Abstract
A group of finches were accidentally overdosed with ronidazole, a 5-nitroimidazole used for treatment of trichomoniasis. Finches developed neurologic signs on the third day of treatment and were euthanized (or died). Three finches were submitted for necropsy. Focal necrosis of the cerebellar nucleus was seen in all 3 birds, as characterized by neuronal necrosis, vacuolation of the neuropil, gemistocytic astrocytosis, hemorrhage, and axonal swelling (spheroids) with demyelination. The liver from 1 finch was analyzed for ronidazole and its metabolite, 2-hydroxymethyl-1-methyl-5-nitroimidazole, by high-performance liquid chromatography–mass spectrometry. Ronidazole was detected in the liver tissue at 2,700 ng/g (wet weight), and 2-hydroxymethyl-1-methyl-5-nitroimidazole was detected at 140 ng/g (wet weight).
Keywords
An owner of 40 resident finches purchased an additional 11 finches from multiple vendors at a bird market. Species in both groups included Lady Gouldian finches (Erythrura gouldiae), society finches (Lonchura striata), and owl finches (Taeniopygia bichenovii). The new finches were housed in a separate room, 1 to 4 per cage. The newly purchased finches became unthrifty, developed creamy orange diarrhea within 1 month of purchase, and were presented for veterinary care. Birds were diagnosed with trichomoniasis and treated successfully with the recommended avian dose of ronidazole (1-methyl-2-carbamoyloxymethyl-5-nitroimidazole; Ronivet-S, Vetafarm, Australia), 400 mg/liter in drinking water made fresh daily for 7 days. 11,24 The owner then decided to treat all the other finches in the collection. The information obtained by the owner from the Internet recommended 4 g ronidazole (Ronivet-S) in 4 liters (1 g per liter) of water for finches. The owner made up a dose of 4 g per quart of water (4 g/0.946 liter, or 4,200 mg/liter) and administered this as the only water source available to the original 40 finches in the collection. Nine society finches and 2 Lady Gouldian finches developed neurologic signs and/or died 3 days after treatment began. Neurologic signs included trembling, incoordination, ataxia, and fluttering on the cage floor. Birds were euthanized 2 to 3 days after clinical signs began.
Three dead society finches (Nos. 1, 2, and 3) were submitted for necropsy to the California Animal Health and Food Safety Laboratory System, Davis. All 3 finches (1 male and 2 females) were in good postmortem condition. All birds had excessive subcutaneous and abdominal fat. No gross lesions were evident on postmortem examinations of all 3 finches. A complete diagnostic evaluation was performed on each bird. No bacterial growth was obtained from aerobic cultures of the livers and lungs on split MacConkeys/blood agar plates. Salmonella was not isolated in tetrathionate broth inoculated with feces from each bird. Oropharyngeal swabs were negative for avian influenza matrix gene by reverse transcription polymerase chain reaction. Heavy metal analysis of the livers of all 3 birds—inclusive of lead, manganese, iron, mercury, arsenic, molybdenum, zinc, copper, and cadmium—revealed levels within acceptable limits except for 1 bird, which had a low liver zinc concentration of 19 mg/kg (wet weight). Fluorescent antibody test results were negative for Chlamydophila psittaci on impression smears of the livers, spleens, and air sacs.
