layer appears to be affected first; then, as the disease progresses, the outer plexiform layer accumulates PrPSc (abnormal prion protein associated with spongiform encephalopathy). 7 Further evidence suggests that as PrPSc accumulates, there are morphologic changes in retinal ganglion cells, rod bipolar cells, and Müller glia that are associated with functional abnormalities. 28 The spread of PrPCWD in the retina of elk may be similar to that of sheep.
The amino acid sequence of the PRNP gene at codon 132 plays a role during the incubation period of elk developing CWD. The 132MM genotype is the most common in free-ranging and captive elk, and it is associated with an estimated incubation period of approximately 2.5 years. The 132ML genotype is not uncommon in captive and free-ranging elk. 24 CWD was identified in this genotype at a frequency in proportion to its abundance in the entire sampled population in a recent study. 24 The experimental incubation period in elk of this genotype was approximately 3.5 years. Finally, 132LL elk are rare in free-ranging and captive elk (< 4%) and have an experimental incubation period of approximately 5 to 6 years or longer. 8,22 The elk in this study (except for the two 132LL elk) had been incubating the disease for an unknown period, and the number of times these elk were naturally exposed was not known.
There were 2 patterns of PrPCWD immunoreactivity in the retinal layers among the 132MM, 132ML, and 132LL elk. Intracytoplasmic accumulation of PrPCWD immunoreactivity within neurons of the ganglion cell layer of the retina was not found in the 132MM elk, but a few neurons with intracytoplasmic PrPCWD immunoreactivity were detected in the 132ML elk. Intracytoplasmic PrPCWD immunoreactivity granules within neurons within the ganglion cell layer were a prominent feature in the 132LL elk. This pattern was identical with the mAb99 and mAbP4. It also appeared that even though the overall severity of retinal degeneration was minimal, it was nearly nonexistent in the 132MM elk and only sporadically observed in the 132ML and 132LL elk. In mouse models, retinal degeneration was noted in mice expressing PrPc (the normal cellular isoform of the prion protein) in astrocytic, microglial, and neuronal cells in the retina but not in mice expressing PrPc solely in neurons, thereby suggesting that microglial/astrocytic prion protein expression may be required for scrapie-induced retinal damage. 17,18 In regard to the elk in this study with naturally occurring CWD, data suggest that the lack of intraneuronal PrPCWD immunoreactivity in the 132MM elk indicates minimal neuronal PrPC expression. However, if leucine is incorporated into the PRNP gene, then neuronal PrPC expression is increased and, therefore, PrPCWD. Likewise, the 132LL genotype may be associated with higher neuronal PrPC expression.
The second difference was the detection of small aggregates of PrPCWD located on the inner surface of the outer limiting membrane in 132LL elk but not in 132MM or 132ML elk. This pattern was observed with mAb99 but not with mAbP4. The outer limiting membrane (or external limiting membrane) is not actually a membrane but a series of junctional complexes between the photoreceptor cells and the Müller cells. The outer limiting membrane is formed by the distal ends of the Müller cells that attach to the photoreceptor cells by zonula adherens–type junctions. The filamentous material associated with these intercellular zonula adherens junctions with light microscopy appears to be a membrane. 5,15 This PrPCWD immunoreactivity at the inner surface of the outer limiting membrane suggests that this junction can impede movement of PrPCWD to some degree in the 132LL elk but not in the 132MM and 132ML elk. This may reflect a functional difference between the Müller cells of 132MM and 132ML elk and those of the 132LL elk.
Further epitope mapping with antibodies specific for the amino and carboxyl termini of the processed prion protein will be needed to demonstrate a difference in intracellular processing of abnormal PrPCWD in tissues of the visual pathway in 132LL elk. We have reported a novel proteinase K cleavage site in PrPCWD extracted from brain homogenate from 132LL elk. 23 In this study, we demonstrate the novel processing in vivo in ganglia of the retina. The reason for these differences was not determined, but it may relate to the mechanisms by which the leucine plays a role in the metabolism or expression of PrPCWD or PrPC within ganglia and Müller cells.
Data from this study suggest that the retina of elk may be affected in the latter stages of CWD, as similar to the findings of white-tailed deer in which PrPCWD was detected in only 4 of 63 CWD-positive white-tailed deer; all 4 white-tailed deer were in advanced disease based on obex scoring. 16
Prions in the nervous system have been found to localize in the presynaptic junctions in the brain, thereby suggesting that prions may have a function at the synaptic junctions in humans suffering with sporadic and variant Creutzfeldt–Jakob disease. 11,20 This may explain why the inner and outer plexiform layers of the retina were affected first and had most of the detectable PrPCWD immunoreactivity. In domestic sheep, PrPSc has been reported to be heavily concentrated in the synaptic layers of the retina (inner and outer plexiform layers). 7,28
In the terminal stages of CWD, there is heavy immunoreactivity in neuronal regions of the brain and retina but low immunoreactivity in such regions the optic nerve, optic chiasm, optic tract, and optic radiations. This finding may be due to the lack of synaptic junctions in these predominately large axonal tracts; therefore, these regions would not produce as much PrPC and, as such, not support as much PrPCWD production or accumulation.
Literature describing clinical signs of CWD has not mentioned visual impairment. 32,37,38 Elk in terminal stages of CWD often appear to see movement, but if one walks slowly or does not move quickly near an affected elk, the elk does not seem to pay much attention (personal observations, T.R.S.). This behavior has been attributed to lesions in the central nervous system rather than in the eye. The literature does suggest that abnormal prion alters localization of synaptic proteins. 25 Contributing to this behavior, by causing visual impairment in elk in the latter stages of CWD, is the accumulation of PrPCWD within the retina—especially in the inner and outer plexiform layers—with neuronal degeneration of the neurons of the ganglia cell layer, as well as PrPCWD accumulation and spongiform degeneration of the nuclei of the visual pathways within the brain.
Footnotes
Acknowledgements
We would like to thank the owners of the ranch-raised elk (Dennis and Stephanie White, Velvet Ridge Ranch, Fort Collins, CO, and two anonymous owners) for allowing us to do rectal biopsies on their elk and, if positive cases were found, euthanizing and necropsying the suspected positive elk. We would like to thank the National Park Service (Rocky Mountain National Park, Estes Park, CO) for allowing us to perform rectal biopsies on elk and allowing us to necropsy positive elk. We wish to thank Scott Ratchford for collecting the eyes from the free-ranging elk. We wish to thank Vicki Jameson, Tracy Nichols, Chrystal Meyerett, Tara Ruby, Amy Graham, Danielle Lagana, and David Walter for assisting in the necropsies of these elk. We wish to thank Bob Zink, Todd Bass, and Bruce Cummins for cutting and staining the tissues. We would like to thank Linda Hamburg and Desiree Lesiak for genotyping these elk. We greatly appreciate the help from Jay Oaks and Charlie Kerlee with the photomicrografts. This project was partially funded by National Park Service project No. CSURM-98, Cooperative Agreement No. H1200040001, task order No. 123400070064; US Department of Agriculture, Agricultural Research Service, Cooperative Agreement No. 58-5348-8-275, project No. 5348-32000-026-105; and Colorado State University Diagnostic Laboratory, College of Veterinary Medicine and Biomedical Sciences, Colorado State University, Fort Collins, CO.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
