Abstract
This case report describes an exceptionally rare occurrence of severe acute respiratory distress syndrome following laparoscopic appendectomy in a previously healthy 57-year-old male. The patient presented with acute appendicitis and underwent an uncomplicated surgical procedure. Approximately 4 h postoperatively, he developed rapid respiratory deterioration with severe hypoxemia, fulfilling the Berlin criteria for severe acute respiratory distress syndrome (partial pressure of arterial oxygen/fraction of inspired oxygen ratio, 99.2). Other causes, including aspiration, pulmonary embolism, and cardiac failure, were excluded. The patient was managed in the intensive care unit with lung-protective ventilation in accordance with the Acute Respiratory Distress Syndrome Network (ARDSNet) protocol along with antibiotics and corticosteroids. He demonstrated rapid clinical improvement, was extubated on postoperative day 7, and discharged on postoperative day 8. This case highlights that severe acute respiratory distress syndrome, although rare, can complicate routine surgical procedures and underscores the importance of early recognition and multidisciplinary management in achieving favorable outcomes.
Keywords
Introduction
Acute appendicitis is one of the most common causes of emergency abdominal surgery. 1 The first appendectomy was performed in New York in 1886, and since then, this procedure has become one of the most frequently performed emergency surgeries. Due to the risk of progression to advanced pathophysiological states, timely appendectomy has long been considered the standard treatment for acute appendicitis. 2
Laparoscopic appendectomy is currently the preferred surgical technique for acute appendicitis because it is associated with lower rate of surgical site infection (SSI), shorter hospital stays, and superior cosmetic outcomes.2,3 Although appendectomy is generally regarded as a minimally invasive procedure, potential postoperative complications may occur, including hemorrhage, intra-abdominal abscess (IAA), fecal fistula, and bowel obstruction. 4
Acute respiratory distress syndrome (ARDS) is an acute form of respiratory failure characterized by pulmonary edema resulting from increased alveolar–capillary permeability secondary to severe inflammation. It can develop following various underlying conditions and traumatic events, including sepsis and severe pneumonia. 5 Symptoms typically develop within 7 days after the inciting event and often progress rapidly. 6 Notably, ARDS is an exceedingly rare postoperative complication of appendectomy, with only a few similar cases reported in the literature.
Herein, we report a rare case of a previously healthy, middle-aged adult with acute appendicitis who developed severe ARDS following laparoscopic appendectomy. Written informed consent for this case report, including the use of images and publication, was obtained.
Case presentation
Patient history and initial presentation
A 57-year-old male presented to the emergency department with a 2-day history of right lower abdominal distension and pain that progressively worsened, accompanied with fever that began 1 day prior to admission. The patient denied experiencing nausea or vomiting. He was 178 cm tall, weighed 87 kg (body mass index (BMI), 27.5 kg/m2), and had a 30-pack-year smoking history (20 cigarettes/day, ongoing). On initial physical examination, his temperature was 39.7°C, heart rate was 118 bpm with sinus tachycardia, and systolic blood pressure ranged from 140 to 150 mmHg. Notably, he exhibited marked tenderness, rebound tenderness, and mild guarding at McBurney’s point in the right lower quadrant. The colonic gas test and psoas sign were positive. Laboratory studies revealed an elevated white blood cell count of 15.98 × 10³/µL, neutrophil count of 13.01 × 10³/µL (with an 81.4% ratio), and a high-sensitivity C-reactive protein (CRP) level of 117.45 mg/L. Preoperative abdominal computed tomography (CT) revealed an enlarged appendix with surrounding exudation (Figure 1). At the patient’s strong request, a chest CT was performed because of his significant smoking history to comprehensively screen for potential cardiopulmonary comorbidities; the findings were unremarkable (Figure 2). Preoperative assessment confirmed no specific contraindications to laparoscopic appendectomy (e.g. inability to tolerate pneumoperitoneum or severe coagulopathy), and the patient was classified as having American Society of Anesthesiologists (ASA) physical status IIE. Given that the patient was a middle-aged male with previously good health status, no history of urinary retention, and an anticipated short operative time (actual duration, 1.5 h), a urinary catheter was not routinely inserted in accordance with evidence-based guidelines to reduce associated infection risks. 3 After obtaining written informed consent from the patient and his family, preoperative preparations were completed, and the patient was transferred to the operating room. Cefuroxime (1.5 g) was administered intravenously 30 min preoperatively as prophylactic antibiotic coverage.

