Abstract
This article describes 11 cases of neuronal embryonal neoplasia in captive adult teleost fish. Neoplasms were located within 1 or both eyes of 8 fish and the skin of 3 other fish. Ocular neoplasms most often presented as unilateral or bilateral exophthalmia. Seven ocular and 1 cutaneous mass were composed of small triangular (carrot-shaped) neoplastic cells with Flexner-Wintersteiner-type rosette formation. Mass location and histologic and ultrastructural features were suggestive of retinoblastomas. One ocular mass was composed of ribbons and rosettes of neoplastic cells with multiple areas of neuronal differentiation and was diagnosed as a teratoid medulloepithelioma. A cutaneous mass from an electric eel (Electrophorus electricus) consisted of rosettes and streams of elongate neoplastic cells. The epidermal electroreceptor (ampullary) organ was considered as an origin. Although distant metastases were not observed, neoplasms were generally locally aggressive with postexcision recurrence. There was occasional spread to or de novo occurrence within the contralateral eye.
A number of neoplastic disorders have been reported in fish. Some well-documented neoplasms include viral-induced dermal sarcomas in walleyes and aflatoxin-associated hepatocellular adenomas and carcinomas in rainbow trout as well as a variety of sporadic tumors in numerous species. 1,2,12 This report details spontaneous ocular and cutaneous tumors in multiple fresh and marine teleost species with features characteristic of neuronal embryonal tumors.
Neuronal embryonal tumors arise from neuroectodermal progenitor cells within subependymal matrix layers and have morphology and aggressive behavior in common. 10 Tumor types include medulloepitheliomas, neuroblastomas, retinoblastomas, ependymoblastomas, and primitive neuroectodermal tumors (PNETs). PNETs are derived from pluripotent germinal neuroepithelial cells and may be further classified on the basis of anatomic location as medulloblastomas, cerebral PNETs, or peripheral neuroectodermal tumors. Note that classification schemes may differ because the cell origin of these tumors is often not well understood.
In humans, neuronal embryonal tumors often arise within the first decade of life. Chromosomal deletions and/or MYCN gene amplification have been noted in affected children and may play a role in pathogenesis in humans. 3 Retinoblastomas predominantly occur following inactivation of both RB1 (retinoblastoma susceptibility protein) tumor suppressor gene alleles. 13
Materials and Methods
Medical records, archived tissues, and histologic sections were evaluated as collected between 2001 and 2009 from 11 adult fish of 11 different species (Table 1) and housed at one of two facilities. Tissues taken at necropsy consisted of whole or representative sections of all major organs, including skin or body wall and bone. Smaller fish were fixed whole in formalin and bisected along the median plane for histology. Tissues from all fish were fixed in 10% neutral buffered formalin, routinely processed for histology, sectioned at 5 μm, and stained with hematoxylin and eosin. Only enucleated eyes were submitted from the white-lined catfish (fish No. 5) and tinfoil barb (fish No. 8). Eyes were not taken from the cichlid (fish No. 9), and gonads were not present on sections from fish No. 4. Complete tissue sets were available from all other fish.
Transmission electron microscopy was used to further evaluate one of the ocular masses (fish No. 6) and one of the cutaneous masses (fish No. 11). Karnovsky-fixed tissues were postfixed in 2% osmium tetroxide, dehydrated, and embedded in epoxy resin. Sections were cut at 0.35 μm and stained with toluidine blue and basic fuschin for preliminary light microscopic review. Selected samples were then sectioned at 80 to 90 nm, stained with uranyl acetate and lead citrate, and examined with a Hitachi H600 transmission electron microscope (Hitachi America, Ltd, Brisbane, CA).
Results
Eleven cases were identified over an 8-year period. Ten cases came from a single institution with a yearly average population of more than 20,000 individual teleost fish. Information regarding species is recorded in Table 1. In sum, 4 fish were female, 4 were male, and 3 were of unknown sex. The most common clinical presentation was exophthalmia of the affected eye. In one case (fish No. 1), anorexia was noted, possibly related to visual deficits secondary to neoplastic invasion of both eyes. Fish No. 7 had diffuse epicarditis and multiple mesenteric granulomas with intralesional bacteria. In all other cases, there were no significant gross or histologic lesions aside from the primary neoplastic masses.
