Abstract
A 4-mo-old northern red-shouldered macaw (Diopsittaca nobilis) was admitted to the veterinary hospital of the Arruda Câmara Zoo, in the State of Paraiba, Brazil, for investigation of an orbital mass. Given rapid progression and lack of response to treatment, the bird was euthanized, and an autopsy was performed. Histologically, the mass consisted of a retrobulbar invasive tumor characterized by tubular and rosette-like structures, with interspersed heteroplastic tissues, such as aggregates of neuroglial cells and islands of hyaline cartilage. The tumor was immunopositive for pancytokeratin, GFAP, NSE, and S100. These findings were compatible with an ocular teratoid medulloepithelioma, a neoplasm best described in humans but also reported rarely in young cockatiels and African Grey parrots.
Ocular lesions are commonly documented in birds but reports of primary ocular neoplasms are scarce. Reported tumors include undifferentiated carcinomas, melanocytic tumors, intraocular osteosarcomas, multicystic adenomas, pigmented iridociliary adenomas, ganglioneuromas, and teratoid medulloepitheliomas. 23
Medulloepitheliomas are embryonal tumors that can affect the central nervous system (CNS), the neuroepithelial structures of the eye, and the optic nerve. In humans, they are rare congenital tumors that usually affect children. 16 Medulloepitheliomas are also rare in domestic animals and are most frequently reported in horses.2,3,7,17,28,29 These neoplasms have also been described in several other species, including llamas, a cat, dogs, and a goldfish.10,12,14,15 Ocular forms are uncommon in birds, and all reports of medulloepithelioma in avian patients are from psittacine birds, specifically cockatiels and African Grey parrots.4,23,25
Here we describe the gross, microscopic, and immunohistochemical features of an ocular teratoid medulloepithelioma in a northern red-shouldered macaw (Diopsittaca nobilis). In addition, we reviewed the literature relevant to this neoplasm.
A 4-mo-old northern red-shouldered macaw was kept at the João Pessoa Zoo in northeastern Brazil. Zookeepers noted that the bird had a slight swelling of the periocular region and redness of the conjunctiva of the left eye. The bird was hospitalized and treated with tobramycin and ketoprofen eye drops, but the swelling rapidly progressed, accompanied by mucohemorrhagic discharge. By day 7, a large mass covered by intact skin protruded from the orbit, with perforation of the cornea (Fig. 1). Abscess, fungal granuloma, and neoplasia with consequent exophthalmia were considered possible differentials. Given the ongoing enlargement of the mass, the macaw was euthanized after 15 d of hospitalization. The mass was collected, immersed in 10% neutral-buffered formalin, and sent to the Federal University of Paraiba. Unfortunately, the carcass was not sent for autopsy.

Ocular medulloepithelioma in a young northern red-shouldered macaw. A large mass covered by intact skin protrudes from the orbit. The cornea is dark red and roughened. Day 15 of observation.
The sample sent for examination consisted of a 5-cm diameter, firm, gray mass that obliterated the orbit, causing marked compression of the globe. The cut surface was brown, interspersed throughout with scattered areas of black discoloration. Three representative sections of formalin-fixed tissue were processed routinely for histology and stained with hematoxylin and eosin and alcian blue stains.
Histologically, most of the tissue consisted of a non-encapsulated, infiltrative neoplasm composed of small tubules and cords of epithelial cells within a small amount of fibrovascular stroma (Fig. 2). In some areas, the neoplasm consisted of single-to-multiple layers of cuboidal-to-columnar cells arranged in tubules and occasional rosettes, which were consistent with both Flexner–Wintersteiner rosettes (Fig. 3) and Homer Wright rosettes (Fig. 4). Neoplastic cells had indistinct cell borders, a small-to-moderate amount of eosinophilic cytoplasm, and ovoid hypochromatic nuclei with 1 or 2 distinct nucleoli. Anisokaryosis was mild-to-moderate, and mitoses were relatively frequent (15 in an area of 2.37 mm2). Multifocally, neoplastic cells had brightly eosinophilic cytoplasm with occasional accumulations of extracellular brightly eosinophilic material interpreted as keratin (squamous differentiation). Some areas of the neoplasm were more variably cellular and less differentiated, composed of small spindle-shaped cells with a minimal-to-small amount of cytoplasm, and cells with round nuclei, conspicuous nucleoli, and scant cytoplasm. These were interpreted as areas of neuroglial differentiation. Rare islands of hyaline cartilage were interspersed among groups of neoplastic cells (Fig. 2). The neoplasm had large foci of necrosis. The globe was collapsed, and the uveal tract was the only recognizable intraocular structure. The cornea was perforated and replaced by a thick layer of degenerate heterophils and extravasated erythrocytes with embedded foreign material and abundant bacterial colonies. The globe was encircled by abundant fibrous connective tissue, which was variably expanded by hemorrhage and a heterophilic infiltrate. Cystic areas scattered throughout the tumor were filled with alcian blue–positive, amorphous, basophilic material.
