Abstract
Objectives
This network meta-analysis aimed to assess the safety profiles of seven commonly used oral antimuscarinic drugs (darifenacin, fesoterodine, imidafenacin, oxybutynin, propiverine, solifenacin, and tolterodine) in patients with overactive bladder (OAB).
Methods
PubMed, Cochrane Library, EMBASE, CNKI, and Wanfang databases were searched for randomized controlled trials (RCTs). Studies comparing one or more antimuscarinic drugs for treating OAB with reported adverse effects (AEs) were eligible. Data were extracted, and a network meta-analysis was performed by two authors independently.
Results
Forty-five RCTs and 124,587 patients were included. The results demonstrated that tolterodine had better safety outcomes for 7 out of 12 major AEs, including dry mouth, constipation, urinary retention, dizziness, urinary tract infection, dry eyes, and dry skin. Darifenacin, fesoterodine, imidafenacin, oxybutynin, and solifenacin presented comparable safety profiles.
Conclusions
Tolterodine may be preferable as it showed a reduced association with important AEs. Darifenacin, fesoterodine, imidafenacin, oxybutynin, and solifenacin have similar safety profiles in treating patients with OAB. Taken together, this analysis provides a valuable overview of the therapeutic safety for oral antimuscarinic drugs and is useful for personalized medicine in patients with OAB.
Keywords
Introduction
Overactive bladder (OAB) syndrome is a common symptom syndrome defined as urinary urgency, typically with frequency and nocturia, and patients present with or without urinary incontinence. 1 As neuronal and non-neuronal acetylcholine plays an important role in bladder function, antimuscarinic drugs are widely used for treating patients with OAB. 2 However, due to the distribution of muscarinic receptors outside the bladder, there are several unfavorable adverse effects (AEs) associated with these drugs, such as dry mouth, constipation, urinary retention, and blurred vision. These AEs have a negative impact on the quality of life for patients with OAB and might limit their adherence to these drugs. 2 Additionally, different antimuscarinic products are reported to have similar clinical effectiveness profiles (improvement in incontinence per 24-hour period, frequency, and voided volume). 2 This makes the safety profiles of each drug a critical factor in the personalized drug selection for patients with OAB.3,4 Although several studies have reported safety outcomes,3–10 the differences in the overall therapeutic safety profiles among different antimuscarinics are unclear and need to be updated. In this study, we conducted a network meta-analysis of the medical literature to compare the safety profiles of seven commonly used antimuscarinic drugs (darifenacin, fesoterodine, imidafenacin, oxybutynin, propiverine, solifenacin, and tolterodine).
Materials and methods
Data sources
Two investigators independently searched the following electronic databases (from inception until March 2020): PubMed, the Cochrane Library, Embase, CNKI, and Wangfang. Only Chinese and English papers were accepted. The following keywords were included: “overactive bladder”, “urinary incontinence”, “darifenacin”, “fesoterodine”, “imidafenacin”, “oxybutynin”, “propiverine”, “solifenacin”, and “tolterodine”.
Eligibility criteria
The inclusion criteria were as follows: (1) a randomized clinical trial design and compared an antimuscarinic drug with placebo or other antimuscarinic drugs in patients diagnosed with OAB; (2) antimuscarinic drugs included darifenacin, fesoterodine, imidafenacin, oxybutynin, propiverine, solifenacin, or tolterodine; (3) reported safety outcomes, such as dry mouth, constipation, urinary retention, and blurred vision. Reviews, meta-analyses, observational studies, and pilot studies were excluded. Multi-arm studies were allowed. There were no restrictions on the sex of patients or the cause of OAB.
Data extraction and analysis
All data were assessed by two investigators independently, and disagreements were resolved by a third author. Baseline characteristics (sample size, age, and intervention), outcome data, and quality information were extracted from eligible studies.
Data analysis was conducted with Stata 15.0 (StataCorp LLC, College Station, Texas, USA). 11 The results are presented as standard mean differences and 95% confidence intervals. To obtain differences between each treatment with direct and indirect evidence, a network meta-analysis in a frequentist framework was used to analyze each outcome. P values <0.05 were considered statistically significant. The treatments were ranked for each outcome. 12 Inconsistency was assessed by calculating the inconsistency factors. 13 This study followed the PRISMA guidelines. 14 This trial was retrospectively registered at INPLASY (https://inplasy.com/) with the registration number 202170095. Ethical board approval and informed consent were not applicable because this research did not contain any studies with human participants or animals performed by any of the authors.