Species, Clinical History, and Gross and Microscopic Findings of 11 Cases of Neuronal Embryonal Tumors in Captive Fishes a
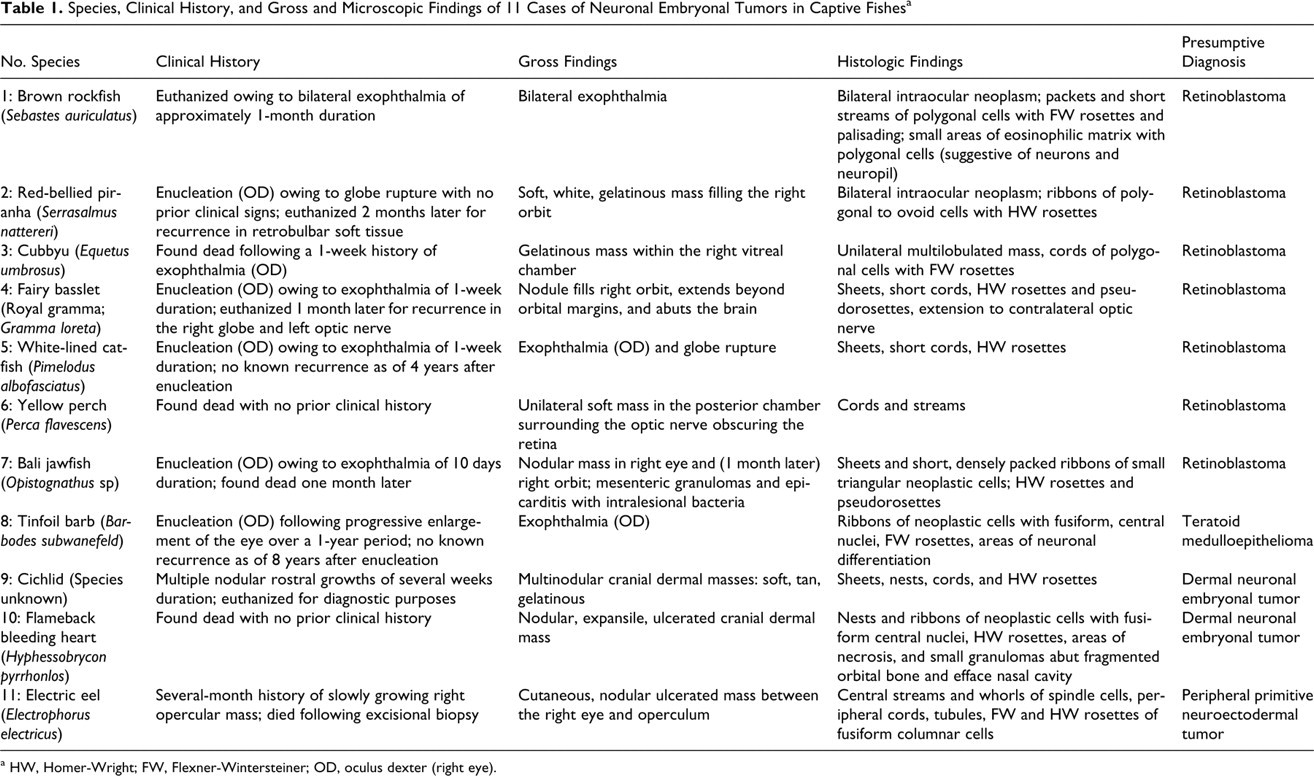
a HW, Homer-Wright; FW, Flexner-Wintersteiner; OD, oculus dexter (right eye).
Masses were firm, gelatinous, and pale grey and grossly appeared to be solid (Fig. 1). Ocular masses were within the vitreous chamber and effaced portions of the retina and choroid. Some also extended to and infiltrated the posterior and anterior chambers, optic nerve, and retrobulbar soft tissue. In some cases, the lens was absent (possible artifact owing to histologic sectioning). When present, the lens occasionally abutted the neoplastic masses but did not appear to be otherwise affected.
