Abstract
Chronic wasting disease (CWD) is an infectious transmissible spongiform encephalopathy of cervids associated with the presence of a misfolded prion protein (PrPCWD). Progression of PrPCWD distribution has been described using immunohistochemistry and histologic changes in a single section of brain stem at the level of the obex resulting in scores from 0 (early) to 10 (terminal) in elk with naturally occurring CWD. Here we describe the spread and distribution of PrPCWD in peripheral tissues and spinal cord in 16 wild and 17 farmed Rocky Mountain elk (Cervus elaphus nelsoni) with naturally occurring CWD and correlate these findings with obex scores. Spinal cord and approximately 110 peripheral tissues were collected, processed, stained with hematoxylin and eosin, and immunolabeled with the anti-prion protein monoclonal antibody F99/97.6.1. The medial retropharyngeal and tracheobronchial lymph nodes were the first tissues to accumulate PrPCWD, followed by other lymphoid tissues, myenteric plexus, spinal cord, and finally tissues outside of the lymphatic and neural systems. However, the only significant histological lesion observed was mild spongiform encephalopathy in the dorsal column of the lower spinal cord in elk with an obex score of ≥9. Initial exposure to CWD prions may be through the respiratory system and spread appears to occur primarily via the autonomic nervous system. Therefore, we suggest using obex scores as a proxy for stage of disease progression and verifying with key peripheral tissues.
Chronic wasting disease (CWD) is an infectious transmissible spongiform encephalopathy of wild and farmed cervids.27,63,68,69 Immunohistochemistry (IHC) is the gold standard used to identify the putative infectious agent, a protease resistant isoform of the prion protein (PrPCWD).49,64 Immunohistochemical labeling of PrPCWD, or immunoreactivity (IR), has been identified in brain and head lymphoid tissues in free-ranging and farmed cervids including Rocky Mountain elk (Cervus elaphus nelsoni) 63 , mule deer (Odocoileus hemionus), 63 white-tailed deer (Odocoileus virginianus),27,63 moose (Alces alces), 5 reindeer (Rangifer tarandus tarandus),7,42 and red deer (Cervus elaphus elaphus). 3 The presence of PrPCWD IR has been documented throughout the brain and peripheral tissues in wild and experimentally infected mule deer but not in elk.14,65
A unique pattern of spread of PrPCWD and spongiform encephalopathy has been described in a single section of the obex of the medulla oblongata in elk with naturally occurring CWD that allows categorization of disease progression from extremely early to terminal cases. 61 There may also be a unique pattern of spread of PrPCWD throughout the peripheral tissues in elk correlated with obex scores. The obex scoring protocol may be useful to predict and estimate the presence, abundance, sequence, and timing of accumulation of PrPCWD in peripheral tissues.
The genotype of elk with CWD is important and has been related to the incubation period in experimentally infected elk. The 132MM genotype is the most common in wild and farmed elk and is associated with an estimated incubation period of approximately 2 years in experimentally infected elk. 15 The 132ML genotype is not uncommon in wild and farmed elk, 50 and the incubation period is approximately 3.5 years. 15 The 132LL genotype is rare (<4%) in free-ranging and farmed elk and is associated with a prolonged experimental incubation period of approximately 5–6 years or longer.15,50,51 Genetic factors have also been documented to influence the incubation period in mule deer. 14
The distribution of PrPCWD IR in peripheral tissues of naturally infected preclinical and clinically affected Rocky Mountain elk has not been described. Detection of PrPCWD in peripheral tissues has received little attention but is important because this information could give insight into the pathogenesis and spread of the PrPCWD throughout the body of CWD-positive elk.
The objectives of this investigation were to delineate the distribution of PrPCWD IR in peripheral tissues and spinal cord of elk with naturally occurring CWD and to compare these findings with obex scores to estimate the sequence of PrPCWD accumulation during the course of infection. In addition, we sought information that would suggest plausible routes of exposure to the abnormal prion.
Materials and Methods
Two groups of CWD-positive Rocky Mountain elk, 1 wild (n = 16) and the other farmed (n = 17), were used in this investigation. The wild elk were from Rocky Mountain National Park (40°22′30″/105°30′00″) in north-central Colorado; the farmed elk were from 5 ranches located in northern Colorado (Supplemental Table 1).
Wild Elk
During December and January of 2008–2011, 79 apparently healthy wild Rocky Mountain elk were chemically immobilized and euthanized for management and research purposes. 43 Carcasses were submitted to the Colorado State University Diagnostic Medical Center, Fort Collins, Colorado, for postmortem examination within 2–5 hours of death.
Neural and approximately 110 non-neuronal tissues were collected from each elk and fixed in 10% neutral buffered formalin. Tissues included the nervous (brain, spinal cord, vagus nerve, axillary nerve, sciatic nerve, and small peripheral nerves), lymphoid, endocrine, cardiovascular, digestive, urogenital, respiratory, integument, and special sensory (eyes) systems. One middle incisor tooth was taken for determination of age using cementum annuli counting. 28 The brain stem at the level of the obex, medial retropharyngeal lymph nodes, and pharyngeal tonsils were examined in all elk with hematoxylin and eosin (H&E) stain and immunolabeled with the anti-prion protein monoclonal antibody F99/97.6.1 to detect the presence or absence of the CWD prion. Sixteen of the 79 wild elk were diagnosed positive for CWD based on detection of PrPCWD IR in at least 1 of the above tissues and were included in this study (Supplemental Table 1).
A section of obex was scored on each CWD-positive elk. The obex score is a grading system that uses the progressive increase in the presence and accumulation of PrPCWD IR in 10 nuclei, 10 white matter tracts, glia limitans, and ependyma combined with the progressive increase and severity of spongiform degeneration in 10 nuclei in the brain stem at the level of the obex. 61 This procedure can delineate the disease progression from 0 (extremely early, lymphoid positive/brain negative) to 10 (terminal) stages. 61
The appearance of PrPCWD IR in tissues was characterized by bright red granular material. Lymph and hemal nodes were scored on a scale of 1–3; score 1+ (mild) less than 10% of the follicles positive, score 2++ (moderate) between 10% and 25% of the follicles positive, and score 3+++ (strong) more than 25% of the follicles positive (Supplemental Figs. 1a–c). The scoring of the nonlymphoid tissues was difficult to quantify but was done according to organ type and subjectively graded on a scale of 1–3; score 1+ (mild), 2++ (moderate), and score 3+++ (strong).
One wild female elk was euthanized after being hit by a car. This elk had no detectable PrPCWD IR within lymph nodes of the head and the obex and was used as 1 of the 3 negative controls. All tissues examined in the CWD-positive elk were also examined in the control elk.
Farmed Elk
Between 2005 and 2011, approximately 1700 rectal biopsies were taken from approximately 700 farmed elk from 5 elk ranches located in northern Colorado to validate the utility of the rectal biopsy to detect CWD in nonclinical elk. 62 Seventeen of these elk had PrPCWD IR in the lymphoid follicles of the rectal biopsy tissues. These elk were classified as positive for CWD and were included in this study. These elk were immobilized and euthanized as described for the wild elk and submitted to the Colorado State University Diagnostic Medical Center for a postmortem examination. Postmortem intervals were approximately 1–3 hours except for 1 elk that was found freshly dead. The same tissues were collected, stained and immunolabeled, and examined from the farmed elk as from the wild elk. Ages were determined via cementum annuli counting, as in the wild elk. 28 Two farmed female elk that had traumatic injuries were euthanized. These elk had no detectable PrPCWD IR in the obex, medial retropharyngeal lymph nodes, or palatine tonsil and were used as negative controls.
