Abstract
The Wnt/β-catenin signal transduction pathway is important in many developmental processes and during tumorigenesis. β-Catenin acts as a signal transducer. To investigate whether the Wnt/β-catenin signal transduction pathway is involved in canine cutaneous melanomagenesis, 18 formalin-fixed paraffin-embedded canine cutaneous melanotic tumor tissues were examined. For cloning and sequencing of the full-length canine ctnnb1 gene encoding β-catenin, conserved sequences of the human and mouse ctnnb1 gene were used to design the primers. For analysis of expression and translocation of β-catenin in canine cutaneous melanotic tumors, semiquantitative reverse transcription polymerase chain reaction (RT-PCR) and immunohistochemistry were performed. The canine ctnnb1 sequence showed a high degree of similarity to those of human and mouse. Semiquantitative RT-PCR showed a substantial increase in expression of ctnnb1 mRNA in canine cutaneous melanotic tumors compared to normal canine melanocytes, regardless of whether the tumor was benign or malignant. Immunohistochemistry revealed cytoplasmic accumulation of β-catenin in melanotic tumors. In melanoma tissues, nuclear translocation of β-catenin was also observed. The present study demonstrated that abnormal intracellular accumulation and substantially increased expression of β-catenin are involved in canine cutaneous melanotic tumor.
Keywords
Cutaneous melanotic tumor is a common skin tumor in dogs, accounting for 5% to 7% of all canine skin tumors. 1,20 The tumor originates from the transformation of melanocytes, which are present mainly in the epidermis and hair follicles. The transformed melanocytes lose normal contact with the surrounding keratinocytes and proliferate into the surrounding tissues. 23 Breed has been reported to be prognostically significant in the development of melanotic tumor: More than 75% of melanotic tumors in Doberman Pinschers and Miniature Schnauzers are behaviorally benign, whereas 85% of melanotic tumors in Miniature Poodles are malignant. 9 Some investigations into the molecular and genetic basis of canine cutaneous melanotic (CCM) tumor have been made, 7,12,15,19 but the cause of the tumor is largely unknown.
The Wnt/β-catenin signaling pathway is important in many developmental processes, including the formation of neural crest–derived melanocytes. 14 β-Catenin, which is encoded by the gene ctnnb1, acts as a signal transducer. In neural tube formation during embryogenesis, this pathway has been implicated in the migration and differentiation of melanoblasts in a β-catenin-dependent manner. 8,11 Following embryogenesis, Wnt signaling ceases, and β-catenin acts as a component of the cadherin–catenin adhesion complex in the plasma membrane. The cytoplasmic concentration of β-catenin is kept low by phosphorylation of the protein, followed by degradation. Dysregulation of the Wnt/β-catenin signaling pathway is associated with uncontrolled cell proliferation and differentiation caused by the accumulation of β-catenin in the cells. 13,18,21 As of yet, the participation of the Wnt/β-catenin pathway in CCM tumor has not been investigated.
The purpose of this study was to examine the translocation and expression of β-catenin in CCM in comparison to normal controls. We propose that translocation and altered expression of β-catenin occurs in CCM and may play a crucial role in the pathogenesis of this disease.
Materials and Methods
Melanoma Patient Information
This study was performed on the patients referred to Veterinary Teaching Hospitals in South Korea. Their cases were diagnosed histopathologically as CCM. The diagnosis between melanocytoma and melanoma was made from anisocytosis, anisokaryosis, invasiveness, and mitotic rate. The mean age of all patients with CCM was 9.4 ± 3.4 years old (range, 2–16 years). The mean age of all patients with benign tumor (n, 16) was 8.9 ± 3.1 years old (range, 2–14 years). Ten dogs were male: 8 intact and 2 neutered. Six dogs were intact females. One patient with malignant tumor was a 12-year-old intact male, and the other patient with malignant tumor was a 16-year-old intact female.
Cloning of Full-Length Canine ctnnb1 cDNA
As a preliminary study for examination of the expression level of canine ctnnb1 mRNA, the full-length canine ctnnb1 cDNA sequence was determined. Total RNA was extracted from normal canine testis tissue using TRIzol Reagent (Invitrogen Life Technologies, Inc, Carlsbad, CA). The concentrations of total RNA extracted were determined by measuring the absorbance at 260 nm. Total RNA (0.5 μg) was inserted by reverse transcription into single-stranded cDNA using random hexamers (Invitrogen) and the Superscript First-strand Synthesis System of a reverse transcription polymerase chain reaction (RT-PCR) kit (Invitrogen).