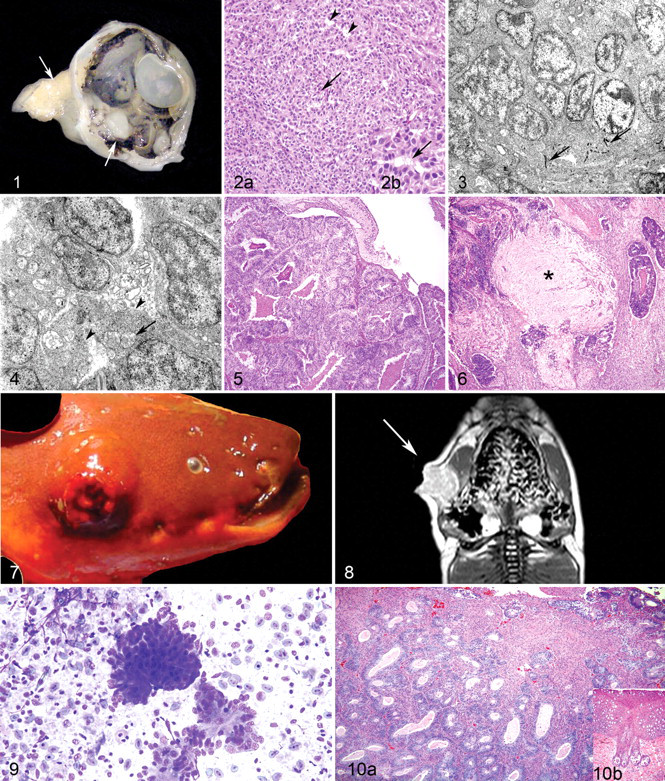
Table 1 outlines the histologic features of the ocular masses. In general, masses were infiltrative, nonencapsulated, poorly demarcated, multilobulated, and composed of densely cellular, variably well-defined short streams, ribbons and packets of spindle to carrot-shaped neoplastic cells on a fine fibrovascular stroma. There were multifocal palisading, scattered rosettes, and pseudorosette formations (Fig. 2A), and cells were intermittently joined by zonula adherens (Figs. 3, 4). Both Flexner-Wintersteiner and Homer-Wright rosettes were present. Flexner-Wintersteiner rosettes are characterized by colorless lumina, whereas Homer-Wright rosettes are arranged around central amorphous and/or finely fibrillar eosinophilic material. In these fish, some rosettes had short, irregular extension of neoplastic cell cytoplasm into lumina. These cytoplasmic extensions resembled fleurettes, and were suggestive of areas of photoreceptor differentiation (Figs. 2B, 4). Mitotic figures ranged from 1 to 6 per ten 400× fields. Within some of the largest tumors were one or more areas of necrosis with variable lymphocytic inflammation. Ultrastructurally, neoplastic cells had moderate numbers of mitochondria (often concentrated apically), abundant rough, endoplasmic reticulum, and variable apical cytoplasmic extensions into rosette lumina. Fish Nos. 1 and 8 had multifocal accumulations of abundant, finely granular pale eosinophilic matrix suggestive of neuropil (Fig. 6). Histologic features of the neoplasm in fish No. 8 were distinct from ocular masses in the other fish. Morphology differed in that neoplastic cells had central fusiform nuclei and were arranged in ribbons with small areas of neuronal differentiation (Figs. 5, 6). These areas consisted of foci of fibrillar eosinophilic material that recapitulated the neuropil with scattered, small polygonal cells that resembled neurons. Polygonal cells had moderate amounts of eosinophilic cytoplasm, distinct cytoplasmic borders, large round nuclei, and prominent round central magenta nucleoli.
