Abstract
Two groups of vespertilionid bats were collected from affected hibernacula. In group 1 (n, 14; pathology and microbiology), the average body weights of all species were at the lower limit of published ranges. Twelve bats (86%) had mycotic growth in the epidermis, hair follicles, and sebaceous glands. Geomyces destructans, with its characteristic curved conidia, was observed microscopically, cultured, and confirmed by polymerase chain reaction. Dermatitis and mural folliculitis was nil to mild. When focally coinfected with Gram-negative bacteria, there was necrosis and pustules. Fat stores were little to abundant in 12 bats (86%) and nil in 2. Thirteen bats (93%) had pulmonary congestion and 7 (50%) had bone marrow granulocytosis. In group 2 (n, 24; liver elements), 3 bats (13%) had potentially toxic lead levels and 1 (4%), potentially toxic arsenic level. There was no evidence of major organ failure or consistent element toxicity.
Hibernating bats in the Northeastern United States have been dying in inordinate numbers for unknown reasons. Because a large proportion of these dying bats have a white fungal growth on the face, particularly on the muzzle and ears, this mortality syndrome has been designated white-nose syndrome (WNS). 13 It was first reported from a cave in New York State in February 2006. During the winters of 2007 and 2008, the disease spread throughout the caves of the Northeastern United States. In 2009, it was reported in Pennsylvania and Virginia. A reduction of the bat population at 2 hibernacula in New York was estimated to be at least 75%. 2
Affected species included little brown (Myotis lucifugus), northern long-eared (Myotis septentrionalis), eastern pipistrelle (Perimyotis subflavus), and big brown (Eptesicus fuscus). Abnormal behavior has been observed in affected colonies, with bats flying out of their caves in the middle of winter and during daylight hours. They have been described as emaciated and dehydrated. 13 Bats that were examined had minimal fat stores and erosive epidermal lesions on the muzzle and wings, with a white fungus, Geomyces destructans. This fungus produces a curved conidia; it is psychrophile; and it grows or can be maintained year-round at the temperatures found in the Northeastern region hibernacula. 2,10
In the past, high mortalities of bats have been observed in the summer owing to deliberate organochlorine spraying of nursery colonies. 14 Other reports of similarly scaled massive die-offs are rare and remain unexplained. 3 Infectious diseases known to be present in Northeastern bat populations, such as rabies or histoplasmosis, do not cause such high mortalities. 19
The study was conducted to identify the lesions and any abnormal mineral and metals levels associated with the syndrome.
Sample Collection and Methods
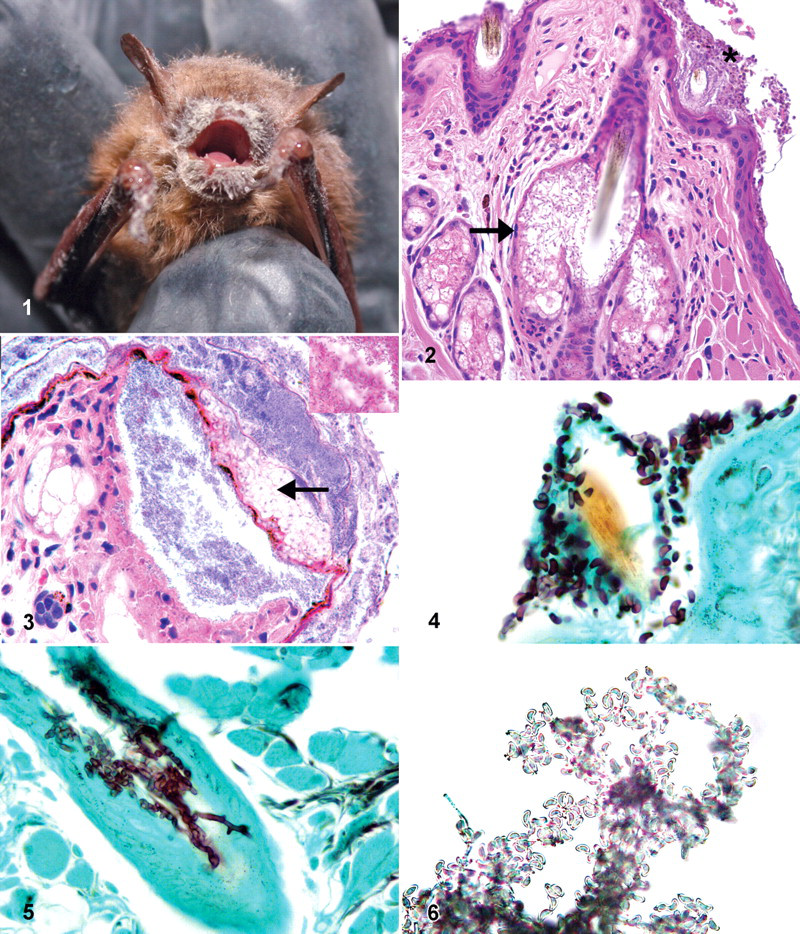
Hibernating bats were collected from hibernacula in New York and Connecticut during February and March of 2008 and 2009. Animals that were collected were dead or moribund and had grossly visible white deposits on the face (Fig. 1). Moribund animals were euthanized with CO2. The bats collected were randomly separated into 2 groups, one for postmortem and microbiological examination (14 bats) one for mineral and metal determination (24 bats). All specimens were held at 5°C until necropsy.