Results
Study selection and characteristics
Forty-five articles were selected from 248 articles based on the inclusion and exclusion criteria. This resulted in 45 eligible trials with 124,587 patients (Ref 1–45, Appendix Table A.1). The selection process is shown in Figure 1.

Flowchart detailing data selection.
The baseline characteristics of the included studies are detailed in Table 1. Ten studies were performed in female patients only, accounting for 22% of the total studies. Twelve weeks was the most common length of treatment (18/45, 40%). Five studies were shorter than 4 weeks, 13 were between 4 to 12 weeks, and 9 were longer than 12 weeks. Thirty-two studies (71%) used fixed doses during the trial.
Baseline characteristics of included studies.
ER, extended release; IR, immediate release.
The safety outcomes were extracted from the eligible studies and used for network meta-analysis. The evaluated safety outcomes included dry mouth, constipation, urinary retention, blurred vision, dizziness, headache, urinary tract infection, dry eyes, dry skin, abdominal pain, dyspepsia, and dysuria. The results are detailed as follows.
Dry mouth
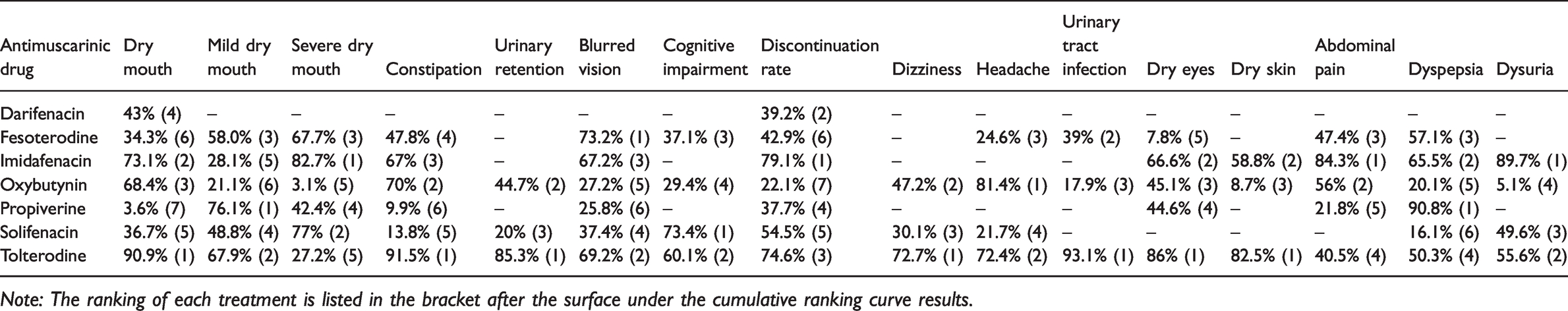
The results for dry mouth for the seven antimuscarinic drugs were available from 36 studies (Appendix Table A.2). Tolterodine was ranked the best treatment for this safety outcome with a 60.5% probability of being the best, followed by imidafenacin (36.8%), fesoterodine (1.0%), darifenacin (0.6%), solifenacin (0.5%), oxybutynin (0.4%), and propiverine (0.2%) (Table 2). There were 21 different pairwise comparisons in total. Among the pairwise comparisons, 14 were direct comparisons (Appendix Figure A.1), and the remaining 7 (darifenacin vs. fesoterodine, imidafenacin, oxybutynin, or propiverine and propiverine vs. fesoterodine, oxybutynin, or tolterodine) were indirect comparisons. Pairwise comparisons demonstrated that tolterodine was significantly superior to darifenacin, fesoterodine, and oxybutynin, imidafenacin was significantly superior to oxybutynin and propiverine, and fesoterodine and solifenacin were significantly superior to oxybutynin (P < 0.05) (Figure 2a).
Ranking of each antimuscarinic drug for the adverse effect outcomes.

Note: The ranking of each treatment is listed in the bracket after the surface under the cumulative ranking curve results.