Masses in 3 fish (Nos. 9–11) appeared to be limited to the epidermis and dermis of the cranium. On the rostrum of fish No. 9 were 3 closely apposed 6-mm-diameter nodules that abutted portions of the subjacent cranial bone. The gross and histologic appearances were similar to the masses in fish Nos. 1–7. Overlying a fragmented orbital bone of fish No. 10 was a large, markedly expansile, nonencapsulated dermal mass with histologic features similar to those of fish No. 8, although lacking neural differentiation.
The third dermal mass was from an electric eel (fish No. 11) with a several-month history of a slowly growing cutaneous mass between the right eye and right operculum (Figs. 7–10). The mass was 3 cm 3 ulcerated, firm, and nodular. On cut surface was a small amount of central thick yellow fluid. A Diff-Quik-stained impression smear of the mass was highly cellular with variably sized, cohesive clusters of polygonal to elongate cells occasionally arranged in rosettes (Fig. 9). On histologic examination, the mass was partially encapsulated and infiltrative, and it consisted of broad, branching trabeculae and irregular islands supported by a scant fibrovascular stroma (Fig. 10A). Neoplastic cellular morphology varied. There were tubules and rosettes (both Flexner-Wintersteiner and Homer-Wright type) of tall columnar to elongate and bipolar cells. Tubules were often filled with proteinaceous material. Between the tubules and rosettes were poorly defined streams and whorls of spindle-shaped neoplastic cells with fibrillar cytoplasm and indistinct cell borders. Among both cell types, mitotic figures were rare, with an average of less than 1 per 400× field. Ultrastructurally, both cell types had indistinct cell borders. Cells in tubules had numerous apical, short cytoplasmic extensions, often adjacent to approximately 120-nm-wide electron-dense tubules and structures that appeared to be recapitulating the terminal web. Spindle cells had numerous cytoplasmic ribosomes, few organelles, and intermittent desmosomes. Within other sections of epidermis and dermis from the head and body, multiple normal sensory receptors with an onion bulb–like appearance were noted (Fig. 10B).
The neoplasm was initially diagnosed with an incisional biopsy. Three months later, the size of the mass had increased to 6 × 6 × 3 cm. With magnetic resonance imaging, the mass was shown to be confined to the skin with no ocular invasion (Fig. 8). The mass was surgically removed and excision appeared complete. The eel died shortly after surgery. Necropsy confirmed complete excision of the mass and there were no metastases. The cause of death was undetermined.
Other than fish No. 11, metastasis or recurrence occurred in 3 of 5 fish treated surgically. A neoplasm histologically identical to the previously excised mass was diagnosed in the contralateral eye of fish No. 4 one month after surgery. The fish subsequently became lethargic, then nonresponsive, and was euthanized. In two other cases (fish Nos. 2 and 7), there was recurrence of the masses in the ipsilateral orbital soft tissues 1 to 2 months after enucleation. Fish No. 2 was euthanized following recurrence, owing to quality-of-life concerns. Bacterial septicemia was the cause of death in fish No. 7. The ocular mass seemed the most likely bacterial portal of entry, given that there were no other significant gross or histologic lesions.
Discussion
Although tumors among these fish differed somewhat from one another, cell morphology and tumor architecture were overall consistent with neuronal embryonal tumors. Characteristic features of neuronal tumors present within these neoplasms included elongate to carrot-shaped neoplastic cells, variable palisading rosette formation, high cellularity, and one or more areas of necrosis. 6
In humans, characteristics such as signalment, location, histologic features, immunohistochemisty, and ultrastructure are used to help determine the tumor type. Regardless of the subtype, neuronal embryonal tumors label variably for multiple neuronal markers, including synaptophysin, neuron-specific enolase, and S-100. In humans, additional labeling with retinal S-antigen, retinol-binding protein, and rod or cone opsins have been observed in some retinoblastomas but again depend on the level of differentiation. Medulloepitheliomas and PNETs are typically vimentin positive. 6,19
Among the cases in this report, further classification is hindered by the often-anaplastic nature of the tumors as well as the difficulty in validating mammalian immunohistochemical techniques in fish. Many neoplasms in these cases were considered to be anaplastic with small, disorganized neoplastic cells and vague or nonexistent rosettes. Immunohistochemical staining for synaptophysin, vimentin, cytokeratin, and neuron-specific enolase were attempted but were largely nondiagnostic with a lack of reliable internal positive controls. Failure of immunoreactivity was possibly due in part to the lack of differentiation. Other complicating factors included decalcification during initial processing and, for many of the cases, prolonged time spent in formalin.