Chest X-ray on admission.
Abdominal CT scan on admission. CT: computed tomography.
Surgical and anesthesia procedures
The surgery lasted approximately 1 h and 30 min, with an estimated blood loss of 10 mL and an intraoperative infusion of 800 mL of crystalloid solution. The removed appendix was submitted for pathological examination, which confirmed acute suppurative appendicitis with periappendicitis (Figure 3). Following surgery, the patient underwent tracheal extubation after the resumption of adequate spontaneous respiration and the patient’s ability to follow commands. Subsequently the patient was transferred to the postanesthesia care unit (PACU). The patient was discharged from the PACU to the general ward only after meeting standardized discharge criteria, including full consciousness, stable vital signs, and an Aldrete score of ≥9.

Histopathological examination of the resected appendix (H&E staining, 100×).
At 13:20, upon arrival at the ward, the patient was conscious and responsive. Vital signs were stable, with a respiratory rate of 19 breaths/min, pulse rate of 108 bpm, blood pressure of 127/71 mmHg, and oxygen saturation of 100%. A real-time physiological monitoring system based on the Early warning score (EWS) was established. Continuous electrocardiogram (ECG) monitoring, low-flow nasal cannula oxygen supplementation, and hourly assessments (including blood pressure, pulse, respiratory rate, and oxygen saturation) were instituted. Strict 24-h fluid intake and output records were maintained to guide fluid management. The patient was kept nil per os and treated with 4.5 g intravenous piperacillin/tazobactam every 8 h, omeprazole for gastric protection, appropriate analgesia, and nutritional supplementation. In addition, close attention was paid to abdominal signs and the characteristics of the drain output.
Postoperative course and clinical deterioration
At 17:00, the patient’s oxygen saturation gradually decreased to 92%. Despite increasing the oxygen flow via nasal cannula, minimal improvement was noted. By 17:05, he developed marked tachypnea, muscle twitching, and began coughing up copious yellow sputum. Concurrently, his blood pressure rose to 167/103 mmHg, heart rate increased to 150 bpm, and oxygen saturation dropped further to 80%. The nurse immediately repositioned the patient’s head, performed suctioning, transitioned him to face mask oxygen, and notified the on-call physician. Urgent blood tests were ordered, including coagulation studies, complete blood count, renal function tests, electrolytes, cardiac troponin, B-type natriuretic peptide (BNP), and arterial blood gas analysis. Intravenous dexamethasone phosphate (10 mg) was administered. Arterial blood gas analysis revealed a partial pressure of arterial oxygen (PaO2) of 45.2 mmHg, pH 7.08, partial pressure of carbon dioxide (PaCO2) of 42.7 mmHg, actual bicarbonate of 11.9 mmol/L (standard bicarbonate, 11.5 mmol/L), actual base excess of −7.7 mmol/L (standard base excess, −18.1 mmol/L), glucose concentration of 15.8 mmol/L, lactate concentration of 13.27 mmol/L, and an elevated D-dimer level of 10.56 mg/L, indicating a hypercoagulable state.