The conserved sequences of the ctnnb1 mRNA of human (GenBank accession No. NM_001904) and mouse (No. NM_007614) were used to design the primers. Each primer was designed to contain overlapping sequences of 150 to 200 base pairs (bp) at both ends of the polymerase chain reaction (PCR) products. Table 1 shows the sequences of the primers used. The conditions of the designed primers were verified by DNAMAN software, version 4.16 (Lynnon Corp, Quebec, Canada). To amplify the ctnnb1 gene, PCR amplifications were performed as follows: 1 cycle at 94°C for 5 minutes; 35 cycles at 94°C for 30 seconds, at 49° to 57°C for 30 seconds for annealing (Table 1), and then at 72°C for 1 minute; and a final cycle at 72°C for 5 minutes. Both ends of the cDNA were amplified by a series of rapid amplifications of cDNA ends (RACE) PCR experiments using a 5′-RACE kit or 3′-RACE kit (Invitrogen). The sizes of the resultant PCR products were analyzed by 2% agarose gel electrophoresis.
Oligonucleotide Sequences for Cloning of ctnnb1
a Primers used for rapid amplifications of cDNA ends polymerase chain reaction.
All PCR products, including the products of the 5′ and 3′ ends of canine ctnnb1 cDNA, were sequenced using an ABI PRISM Bigdye Terminator Cycle Sequencing Ready Reaction Kit, version 5.1 (PE Applied Biosystems, Foster City, CA). Finally, all identified sequences were merged (with the overlapped sequences excluded) and compared with the sequences of human and mouse using CLC Sequence Viewer, version 4.6.2 (CLC bio, Aarhus, Denmark).
Semiquantitative RT-PCR of ctnnb1 mRNA
Total RNA was extracted from normal melanocytes (n, 7) and formalin-fixed paraffin-embedded (FFPE) CCM tissues (n, 18). With a laser capture microdissection (LCM) system (PE Applied Biosystems), normal melanocytes were obtained from 7 different normal skin tissues of Beagles. For total RNA extraction, TRIzol Reagent (Invitrogen) was used for normal melanocytes, and PureLink FFPE Total RNA Isolation Kit (Invitrogen) was used for FFPE tissues consisting of tumor cells only. In FFPE tissues consisting of tumor cells with surrounding normal tissues, the area of the tumor was resected using the LCM system (MDS Analytical Technologies, Toronto, Canada); then, the Arcturus Paradise Extraction and Isolation Components Kit was used (MDS Analytical Technologies). The concentration of total RNA extracted was determined by measuring the absorbance at 260 nm.
Total RNA (0.5 μg) was inserted by reverse transcription into single-stranded cDNA using random hexamers (Invitrogen) and the Superscript First-strand Synthesis System of the RT-PCR kit (Invitrogen). To determine the conditions for logarithmic-phase PCR amplification of the target mRNA, cDNAs made from 0.5 μg of total RNAs were amplified using different numbers of cycles. The chloramphenicol acetyltransferase gene was used to rule out the possibility of RNA degradation. A linear relationship between PCR products and amplification cycles was observed for the target mRNAs. The template cDNA (1 μl) was amplified by PCR in a reaction volume of 50 μl containing 10mM Tris-HCl (pH 8.5), 50mM KCl, 2mM MgCl2, deoxynucleotide triphosphates (0.2mM each), and a 0.5 unit of Taq polymerase (Thermo Fisher Scientific, Cheshire, UK). Glyceraldehyde-3-phosphate dehydrogenase (GAPDH) was used as an internal control. The oligonucleotide sequences for GAPDH were 5′-GCCCTCAATGACCACTTTGT (sense) and 5′-TCCTTGGAGGCCATGTAGAC (antisense); the oligonucleotide sequences for ctnnb1 were 5′-CTGGTGACAGGGAGGACATT (sense) and 5′-CATAGTGAAGGCGAACAGCA (antisense).