Of the 14 animals collected for postmortem analysis, 6 were collected alive and 8 were dead. All were measured (nose to distal perineum) and weighed. Animals were necropsied, and an impression from the brain was submitted for rabies immunofluorescence testing. Tissues were fixed in 10% neutral buffered formalin and processed routinely for histopathology examination. Skin swabs from the faces of 3 animals were cultured for fungus on Sabouraud’s dextrose agar at 5° and 30°C, with 80% moisture for a minimum of 2 weeks. Two colonies from different animals had a morphology consistent with the one observed in the histologic skin lesions and were selected for molecular analysis. Fungal DNA was detected by polymerase chain reaction, using primer sets that target the noncoding internal transcribed spacer region 1 and 2 of the 26S ribosomal RNA gene—primer set ITS1 (5′-TCCGTAGGTGAACCTGCGG-3′) / ITS2 (5′-GC TGCGTTCTTCATCGATGC-3′) and primer set ITS3 (5′- GCATCGATGAAGAACGCAGC-3′) / ITS4 (5′-TCCTCCGCTTATTGATATGC-3) respectively—using conditions previously described. 5 Sequence identities were determined using the National Center for Biotechnology Information software Basic Local Alignment Search Tool.
Mineral and Toxicologic Testing
The presence of the fungus in the skin lesions was confirmed by direct microscopy of skin scrapings. Animals were necropsied and liver was collected. Wet weight mineral and metal analysis was performed by inductively coupled plasma mass spectroscopy. The dry weight concentration was estimated by multiplying the wet weight result by a factor of 4, a widely used conversion factor. 16
Results
Fourteen bats, 8 males and 6 females, were examined. Table 1 presents the biometry and histopathology findings. At the time of necropsy, 5 bats had one or more 1- to 2-mm delicate white deposits on the fur of the face, and of these, 2 also had deposits on body and wings (Fig. 1). Internal organs of all 14 were free of macroscopic lesions and all were rabies negative. In the skin of 12 bats, particularly on the face but also randomly on the body and wings, there were aggregates of fungal hyphae and keratinic debris adherent to the epidermis, variably mixed with different species of bacteria (Fig. 2). Hyphae extended into the stratum corneum and along hair follicles, colonizing the root sheaths and associated sebaceous glands (Fig. 2). On the nonhaired skin (eg, wings), fungal hyphae, when present, were focally pasted at the surface or between thin layers of the stratum corneum. Neutrophilic infiltrates in the hair follicle interface or dermis were usually sparse. In 3 bats, the stratum corneum, as well as the dermal–epidermal interface, was split and formed spaces that were colonized by fungus or Gram-negative bacilli. In these cases, there was necrosis of the epidermis with a marked focal neutrophilic infiltrate (Fig. 3). Careful examination of the fungal morphology in the superficial epidermis revealed clusters of 2- × 6-µm curved conidia attached to the hyphae (Fig. 4). In tissues, fungal hyphae were slender (2 to 3 µm in diameter), with parallel nonpigmented walls, septation, and right-angle branching (Fig. 5). In dead animals, hyphae often extended deeper from root sheaths and sebaceous glands into the surrounding tissues. In moribund animals, the fungus most often did not traverse the basement membrane of the root sheaths or sebaceous glands. Thirteen bats had marked pulmonary congestion with abundant intravascular neutrophils. Five of these had small foci of neutrophils in alveolar septae or spaces, together with edema material. Seven bats had marked bone marrow granulocytosis. All bats had adipose tissue composed of cells with a single large lipid globule and a marginal compressed nucleus (white fat). The interscapular adipose tissue, composed of adipocytes with multiple lipid droplets and a central nucleus (brown fat), contained fine to large lipid vacuoles in 12 bats and no appreciable lipid vacuoles in 2. Ten of the 14 bats had gastrointestinal flukes; 5 had gastrointestinal nematode larvae; 1 had intestinal cestodes morphometrically consistent with Hymenolepsis christensii; 2 had arthropod ectoparasites.