According to the Berlin definition, the patient was diagnosed with severe ARDS based on the following criteria: (a) acute onset within 3.5 h of a known clinical insult (intra-abdominal infection/surgery); (b) chest CT and bedside X-ray showing bilateral pulmonary infiltrates not fully explained by cardiac failure or fluid overload; (c) impaired oxygenation with a PaO2/fraction of inspired oxygen (FiO2) ratio of 99.2 (measured under face mask oxygen at 7 L/min, FiO2 ≈ 0.5, PaO2 = 49.6 mmHg), meeting the criteria for severe ARDS (PaO2/FiO2 ≤ 100); and (d) absence of evidence for cardiac etiology, supported by no cardiac history, normal BNP levels, and absence of cardiomegaly or signs of vascular congestion on imaging. The initial diagnosis was acute hypoxemic respiratory failure complicated by ARDS. A rescue protocol was activated, and an anesthesiologist was called for bedside tracheal intubation, with respiratory and intensive care unit (ICU) teams also alerted.
At 17:20, the patient received 80 mg intravenous methylprednisolone succinate. Sedation was maintained with propofol, and assisted ventilation was provided using a manual resuscitation bag. A urinary catheter was inserted, yielding 50 mL of dark yellow urine. At that time, the patient’s temperature was 40.5°C, blood pressure 124/61 mmHg, heart rate 120 bpm, and oxygen saturation was stabilized at 90%–92% with assisted ventilation. Following stabilization, he was transferred to the ICU for further management.
ICU management
Upon ICU admission, the patient’s vital signs were as follows: temperature, 37.8°C; heart rate, 101 bpm; respiratory rate, 18 breaths/min; and blood pressure, 90/31 mmHg. He remained sedated and under analgesia. Lung auscultation revealed coarse breath sounds with bilateral wet rales, whereas cardiac examination showed a regular rhythm with no pathological murmurs. The abdominal incision dressing was dry, with minimal serosanguinous drainage from the intra-abdominal drain, and the abdomen was soft. No significant edema was noted in the lower extremities. Emergent chest CT demonstrated bilateral pleural effusions and lower lobe atelectasis (Figures 4 and 5).

Chest CT scan on admission showing bilateral pleural effusions and atelectasis. CT: computed tomography.
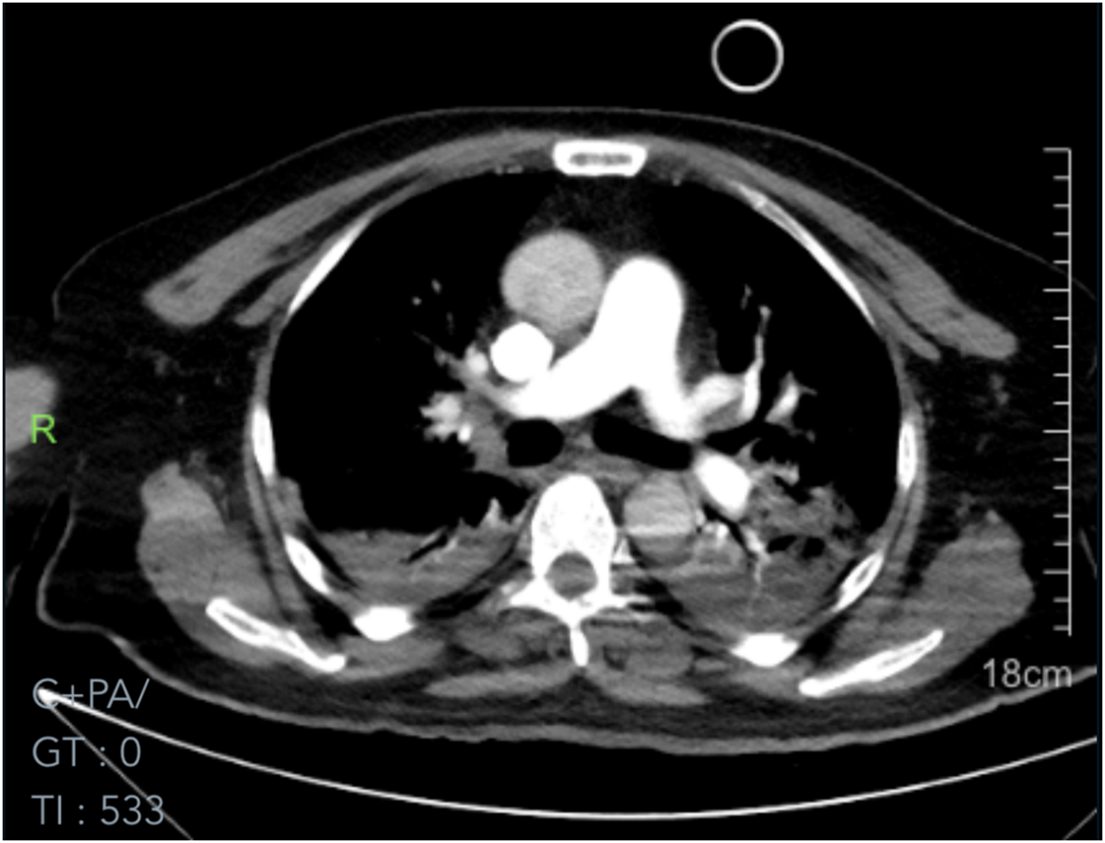
Additional view of the admission chest CT scan (further demonstrating bilateral pleural effusions and atelectasis). CT: computed tomography.