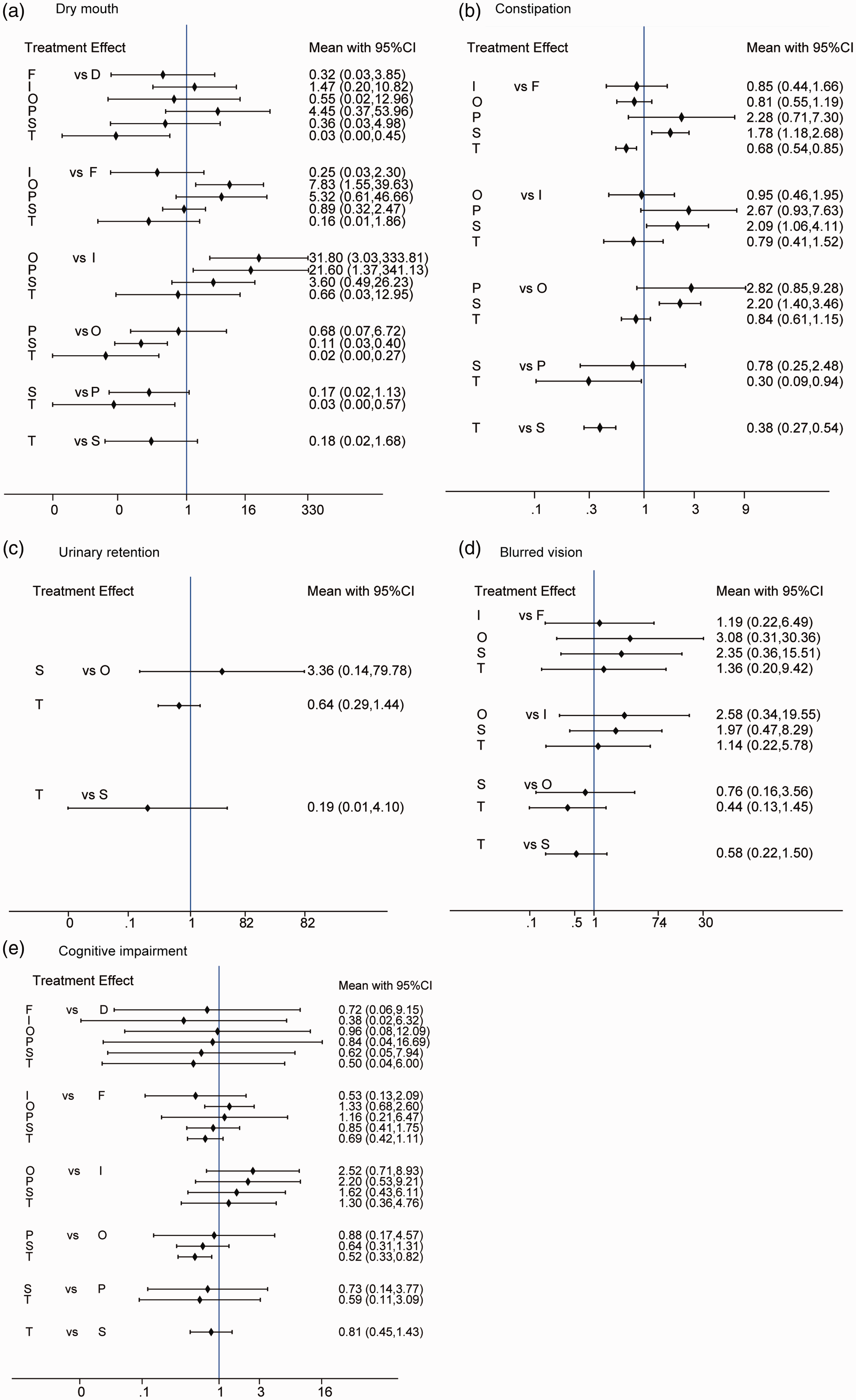
Forest plot of comparisons among different antimuscarinic drugs for adverse effects. a. dry mouth, b. constipation, c. urinary retention, d. blurred vision, and e. cognitive impairment. The mean is the value of ([the ratio of left drug]/[the ratio of right drug]) for the indicated outcome. Error bars indicate the 95% confidence interval (CI) for each comparison. If the error bar crosses with the vertical line (X = 1), there is no significant difference between the two drugs compared. If there is an intersection between the error bar and the vertical line (X = 1), a significant difference exists between the two drugs compared
The rate of mild dry mouth for each drug included was also evaluated for usable studies. Propiverine showed the lowest incidence of dry mouth (the probability of being the best was 58.2%), followed by tolterodine (24.6%), fesoterodine (12.2%), solifenacin (3.5%), imidafenacin (1.0%), and oxybutynin (0.5%). We then analyzed the rate of severe dry mouth, and the results showed that imidafenacin had the highest rate of this AE (50.2%), followed by solifenacin (35.1%), fesoterodine (12.1%), propiverine (2.6%), oxybutynin (0.0%), and tolterodine (0.0%) (Table 2).