Tissue Processing and Evaluation
Tissues from all organ systems from CWD-positive and CWD-negative control elk were processed for evaluation. Tissues were fixed in 10% neutral buffered formalin, trimmed for histology, sectioned at 5 µm, stained with H&E, and immunolabeled with the anti-prion protein monoclonal antibody F99/97.6.1 (mAb 99) using the Ventana platform. 64 Positive and negative control slides containing lymphoid tissue and brain were immunolabeled with each run of 30 slides. Approximately 50–60 slides were prepared from each elk.
Genetics
The amino acid sequence of the prion protein gene (PRNP gene) at codon 132 influences the incubation period of CWD in elk.15,51 The open frame of the PNRP gene was determined for each elk and reported as 132MM, 132ML, or 132LL. 50 The protocol for the precise methodology of sequence analysis applied to these elk has been published in detail. 50
Variation in prion strains has been described.16,44,47,52 While strain investigation of the prion in these animals would be interesting, it was outside of the scope of this work.
Results
Sixteen wild and 17 farmed CWD-positive elk were identified and used in this investigation. Eleven of the 33 CWD-positive elk (1 wild and 10 farmed) were in poor body condition, had serous atrophy of fat surrounding the kidneys, coronary vessels of the heart, within the bone marrow, behind the eyes, and around the spinal cord and were in the latter stages of CWD. In addition, on gross exanimation, these elk had mild megaesophagus, a rumen filled with feed, small and large intestine contained a small volume of contents, and a dilated rectum filled with firm pellets. Elk with these gross lesions were in terminal stages of CWD and had an obex score of 9 or 10. The remaining 22 elk were subjectively considered to be in good to fair body condition, based on the amount of subcutaneous and abdominal fat. The 1 farmed elk that was found freshly dead had a subacute suppurative bronchopneumonia (case 28).
Demographic features were similar between the groups of wild and farmed elk. The 16 wild elk were all females. Eleven of the 16 wild elk had the132MM genotype and 5 were 132ML (Supplemental Table 1). The age range of the 11 wild 132MM elk was 2–16 years with an average age of 6.4 years. The age range of the 5 wild 132ML elk was 5–8 years with an average age of 6.4 years. Of the 17 farmed elk, 15 were 132MM genotype (5 males and 10 females) and 2 were 132ML genotype (1 male and 1 female). The ages of the fifteen 132MM farmed elk ranged from 1.5 to 13 years with an average age of 4.5 years. The ages of the two 132ML farmed elk ranged from 6 to 7 years with an average age of 6.5 years. Six of the wild elk and 4 of the farmed elk were pregnant. Elk with a 132LL genotype was not identified in either wild or farmed elk (Supplemental Table 1).
Obex scores in the wild elk ranged from 0 (extremely early cases/lymphoid positive/brain negative) to 9 (near terminal) and from 1 (extremely early cases) to 10 (terminal) in the farmed elk, thereby providing a wide range of scores for evaluation of peripheral tissues and spinal cord. Appreciable differences in the appearance, distribution, and abundance of PrPCWD IR were not found between wild and farmed elk with similar obex scores; therefore, the results between the 2 groups of elk were combined.
The anatomical structures discussed below have not been described in elk. Therefore, literature describing these anatomically structures in domestic cattle, sheep, goats, and dogs were used and assumed to be similar in elk.19,20,31–36
Control Elk
All 3 negative control elk had the132MM genotype and were free of PrPCWD IR in all tissues examined.
Obex Score 0
Five female elk (cases 1–5) had an obex score of 0. Four had the PRNP genotype of 132ML and 1 had the PRNP of 132MM. The ages of these 5 elk ranged from 5 to 10 years with an average of 7 years. These elk had mild to moderate PrPCWD IR in the medial retropharyngeal lymph node with no detectable immunolabeling in 2 sections of the parasympathetic nucleus of the vagus nerve (PSNV), formally called the dorsal motor nucleus of the vagus. 31 Occasionally, only 1 of the 2 sections of this lymph node was positive in the 132ML elk. In addition to the medial retropharyngeal lymph node, lymphoid follicles with mild labeling were found in the parotid lymph node in 1 of the 132ML elk. The one 132MM elk had PrPCWD IR in the medial retropharyngeal, prescapular, tracheobronchial, and ruminal lymph nodes. Few follicles were positive in these nodes except for the tracheobronchial node in which approximately half of the 100 follicles were positive. The PrPCWD IR was characterized by red granular staining within the lymphoid follicles (Fig. 1a, b). All other tissues in these 5 elk were free of PrPCWD IR (Table 1).
Summary of multiple tissues in which PrPCWD IR was detected in 33 Rocky Mountain elk with naturally occurring CWD as classified according to their obex scores.
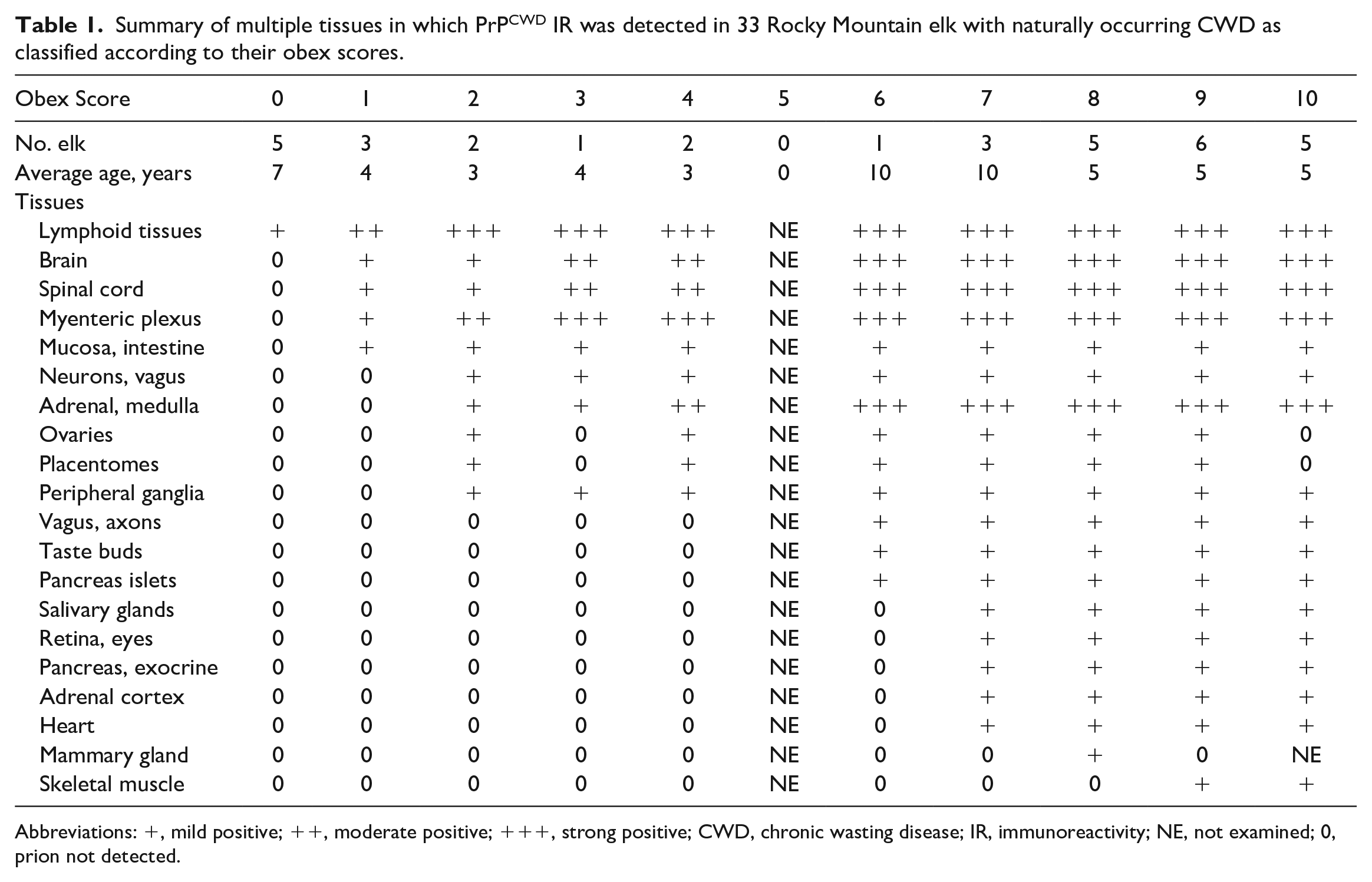
Abbreviations: +, mild positive; ++, moderate positive; +++, strong positive; CWD, chronic wasting disease; IR, immunoreactivity; NE, not examined; 0, prion not detected.