Following the Acute Respiratory Distress Syndrome Network (ARDSNet) protocol, lung-protective ventilation was initiated using low tidal volumes and high positive end-expiratory pressure (PEEP). The patient was managed on mechanical ventilation in pressure control mode (pressure, 12 cmH2O; PEEP, 8 cmH2O; FiO2, 70%), which resulted in an oxygen saturation of 97%. Additionally, he received 0.5 g imipenem/cilastatin every 8 h, and vasopressor support was provided with metaraminol as needed. Other supportive measures included omeprazole for gastric protection, ambroxol for secretion clearance, adenosylmethionine for liver protection, potassium supplementation, and nutritional support. Sedation and analgesia were titrated to maintain a Richmond Agitation-Sedation Scale (RASS) score between −2 and 0 and a Critical-Care Pain Observation Tool (CPOT) score between 0 and 1. Blood glucose was maintained between 8 and 12 mmol/L.
A repeat arterial blood gas analysis on 100% FiO2 showed a PaO2/FiO2 ratio of 210.5 mmHg, pH 7.235, PaCO2 of 48.4 mmHg, PaO2 of 227.7 mmHg, bicarbonate of 18.6–19.3 mmol/L, base excess of −7.1 mmol/L, and lactate of 2.9 mmol/L. Laboratory analyses revealed a white blood cell count of 14.0 × 10³/µL with 93.9% neutrophils and 2.5% lymphocytes, a red blood cell count of 5.18 × 1012/L, hemoglobin of 153 g/L, and a CRP level exceeding 200 mg/L; other biochemical parameters were unremarkable.
On postoperative day 2, while still sedated and mechanically ventilated (with FiO2 reduced to 40%), the patient’s oxygen saturation was maintained at 97%, temperature was 38.5°C, heart rate 99 bpm, and blood pressure was sustained at 146/76 mmHg on low-dose metaraminol infusion. Lung auscultation continued to reveal coarse breath sounds with bilateral wet rales. The abdominal incision and drain output remained stable. Gradually, the patient’s clinical status improved. Dynamic changes in postoperative inflammatory markers are shown in Figure 6. CRP levels peaked early after surgery and then decreased rapidly, whereas the white blood cell count returned to the normal range by postoperative day 3 (Figure 6).
