Abstract
Objective
To evaluate the prognostic value of the uric acid-to-high-density lipoprotein cholesterol ratio in predicting postoperative survival in patients with hepatocellular carcinoma and to develop a nomogram to assist in clinical decision-making and patient management.
Methods
A total of 422 patients who underwent hepatectomy at Meizhou People’s Hospital between May 2011 and January 2023 were included. The optimal cutoff value for uric acid-to-high-density lipoprotein cholesterol ratio was determined using the R package ‘survminer,’ and patients were stratified into two groups accordingly. The prognostic significance of uric acid-to-high-density lipoprotein cholesterol ratio was evaluated using Kaplan–Meier survival analysis and Cox proportional hazards regression models. A prognostic nomogram was subsequently constructed. The predictive performance of the nomogram was assessed using the concordance index, and its clinical utility was evaluated via decision curve analysis.
Results
The optimal cutoff value for uric acid-to-high-density lipoprotein cholesterol ratio was 253.9. Survival analysis demonstrated significantly longer survival in the low-uric acid-to-high-density lipoprotein cholesterol ratio group. Cox regression analysis identified uric acid-to-high-density lipoprotein cholesterol ratio, advanced lung cancer inflammation index, and tumor–node–metastasis classification stage as independent prognostic factors for postoperative survival in patients with hepatocellular carcinoma. A predictive nomogram incorporating these variables was developed and validated using calibration curves, time-dependent receiver operating characteristic analysis, and decision curve analysis. The results confirmed that the nomogram demonstrated superior predictive accuracy and greater net benefit compared with models based on any of the individual variables alone.
Conclusions
Uric acid-to-high-density lipoprotein cholesterol ratio may serve as a potential prognostic biomarker for postoperative survival in patients with hepatocellular carcinoma. The established nomogram may provide a practical tool for prognostic assessment in clinical practice.
Keywords
Introduction
According to the latest Global Burden of Disease study, liver cancer remains the seventh leading causeof cancer-related mortality worldwide. 1 Among its diverse histological subtypes, hepatocellular carcinoma (HCC) represents the most prevalent form. Hepatic resection continues to be a primary therapeutic modality for HCC. Despite comprehensive preoperative evaluation, including extensive serological biochemical assays and advanced imaging examinations, a substantial proportion of patients experience tumor recurrence or distant metastasis within 5 years postoperatively. Consequently, identifying high-risk patients is critical for improving long-term outcomes following hepatectomy.
Established etiological factors for HCC include chronic hepatitis B virus (HBV) and hepatitis C virus infections, alcohol-related liver disease, and nonalcoholic fatty liver disease (NAFLD). The inflammatory burden associated with these conditions promotes compensatory hepatocyte proliferation and ultimately contributes to HCC development. 2 Notably, NAFLD represents the most rapidly increasing etiological factor for HCC, with an age-standardized increase in incidence of 33.2% between 2010 and 2019. 3 Furthermore, elevated serum uric acid (SUA) levels are significantly associated with tumor progression, recurrence, and poor prognosis in patients with HCC.4,5 Recent evidence from two independent cohort studies has demonstrated prevalent dysregulation of urate metabolism in patients with HCC, with peripheral blood uric acid (UA) levels showing significant prognostic associations. 5 Specifically, preoperative SUA levels serve as a reliable surrogate marker of oxidative stress in HBV-related HCC. 6
The uric acid-to-high-density lipoprotein cholesterol ratio (UHR), an emerging metabolic–inflammatory index, has been validated in multiple studies as a novel biomarker of metabolic dysregulation and systemic inflammation and shows strong correlations with various hepatic pathologies.7,8 Notably, accumulating evidence indicates that UHR is significantly correlated with NAFLD progression. As an indicator of systemic metabolic–inflammatory status, UHR has demonstrated significant prognostic value in diabetic cohorts.9,10 Further studies have shown that UHR is also associated with diabetic complications, including diabetic retinopathy and diabetic nephropathy.11,12 Additionally, UHR has been linked to inflammatory conditions such as thyroiditis and hypertension13,14 as well as metabolic syndrome and cardiovascular diseases.15,16 This body of evidence supports translational investigation into the potential of UHR as a posthepatectomy biomarker for HCC. Prospective validation of the utility of UHR in predicting long-term survival outcomes following HCC resection is therefore warranted.