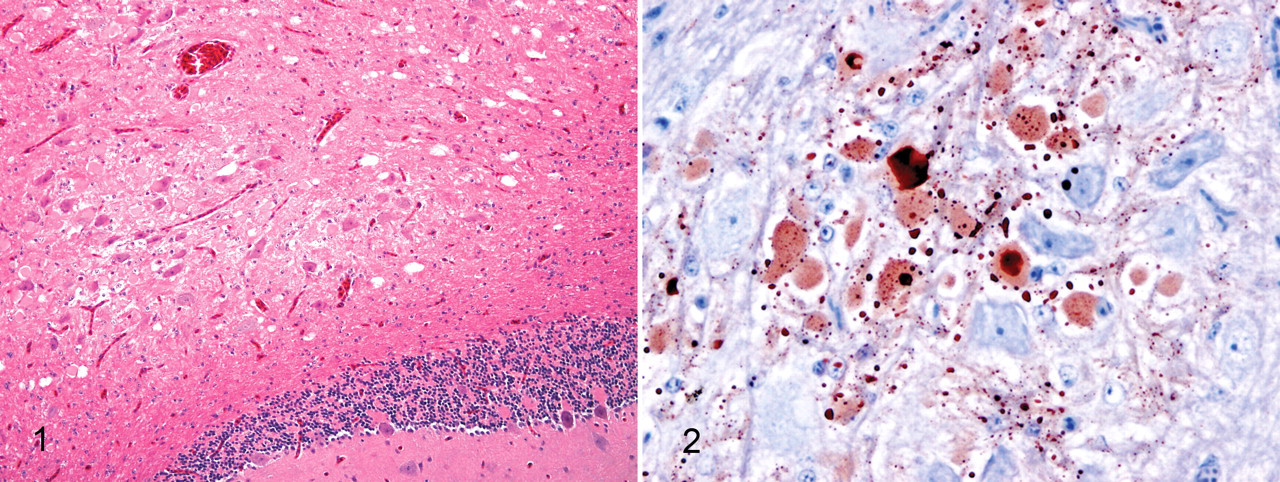
Portions of brain (parasagittal), heart, liver, kidneys, lungs, spleen, intestines, skeletal muscle, trachea, proventriculus, gizzard, esophagus, sciatic nerves, reproductive organs (oviduct and ovary from 2 birds and testes from 2 bird), thyroid gland, parathyroid glands, bone marrow, and adrenal gland were immersed in 10% buffered neutral formalin. Tissues were embedded in paraffin, and 4- to 5-μm sections were cut and stained with hematoxylin and eosin and examined by light microscopy. All 3 birds (Nos. 1, 2, and 3) had focal necrosis restricted to the cerebellar nucleus, characterized by neuronal necrosis, vacuolation of the neuropil, and gemistocytic astrocytosis (Fig. 1 ). Bird No. 1 also had rare foci of hemorrhage and axonal swelling (spheroids) with demyelination. Immunostaining of the brain of bird No. 2 with mouse monoclonal antibody SMI 31 (1:2,000 dilution; Covance Corp, Princeton, NJ) demonstrated aberrant phosphorylation of neurofilaments in labeled necrotic neurons and their processes restricted to the affected cerebellar nucleus. There were also neurons in the cerebellar nucleus that were morphologically intact and immunonegative (Fig. 2). Serial and step sections of the remaining sagittal half of the paraffin-embedded brain were examined microscopically. The other sagittal half of the brain (in formalin) was cut into 1- to 2-mm transverse sections, and the entire half brain was embedded, cut, and stained, with serial sections examined by light microscopy. Focal necrosis in the cerebellar nucleus was the only lesion seen in the brains in all 3 birds. Miscellaneous incidental findings included mild to moderate discrete enterocyte necrosis in the small intestines and mild focal necrosis in the skeletal muscle of the leg in 1 bird and mild hepatic lipidosis in another bird.
A tentative diagnosis of ronidazole toxicosis was made given the history, the clinical signs, and the lesion in the cerebellar nucleus in each of the 3 finches. Frozen liver from 1 finch (No. 1) collected during the postmortem examination was analyzed for ronidazole and its metabolite 2-hydroxymethyl-1-methyl-5-nitroimidazole (HMMNI) by high-performance liquid chromatography–mass spectrometry, modifying a previously published procedure. 28 Briefly, aliquots of liver tissue (0.5 g of control liver, 0.08 g of liver from bird No. 1) were fortified with d3-ronidazole and d3-HMMNI internal standards and homogenized in 5 ml of ethyl acetate. Each homogenate was centrifuged and the supernatant decanted into a clean tube and evaporated to dryness under nitrogen. Each extract was reconstituted in 5 ml of hexane. The hexane was extracted with 3 ml of 0.1 N hydrochloric acid, and the resulting aqueous layer was cleaned up by solid phase extraction using a mixed-mode C8/cation-exchange cartridge. Extracts were analyzed using a model 4000 Q TRAP triple-quadrupole mass spectrometer (Applied Biosystems, Foster City, CA) connected to a Paradigm high-performance liquid chromatograph (Michrom Bioresources, Auburn, CA). The chromatograph was fitted with a 150- × 4.6-mm Zorbax Eclipse XDB-C8 high-performance liquid chromatography column (Agilent Technologies, Santa Clara, CA). Gradient elution using 0.1% formic acid in water and 0.1% formic acid in methanol gave retention times of 3.75 minutes for ronidazole and 3.90 minutes for HMMNI. The mass spectrometer was run in multiple reaction-monitoring mode, with 1 precursor ion and 