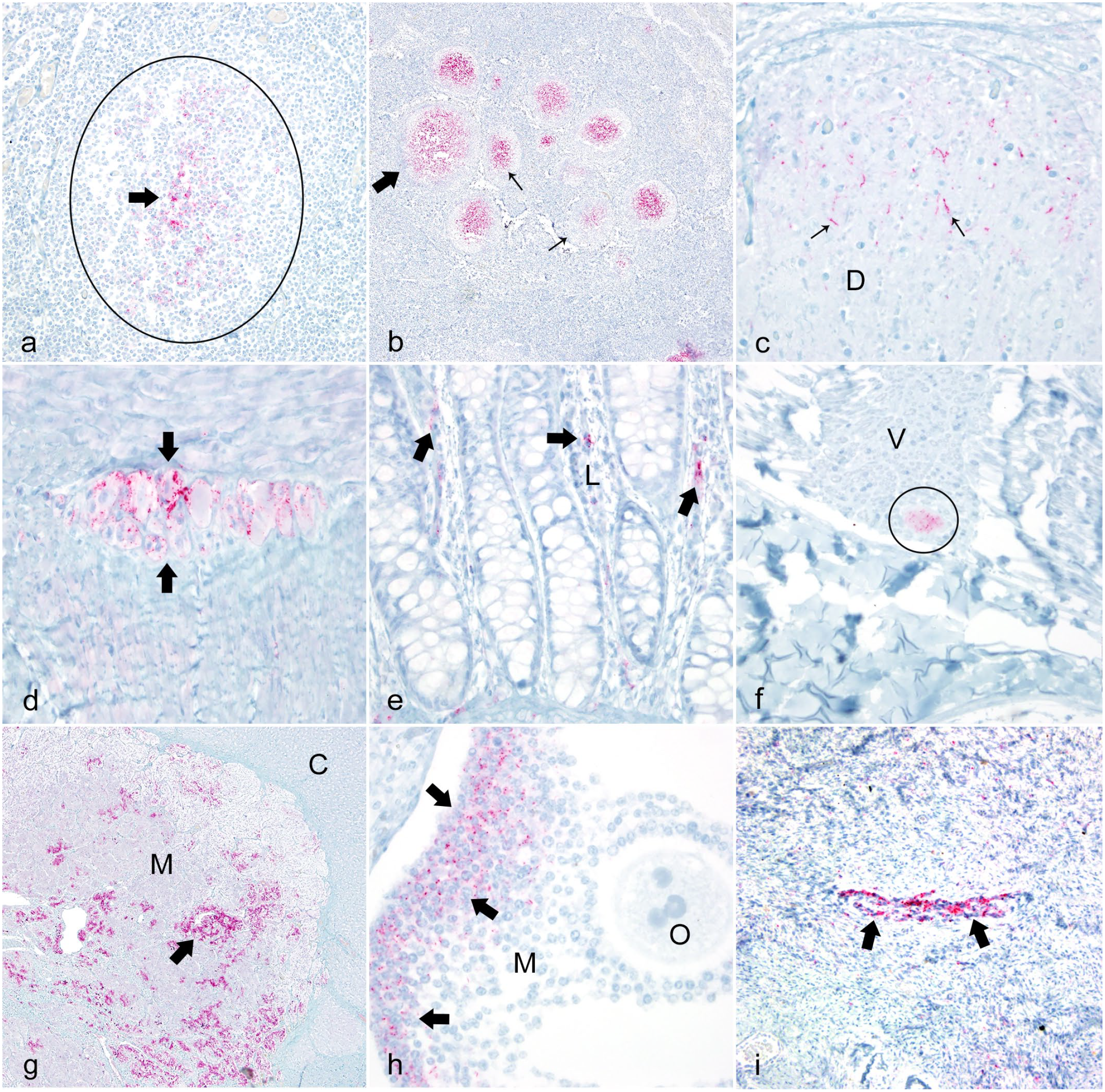
Photomicrographs documenting positive chronic wasting disease (CWD) prion protein immunoreactivity (PrPCWD IR) in Rocky Mountain elk with natural occurring CWD in multiple tissues. Positive labeling is characterized by red chromogen granules. Immunohistochemistry with streptavidin-alkaline phosphatase method and hematoxylin counterstain.
Obex Score 1
Three elk (two 132MM and one 132ML genotype, cases 6–8) had an obex score of 1. These elk had mild PrPCWD IR on the periphery of a few neurons in the ventral lateral aspects of the middle region of the PSNV. PrPCWD IR was more pronounced and widespread in both internal and peripheral lymphoid tissues in the 2 elk with the 132MM genotype. A typical section of lymph node in these elk contained approximately 20–100 lymphoid follicles depending on the size, direction of cut, and location of the node, while a typical section of hemal node contained approximately 4–15 lymphoid follicles depending on the size. Sections of lymph and hemal nodes of the head, thoracic cavity, abdominal cavity, and peripheral locations contained PrPCWD IR in the 132MM elk; however, the distribution of PrPCWD IR varied in multiple sections of the same node. Occasionally, only 1 or 2 positive follicles could be found in 2 sections of the same node. Even though most of the lymph and hemal nodes had detectable PrPCWD IR, the pattern of positive follicles was patchy, similar to the elk with an obex score of 0. Nevertheless, the most consistent strongly affected nodes containing PrPCWD IR were the medial retropharyngeal and tracheobronchial nodes. Lymphoid PrPCWD IR was mild in the 132ML elk and was only found in the tracheobronchial lymph node. All other locations including the medial retropharyngeal node and the gut-associated lymphoid tissues (GALTs) were free of detectable PrPCWD IR. Palatine tonsils were free of PrPCWD IR in all 3 elk with an obex score of 1 (Supplemental Table 2).
Positive lymphoid follicles were found in the submucosa of the ileum and cecum and the adjacent mesenteric lymph nodes in the two 132MM elk, but not in the 132ML elk. PrPCWD IR was not detected in any of the other peripheral tissues in these 3 elk (Table 1 and Supplemental Table 2).
One of the 132MM elk had mild PrPCWD IR within the dorsal column of the upper sacral spinal cord, whereas the other 132MM elk had mild PrPCWD IR in the dorsal and intermediolateral columns of the lumbar and thoracic regions of the spinal cord (Fig. 1c). PrPCWD IR was not present in the spinal cord in the one 132ML elk (Table 1 and Supplemental Table 3).