The present study investigates the prognostic value of UHR and establishes validated risk-stratification criteria to facilitate early identification of high-risk patients, thereby guiding timely clinical intervention.
Materials and methods
Inclusion population
A retrospective cohort of 422 adult patients who underwent hepatic resection for histologically confirmed HCC was enrolled at Meizhou People’s Hospital between May 2011 and January 2023. This study was approved by the Institutional Ethics and Indications Committee and was conducted in accordance with the Declaration of Helsinki. All study procedures were performed exclusively at Meizhou People’s Hospital. The need for informed consent was formally waived due to the retrospective nature of the study. All patient information was deidentified. The reporting of this study conforms to the Strengthening the Reporting of Observational studies in Epidemiology (STROBE) guidelines. 17
Inclusion and exclusion criteria
The inclusion criteria were as follows: (a) consecutive patients primarily diagnosed with HCC who underwent hepatic resection at Meizhou People’s Hospital; (b) treatment-naïve status, defined as no prior antitumor therapies, including transcatheter arterial chemoembolization (TACE), radiotherapy, targeted molecular therapy, or other systemic anticancer treatments; and (c) histopathological confirmation of HCC after resection.
The exclusion criteria were as follows: (a) incomplete follow-up data or recurrence-free survival <90 days postoperatively; (b) histopathological diagnosis of combined hepatocellular–cholangiocarcinoma (cHCC-CCA) or the presence of synchronous extrahepatic malignancies; and (c) noncurative resection (Figure 1).

Flowchart of the study.
Data source
All cases were obtained from the medical records database of Meizhou People’s Hospital. The collected clinical characteristics included age, sex, body mass index (BMI), comorbidities (hypertension and diabetes), hepatitis B surface antigen (HBsAg) status, cirrhosis, portal hypertension, Child–Pugh score, and tumor-related variables (tumor number, size, capsule status, vascular invasion, microvascular invasion (MVI), histological grade, and tumor–node–metastasis classification (TNM) stage). Serological parameters included albumin (ALB), total bilirubin (TBil), gamma-glutamyl transpeptidase (GGT), and alanine aminotransferase (ALT). Surgical variables included anatomical resection, surgical approach, major resection, and intraoperative blood loss. Survival data included overall survival (OS) and the advanced lung cancer inflammation index (ALI). All enrolled cases were staged according to the ninth edition of the TNM staging system issued by the International Union Against Cancer. According to previous literature, UHR was calculated using the established formula: UHR = UA (mg/dL)/High-Density Lipoprotein Cholesterol (HDL-C) (mg/dL). The study endpoint was OS, defined as the interval from the date of curative resection to death from any cause or the last confirmed follow-up.
Statistical analysis
Univariate and multivariate Cox proportional hazards regression models with a forward stepwise procedure were used to evaluate the associations between clinical characteristics and UHR. The analysis was performed using the R package ‘survival’ (version 3.5-5). Survival curves were generated using the Kaplan–Meier method and compared using the log-rank test. A nomogram was constructed incorporating the independent prognostic factors identified in the multivariate analysis. Calibration curves were generated using bootstrap resampling with 1000 iterations to assess the performance of the nomogram. Time-dependent area under the curve (AUC), concordance index (C-index), and decision curve analysis (DCA) were calculated for TNM stage, UHR, and ALI, with comparative performance analyses of TNM alone, UHR, ALI, and the combined UHR–ALI model.
All statistical analyses were performed using R version 4.3.3 (http://www.r-project.org). The optimal cutoff value for UHR was determined using the ‘survminer’ package (version 0.4.9). Continuous variables were expressed as mean ± standard deviation (SD) or median (interquartile range), as appropriate, and categorical variables were presented as frequencies (percentages). Group comparisons were performed using the Mann–Whitney U test or independent-samples t-test for continuous variables and the Pearson χ2 or Fisher’s exact tests for categorical variables. A two-sided p value <0.05 was considered statistically significant.
Language
We used the Deepseek R1 large language model for language polishing and refinement of this article.