The presence of Flexner-Wintersteiner rosettes in many of the fish tumors was considered a key feature because these structures are fairly specific for retinoblastomas, pineoblastomas, medulloepitheliomas, and olfactory neuroblastomas (esthesioneuroblastomas). 4,16 As the ocular tumors were largely within the vitreous chamber with little or no remaining normal retinal structures, a retinal origin (retinoblastoma) seemed likely. Retinoblastomas can arise from any of the nucleated retinal layers. Typical histologic features include a hyperchromatic nucleus with scant cytoplasm and the formation of Flexner-Wintersteiner rosettes surrounded by a limiting membrane (presumably corresponding to the retinal external limiting membrane). 13 Well-differentiated tumors may have photoreceptor differentiation characterized by rosettes with luminal projections of club-shaped bodies (fleurettes) filled with elongate mitochrondria. 16 Among some of the ocular masses in this report, there were similar projections of mitochondria-filled cytoplasm into rosette lumens that could be interpreted as primitive fleurettes (Figs. 2, 4).
Retinoblastomas in domestic mammals are rare, with individual cases reported in a dog and a llama. 8,15 In fish, retinoblastomas have been sporadically diagnosed. Published literature includes isolated cases in a porkfish (Anisotremus virginicus), a brown bullhead (Ictalurus nebulosus), and a spring cavefish (Chologaster agassizi). 7,14 The diagnosis of retinoblastoma in those cases was made on the basis of typical histologic features (rosette-forming neuroblastic cells) and location within the posterior globe. Additional features in the cavefish included rosettes with apical material reminiscent of fleurettes. 7 As with the cases in this report, there was no evidence of metastasis.
In the cases in this report, all tumors were slow-growing, and distant metastases were not present. Recurrence and spread to the contralateral optic nerve occurred in one of the enucleated cases (fish No. 4) within 1 month of the initial surgical excision. Although it was possible that tumor growth in the contralateral eye was a de novo occurrence, the neoplasm extended along the optic nerve and into the meninges, which seemed more suggestive of direct extension from the primary mass.
The tumor in fish No. 8 had neural differentiation suggestive of teratoid tumors. This mass was diagnosed as a well-differentiated medulloepthelioma. Medulloepitheliomas may arise from undifferentiated cells of the ciliary body but also may originate in the posterior chamber. 5,11 These tumors are uncommon in domestic mammals. Most reported cases are in horses, in which the tumors are locally aggressive but uncommonly metastasize. 5 In fish, ocular cases have been documented in a goldfish (Carassius auratus) and multiple Japanese medaka (Oryzias iatipes). 9,11 The goldfish medulloepithelioma was a sporadic case, and neural differentiation was not observed. Tumors in the Japanese medaka were of the teratoid variant and arose following brief, experimental exposure to methylazoxymethanol acetate. In all cases (and similar to those in this study), the neoplasms were locally invasive, but metastasis was not a feature. Medulloepitheliomas differ from other embryonal tumors in histologic appearance and genetic pathways. They are derived from primitive medullary epithelium of the neural tube and may differentiate along neural, glial, or mesenchymal lines. In humans, ocular medulloepitheliomas most commonly arise from the ciliary body and are typically malignant. 10 The histologic appearance is characteristic, with formation of tubules and papillary structures composed of columnar to pseudostratified neoplastic cells aligned along a basement membrane. The teratoid variants contain cartilage, glial tissue, and/or skeletal muscle. Ultrastructurally, there are extensive primitive lateral cell junctions and basal lamina on the outer epithelial surface. 13
In fish No. 1, small areas within the neoplasm resembling foci of neuropil differentiation were observed; however, histologic features of the mass were not consistent with a medulloepithelioma. A diagnosis of retinoblastoma was made on the basis of microscopic appearance and tumor location, but a poorly differentiated neuroblastoma is another differential to consider.