PCR products were separated on a 2% agarose gel, stained with ethidium bromide, and photographed under ultraviolet illumination. The photograph was analyzed using SigmaScan Pro, version 6.0.0 (SPSS, Chicago, IL).
Immunohistochemistry of β-catenin
β-Catenin expression in FFPE CCM tissues was examined by immunohistochemistry (IHC). As a control, the skin tissues of normal Beagles were used (n, 3). Sections (4 μm) were deparaffinized in xylene and hydrated in descending grades of ethanol (100%, 95%, 90%, and 80%). Antigen retrieval was performed in citrate buffer (pH 6.0) for 10 minutes at 120°C using an autoclave. After 20 minutes of cooling, endogenous peroxidase activity was blocked with 3% hydrogen peroxide at 45°C for 5 minutes. After washing with Tris-buffered saline (TBS) Tween buffer, a nonserum protein block was applied for 5 minutes at 45°C (Ultra V Block, Lab Vision Corporation, Fremont, CA). Sections were subsequently incubated with mouse monoclonal β-catenin antibody at a 1:100 dilution (No. sc-7963, Santa Cruz Biotechnology, Inc, Santa Cruz, CA). After being washed with TBS Tween buffer, the sections were incubated with a biotinylated secondary antibody (anti-mouse, goat, and rabbit immunoglobulin G) (Dako North America, Inc, Carpinteria, CA) at 45°C for 10 minutes and further incubated with streptavidin conjugated to horseradish peroxidase at 45°C for 10 minutes. As a chromogen, diaminobenzidine or 3-amino-9-ethylcarbazole (Thermo Fisher Scientific) was used, and the sections were counterstained with Gill’s hematoxylin, followed by mounting with a coverslip.
Statistical Analyses
All data were analyzed by Student t-test (for comparing 2 different groups) or the Kruskal–Wallis test with Bonferroni correction (for comparing three different groups). All statistical analyses were performed with SPSS for Windows. P < .05 was considered statistically significant.
Results
Cloning of Full-Length Canine ctnnb1