Results
Baseline characteristics
This retrospective analysis included 422 patients with HCC, comprising 373 males and 49 females. The mean age was 58 years, and the median age was 57.5 years. Using the ‘survminer’ R package, the optimal cutoff values for UHR and ALI in this cohort were determined to be 253.9 and 27.7, respectively. Based on the UHR threshold, patients were stratified into high-UHR (n = 208) and low-UHR (n = 214) groups. The correlations between clinical characteristics and UHR are summarized in Table 1. UHR levels were significantly associated with BMI and total cholesterol (TC) (p < 0.05). However, no significant associations were observed between UHR and other established adverse tumor prognostic factors, such as alpha-fetoprotein (AFP) level, tumor grade, or microvascular invasion (p > 0.05).
Baseline characteristics of included patients.

ALB: albumin; AFP: alpha-fetoprotein; ALT: alanine aminotransferase; ALI: advanced lung cancer inflammation index; BMI: body mass index; GGT: glutamyl transpeptidase; Glu: glucose; HDL-C: high-density lipoprotein cholesterol; LDL-C: low-density lipoprotein cholesterol; MVI: microvascular invasion; OS: overall survival; TBil: total bilirubin; TC: total cholesterol; TG: triglycerides; UHR: uric acid-to-high-density lipoprotein cholesterol ratio; SD: standard deviation.
p values considered statistically significant at <0.05.
Kaplan–Meier survival analysis and independent prognostic value of UHR
Patients in the low-UHR group demonstrated significantly better OS than those in the high-UHR group (p = 0.01). The hazard ratio (HR) for UHR was 0.618 (95% confidence interval (CI): 0.427–0.894; Figure 2(a)). Further stratification by TNM stage showed that low UHR was significantly associated with improved OS in patients with TNM stage III–IV disease (Figure 2(c)). In contrast, no significant difference in OS between UHR groups was observed among patients with TNM stages I–II disease (Figure S1J).

Kaplan–Meier survival curves for different factors. (a–h) Kaplan–Meier curves for OS stratified by different factors. ALI: advanced lung cancer inflammation index; OS: overall survival; TNM: tumor–node–metastasis classification; UHR: uric acid-to-high-density lipoprotein cholesterol ratio.
Univariate and multivariate Cox regression analyses were performed to assess the associations between clinical characteristics and OS (Table 2). The results identified UHR, TNM stage, and ALI as independent prognostic factors for OS. Among these variables, ALI and TNM stage were strongly associated with OS (p < 0.001). Subsequent subgroup analysis further demonstrated that low UHR was consistently associated with improved OS across all predefined subgroups (HR < 1) (Figure 3).