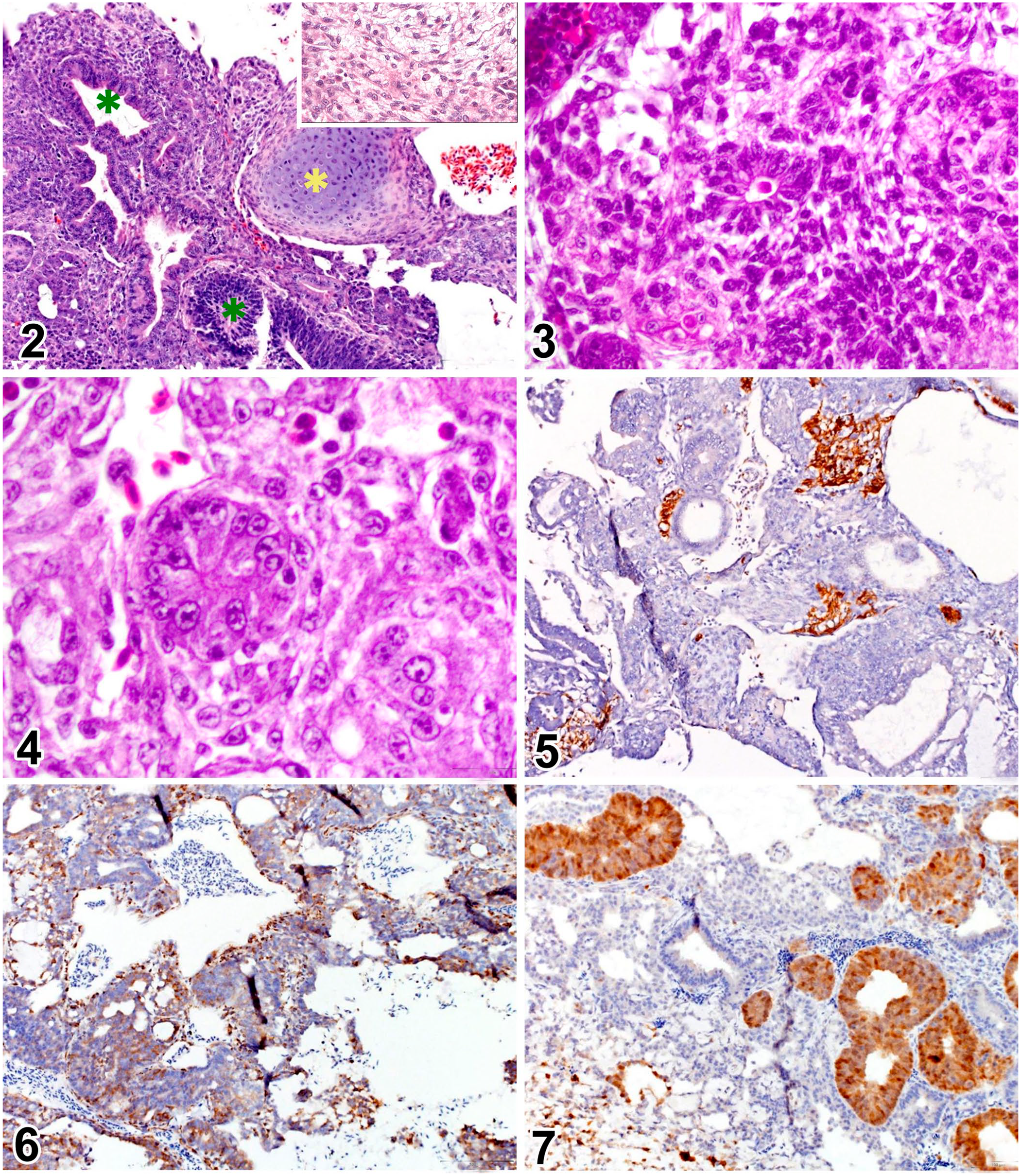
Histologic and immunohistochemical features of an ocular teratoid medulloepithelioma in a young macaw.