Masses in the cichlid (fish No. 9), flameback bleeding heart (fish No. 10), and electric eel (fish No. 11) appeared to be cutaneous in origin. The histologic appearance of the mass in fish No. 9 was similar to that of the previously described retinoblastoma-like neoplasms. Eyes were not assessed histologically, but given the location of the mass, an ocular origin was possible. Alternatively, the masses may have arisen from the lateral line, a sensory organ that extends from the rostrum to the tail. Lateral-line sensory structures are clusters of neuroepithelial cells called neuromasts and are similar to hair cells of the inner ear. In this case, direct extension from the lateral line was not visible. The neoplasm in fish No. 10 was close to the eye and brain and largely effaced the nasal cavity. An ocular origin could not be confirmed with additional sectioning, and although microscopic features were suggestive of a medulloepithelioma, a differential diagnosis of olfactory neuroblastoma should be considered.
The electric eel had a large infiltrative opercular mass not associated with either eye or the brain, as confirmed histologically and by antemortem magnetic resonance imaging. This tumor differed from the ocular tumors in that there were two populations of fairly well-differentiated neuroepithelial cells. Structures with an onion bulb–like appearance in sections of normal skin corresponded to ampullary electroreceptors described in other gymnotiform species. Ampullary organs are composed of receptor cells and supporting cells, and both cell types are thought to arise from primordial cells within the stratum germinativum. 18 Pluripotentiality of the neoplastic cells could account for the two histologically distinct cell populations. Because of the location, structure, and presumed origin, this neoplasm was diagnosed as a peripheral PNET. PNETs have been reported in the skin of young coho salmon (Oncorhynchus kisutch) in association with the lateral line, another sensory structure. 19 In these cases, the tumors were histologically more similar to those noted in the eyes of these fish and the skin of fish No. 9, although rosettes were not observed.
Overall, cases of neuronal embryonal neoplasia in teleost fish occurred infrequently. Underlying genetic abnormalities may be implicated. Toxin exposure seemed unlikely given the well-controlled environment and sporadic nature of the cases. Adult teleost fish retain the ability to regenerate ocular neuroepithelial tissue in response to injury and can in fact replace retinal neurons and rods from a retinal source of precursor cells. This response is sometimes overexuberant and may increase opportunities for neoplastic transformation of neuroectodermal progenitor cells in fish. 17
In summary, neuronal embryonal neoplasia can occur in a variety of fish species, and many have features suggestive of retinoblastoma. Clinical signs of progressive unilateral or bilateral exophthalmia should merit a differential diagnosis of embryonal neoplasia. Underlying genetic abnormalities and toxin exposure have been documented in association with ocular embryonal neoplasms in humans and some fish species, but no unifying factor could be identified in these cases. Cases seemed to be sporadic with an extremely low incidence. Additionally, an electroreceptor origin should be considered in similar tumors occurring in the skin of gymnotiform species. Recurrence following surgical excision underscored the locally aggressive nature of the retinoblastoma-like neoplasms, but metastasis was not a feature. Additional immunohistochemical studies may aid in further classifying these neoplasms.
Footnotes
Acknowledgements
We would like to acknowledge the Animal Health Care staff of the John G. Shedd Aquarium and Lincoln Park Zoo for their essential support in compiling case materials and photos, Jane Chladny and the University of Illinois Veterinary Diagnostic Laboratory’s histology laboratory for slide preparation, Lou Ann Miller of the University of Illinois Center for Microscopic Imaging for assistance with electron microscopy, Drs Karen Terio and Kathleen Colegrove for reviewing the article and images, and the Zoological Pathology Program and Shedd Aquarium staff and students who assisted with the necropsies included in this study.
The authors declared no potential conflicts of interest with respect to the authorship and/or publication of this article.
The authors received no financial support for the research and/or authorship of this article.