Univariate and multivariate analyses in patients.
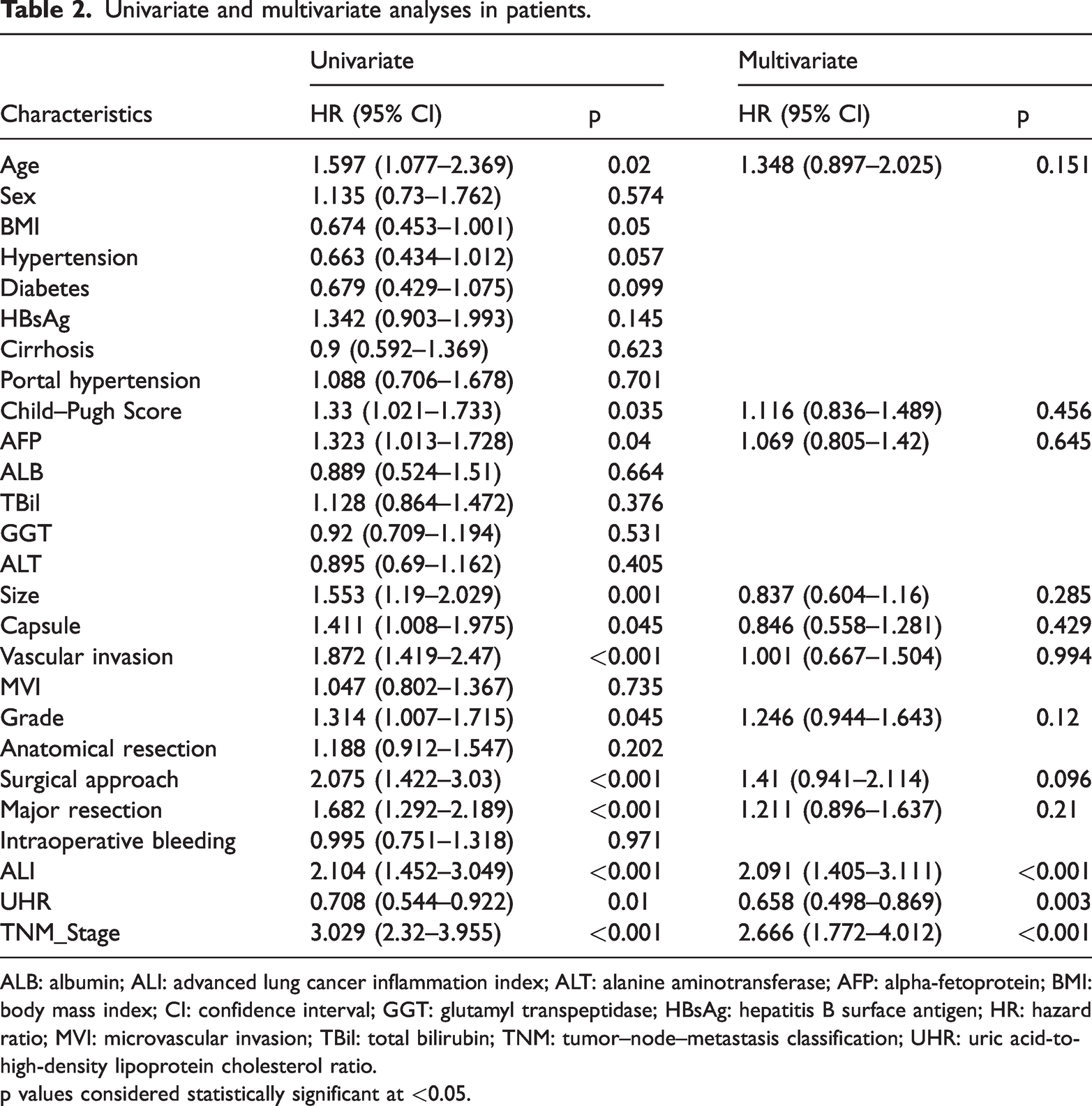
ALB: albumin; ALI: advanced lung cancer inflammation index; ALT: alanine aminotransferase; AFP: alpha-fetoprotein; BMI: body mass index; CI: confidence interval; GGT: glutamyl transpeptidase; HBsAg: hepatitis B surface antigen; HR: hazard ratio; MVI: microvascular invasion; TBil: total bilirubin; TNM: tumor–node–metastasis classification; UHR: uric acid-to-high-density lipoprotein cholesterol ratio.
p values considered statistically significant at <0.05.

Forest plot of subgroup survival analysis using univariate Cox regression in HCC. The horizontal axis represents the HR, with higher values indicating greater risk; the vertical axis shows the respective subgroups. The interaction p value was calculated using the likelihood ratio test to assess the consistency of the proportional hazards effect of UHR across subgroups. ALB: albumin; ALI: advanced lung cancer inflammation index; ALT: alanine aminotransferase; AFP: alpha-fetoprotein; BMI: body mass index; GGT: gamma-glutamyl transpeptidase; HBsAg: hepatitis B surface antigen; HCC: hepatocellular carcinoma; HR: hazard ratio; MVI: microvascular invasion; TBil: total bilirubin; TNM: tumor-node -metastasis classification; UHR: uric acid-to-high-density lipoprotein cholesterol ratio.
Survival analysis based on ALI
The prognostic value of ALI was evaluated in the present cohort. ALI demonstrated significant predictive value for OS. Patients with high ALI had significantly better OS than those with low ALI (HR: 2.096; 95% CI: 1.387–3.167; p < 0.001; Figure 2(b)). Furthermore, this association remained significant in both early-stage and advanced-stage tumor subgroups (Figure 2(e) and (f)).
The combined use of ALI and UHR was further assessed for predicting OS in patients with HCC. Regardless of disease stage (early stage, TNM I–II; or advanced stage, TNM III–IV), patients with high ALI and low UHR consistently exhibited superior OS compared with those with other biomarker combinations (Figure 2(g) and (h)).
Development and evaluation of the nomogram and DCA
To visually present the predictive outcomes, a nomogram incorporating the main variables—UHR, ALI, and TNM stage—was developed (Figure 4(a)). This prognostic model was subsequently applied to predict survival in patients with HCC undergoing surgical resection. An interactive version of the nomogram is publicly available at: https://liaojiapeng.shinyapps.io/dynnomapp/.