PrPCWD IR was found in the myenteric and submucosal plexuses of the jejunum, ileum, cecum, spiral colon, and rectum in both 132MM elk (Fig. 1d) but was only found in the myenteric plexuses of the rumen in the 132ML elk. Granules of PrPCWD IR were found within the lamina propria of the cecum in one 132MM elk and in the cecum, spiral colon, terminal colon, and rectum in the other 132MM elk (Fig. 1e), but not in the 132ML elk. PrPCWD IR appeared to be associated with the short postganglionic fibers leaving the myenteric and submucosal parasympathetic ganglia and entering the lamina propria of the adjacent mucosa (Table 1, Supplemental Table 4).
Obex Score 2
Two female elk (cases 9–10) with a 132MM genotype had an obex score of 2 with moderate PrPCWD IR within the PSNV. PrPCWD IR was relatively widespread in both internal and peripheral lymph and hemal nodes in both elk. The overall amount of PrPCWD IR within lymphoid tissues was more abundant in elk with an obex score of 2 as compared with elk with an obex score of 1. As in the elk with an obex score of 1, the distribution of PrPCWD IR varied greatly even in multiple sections of the same lymph node and the pattern of positive follicles was multifocally distributed throughout the node. Palatine tonsils were free of PrPCWD IR, but the GALTs were heavily positive in both elk. PrPCWD IR was detected in the spleen of both elk but was not detected in the thymus (Table 1, Supplemental Table 2).
One of these elk (case 9) had mild PrPCWD IR within the dorsal column of the lower lumbar/sacral spinal cord, whereas the other (case 10) had mild PrPCWD IR in the dorsal and intermediolateral columns of the lumbar and thoracic spinal cord and mild PrPCWD IR on the surface of large neurons in the dorsal root ganglia of the lower lumbar spinal cord. Neither elk had PrPCWD IR within the cervical spinal cord. In addition to the PrPCWD IR documented in elk with an obex score of 1, PrPCWD IR was located within or on the periphery of a few neurons in the vagus nerve of 1 elk (Fig. 1f) but was not found in the other elk. Even though PrPCWD IR was detected on the cell membranes of neurons within the vagus nerve, PrPCWD IR was not detected within or between these large, myelinated axons of the vagus nerve (Table 1, Supplemental Table 3).
PrPCWD IR was found in the myenteric and submucosal plexuses in 5 locations (jejunum, ileum, cecum, terminal colon, and rectum) in 1 elk (case 9) and 9 locations (esophagus, rumen, abomasum, jejunum, ileum, cecum, spiral colon, terminal colon, and rectum) in the second elk (case 10). Small granules of PrPCWD IR were found within the lamina propria in 2 locations (cecum and rectum) in the first elk and in 6 locations (jejunum, ileum, cecum, spiral colon, terminal colon, and rectum) in the second elk (Table 1, Supplemental Table 4).
PrPCWD IR was detected in the adrenal medulla in 1 elk (Fig. 1g). One elk was pregnant with an estimated 4- to 5-month-old fetus. Moderate PrPCWD IR was detected in the membrana granulosa of secondary follicles in the ovaries (Fig. 1h). A few follicles had mild PrPCWD IR in the theca externa but not in the theca interna. Moderate PrPCWD IR was detected in medium (Fig. 1i) and small autonomic nerves in the cortex of the ovary. Mild to moderate PrPCWD IR was detected within the trophoblastic fetal cells multifocally in the chorioallantois villi in the placenta (Fig. 2a). PrPCWD IR was not found in the brain and lymphoid tissues of this fetus using IHC. The second elk was not pregnant and PrPCWD IR was not found in the ovaries or uterus (Table 1, Supplemental Table 5).
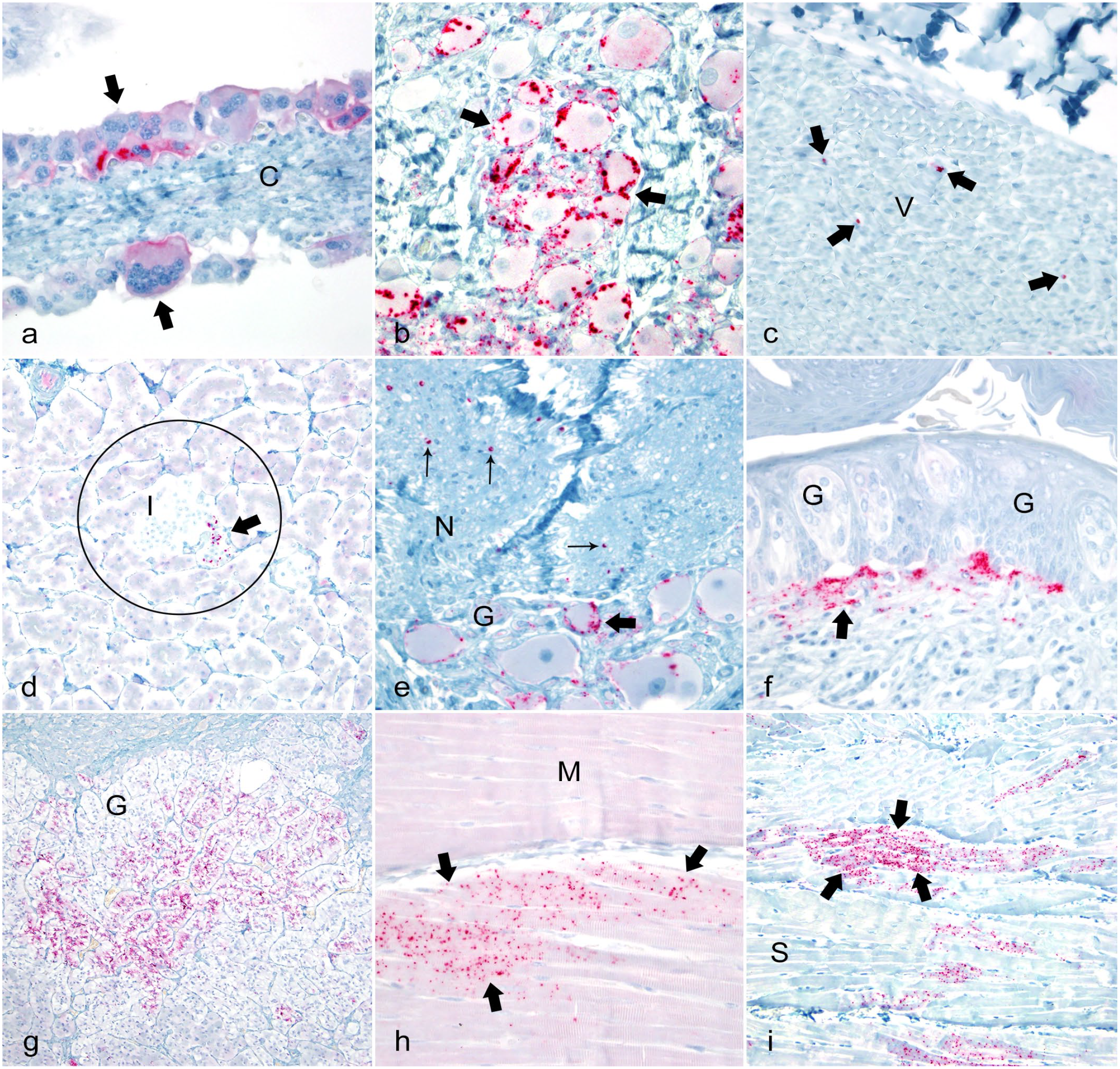
Photomicrographs documenting positive chronic wasting disease (CWD) prion protein immunoreactivity (PrPCWD IR) in Rocky Mountain elk with natural occurring CWD in multiple tissues. Positive labeling is characterized by red chromogen granules. Immunohistochemistry with streptavidin-alkaline phosphatase method and hematoxylin counterstain.