Biometry and Histopathologic Findings in 14 Hibernating Bats With White-Nose Syndrome in the Northeastern United States
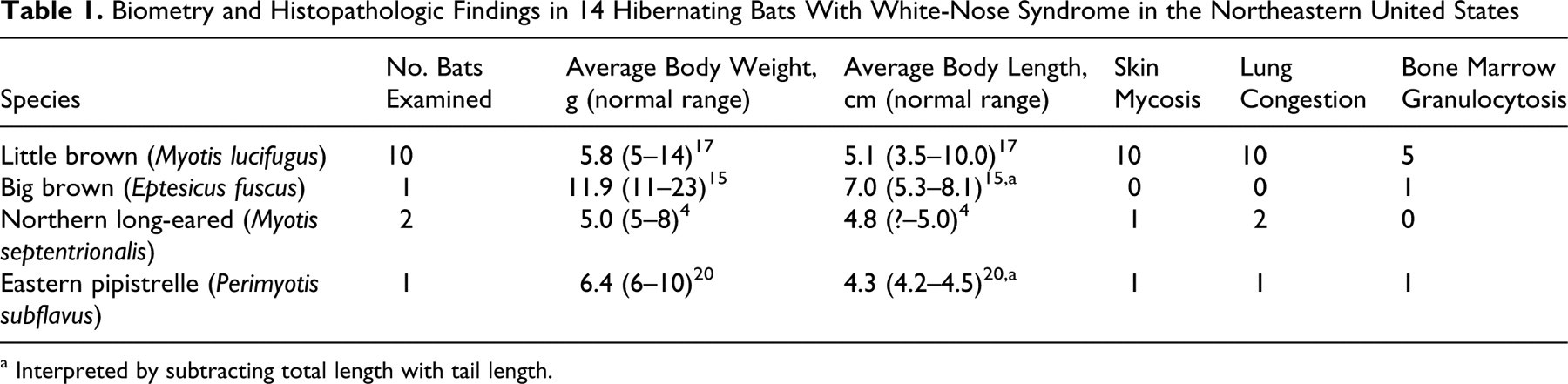
a Interpreted by subtracting total length with tail length.
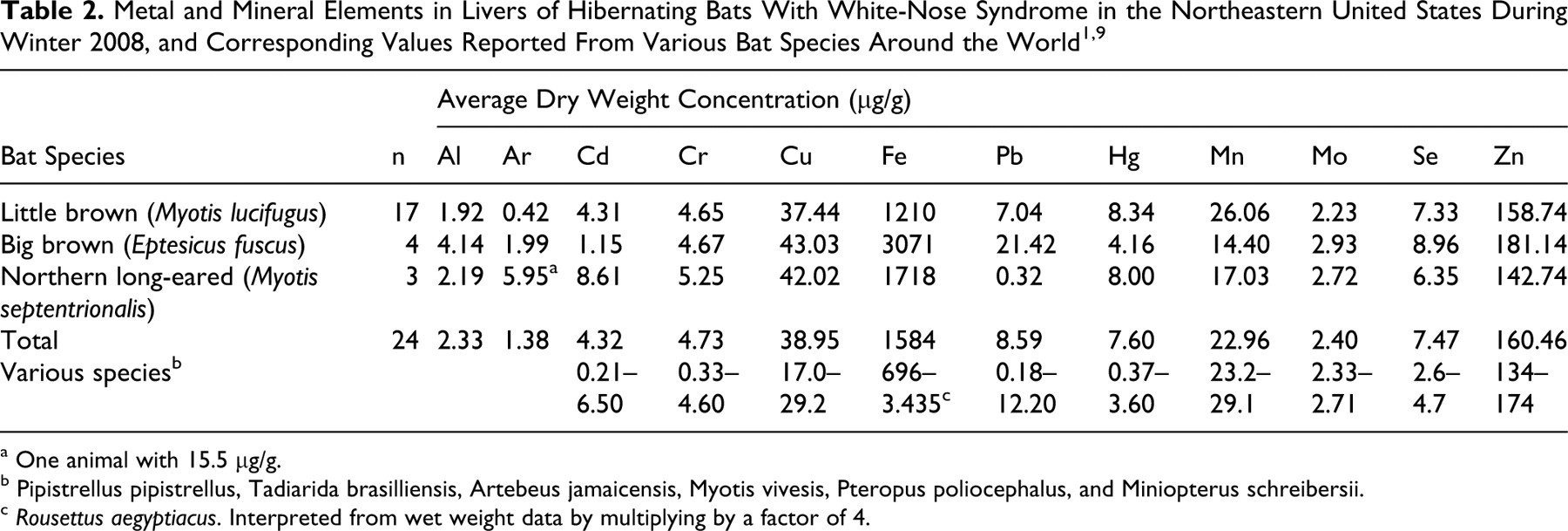
Cultures of the skin samples held at 5°C grew white pinpoint colonies in 7 days that expanded and turned smokey green at the center. The microscopic morphology of these colonies (Fig. 6) was consistent with the morphology of the fungus in histologic sections. The molecular analysis of the 2 isolates produced an ITS1 255–base pair (255-bp) amplicon that showed 100% sequence similarity to GenBank accessions of G destructans and Geomyces pannorum. ITS2 337-bp amplicon (CVMDL ITS2) showed > 99% identity with Geomyces spp, including the IFS-2009a isolated from bats with WNS (Fig. 7). Other species that grew at 5° and/or 30°C were Cladosporium spp, Penicillium spp, Trichophyton terrestre, Aspergillus terreus, and Mucor spp. These fungi were morphologically different from the fungus observed in histologic sections. Table 2 presents livers elements, with corresponding published “normal” values for various bat species.