Nomogram and calibration curves for predicting OS in patients with HCC. (a) Nomogram constructed based on four independent prognostic factors in HCC. (b) Calibration curves for internal validation of the nomogram. (c) Time-dependent AUC curves of the nomogram. (d) Time-dependent AUC curves and concordance index comparisons of the nomogram and other model. (e–g) DCA curves of the nomogram. AUC: area under the curve; DCA: decision curve analysis; HCC: hepatocellular carcinoma; OS: overall survival.
The predictive performance of the nomogram was evaluated using the concordance index (C-index) and calibration curves. The calibration curves demonstrated good agreement for 1-year OS prediction, with relatively lower consistency for 3-, 5-, and 8-year OS predictions (Figure 4(b)). The nomogram achieved a C-index of 0.736 (95% CI: 0.708–0.805), which was higher than that of UHR alone (C-index = 0.57) and ALI alone (C-index = 0.56).
Time-dependent AUC analyses were further performed to assess predictive accuracy. The AUC values of the UHR model for predicting 1-, 3-, 5-, and 8-year OS were 0.49, 0.59, 0.62, and 0.58, respectively. For the ALI model, the corresponding AUC values were 0.61, 0.61, 0.60, and 0.59. In comparison, the nomogram demonstrated superior discriminative ability, with AUC values of 0.76, 0.78, 0.77, and 0.70 for 1-, 3-, 5-, and 8-year OS, respectively. The integrated time-dependent AUC analysis further confirmed that the nomogram consistently outperformed the single-factor models at all evaluated time points (Figure 4(d)).
Finally, DCA was conducted to assess the clinical utility of the model. The results indicated that the nomogram provided greater net benefit in predicting 5-year survival probability compared with alternative strategies (Figure 4(e–g)).
Discussion
The UHR has recently been proposed as a novel inflammatory and metabolic biomarker that integrates two pathological components: dysregulated UA metabolism and lipid abnormalities. 18 However, its prognostic value has not been fully elucidated in patients with HCC undergoing surgical resection. In this retrospective study of 422 patients with HCC who underwent hepatectomy, we evaluated the prognostic significance of UHR for OS. Our results showed that a low preoperative UHR was associated with lower bilirubin levels. Survival analysis demonstrated significantly prolonged OS in patients with low UHR, and multivariate analysis confirmed UHR as an independent prognostic factor for OS. We further developed a combined prognostic model incorporating UHR, ALI, and TNM stage, all of which were identified as independent risk factors. Time-dependent AUC analysis and DCA indicated that the integrated model provided improved predictive accuracy and greater clinical net benefit for OS prediction.
In patients with HCC, abnormal serum UA levels have been associated with tumor progression, recurrence, and prognosis. One study reported that elevated preoperative UA levels were positively correlated with postoperative recurrence in patients with HBV-related HCC. 6 Hyperuricemia also contributes to the development of NAFLD, a recognized precursor of HCC. Chronic NAFLD may progress to liver cirrhosis and ultimately to HCC.19,20 Within the HCC tumor microenvironment, enzymes involved in urate metabolism—such as xanthine oxidoreductase (XOR)—can promote inflammatory responses by modulating macrophage polarization. XOR catalyzes UA production during purine metabolism and facilitates the polarization of tumor-associated macrophages toward a pro-inflammatory phenotype. 21 This mechanism may contribute to HCC progression. Collectively, these findings suggest that alterations in UA levels may serve as biomarkers reflecting the extent of hepatic injury and inflammation.
Reduced HDL-C levels impair reverse cholesterol transport, resulting in cholesterol accumulation in hepatocytes and promoting tumor proliferation and metastasis.22,23 A large cohort study (n = 459,619) conducted by Su Youn Nam et al. 24 demonstrated that, compared with individuals with HDL-C levels of 50–59 mg/dL, those with HDL-C <50 mg/dL had an 83% higher risk of liver cancer (adjusted HR = 1.83; 95% CI: 1.65–2.04), indicating that low HDL-C is a significant risk factor for HCC. Patients with HCC frequently exhibit intrahepatic cholesterol accumulation accompanied by decreased circulating HDL-C. Estrogen has been shown to increase HDL-C levels by activating lecithin–cholesterol acyltransferase (LCAT), thereby inhibiting cholesterol synthesis and potentially reducing the risk of HCC. This pathway may represent a potential biomarker for HCC prognosis.