2 product ions monitored for each analyte. For ronidazole, the precursor ion was at m/z 201, and the product ions were at m/z 140 and m/z 110. For HMMNI, the precursor ion was at m/z 158, and the product ions were at m/z 140 and m/z 94. Single precursor–product ion transitions from m/z 204 to m/z 143 and from m/z 161 to m/z 143 were monitored for d3-ronidazole and d3-HMMNI, respectively. Quantitation was by the internal standard method using 5-point calibration curves ranging from 10 to 500 ng/ml. Limits of detection were 14 ng/g, and limits of quantitation were 35 ng/g for both compounds owing to the small sample size available. Response was linear for both compounds with r 2 values > .998. Ronidazole was detected in the finch liver tissue at a concentration of 2,700 ng/g (wet weight), and HMMNI was detected at 140 ng/g (wet weight). These results are in agreement with studies in turkeys 17 and chickens 1 that showed that HMMNI concentrations are considerably lower than 10% of the ronidazole parent compound concentrations.
Ronidazole (1-methyl-2-carbamoyloxymethyl-5-nitroimidazole) is a nitroimidazole derivative similar to metronidazole. In avian species, oral administration of ronidazole is used in the treatment of trichomoniasis, histomoniasis, cochlasomiasis, hexamitiasis, and giardiasis. 11-13,24 Recommended dose for treatment of clinical infections in avian species is 400 mg/liter in water. 11,24 Dietary ronidazole has been demonstrated to improve growth in turkeys. 15 Metronidazole (179 mg/kg body weight) and ronidazole (60 mg/liter water) were found to be effective and safe treatments in finches with subclinical infection with Cochlasoma spp. 6 The suggested mechanism of antimicrobial activity depends on the ferrodoxin-mediated reduction of the target organism nitro group, with generation of reactive metabolites that interact with DNA and other macromolecules leading to inhibition of nucleic acid and protein synthesis. 2,13 The cytotoxic mechanism of nitroimidazoles in protozoa also occurs in hypoxic mammalian cells in vitro. 8
In mammals, ronidazole has the same metabolism under anaerobic conditions as metronidazole. 16 Metabolism of ronidazole and other nitroimidazole drugs varies in turkeys and chickens, however. Dimetridazole and ipronidazole are extensively metabolized to hydroxylated nitroimidazole metabolites, whereas ronidazole is excreted mainly as a parent compound in turkeys and chickens. 1,17 Analysis of the liver of finch No. 1 demonstrated that metabolism of ronidazole in finches is likely similar. The 5-nitroimidazoles are widely distributed in the tissues of some animal species, including the brain. 4,23,27
Clinical and experimental ronidazole neurotoxicoses have been reported in rats, 10,25 dogs, 10,26 and cats. 14,22 Neurotoxicity of 5-nitroimidazoles in mammals has been theorized to be due to formation of superoxide radicals, hydrogen peroxide, nitrofurantoin radicals, and semiquinone radicals from catecholamine reduction of these drugs. 13,16,19 The mechanism of action of metronidazole neurotoxicosis may involve modulation of the inhibitory neurotransmitter γ-aminobutyric acid receptor within the cerebellar and vestibular systems. 5 Microscopic lesions in a cat with putative ronidazole neurotoxicosis were confined to the brain stem. 14 The reticular formation of the central brain stem was predominantly affected with necrotic foci in the medullary and pontine reticulospinal tracts and rubrospinal tract.