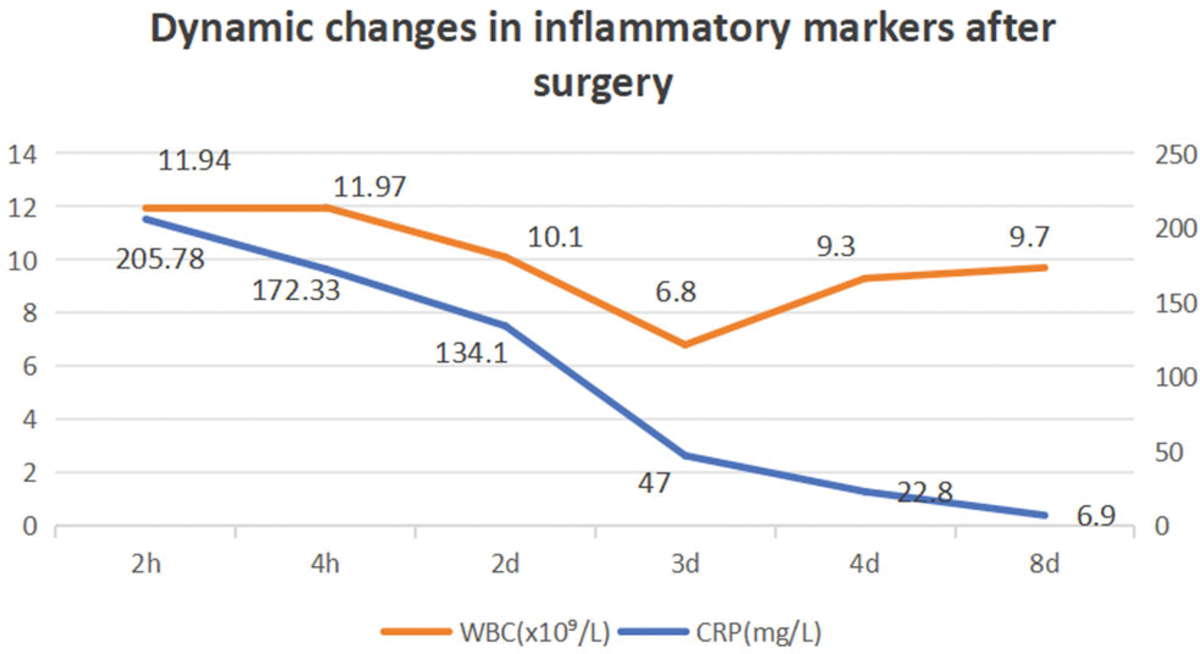
Dynamic changes in postoperative inflammatory markers (C-reactive protein and white blood cell count).
Outcome
On postoperative day 7, the patient was successfully extubated. Following adherence to the ARDSNet protocol, he recovered adequately and was discharged on postoperative day 8. At a 1-month follow-up, he had resumed normal daily activities, achieved a 6-min walk test distance of 500 m, and reported no cough, sputum production, or reduced exercise tolerance.
Discussion
Acute appendicitis is a common emergency department presentation, typically curable through surgery or appropriate antimicrobial therapy. 4 Globally, the overall incidence of appendicitis or complications arising from appendectomy is estimated at approximately 100–150 cases per 100,000 person-years. 7 Laparoscopic appendectomy is typically considered a minimally invasive procedure, with postoperative complication rates ranging from 3.0% to 28.7%. These complications can include small bowel obstruction (0%–1.9%), SSI (1.2%–12.0%), IAA (1.6%–8%), stump leakage, or retained appendicitis. Literature suggests that patients with complicated appendicitis experience higher complication rates. 8
The precise incidence and risk factors for postoperative pulmonary complications in patients undergoing appendectomy have not been extensively studied. Factors such as the laparoscopic approach and prolonged operative time may increase the risk of pulmonary complications. Preexisting pulmonary conditions, including smoking, chronic obstructive pulmonary disease (COPD), acute upper respiratory tract infections (URTI), and bronchial asthma (BA), are associated with a higher risk of postoperative pulmonary complications. Patients with baseline pulmonary problems are more vulnerable to bronchospasm, atelectasis, and lung infections following airway instrumentation. 9
Atelectasis during and after surgery can impair gas exchange and respiratory mechanics to varying degrees. In severe cases, lung collapse may result in postoperative respiratory failure, pneumonia, and overall poorer clinical outcomes. The clinical risk factors for atelectasis are multifactorial, including patient-related, anesthetic-related, and surgery-related factors. Patient-related factors include BMI >25 kg/m2, age-related small airway closure, acute pneumonia, noninflammatory pulmonary edema, and diaphragmatic or respiratory muscle dysfunction, such as muscle atrophy and neuromuscular diseases. Anesthetic-related factors include deep anesthesia, use of muscle relaxants, diaphragmatic nerve blockade, high inspired oxygen concentration, use of nitrous oxide, small tidal volume ventilation without adequate PEEP, residual neuromuscular blockade, opioid- or sedative-induced respiratory depression, fluid overload, and transfusion-related acute lung injury. Surgery-related factors include patient positioning (Trendelenburg, supine, or lateral decubitus); pneumoperitoneum; single-lung ventilation; extracorporeal circulation; prolonged operative time; and type of surgery, with prolonged cardiothoracic, upper abdominal, or laparoscopic procedures as well as open thoracic or abdominal surgeries with severe postoperative pain, being particularly implicated. Notably, the prone position during surgery may reduce the risk of atelectasis. 10