Compared with individual biomarkers such as UA or HDL-C, UHR integrates both parameters and is considered a novel indicator of inflammation and metabolic dysregulation, reflecting systemic metabolic–inflammatory status.9,10 Evidence suggests that in patients with insulin resistance, UHR—together with visceral obesity—may contribute to the progression of metabolic dysfunction–associated steatotic liver disease (MASLD) to HCC. 25 Although both serum UA and HDL-C levels have demonstrated prognostic value in patients with HCC, UHR, as a composite index incorporating both components, is also likely to have value in predicting postoperative survival. However, direct evidence supporting UHR as a predictive biomarker in patients with HCC undergoing hepatectomy remains limited. The present study provides clinical evidence supporting UHR as a novel prognostic marker in this population.
This retrospective study demonstrated that a low preoperative UHR level was independently associated with significantly prolonged OS after hepatectomy. Multivariate Cox regression analysis confirmed UHR as an independent prognostic predictor. Compared with conventional liver function assessment tools, such as the Child–Pugh grade, or tumor-specific markers such as AFP, UHR offers several advantages: it is readily available from routine preoperative blood tests, is cost-effective, and provides a continuous variable for risk stratification. Our analysis showed that UHR retained independent predictive value after adjustment for established prognostic factors, including Child–Pugh grade, AFP, and tumor stage, indicating that it provides additional prognostic information. For example, among patients with Child–Pugh grade A, UHR further identified a high-risk subgroup. This finding may have clinical implications for refining preoperative risk assessment and guiding postoperative surveillance.
The prognostic value of ALI as a systemic inflammatory marker in patients with HCC was first reported by Wen et al. 26 Subsequently, Qiu et al. 27 conducted a larger cohort study (n = 1282) using propensity score matching to reduce confounding and further confirmed the prognostic utility and robustness of ALI in HCC. In our study, ALI was included as a prognostic variable and consistently demonstrated significant predictive value (HR: 2.096; 95% CI: 1.387–3.167; p < 0.001), in agreement with previous findings. Moreover, the combined use of UHR and ALI showed prognostic value in both early- and advanced-stage patients, with particularly strong predictive performance observed in those with advanced-stage disease (TNM III–IV).
Numerous prognostic models have been developed to predict postoperative outcomes in patients with HCC. These include the widely used Barcelona Clinic Liver Cancer (BCLC) staging system as well as molecular markers such as circulating tumor cells (CTC), 28 and genetic or protein biomarkers (e.g. HOXB4 and CCDC110).29,30 However, some studies have suggested that the BCLC staging system may underestimate risk heterogeneity associated with continuous variables such as tumor diameter.31,32 In addition, CTC detection and genetic testing are costly, impose a substantial financial burden on patients, and are often unavailable in resource-limited settings due to limited technical infrastructure. In contrast, the nomogram developed in our study incorporates routinely available clinical variables—UHR, ALI, and TNM stage—all of which can be obtained through standard clinical testing. To enhance clinical applicability and facilitate risk stratification, we have made this model freely available as an online tool (https://liaojiapeng.shinyapps.io/dynnomapp/), enabling clinicians to generate individualized prognostic estimates by entering patient data. This approach may improve the efficiency of clinical decision-making.