Obex Score 3
One elk with a 132MM genotype (case 11) had an obex score of 3. This elk had a mild to moderate PrPCWD IR within the dorsal and intermediolateral columns of the lumbar, thoracic, and cervical spinal cord. This elk also had mild PrPCWD IR in the isthmus and between large myelinated axons of the white matter tracts of the lumbar spinal cord, but not in the thoracic or cervical regions. In addition to PrPCWD IR in the same tissues as elk with an obex score of 2, this elk also had moderate PrPCWD IR on the surface of neurons (assumed to be the synaptic junctions) within the celiac ganglia (Fig. 2b). This elk was pregnant, but PrPCWD IR was not found in the ovary, placentomes, or fetus. The overall amount of PrPCWD IR was slightly more abundant in these tissues as compared with elk with an obex score of 2 (Table 1).
Obex Score 4
Two elk with a 132MM genotype (cases 12–13) had an obex score of 4. PrPCWD IR was located in all the same peripheral tissues as found in the elk with an obex score of 3, but immunolabeling appeared to be more abundant. One of these elk had PrPCWD IR in the ventral column of the lumbar region of the spinal cord. One elk was pregnant and had mild PrPCWD IR in the ovaries, including the luteal cells and the fetal trophoblasts of the placentomes. PrPCWD IR was not detected in the fetal brain or lymphoid tissues using immunolabeling.
Obex Score 5
Elk with an obex score of 5 were not identified during this study.
Obex Score 6
One female elk with a 132MM genotype (case 14) had an obex score of 6. In addition to the locations where PrPCWD IR was detected in elk with an obex score of 4, mild PrPCWD IR was detected on the periphery, or between the large, myelinated axons, in the vagus nerve (Fig. 2c). Mild PrPCWD IR was detected in the islets of Langerhans in the pancreas (Fig. 2d), but not within the acini. Mild to moderate PrPCWD IR was detected in autonomic ganglia in the submandibular salivary glands (Fig. 2e) and the tongue. Strong PrPCWD IR was detected in the small nerves at the base of the circumvallate papillae taste buds and mild PrPCWD IR was detected in the terminal ends of these nerves within the bundles of gustatory and sustentacular cells (Fig. 2f). Mild PrPCWD IR was detected in the paravertebral ganglia of the sympathetic trunk and within the stellate ganglia. This elk was pregnant and mild PrPCWD IR was detected in the ovary and placentomes, but not in fetal tissues (Table 1, Supplemental Table 3).
Obex Score 7
Three female elk with a 132MM genotype (cases 15–17) had an obex score of 7. In addition to the locations of PrPCWD IR found in the elk with lower obex scores, mild immunolabeling was detected around neurons of the trigeminal ganglion in 1 of these elk. PrPCWD IR was detected within the retina and optic nerve. 60 PrPCWD IR was detected in the adrenal cortex in 2 elk (Fig. 2g). Mild PrPCWD IR was detected in the myocardial cells in the left free ventricular wall of the heart in 1 elk (Fig. 1g), but not in the Purkinje fibers, the right free ventricular wall, or the right or left atria. Mild PrPCWD IR was detected within the pancreatic acinar cells in 1 elk. Two of these elk were pregnant. Mild PrPCWD IR was detected in the ovaries and within the trophoblasts of the placentomes. Mild PrPCWD IR was detected in parenchymal cells of the submandibular salivary glands in 2 elk.
Obex Score 8
Five elk, 4 (3 females and 1 male) with a 132MM genotype and 1 female with a 132ML genotype (cases 18–22), had an obex score of 8. PrPCWD IR was detected in similar tissues as detected in elk with obex score 7 with the additional finding of a few granules of PrPCWD IR in the mammary glands in 1 of the 132MM elk. Mild PrPCWD IR was detected in the ovaries and in the trophoblasts of the placentomes in one of the two 132MM pregnant elk, but not in fetal tissues.
Obex Score 9
Six elk (4 females and 2 males) with a PRNP 132MM (case 23–28) had an obex score of 9. In addition to the locations PrPCWD IR was detected in elk with obex score 8, mild PrPCWD IR was detected in skeletal muscle myocytes in cervical muscles, biceps femoris, semitendinosus, and diaphragm in 1 elk, but not in the other 5 (Fig. 2i). PrPCWD IR was not detected in the nerves supplying these muscles. One of these elk had mild spongiform degeneration in the dorsal column of the lumbar spinal cord.
Obex Score 10
Five elk, 4 (2 females and 2 males) with a 132MM genotype and 1 male with a 132ML genotype (cases 29–33), had an obex score of 10. These were terminal cases and the distribution of PrPCWD IR was typified by widespread immunolabeling. All tissues demonstrating PrPCWD IR in elk with an obex score of 9 were also positive in elk with an obex score of 10, but tissues had slightly more labeling. Spongiform degeneration was also found in the dorsal columns of the spinal cord in 2 of the 132MM elk. These elk were showing clinical signs and had gross lesions typical of terminal cases of CWD (Table 1).
Numerous, but not all, tissues in elk with terminal CWD exhibited PrPCWD IR. Tissues free of PrPCWD IR in the terminal stage included the nasal mucosa covering the ethmoid or nasal turbinates, trachea, large to small bronchi and alveoli of the lungs, parenchyma and bile ducts of liver, large myelinated nerves (axillary and sciatic) except for the vagus nerve, medium and small peripheral nerves (exceptions: small autonomic nerves of the submucosa of the tongue, intestinal tract, and ovary), muscular walls of blood vessels, kidney, urinary bladder, thyroid gland, thymus, bone marrow, cortical bone, seminiferous tubules and spermatids of the testis and epididymis, epidermis of the skin, hair follicles, tarsal glands, and the large sebaceous gland of the tail (specific gland in elk).
Discussion
This investigation provides the first description of PrPCWD distribution in peripheral tissues and spinal cord from extremely early to terminal cases of naturally occurring CWD in elk. This work was made possible by correlating progressive obex scores with detection of PrPCWD IR in peripheral tissues. This information provides insight into the pathogenesis and progressive spread of PrPCWD throughout the body. A longitudinal study design in which elk were sampled through time following known disease exposure would be ideal; however, such an approach can only be performed with experimental challenges that could introduce bias, such as, route of exposure, volume of dose, number of exposures, age of animals, and perhaps even nutrition. Even though the number of elk in this study was small (n = 33), this information is valuable, as it is rare to necropsy naturally infected elk with a wide range of obex scores (0–10) with minimal autolysis.
One limitation of this study is that the incubation times that correlate with the obex scores is not known; however, suggested time frames of the obex scores were obtained from the examination of retropharyngeal lymph nodes and obex from an investigation that was done in 2008 by Dr Scott Wright (retired, at the time of the project, he was working with the United States Geological Survey).70 He allowed 1 of the authors (TRS) to examine the obex and retropharyngeal lymph nodes from 10 orally infected elk with CWD-positive elk brain. This study included three 132MM elk euthanized at 6 months postinfection, 2 elk had obex scores of 0 and 1 elk had an obex score of 1; two 132MM elk euthanized at 12 months postinfection had obex scores of 2 and 3; 3 elk euthanized at 18 months of which two 132MM genotype elk had an obex score of 7 and 1 elk with a 132ML genotype had an obex score of 4; and, finally, two 132MM elk euthanized at 24 months had obex scores of 8 and 9. Even with limited numbers, this project suggests a rough estimation of the time frames for the different obex scores. These data also demonstrate that the progression of PrPCWD in 132MM elk progresses more rapidly as compared with 132ML.