Immunohistochemistry was performed for further characterization of the neoplasm. The antibodies used were glial fibrillary acidic protein (GFAP; Z0334, 1:500; Dako), S100 protein (Z0311, 1:4,000; Dako), neuron-specific enolase (NSE; M0873, 1:150; Dako), and pancytokeratin AE1-AE3 (M3515,1:40; Dako). Tissues from a blue-and-gold macaw (Ara ararauna) were used as a control. The cells in the areas of neuroglial differentiation were strongly immunoreactive for GFAP, the cells forming some of the multilayered tubules and solid nests had positive immunolabeling for S100 and cytokeratin, and virtually all cells expressed NSE (Figs. 5–7). Based on these findings, the diagnosis was malignant teratoid medulloepithelioma.
The classification of human embryonal tumors of the CNS has undergone substantial modification over the past decade, especially with the release of the most recent 2016 WHO classification. 19 The term primitive neuroectodermal tumor (PNET), which used to encompass the medulloepitheliomas, was removed from the diagnostic lexicon. Embryonal tumors that display amplification of the C19MC region on chromosome 19 (19q13.42), including embryonal tumor with abundant neuropil and true rosettes (ETANTR), ependymoblastomas (EBL), and some medulloepitheliomas are now under the umbrella term embryonal tumor with multilayered rosettes (ETMR), C19MC-altered. 19 Tumors with histologic characteristics of medulloepithelioma that do not display C19MC expression should continue to be called medulloepithelioma.19,27 This new classification is restricted to humans and is not currently applicable to animals.
Embryonal tumors of the CNS are rare in animals and are reported most commonly in dogs. 9 There are only a few reports of these tumors in birds, including a cerebral neuroblastoma in a budgerigar (Melopsittacus undulatus), 5 a cerebellar medulloblastoma in an umbrella cockatoo (syn. white cockatoo; Cacatua alba), 6 and a poorly differentiated embryonal tumor in the brain of a domestic goose (Anser anser). 20
Intraocular medulloepitheliomas are congenital tumors that originate from primitive neuroectodermal cells and therefore have the same cellular origin as CNS medulloepitheliomas; however, in humans, the intraocular tumors usually have a better prognosis than their cerebral counterparts.4,27 The vast majority of intraocular medulloepitheliomas originate from the nonpigmented epithelium of the ciliary body and are named ciliary body medulloepitheliomas (previously known as diktyomas given their net-like histologic features). 27 Less frequently, intraocular medulloepitheliomas originate from the primitive medullary epithelium of the iris, retina, and optic nerve. 13 Most (75–80%) intraocular medulloepitheliomas occur in children, and they are the second most common primary intraocular tumor of children after retinoblastoma. 27 Although they are congenital neoplasms, their clinical manifestation may be delayed for years. 27
In humans, the tumors are typically cystic and range in color from white to gray to yellow. 25 Associated clinical signs may include glaucoma, cataract formation, poor vision, blindness, leukocoria, exophthalmos, proptosis, buphthalmos, and iritis. 27 Intraocular medulloepitheliomas are unilateral in most cases and, given their slow growth, may not become noticeable until they protrude into the anterior chamber or invade surrounding structures. 27 These tumors are usually only locally aggressive, and enucleation can be curative; however, when the diagnosis is delayed, the tumor can spread through adjacent structures, including the optic nerve, consequently gaining access to the brain. 11
On the other hand, in veterinary medicine, the most commonly associated clinical signs include buphthalmia,4,14,26 exophthalmia,2,25glaucoma,1,21 anterior uveitis and corneal edema, 10 and leukocoria. 4 Medulloepitheliomas usually manifest at a young age in animals, and a history of trauma is commonly reported.4,15,25 As seen in our case, once the first clinical signs are noted, the disease progresses rapidly.4,29 The buphthalmia and the mucohemorrhagic discharge seen in our case have also been described in cockatiels with medulloepithelioma. 4