It should be noted that, although our study is the first to propose the potential prognostic value of UHR in patients after HCC resection, several limitations should be acknowledged. First, this was a single-center retrospective study, which may limit the generalizability of our findings because of potential selection bias and geographic and ethnic homogeneity. The relatively strict exclusion criteria, particularly the requirement of a minimum 90-day OS and recurrence-free survival, may have introduced selection bias and limited the evaluation of ALI in short-term survival outcomes. Nevertheless, these criteria were retained to reduce potential confounding from perioperative complications, residual tumor after resection, and nonstandardized follow-up. In addition, the proposed UHR cutoff value was derived from a single-center cohort without external validation or bootstrapped internal validation to assess reproducibility. Therefore, large-scale, multicenter prospective studies are required to validate the robustness of this model. Second, inherent confounding bias associated with retrospective analyses may have affected the reliability of our findings. Variables such as age and sex may still have exerted residual effects. Although Cox regression and subgroup analyses were performed to adjust for these factors, we did not account for external variables such as socioeconomic status. Moreover, as a metabolic and inflammatory marker, UHR may be influenced by diet, renal dysfunction, and medications that affect UA or HDL-C levels. It should also be noted that multiple statistical comparisons were performed in the between-group analysis of baseline characteristics (Table 1) without adjustment for multiple testing. Therefore, p values—particularly those between 0.01 and 0.05—should be interpreted cautiously and regarded as exploratory. Further studies are needed to address these issues. Third, although our findings indicate a potential association between UHR and postoperative survival in HCC, we did not evaluate interventional strategies based on preoperative UHR levels to improve outcomes. In addition, as UHR is an inflammation-related index, our analysis was limited by the absence of other commonly used inflammatory markers, such as C-reactive protein and erythrocyte sedimentation rate. Future studies incorporating these parameters may further clarify the relationship between UHR and postoperative prognosis in HCC. Fourth, due to the retrospective design, serial longitudinal measurements were unavailable. Therefore, time-dependent receiver operating characteristic analyses based on dynamic changes or trajectory-based prediction models could not be performed. Although baseline UHR reflects the preoperative metabolic–inflammatory state and shows independent prognostic significance, a single measurement cannot capture biological changes during treatment and disease progression. Future prospective studies should incorporate scheduled postoperative blood sampling—such as at 1, 3, and 6 months and at recurrence—to evaluate dynamic UHR patterns and develop more accurate prediction models that integrate temporal changes. Finally, it should be noted that the data collection for this study spanned a considerable duration. This extended timeframe may have introduced unmeasured confounding related to changes in surgical techniques, perioperative care, and adjuvant treatment strategies. Therefore, these findings require further validation in future multicenter prospective studies.
Conclusions
This study is the first to demonstrate a potential association between preoperative UHR and postoperative survival in patients with HCC. The established nomogram may assist clinicians in estimating postoperative survival and provide a reference for formulating treatment strategies. UHR may serve as a potential prognostic biomarker for postoperative outcomes in patients with HCC.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605261429828 - Supplemental material for Prognostic utility of the serum uric acid-to-high-density lipoprotein cholesterol ratio following hepatectomy for hepatocellular carcinoma
Supplemental material, sj-pdf-1-imr-10.1177_03000605261429828 for Prognostic utility of the serum uric acid-to-high-density lipoprotein cholesterol ratio following hepatectomy for hepatocellular carcinoma by Jia-Peng Liao, Di-Kai Liang, Lu-Yun Zhang, Xin Jiang, Xiong Tang, Ji-Wei Xu and Gao-Min Liu in Journal of International Medical Research
Footnotes
Acknowledgments
We thank all participants and staff involved in this study. We also acknowledge the assistance of AI language tools for language editing purposes.
Author contributions
(I) Conception and design: Jia-Peng Liao, Gao-Min Liu, Di-Kai Liang, Lu-Yun Zhang, Xin Jiang, Xiong Tang, and Ji-Wei Xu; (II) Administrative support: Ji-Wei Xu; (III) Provision of study materials or patients: Gao-Min Liu; (IV) Collection and assembly of data: Jia-Peng Liao and Gao-Min Liu; (V) Data analysis and interpretation: Jia-Peng Liao, Gao-Min Liu, Di-Kai Liang, Lu-Yun Zhang, Xin Jiang, Xiong Tang, and Ji-Wei Xu; (VI) Manuscript writing: All authors; (VII) Final approval of manuscript: All authors.
Consent to participate
The ethics committee/institutional review board waived the requirement of written informed consent for participation from the participants or the participants’ legal guardians/next of kin because the need for informed consent was waived for the retrospective study.
Consent for publication
Not applicable.
Data availability statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Declaration of conflicting interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and publication of this article.
Ethical considerations
This study received ethical approval from the Meizhou People’s Hospital IRB (No. 2023-C-95). This is an IRB-approved retrospective study, all patient information was de-identified and patient consent was not required. Patient data will not be shared with third parties.
Ethics statement
The authors are accountable for all aspects of the work and ensure that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The study was conducted by the Declaration of Helsinki (as revised in 2013). All protocols were approved by the Ethics and Indications Committee of the Meizhou People’s Hospital (2023-C-95). The requirement for informed consent was waived for the retrospective study.
Funding
The authors have not declared a specific grant for this research from any funding agency in the public, commercial, or not-for-profit sectors.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