Constipation
Twenty-seven studies were eligible for the evaluation of constipation (Appendix Table A.2). Tolterodine was ranked first with a 62.9% probability of having a lower incidence of constipation, followed by imidafenacin (25.0%), oxybutynin (10.9%), and propiverine (1.2%). Fesoterodine and solifenacin appeared to have higher rates of constipation as the probabilities for being the best were both 0.0% (Table 2). No included darifenacin studies were usable for this safety outcome analysis. There were 15 pairwise comparisons, including 11 direct comparisons (Appendix Figure A.1) and 4 indirect comparisons (imidafenacin vs. oxybutynin and propiverine vs. fesoterodine, oxybutynin, and tolterodine). In pairwise comparisons, tolterodine was statistically superior to fesoterodine, propiverine, and solifenacin, whereas solifenacin had a statistically higher constipation rate than fesoterodine, imidafenacin, and oxybutynin (P < 0.05) (Figure 2b).
Urinary retention
Seven studies reported urinary retention results and were eligible for further analysis (Appendix Table A.2). Tolterodine was reported to have less urinary retention rates than other drugs, with a 72.9% probability of being the best, followed by solifenacin (14.7%) and oxybutynin (12.4%) (Table 2). No darifenacin, fesoterodine, imidafenacin, or propiverine studies reported usable urinary retention outcomes. Thus, three pairwise comparisons were obtained, including two direct comparisons (Appendix Figure A.1) and one indirect comparison (oxybutynin vs. solifenacin). No statistically significant differences were observed between the different drugs, as shown in Figure 2c.
Blurred vision
The usable outcomes for analyzing blurred vision were extracted from 15 trials (Appendix Table A.2). Fesoterodine was ranked the best treatment for this AE, with a 43.8% probability of being the best, followed by tolterodine (23.1%) and imidafenacin (22.4%). Propiverine (5.5%) ranked fourth, oxybutynin (3.3%) ranked sixth, and solifenacin (1.9%) ranked last. No usable data were obtained for darifenacin, as shown in Table 2. In total, 15 pairwise comparisons were obtained, including 7 direct comparisons (Appendix Figure A.1) and 8 indirect comparisons (fesoterodine vs. oxybutynin, propiverine, and solifenacin; imidafenacin vs. oxybutynin and tolterodine; oxybutynin vs. solifenacin; propiverine vs. tolterodine). No significant differences were observed for this outcome (Figure 2d).
Cognitive impairment
There were five available studies for analyzing cognitive impairment (Appendix Table A.2). Solifenacin showed the lowest incidence of cognitive impairment with a 65.7% probability for being the best, followed by tolterodine (18.8%), fesoterodine (14.7%), and oxybutynin (0.8%). No usable data were obtained for darifenacin, imidafenacin, or propiverine (Table 2). Six pairwise comparisons were obtained, including three direct comparisons (Appendix Figure A.1) and three indirect comparisons (fesoterodine vs. solifenacin and tolterodine; solifenacin vs. tolterodine). No significant differences were observed for this outcome (Figure 2e).
Other safety outcomes
In the current study, we also assessed other reported usable safety outcomes. Briefly, oxybutynin was associated with a lower rate of headache, but no significant differences were observed among the four drugs (fesoterodine, oxybutynin, solifenacin, and tolterodine) included. For dyspepsia, propiverine ranked first among the six drugs (fesoterodine, imidafenacin, oxybutynin, propiverine, solifenacin, and tolterodine) included, and no significant differences were observed. Imidafenacin showed less abdominal pain (fesoterodine, imidafenacin, oxybutynin, propiverine, and tolterodine) and dysuria (imidafenacin, oxybutynin, solifenacin, and tolterodine). Tolterodine was ranked the best treatment for dizziness (oxybutynin, solifenacin, and tolterodine), urinary tract infection (fesoterodine, oxybutynin, and tolterodine), dry eyes (fesoterodine, imidafenacin, oxybutynin, propiverine, and tolterodine), and dry skin (imidafenacin, oxybutynin, and tolterodine). We also analyzed the discontinuation rate for each drug included. Our results demonstrated that darifenacin had the lowest discontinuation, followed by oxybutynin and propiverine, which showed comparable discontinuation rates, and the rest of the drugs (fesoterodine, imidafenacin, solifenacin, and tolterodine) with higher discontinuation rates. The details are shown in Table 2.