ARDS represents a diffuse, rapidly evolving inflammatory process affecting both lungs. It results in increased pulmonary vascular permeability, reduced lung compliance, alveolar edema, and tissue injury. 6 ARDS is typically triggered by conditions that induce a widespread inflammatory response, such as sepsis, aspiration, pneumonia, severe trauma, or massive transfusion. Patients with ARDS exhibit numerous signs and symptoms, primarily because of a marked and sudden decline in lung compliance and effective gas exchange. These include severe respiratory distress, tachypnea, tachycardia, and progressive hypoxemia. Typically, these manifestations, which may also include cough, chest pain, and diffuse moist rales, become evident within 7 days following the inciting event and often deteriorate rapidly. 4
Following exposure to ARDS risk factors, pulmonary endothelial injury develops rapidly, resulting in increased vascular permeability. Subsequently, protein-rich fluid, cells, and macromolecules leak into the alveolar space, inactivating surfactant and ultimately forming the characteristic hyaline membranes. 11 According to the Berlin definition, ARDS is classified into three categories based on the degree of hypoxemia: mild (200 mmHg < PaO2/FiO2≤ 300 mmHg), moderate (100 mmHg <PaO2/FiO2 ≤ 200 mmHg), and severe (PaO2/FiO2 ≤ 100 mmHg). 6
During the establishment of the diagnosis of ARDS, a careful differential diagnosis was undertaken to rule out other potential causes of acute respiratory failure and pulmonary infiltrates.
First, the diagnosis of aspiration pneumonia was considered unlikely, as the patient lacked clear risk factors, including smooth induction of anesthesia, absence of intraoperative cough or regurgitation, and rapid postoperative awakening. Furthermore, chest imaging revealed diffuse bilateral pulmonary involvement rather than the segmental or lobar distribution typically seen in gravity-dependent regions affected by aspiration. 12
Second, screening for acute pulmonary embolism revealed no clinical symptoms or high-risk factors for deep vein thrombosis. Importantly, a computed tomography pulmonary angiography (CTPA) performed postoperatively during the episode of acute hypoxemia revealed no filling defects in the pulmonary arteries. According to current authoritative guidelines, this finding is sufficient to essentially exclude the diagnosis of pulmonary embolism. 13
Finally, distinguishing ARDS from cardiogenic pulmonary edema was crucial. The patient was a middle-aged male without underlying cardiovascular disease. His BNP level was within the normal range upon admission, and imaging studies showed no typical signs of cardiogenic edema, such as cardiomegaly, pulmonary vascular congestion, or interstitial edema (e.g. Kerley B lines). These findings were inconsistent with cardiogenic pulmonary edema and instead aligned more closely with the diagnosis of noncardiogenic ARDS.