In elk with an obex score of 0, the medial retropharyngeal and tracheobronchial lymph nodes were the first and most consistently affected with PrPCWD IR. The submandibular, parotid, and the lateral retropharyngeal lymph nodes were affected to a lesser degree. All other peripheral and internal lymph nodes and GALT were free of PrPCWD IR in elk with an obex score of 0. More lymphoid tissues were found positive in the 132MM elk as compared with the 132ML elk. This may suggest that the progression of the prion in 132MM elk is faster as compared with the 132ML elk.
Afferent lymphatic vessels from the medial retropharyngeal lymph nodes primarily drain the nasal and pharyngeal mucosa and larynx.19,58 In addition, the parotid, submandibular, and lateral retropharyngeal lymph nodes also receive afferent lymphoid drainage from the head and oral cavity. The tracheobronchial lymph node primarily receives afferent lymphatics from the trachea and lungs.19,58 Because these nodes were affected before any of the other tissues in the body, perhaps 1 important initial route of infection could be inhalation of dust particles coated with abnormal prion as described in white-tailed deer 47 and cervidized mice. 12 The prion may attach to the nasal, oral, or respiratory mucosa and then be transported to the head and respiratory lymph nodes. These nodes have efferent vessels that enter the blood via the thoracic duct. 58 If prion enters the body via the nasal cavity or lungs, this could explain why the palatine tonsil was not infected in extremely early cases of CWD in these elk, given the palatine tonsils primarily filter ingested and regurgitated plant material and fluids from the rumen.
Early PrPCWD accumulation within the medial retropharyngeal lymph node has been previously reported in elk with naturally occurring CWD.63,59 Similar results have been observed in experimentally infected mule deer in early infections following oral inoculation with heavy doses of CWD-infected brain.14,56,57 PrPCWD accumulation was observed in 1 of the 3 elk experimentally infected via oral inoculation of CWD-infected brain and euthanized at 6 months postinfection. This elk had PrPCWD IR within the medial retropharyngeal lymph node, but PrPCWD IR was not detected in the PSNV or lymph nodes of the head (obex score of 0) including the palatine tonsil.
One potential alternate route for prions from the head lymph nodes and oral cavity to the brain is along nerves. The primary innervation of the retropharyngeal lymph nodes and palatine tonsils is by the glossopharyngeal nerve (IX) which leads to the solitary nucleus of the brain stem.34,31 The first region of the brain to accumulate PrPCWD is the ventrolateral aspect of the middle one third of the PSNV, not the solitary nucleus. Autonomic innervation of the oral cavity is primarily by the trigeminal nerve (V), with small nerves from the facial (VII) and vagus (X).34,31 The nuclei of the trigeminal nerves and facial nerves are affected later in the course of the disease in elk. The sensory and taste neurons of the vagus innervate the posterior aspects of the tongue and originate in the posterior aspects of the PSNV rather than the middle half of the PSNV.19,31,33 Motor nerves of the tongue originate from the neurons of the hypoglossal nucleus (XII), which is affected later in the progression of disease. 61 PrPCWD did not appear to travel in nerves from the nasal or oral cavity and head lymph nodes to the brain.
Three elk had an obex score of 1 which was characterized by mild PrPCWD IR in the PSNV. The PSNV contains preganglionic parasympathetic neurons that send efferent fibers in the vagus nerve and control glandular activities in the pharynx, larynx, trachea, bronchi, and numerous organs in the digestive system. The efferent fibers in the PSNV are topographically organized. The rostral aspect of the nucleus sends efferent parasympathetic nerve fibers to the abdominal organs, the middle aspects of the nucleus send fibers to the abdominal and thoracic organs, and the caudal region sends fibers to cervical glands.19,33 Moreover, the PSNV receives afferent fibers from the lower colon and urinary and reproductive systems via the neurons of the dorsal root ganglia of the sacral spinal cord and sends fibers to the brain, primarily the brain stem and hypothalamus.19,31,33
In 1 of the 132ML elk, PrPCWD IR was detected in the dorsal column of the upper sacral spinal cord. The dorsal column of the spinal cord in this region contains small proprioceptive neurons with axons that receive autonomic fibers from abdominal organs and send fibers mostly anterograde within the spinal cord. Taken in totality, this suggests that PrPCWD invasion into the spinal cord first affects the lower lumbar/upper sacral regions and then progresses antegrade in the spinal cord. This was further documented in elk with higher obex scores. This suggests there is an early connection between peripheral autonomic nerve fibers and spinal cord which could play a role in the pathogenesis of CWD in elk.
In addition to immunolabeling in the head and respiratory lymph nodes, as found in elk with an obex score of 0, the elk with an obex score of 1 had PrPCWD IR in the follicles of thoracic, abdominal (mesenteric and GALT), and peripheral lymph nodes, and the hemal nodes and spleen. PrPCWD IR was found in these locations in both 132MM elk, but not in the one 132ML elk. The finding of PrPCWD IR in numerous peripheral lymph nodes in the 132MM elk and not in the peripheral nodes of 132ML may suggest that the spread, and perhaps the amplification, of prion in peripheral tissues may be slower in 132ML elk. These data are consistent with data demonstrating the incubation period in 132MM elk is faster than in 132ML elk.15,51 The head and pulmonary lymph nodes drain into the thoracic duct and then into the vena cava. Peripheral nodes and spleen of infected elk with an obex score of 1 may have been exposed when prions spread via the blood.
Occasionally, PrPCWD IR can be found in the PSNV without IR in the medial retropharyngeal lymph node. This scenario was found in one 132ML elk with an obex score of 1. Lymphoid PrPCWD IR was only found in the tracheobronchial lymph node in this elk; all other locations, including the medial retropharyngeal node and the GALT, were free of detectable PrPCWD IR. This scenario of bypassing the retropharyngeal lymph nodes has been previously reported. 61 In these cases, perhaps the PrPCWD gains access to the brain directly via nerves from the digestive system rather than the nasal cavity as described in rodents. 6 An alternate route or possible explanation may be that PrPCWD is present in the head and respiratory lymph nodes but was in such low levels that it was not detectable with the IHC technique used in this investigation. Regardless, this emphasizes the need to examine both the medial retropharyngeal lymph nodes, as well as the obex, when trying to diagnose early cases of CWD in elk.