Nucleotide sequence analysis revealed that the resultant cDNA sequence was 3,462 bp long. The full-length cDNA contained a 5′ untranslated region (UTR; 42 bp), an open-reading frame (ORF; 2,346 bp), and a 3′ UTR (1,074 bp), encoding a total of 781 amino acids. The sequence of the canine ctnnb1 ORF was 94.5% and 89.3% homologous to human and mouse ORFs at the nucleotide sequence level and 99.7% and 99.8% homologous at the amino acid sequence level, respectively (Fig. 1 ). In human and mouse, the 69th amino acid was serine, whereas in dog it was threonine. The 706th amino acid in human was alanine, whereas proline was present at this position in dog and mouse. The amino acid sequence targeted by the mouse monoclonal antibody used for IHC is the same in dog and mouse.

Semiquantitative RT-PCR
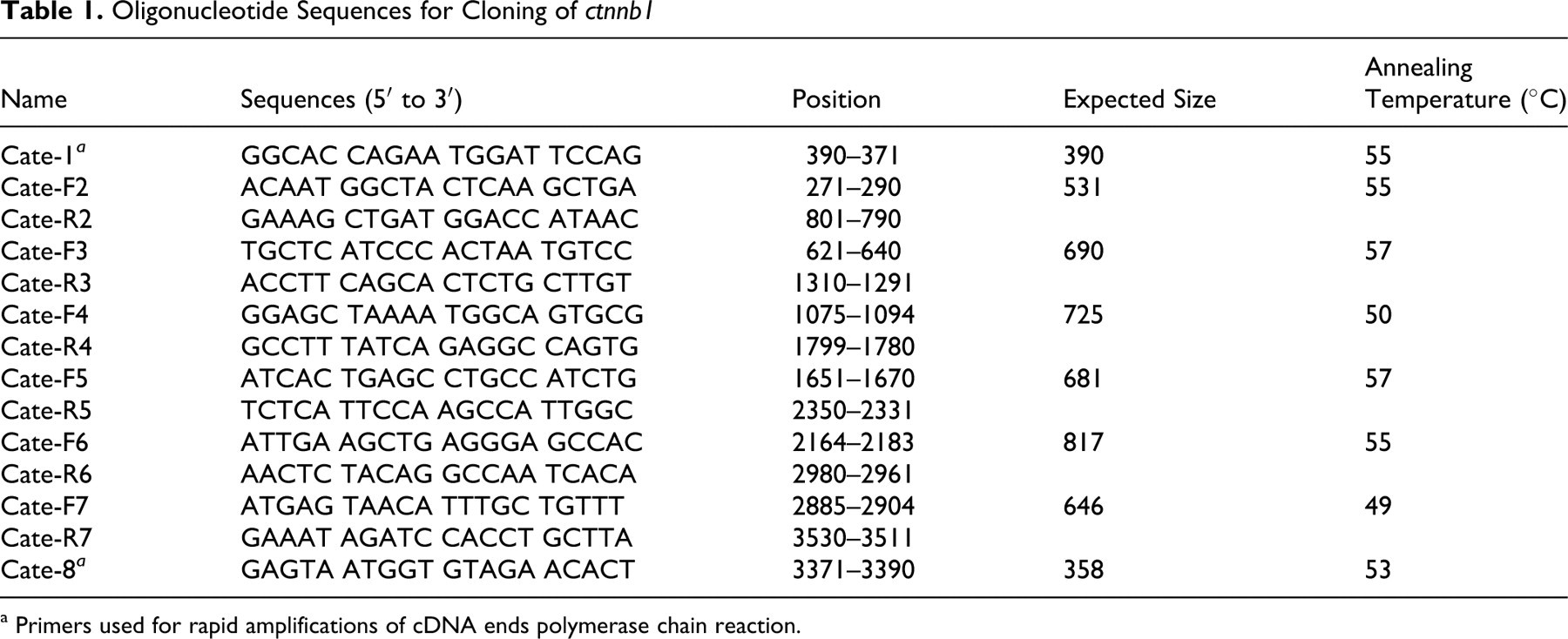
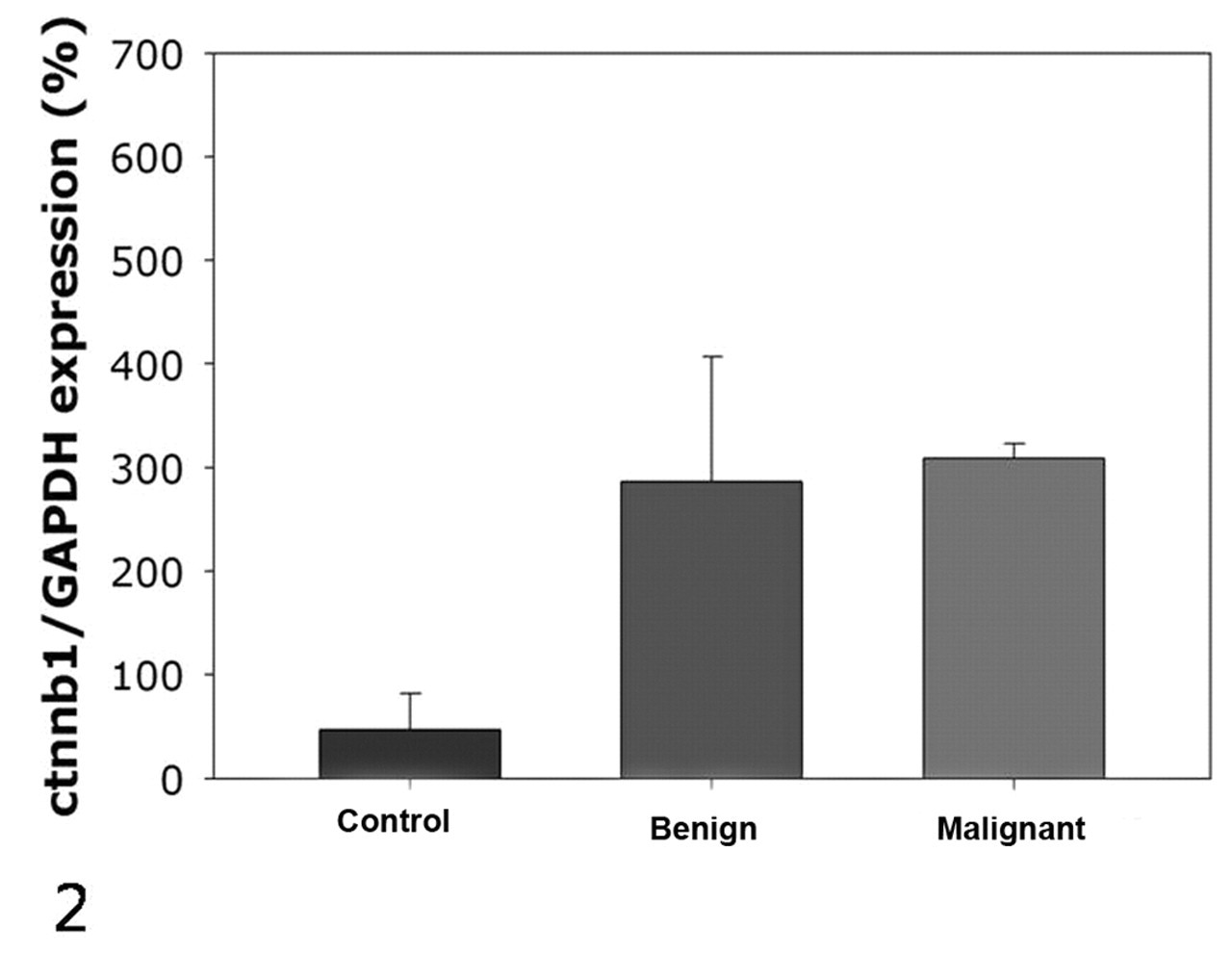
When normalized to GAPDH expression levels, normal melanocytes showed weak expression of β-catenin, whereas all melanoma tissues examined exhibited substantially higher levels of expression. Although only 2 melanomas were examined in our study, the expression levels were not different between melanocytoma and melanoma (Fig. 2) and were not different between male and female dogs (Fig. 3).