In avian species, neurotoxicosis has been reported with administration of other nitroimidazoles or their derivatives (metronidazole, dimetridazole) in turkey poults, 3 goslings, 21 adult pigeons and ducks, 20 and budgerigars. 9 Nervous signs and/or death occurred in 6 budgerigars treated with 800 mg/liter of water for the first 3 days, then 1,000 mg/liter for 2 days. 9 Nervous signs included lethargy and periodic convulsions with rapid fluttering of the wings; birds also fell onto their sides or backs, with occasional opisthotonus. Neuronal necrosis and astrocytosis were seen in the cerebral hemispheres. Dimetridazole (1,000 mg/liter in drinking water) caused nervous signs (day 2) and/or mortality in both 3-week-old ducklings and 2- to 3-week-old goslings, and 500 mg/liter (day 3) caused growth depression and nervous signs (abnormal head attitudes, excess activity, ataxia, vocalization, head pressing and recumbency). 21 No microscopic lesions were seen in the brains of any of the birds except for 1 bird that had “vacuolation in the white matter in the center of the cerebellum.” The author suggested that this may represent malacia but could be artifact. The similar location of the lesions in the gosling and in the finches in our report (Nos. 1, 2, and 3) suggest that the lesion in the gosling was likely malacia and not artifact.
Morphological variability of the cerebellar nuclei is well documented in avian species. 7 The nuclear complex is divided into the nucleus cerebellaris internus, the nucleus cerebellaris intermedius, the nucleus cerebellaris lateralis superior, and the nucleus cerebellaris inferior. 7 The superior vestibular nucleus enters at the level of this nuclear complex. It is suggested that the subdivisions relate to fiber tracts and the size of the cerebellar nuclei correlates with that of the inferior olive. There are numerous afferent tracts to the cerebellar cortex from the brain stem and midbrain nuclei and from the spinal cord. Efferent cerebellar tracts originate in the cerebellar nuclei with pathways to nuclei in the medulla oblongata, spinal cord, and mesencephalon. The cerebellum coordinates interaction of the tail, wings, and legs of birds; thus, intention tremors (“trembling”), falling to the side, fluttering, and ataxia in the birds administered toxic levels of ronidazole correlate with the lesion in the cerebellar nucleus of the brain.
Analysis of the liver from bird No. 1 for ronidazole and its primary metabolite HMMNI confirmed their presence. There are currently no data for liver ronidazole and HMMNI concentrations in clinical cases of toxicosis; however, there are limited data for liver ronidazole and HMMNI concentrations in turkeys given therapeutic doses of ronidazole. 17 In that study, turkeys were given ronidazole via drinking water (120 mg/liter), and liver ronidazole and HMMNI concentrations were determined after a 1-day withdrawal period. The metabolite and the parent ronidazole compound were together present in concentrations below 1 ng/g, relative to the 2,700 ng/g of ronidazole and 140 ng/g of HMMNI detected in the liver from the tested finch. Our findings suggest that toxicologic analysis can be used to confirm exposure to ronidazole in birds to estimate overexposures.
This report of ronidazole toxicosis of finches, at a dose of 4,200 mg/liter in drinking water, demonstrates targeting of the cerebellar nucleus. Examples of neurotoxins that target specific loci in the brain are numerous and include domoic acid in the limbic system 18 and Escherichia coli verotoxin, which causes cerebellar edema, hemorrhage, and alterations of Purkinje cells associated with verotoxin receptors (galabiosyl ceramide). 29 The action of some neurotoxins is associated with the biophysiologic properties of cells in specific loci of the brain, whereas others are related to specialized receptors.
In summary, ronidazole toxicosis should be suspected when focal necrosis of the cerebellar nucleus is the primary lesion seen on microscopic examination of finches with a clinical history of neurologic disease. Liquid chromatography–mass spectrometry analysis of the liver is an effective method for detection and quantitation of ronidazole and the HMMNI metabolite. Based on the kinetics and tissue residue studies of ronidazole, liver (and muscle) samples would be the tissues of choice to submit for analysis from birds with suspected ronidazole poisoning. Additionally, the suspected medication or source material could be analyzed to evaluate its true concentration. A definitive diagnosis of ronidazole toxicosis can be made based on the history of clinical signs and administration, microscopic lesions, and toxicologic procedures used in the birds of this report.
Footnotes
Acknowledgements
We thank Jim Koobs, Mike Manzer, Karen Sverlow, and Diane Naydan for excellent technical assistance.
The authors declared no potential conflicts of interests with respect to the authorship and/or publication of this article.
The authors received no financial support for the research and/or authorship of this article.