Histologically, medulloepitheliomas are characterized by thick multilayered tubular structures with distinct lumens resembling primitive medullary epithelium. 27 They can also have rosettes known as Flexner–Wintersteiner rosettes (cell layers with apical tight junctions and distinct lumens) and Homer–Wright rosettes (more primitive and less distinct structures that lack tight junctions and have indistinct lumens), but neither type of rosette is specific for medulloepithelioma. Medulloepitheliomas are classified as teratoid or non-teratoid based on the presence or absence of heteroplastic tissues, such as striated muscle, neural tissue, and hyaline cartilage. 27 Indicators of malignancy include an invasive growth pattern, high mitotic count, and cellular pleomorphism. 16
The neoplasm in our case had several of the classic histologic features of teratoid medulloepithelioma, including simple and complex rosettes, hyaline cartilage, and neural differentiation. The tubular and rosette-like structures are consistent with the histologic features of intraocular medulloepithelioma observed previously in cockatiels.4,25 The most common heteroplastic tissue in teratoid medulloepithelioma is hyaline cartilage, which has been described previously in teratoid medulloepitheliomas of a cockatiel, 4 dogs, 15 a llama, 10 horses, 17 and children. 27 The mitotic count observed in our case was similar to that in previous reports of malignant medulloepitheliomas, which report 0–30 mitoses in an area of 2.37 mm2 (0–3 mitoses per 400× field).4,10,15 Unfortunately, the carcass of the bird was not submitted for autopsy, so the occurrence of metastasis could not be assessed.
Medulloepitheliomas are possibly the most primitive tumor of the neuroepithelium, and the great variability of immunolabeling patterns probably reflects the diversity of differentiation of the neoplastic cells. 4 The positive immunolabeling for GFAP (Fig. 5) and NSE (Fig. 6) in our case was consistent with that seen in medulloepitheliomas in an African grey parrot, 23 cockatiels, 4 llama, 26 horses, 17 and humans. 11 In contrast, GFAP was negative in another llama, 10 and both GFAP and NSE were negative in a dog. 1 In our case, some neoplastic cells were also positive for S100 (Fig. 7), similar to reports in llamas10,26 and humans. 16 Unlike the reports of medulloepitheliomas affecting llamas,10,26 horses, 17 and dogs, 1 tissues were variably positive for pancytokeratin immunolabeling in our case, similar to reports in some human studies of patchy areas of positive immunolabeling. 16 There are no reports of pancytokeratin immunohistochemistry in other cases of medulloepithelioma in birds,4,23,25 to our knowledge.
Teratoid medulloepitheliomas should be differentiated from teratomas. Both can have heteroplastic tissues; however, teratomas usually exhibit differentiation from all 3 germ layers (endoderm, mesoderm, and ectoderm), producing a wide variety of tissue components, whereas teratoid medulloepitheliomas have a predominance of neuroepithelial differentiation with a few interspersed heteroplastic foci. 30 Most medulloepitheliomas are classified as malignant, but teratomas in birds are usually benign. 22 Most avian teratomas are gonadal or intracelomic tumors, 8 although there are a few reports of ocular teratomas in a kestrel, 18 a heron, 24 and a turkey. 22
In most available reports of medulloepitheliomas in psittacine patients, the birds were 11 mo to 3 y old.4,25 In one report describing the occurrence of this tumor in an African grey parrot, the age of the bird was unknown. 23 The occurrence of this tumor in a 4-mo-old red-shouldered macaw reinforces the importance of including this tumor as a differential diagnosis for a unilateral ocular mass in a young psittacine bird.
Footnotes
Acknowledgements
We thank the Histology Laboratory of the Louisiana Animal Disease Diagnostic Laboratory for the immunohistochemistry.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