Immunohistochemistry
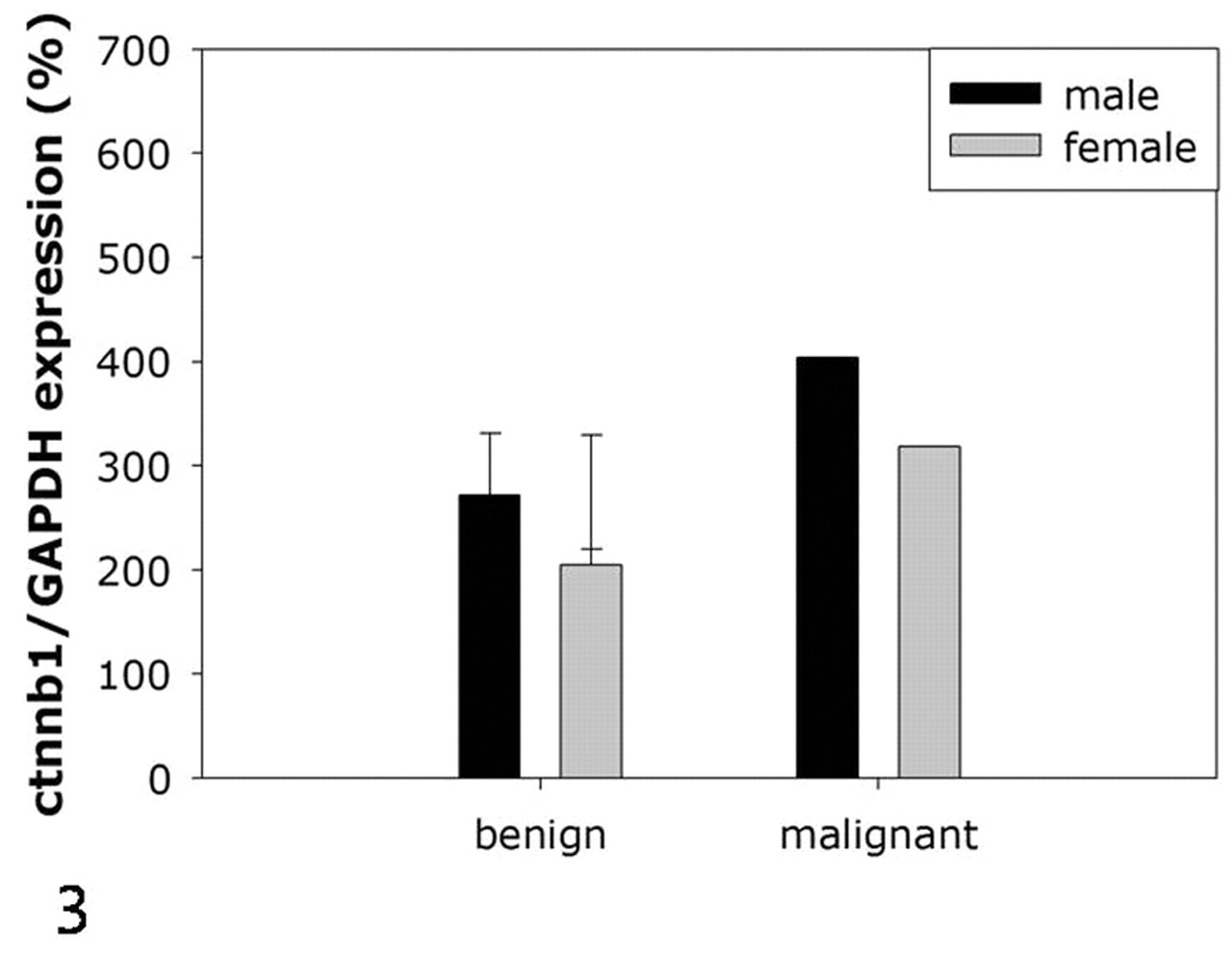
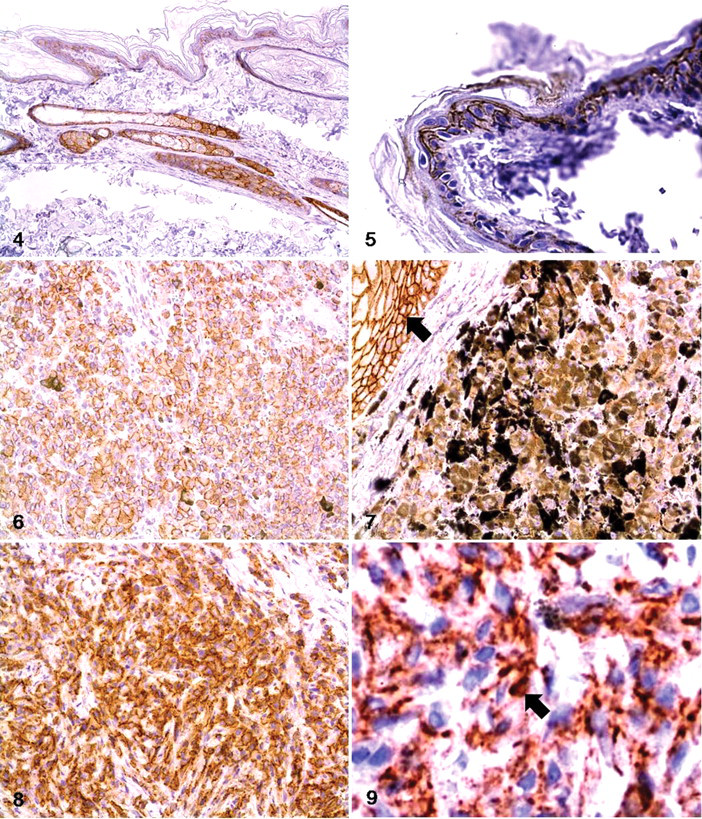
In normal skin, β-catenin was found to be expressed along the borders of epithelial cells and melanocytes. Its expression was more pronounced in the keratinocytes of hair follicles and glandular epithelial cells (Fig. 4). In the epidermis, the basal layer showed higher levels of β-catenin expression than the spinous or cornified layer did (Fig. 5). In melanocytoma, > 90% of cells in all tissues showed cytoplasmic accumulation of β-catenin and a partial loss of expression at the cell borders (Figs. 6, 7). Nuclear translocation of β-catenin was not detected in these cells. In melanoma, the degree of cytoplasmic accumulation and the expression pattern of β-catenin at the cell border were similar to that of melanocytoma (Fig. 8). In addition, β-catenin was observed in < 1% of the nucleus of the malignant cells (Fig. 9).