a One animal with 15.5 µg/g.
b Pipistrellus pipistrellus, Tadiarida brasilliensis, Artebeus jamaicensis, Myotis vivesis, Pteropus poliocephalus, and Miniopterus schreibersii.
c Rousettus aegyptiacus. Interpreted from wet weight data by multiplying by a factor of 4.
Discussion
Similar to cases reported previously, the majority of our cases comprised little brown bats, 2 which are a predominant part of the vespertilionid species in the Northeastern hibernacula of the United States. In the Roxbury mine in Connecticut, for instance, they represented 81% and 70% of the total hibernating bats in 2005 and 2007, respectively, before WNS was observed (unpublished data, Department of Environmental Protection, State of Connecticut).
Body weights of the bats reported here were consistently at the lower end of the normal range, whereas average sizes were mid- to upper-normal range. Previous investigators reported that 65% of their cases had “little or no identifiable” fat stores 2 ; however, the majority of our cases had a significantly higher, appreciable degree of adipose stores. The endoparasitism observed in our cases was consistent with parasite burdens reported previously in little brown bats. 6
G destructans produces a characteristic conidia that can be easily and reliably identified by direct microscopic examination of skin scrapings of the lesions. Cutaneous mycoses typically produce variable lesions depending on agent properties, host response and bacterial superinfection. 11 This fungus has invasive properties that differ from those of dermatophytes in that it extends into the noncorneal layers of the epidermis and sebaceous glands. Hibernation-induced immunodepression may explain a minimal inflammatory host response in many cases. This has been recognized in hibernating red-cheeked ground squirrels (at 3°C to 4°C) that were experimentally exposed to Trichophyton mentagrophytes. 18 We observed significant necrosis and inflammation mainly when there were abundant bacteria. Interestingly, we and others did not observe the fungus in the big brown specimens. 2 When hibernating, big brown bats rest in drier, more ventilated areas than do little browns. 20 Overall, this behavior may have an importance in the exposure to the fungus.
We confirm what others have reported—namely, that there is no evidence of major organ failure. 2 Pulmonary congestion and mild focal pneumonitis were frequent changes. Pulmonary congestion is often an unspecific necropsy finding. However, in a large number of our cases, it was associated with a large number of circulating neutrophils and with bone marrow granulocytosis. One interpretation is that these represent early inflammation.
Mercury and selenium were consistently higher than levels found in other species of bats around the world. 1 These are not excessive considering that mercury is toxic above 20 µg/g and that selenium levels between 4 and 10 µg/g were measured in healthy birds. 8 Three bats had elevated lead, and 1 elevated arsenic, which are potentially toxic levels when compared to those of fruit bats, birds, and domestic animals. 7,12 However, these elevated levels were sporadic, and we did not observe microscopic lesions compatible with toxicity in our postmortem study group.
Footnotes
Acknowledgements
We thank the histology and microbiology staff at the Connecticut Veterinary Medical Diagnostic Laboratory; Salvatore Frasca, Milton Levin, and Radakrishna Sura at the Department of Pathobiology and Veterinary Science; Mohamed Faizal at the University Center for Instructional Media and Technology, University of Connecticut; Jeff Hall at the Utah State Laboratory; Jenny Dickson and Christina Kocer at the Department of Environmental Protection, State of Connecticut; Lisle Eaton at the Pathology Laboratory of St Peters Hospital, Albany, New York; Joseph Okonievski at the Department of Environmental Conservation, State of New York; Joseph Hoyt and Sammi Kadhin at State University of New York, Cobleskill.
The authors declared that they had no conflicts of interests with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.