Discussion
This study compared all seven commonly used antimuscarinic drugs with an emphasis on their safety profiles, which may be a critical factor for personalized medicine in patients with OAB. 3 Based on the results, tolterodine may have better safety outcomes than the other drugs because it ranked first for 7 of 13 analyzed outcomes (dry mouth, constipation, urinary retention, dizziness, urinary tract infection, dry eyes, and dry skin). For blurred vision, a common side effect, fesoterodine may be the best treatment, followed by tolterodine. Moreover, darifenacin, fesoterodine, oxybutynin, propiverine, and solifenacin showed similar safety profiles.
This network meta-analysis allowed a broader assessment of the different antimuscarinic drugs through both direct and indirect comparisons. Based on a previous report, 10 we focused on the more prevalent side effects, including dry mouth, constipation, urinary retention, and blurred vision. Among them, dry mouth is the most frequently reported side effect related to the discontinuation of antimuscarinic drugs. 15 Our findings suggest that tolterodine is superior to other drugs assessed for dry mouth, constipation, and urinary retention. Furthermore, we analyzed the rate of mild and severe dry mouth, and the results were generally consistent and showed that patients treated with tolterodine were more likely to have a mild dry mouth than a severe one. Additionally, our results are consistent with a published review in which tolterodine was reported to be less associated with constipation than other similar drugs. 16 Cognitive impairment is also a noteworthy side effect of antimuscarinic drugs, as a large number of patients with OAB are elderly. In our analysis, we found that solifenacin was the least related to cognitive impairment.
In a previous systematic review, 10 Kessels et al. first summarized the adverse events for antimuscarinics via a network meta-analysis. They found that the overall adverse event profiles were similar for different antimuscarinics, except for orally administered oxybutynin, which may be associated with more unfavorable adverse events. They also reported the safety profiles and a rather detailed assessment for the entire class of antimuscarinics by categorizing the drugs via dosages. As a flexible dose of a drug is often adopted in clinical practice, their criteria on fixed dosage might have some limitations. In the current study, we aimed to provide updated and additional evidence on the safety profiles of different antimuscarinic drugs. During the past decade, AEs have been reported in more detail, as the safety profiles are the most important factor affecting patient adherence to OAB therapies. With a larger number of recent clinical trials included and more emphasis on relevant safety outcomes, we found that tolterodine might have a better overall safety profile than other antimuscarinic drugs.
In this study, our network meta-analysis enabled a broader comparison of different antimuscarinics with a limited number of studies. By focusing on several major AEs, we obtained a simplified summary of the safety profiles for different antimuscarinic drugs. Therefore, this analysis provides a valuable overview of this field and is useful for personalized medicine in patients with OAB. However, we must acknowledge that there are some limitations to our analysis. As mentioned above, we generally assessed the safety profiles for different antimuscarinics, and the differences in dosage, frequency, or formulations were not considered. Second, for some less frequent side effects, only a limited number of trials and small sample sizes were eligible. Therefore, bias might exist. In addition, potential bias may have been caused by missing information in some studies or varied definitions of AEs among trials. Third, because the influences of treatment duration, sex, and age were not considered, the results should be interpreted with caution. Additional studies are needed to provide a sufficient sample size to further explore the safety profiles of antimuscarinic drugs for detailed subgroup analyses. Furthermore, it is notable that the quality of life of a patient should be evaluated with overall consideration of efficacy and tolerability, and it might not be appropriate to select a certain therapy based on either efficacy or tolerability alone.
Conclusions
Our findings suggest that tolterodine may be preferable for over half of the safety outcomes evaluated. However, some of the probabilities for being the best were low, suggesting that the drug is clearly not superior. Fesoterodine and tolterodine may be superior to others for blurred vision. Darifenacin, fesoterodine, oxybutynin, propiverine, and solifenacin may have similar safety profiles in treating patients with OAB.
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