Discussion
In this study, we observed increased expression and intracellular accumulation of β-catenin in the FFPE CCM tissues. In the absence of Wnt signaling, β-catenin acts as part of a cell adhesion complex, and unbound β-catenin in the cytoplasm is recognized and degraded by the GSK-3β/APC/AXIN complex. 6,14,22 GSK-3β recognizes the amino acid sequence encoded by exon 3 of ctnnb1 in β-catenin and initiates phosphorylation, followed by degradation. When the GSK-3β/APC/AXIN complex is transformed or a mutation in exon 3 of the ctnnb1 gene is present, β-catenin is not degraded, and consequently, intracellular accumulation and nuclear translocation occur. 6,13,14 In addition to the dysregulation of the mechanisms that regulate β-catenin degradation, overexpression of some receptor tyrosine kinases can disrupt the cadherin–catenin complex on the cell membrane, leading to the release of β-catenin into the cytoplasm. 4 Finally, persistent activation of the Wnt/β-catenin signaling pathway by excessive Wnt factors also causes deactivation of the GSK-3β/APC/AXIN complex, followed by intracellular accumulation of β-catenin. 14 In our study, the partial loss of membrane expression and the cytoplasmic accumulation of β-catenin revealed by IHC suggests that factors affecting the adhesion of β-catenin to membrane cadherin may induce the cytoplasmic release of β-catenin. Although increased RNA synthesis does not always equate with increased protein synthesis, the increased expression of ctnnb1 mRNA revealed by semiquantitative RT-PCR suggests that increased β-catenin synthesis also has an effect on the cytoplasmic accumulation of β-catenin. In future studies, we plan to examine the relationship between the expression levels of cadherin and β-catenin on the cell membrane in FFPE CCM tissues and to examine the difference between cutaneous melanoma and oral melanoma.
In human cutaneous melanoma cell lines, the exon 3 mutation of ctnnb1 has been considered to be widespread. However, a recent study using surgically extracted human cutaneous melanoma tissues indicated that this mutation rarely occurs. 6,16 The reason for this inconsistency between the findings obtained from cell lines and human cutaneous melanoma tumor tissues is not clear; however, it suggests that the results of studies using cell lines should be interpreted with caution. It is for this reason that our study was conducted using surgically extracted tumor tissues.
Because β-catenin was discovered to be a component of an intercellular adhesion complex, the Wnt/β-catenin signaling pathway has been scrutinized in vertebrates and invertebrate organisms. 2,10,17,25 In our study, we found that the canine β-catenin protein sequences were highly homologous with those of human and mouse. Specifically, we observed that there was a single amino-acid difference in the protein sequences between mouse and dog. It is believed that the structure and physiologic functions of β-catenin in dog are similar to those of human and mouse. In particular, the protein sequence targeted by the mouse β-catenin antibody used in our study is identical in mouse and dog. In IHC, we observed that the IHC results were the same regardless of the application of Ultra V Block, a nonserum protein blocker, 24 which suggests that the antibody used in our study could apply for canine β-catenin without any nonspecific reactions.
Because of the high incidence of dysregulation of the Wnt/β-catenin signaling pathway, a genetically modified mouse model has been developed. 5 However, the location of melanocytes in mice differs from that in the human skin, and the mouse model does not spontaneously develop melanoma. 13 In dogs, melanocytes are mainly present in the basal layer of the epidermis and hair follicle, similar to those in human. In addition, the age of incidence, histopathology, and biological behavior in malignant CCM, which rapidly progresses to the vertical growth phase without the radial growth phase, are similar to the characteristics of human cutaneous nodular melanoma. 3,9,23 This suggests that malignant CCM could be a suitable model for therapeutic trials aimed at correcting alterations of the Wnt/β-catenin signaling pathway.
Footnotes
Acknowledgements
We thank the National Veterinary Research and Quarantine Service, and Professor Jae-Hoon Kim of the Department of Veterinary Pathology of Jeju National University.
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
The authors disclosed receipt of the following financial support for the research and/or authorship of this article: This work was supported by the 2009 Brain Korea Project.