In one 132MM elk with an obex score of 1, PrPCWD IR was detected in the parasympathetic myenteric plexuses of the abomasum, jejunum, ileum, cecum, spiral colon, and rectum. Myenteric plexuses receive afferent autonomic fibers from the vagus nerve.31,32 The overall amount of PrPCWD IR was heavy in the plexuses although there was little PrPCWD IR in the PSNV and the spinal cord in this 132MM elk. Because all these regions were affected, it was not possible to determine which area was infected first; however, these data suggest these plexuses, the PSNV, and dorsal and intermediolateral columns of the lower spinal cord are affected early in the course of disease. PrPCWD IR was also found in the autonomic postganglionic fibers within the lamina propria of the cecum in 1 elk with an obex score of 1. Fibers from these plexuses primarily innervate thin muscle fibers and vessels of the lamina propria of the digestive system.32,38 Given there was PrPCWD IR in myenteric plexuses in many segments of intestine, but PrPCWD IR was only found in the lamina propria of the cecum, we postulate that prion accumulated first in the myenteric plexuses then followed or traveled within these postganglionic fibers into the lamina propria and to the lumen of the intestine. PrPCWD IR associated with these postganglionic autonomic nerve fibers has been reported in the lamina propria of rectal mucosal biopsies in elk with CWD. 62 Thus, fecal shedding probably occurs relatively early in the progression of disease. Prion in the feces has been confirmed using protein misfolding cyclic amplification. 53
The presence of PrPCWD within these myenteric and submucosal plexuses may result in altered function of the entire digestive system including peristalsis, relaxation of the pyloric and ileocecocolic sphincter, and secretions of gastric and pancreatic enzymes. 38 The esophagus of elk in terminal stages of CWD was often mildly dilated and the rectum distended with dry fecal pellets. This condition may be the result of partial failure of the autonomic nervous system in these regions. Prion-infected plexuses may also affect the intestinal microbiota as reported in white-tailed deer by altering normal digestion processes. 41
The source of infection of these autonomic plexuses of the digestive system was not determined, but presumably occurred via the blood because lymph nodes drain into the circulatory system. 19 Another possible route of infection of the myenteric and submucosal plexuses could be via the M cells transporting prion to the GALT then to the plexuses as described in rodents. 6 Perhaps there are multiple routes of infection in these plexuses that have not been described.
Two 132MM elk had an obex score of 2. These 2 female elk had increased PrPCWD IR within the PSNV and peripheral lymph and hemal nodes. This suggests there was perhaps amplification of prion in these tissues. Both elk had PrPCWD IR in the dorsal column of the lower lumbar/upper sacral region of the spinal cord, but 1 also had immunolabeling in the intermediolateral columns in the upper lumbar and thoracic regions. The intermediolateral column of the spinal cord contains mostly medium sized sympathetic autonomic neurons that send fibers to the PSNV. 35 This further suggests that PrPCWD may travel anterograde along autonomic axons from the lower digestive system and enter the spinal cord relatively early in the pathogenesis of CWD.
PrPCWD IR was detected on the cell membranes (assumed to be in the synaptic clefts) 21 of a few large autonomic neurons located within the vagus nerve in elk with an obex score 2. This would further suggest that the prion was able to travel within the parasympathetic autonomic nervous system and accumulate in the synaptic clefts of neurons located within the vagus nerve. However, PrPCWD IR was not detected within Schwann cells or in the axoplasm of these axons. The route of PrPCWD travel in the vagus nerve was not determined.
PrPCWD IR was detected within the medulla of the adrenal gland in elk with an obex score of 2. The medullary cells of the adrenal gland are derived from neural crest ectoderm and are innervated by preganglionic sympathetic fibers; therefore, the medullary cells are modified sympathetic neuronal cell bodies of postganglionic axons with no axons.23,32The amount of PrPCWD IR within the medulla of the adrenal gland became progressively more abundant as the obex score increased to 10. This could affect the animals’ ability to quickly respond to stressors including predators. Mule deer with CWD have increased vulnerability to being killed in vehicular collisions, 30 by mountain lion (Puma concolor) predation, 39 and possibly by hunters. 9 CWD-positive elk also appear to have an increased risk of mortality from other causes including mountain lion predation. 54
In the 1 pregnant elk with an obex score of 2, PrPCWD IR could be detected within the developing ovarian follicles, theca externa, ovarian cortical nerves, and the trophoblastic epithelium of the chorioallantois villi in the placenta. The uterus and ovaries receive autonomic nerve fibers that directly innervate the ovaries.31,36,38 The significance of finding PrPCWD IR in the placentomes and ovaries was undetermined but suggests possible transovarian and/or transplacental transmission. Although published reports on CWD in captive cervids suggest horizontal transmission is most important, 40 vertical transmission may play a role as well and has been documented in white-tailed deer and elk.8,45,55
In addition to the PrPCWD IR in elk with an obex score of 2, elk with an obex score of 3 and 4 had detectable PrPCWD IR on the cell membranes of neurons in the celiac ganglion, presumably in synaptic clefts. 21 Parasympathetic, sympathetic, and nociceptive fibers traverse through the celiac ganglia. Parasympathetic input for the celiac plexus comes from the vagus nerve, while the sympathetic input is from the greater and lesser splanchnic nerves. Branches of the celiac plexus reach their target organs by forming periarterial plexuses surrounding the branches of the abdominal aorta. Thus, the celiac plexus supplies autonomic fibers to the abdominal viscera (pancreas, spleen, kidneys, liver, gallbladder, and small intestine).31,32 This may suggests that prion in elk with an obex score of ≤4 was undergoing a period of amplification and slowly spreading throughout the body via the autonomic nervous system.
Mild, randomly distributed granules of PrPCWD IR were detected on or between the large, myelinated axons of the vagus nerve in the 1 elk with an obex score of 6, although it could not be determined whether the prion was in Schwann cells or within the axoplasm. PrPCWD IR was also detected in small nerves at the base of the large circumvallate papillae of the tongue. If PrPCWD can interrupt synaptic function, 21 the prion may decrease the ability to taste, affecting the selection of foods which could contribute to the emaciation observed in terminal cases. Mild PrPCWD IR was detected within the pancreatic islets of Langerhans. The pancreas receives autonomic fibers from the celiac ganglion. The presence of prion within islets of Langerhans may reduce secretions of enzymes regulating glucose metabolism. 32 However, the degree of PrPCWD IR within the islets of Langerhans was relative minor and probably had little overall effect on metabolism at this stage of disease. Regardless, it appears there may be multiple mechanisms whereby the digestive system is affected which results in weight loss and terminal emaciation.
Eight elk with an obex score of 7 or 8 had the same tissues with detectable PrPCWD IR compared with elk with lower obex scores. Mild PrPCWD IR was detected in the myocardium and found in 1 of 3 elk with an obex score of 7, and 1 of 5 elk with an obex score 8. PrPCWD IR was not detected in Purkinje fibers. The heart receives fibers from the autonomic nervous system,31,38 which is probably how the prion entered the heart. This finding was not unexpected because PrPCWD IR has previously been reported in the myocardium in elk and white-tailed deer in the terminal stages of CWD. 25
Mild PrPCWD IR was detected within the fine fibrovascular stroma encircling salivary acini, probably terminal autonomic nerve fibers. This location was not unexpected because prion has been detected using protein misfolding cyclic amplification in saliva from orally challenged white-tailed deer and mule deer in terminal stages of CWD.17,37
Mild PrPCWD IR was found within the fine fibromuscular stroma encircling the mammary gland acini (probably fine terminal autonomic nerve fibers) in 1 pregnant elk with an obex score of 8. This is not surprising because moderate to heavy PrPCWD IR was found in the supramammary lymph nodes in elk with an obex score ≥3; therefore, transmammary transmission may be possible, although evidence to support transmammary transmission has not been reported in cervids. 40 PrPCWD IR was not detected in the seminiferous or epidydimal tubules of the testes or associated with spermatozoa of mature bull elk. This contrasts with detection of prions using sensitive amplification assays in the semen and reproductive tissues of white-tailed deer, 29 leaving open the question of sexual transmission in elk.
Heavy PrPCWD IR was detected in the retina and optic nerves in elk with an obex score of ≥7. 60 The retina and optic nerve do not have autonomic innervation26,38; therefore, we postulate that the observed PrPCWD was an extension from the brain and not the autonomic nervous system. 38 The iris does have autonomic innervation, but PrPCWD IR was not found in the iris or cornea. The relatively heavy PrPCWD IR in the retina in elk with an obex scores of ≥7 may indicate that these elk may have visual impairment. 60
As elk progressed into obex scores of 9 or 10, distribution of PrPCWD IR was the same but more intense as that compared in elk with an obex score of 8. Mild PrPCWD IR was found in skeletal muscle myocytes in 1 of 6 elk with an obex score of 9 and 2 of 5 elk with an obex score of 10. Prions have been reported in skeletal muscle in terminal cases in deer1,11,25 and elk, 25 as well as in hamsters and mice. 6 The overall amount of PrPCWD IR in skeletal muscle found in these elk was minimal and randomly distributed. The PrPCWD IR appeared to be located within the sarcoplasm and/or on the surface of the sarcolemma perhaps in the synaptic clefts of myoneural junctions as reported in mice and hamsters. 6 Although the overall amount of PrPCWD IR detected in the myocardium in elk with an obex score of 9 or 10 was considered mild, it was found in 2 of 5 elk with a score of 9 and all elk with an score of 10.
Using obex scores as a proxy for stage of disease progression yields inferences into the spread and pathogenesis of naturally occurring CWD in elk. However, regarding the elk studied in this investigation, there are limitations. It was not known at what age these elk were exposed to the CWD prion. It was also not known, but assumed, that these elk were exposed multiple times with small dosages of PrPCWD over an extended period of time as would be expected in a natural setting. It was not known if naturally infected elk show a different disease time course or progression of CWD than what has been reported in experimentally infected cervids following large oral doses of CWD-positive brain.14,15,57 Nevertheless, this study suggests there is a unique progressive spread of PrPCWD throughout spinal cord and peripheral tissues in elk with naturally occurring CWD that can be correlated with obex scores even if the age of exposure, dose, and/or the number of exposures of prion are unknown. Also, data from this investigation suggest the natural route of exposure may be the respiratory system (ie, nasal cavity and/or lungs and spreads through the autonomic nervous system). Variation in observed incubation periods of CWD in elk with different PRNP genotypes 15,51 could be explained, in part, by observations that prions perhaps spread more rapidly and with greater intensity in the 132MM elk as compared with the 132ML elk. Perhaps this is due to the speed of prion amplification in these elk with different genotypes.
Findings from this investigation further indicate the importance of examining medial retropharyngeal lymph nodes, rather than solely the obex, to detect early cases of CWD in elk. 59 Finally, this work provides a map of disease progression throughout the body that has not previously been described. This can assist pathologists, ecologists, and wildlife managers estimate the length of time the disease has been within an individual elk, and possibly within a population, which could inform management options for controlling the disease.
Bovine spongiform encephalopathy (BSE), which has previously been highly relevant in the public health domain, has been compared with CWD, that is, “mad deer disease.” However, when the published pathogenesis of classical BSE in cattle is compared with CWD in elk, there appear to be differences between the diseases. 10 It has been reported that following oral exposure, the BSE agent is believed first to cross the intestinal epithelium via M cells, which are located in the follicle-associated epithelium of the gut and tonsil. These cells specialize in the transport of macromolecules across the epithelium. 18 In cattle during the first 6 and 10 months postinfection, the first cells that accumulate the BSE agent are tingible body macrophages in the tonsils, GALT of the ileocecal junction and the jejunum, and in Peyer’s patches of the ileum.66,67 In contrast to CWD, the first tissues to accumulate PrPCWD are the retropharyngeal and tracheobronchial lymph nodes, not the palatine tonsils or the GALTs.
At 12 and 24 months postinfection, the BSE prion has been reported to increase in the distal ileal GALT in cattle, 22 but in elk, many more tissues have accumulated PrPCWD. During this same time period, the prion agent of BSE is reported to come in contact with the fine nerve fibers of the mucosal plexus of the enteric nervous system. 24 This appears to occur relatively early in elk with an obex score of 1. Also, experimentally infected 132MM elk die at 24 months postinfection.
During 32 and 48 months postinfection, the BSE agent is found in the mesenteric nerves and the cranial celiac-mesenteric ganglion complex and then ascends to the thoracic spinal cord via the sympathetic nervous system (eg, splanchnic nerves) and to the brainstem and the brain via the parasympathetic nervous system (eg, vagus nerve) and nodose ganglion. 4 A proposed alternant pathway for the BSE prion to travel to the nervous system from the GALT is via an interaction with small peripheral nerves or with immune cells. 6 However, the route at which the BSE prion travels from the GALTs to the spinal cord is unknown. 6
From the thoracic spinal cord, the BSE agent spreads rostrally to the cranial medulla and caudally to the cauda equina. 4 In elk (obex score 2), the lumbar/sacral spinal cord is infected before the thoracic spinal cord suggesting the parasympathetic nervous system is infected slighter sooner as compared with the sympathetic nervous system. In elk, it is estimated that the brain and spinal cord are infected approximately 12 months postinfection. This is much earlier in elk as compared with cattle. From the spinal cord, the BSE agent then accumulates in the dorsal root, trigeminal, and cervical ganglia. 2 In elk, the lumbar/sacral spinal cord is affected extremely early and appears to travel anterior. This contrasts with cattle where it starts in the thoracic cord and progresses down the cord. In later stages of BSE, the adrenal glands and sciatic nerve have also accumulated the BSE prion protein. 13 This occurs in elk with low obex scores.
Between 42 and 84 months postinfection, the BSE agent spreads to the spindles of various muscles such as the masseter, the triceps brachii, intercostal muscles, and the semitendinosus. 48 PrPCWD is detected in skeletal muscle in terminal elk with obex scores of 9 and 10. The duration of infection in 132MM elk with an obex score of 9–10 is approximately 24 months, not 42–84 months.
Supplemental Material
sj-pdf-1-vet-10.1177_03009858231173467 – Supplemental material for Distribution of the misfolded isoform of the prion protein in peripheral tissues and spinal cord of Rocky Mountain elk (Cervus elaphus nelsoni) with naturally occurring chronic wasting disease
Supplemental material, sj-pdf-1-vet-10.1177_03009858231173467 for Distribution of the misfolded isoform of the prion protein in peripheral tissues and spinal cord of Rocky Mountain elk (Cervus elaphus nelsoni) with naturally occurring chronic wasting disease by T. R. Spraker, T. Gidlewski, J. G. Powers, T. A. Nichols and M. A. Wild in Veterinary Pathology
Footnotes
Acknowledgements
We thank Katherine O’Rourke for performing all the genotyping in these elk. We thank Todd Bass and AnaMario Benavides Obon for embedding and Bruce Cummings for cutting, staining, and immunolabeling all the sections of tissue. We thank Vicki Jameson, Elizabeth Wheeler, Tara Ruby, Crystal Meterett, and David Walters for assisting in necropsies and collection of tissues.
Authors’ Note
Wright Scott D, PhD, acknowledges that I sent tissues from elk infected with chronic wasting disease (CWD) to Dr Terry Spraker for his analysis. These tissues were part of a collection of tissues housed at the time at the United States Geological Survey National Wildlife Health Center, Madison, Wisconsin.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was partially funded by the National Park Service, ARS/USDA, Pullman, Washington, and the Colorado State University Diagnostic Laboratory, College of Veterinary Medicine, Fort Collins, Colorado.
Sources and Manufacturers
a = Wildlife Pharmaceutics, Fort Collins, CO
Supplemental Material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