In addition to the aforementioned differential diagnoses, the potential contribution of upper airway obstruction leading to hydrostatic pulmonary edema was carefully evaluated. This condition can occur from postoperative residual neuromuscular blockade or sedation, resulting in the generation of significant negative intrathoracic pressure that promotes fluid transudation. However, in the present case, the patient was documented to be fully conscious and responsive upon arrival at the ward. The absence of clinical signs of upper airway compromise, together with this intact mental status, makes a primary hydrostatic etiology unlikely. Consequently, the clinical and ancillary findings are more consistent with inflammation-mediated ARDS secondary to intra-abdominal sepsis rather than negative pressure pulmonary edema.
Although the incidence of trauma-related ARDS appears to have slightly improved, mortality remains high, with adult ARDS mortality rates exceeding 40%.14,15 In the present case, prompt recognition and intervention within 4 h were crucial.
In the postoperative care of patients undergoing laparoscopic surgery under general anesthesia, systematic nursing interventions are crucial in the early identification of ARDS. Standardized protocols, including continuous electrocardiographic monitoring with particular attention to oxygen saturation trends, comprehensive patient and family education regarding respiratory symptoms, and graded nursing rounds conducted every 60 min, are essential. Notably, dynamic analysis of oxygen saturation using advanced monitoring systems facilitates the early detection of progressive hypoxemia (e.g. a drop in peripheral oxygen saturation (SpO2) from 100% to 80% within the first 4 h postoperatively), serving as a key premonitory sign preceding the clinical onset of ARDS.
Treatment options for ARDS include lung-protective ventilation strategies, neuromuscular blockade, prone positioning, inhaled vasodilators, corticosteroids, recruitment maneuvers, and extracorporeal life support. 16 The cornerstone of ARDS management is treating the underlying etiology and optimizing mechanical ventilation. Inadequate management may predispose patients to ventilator-induced lung injury. Although the treatment of hypoxemia typically requires moderate-to-high levels of PEEP, no single PEEP titration strategy has been definitively shown to improve outcomes. 17 In the present case, the patient developed ARDS secondary to acute appendicitis and was managed using the ARDSNet protocol, employing low tidal volumes (6–8 mL/kg) and a lung-protective PEEP regimen. The observed rapid decline in CRP levels (Figure 6) provides strong evidence that the systemic inflammatory response was effectively controlled after source control surgery, consistent with the pathophysiology of sepsis-induced ARDS.
This case can be compared with two previously reported instances in which patients developed septic shock during antibiotic therapy and subsequently required emergency surgery.18,19 These reports suggest that the pathogens associated with appendicitis may exhibit antibiotic resistance, potentially contributing to the development of postoperative ARDS. In one prior report by Ologun et al., 18 the patient received a change in antibiotic regimen along with corticosteroid therapy for ARDS management, a therapeutic approach similar to that employed in our case.
In summary, the successful outcome in the present case depended on several critical factors. First, the surgical team must maintain vigilance for atypical signs of respiratory deterioration during the perioperative period in patients with acute abdominal emergencies. Second, establishing a real-time physiological monitoring system based on EWS in the general surgery ward is recommended. Finally, a standardized postoperative ARDS screening protocol, including dynamic arterial blood gas analysis and bedside lung ultrasound evaluation, must be implemented. This case provides empirical evidence for optimizing perioperative management and underscores the essential role of multidisciplinary collaboration in the early recognition and treatment of severe postoperative complications.
The novelty and significance of this case are evident in several aspects. First, it provides a classic teaching example of ARDS characterized by a fulminant onset but rapid improvement following aggressive treatment. Second, it highlights to clinicians the potential for this life-threatening complication even after routine, low-risk surgery, emphasizing the importance of vigilance and early recognition. Third, our detailed documentation of perioperative management offers a valuable reference for managing similar cases in the future (Table 1).
Reported cases of ARDS after surgery for acute appendicitis.

“−” is defined as “not present or absent” and “+” as “present.”
PMH: past medical history; ARDS: acute respiratory distress syndrome; M: male.
Despite these insights, certain limitations remain. As an isolated case report, a causal relationship cannot be definitively established, for example, whether ARDS was directly induced by the surgical procedure or whether undetected confounding factors, such as occult infection or genetic predisposition, played a contributory role. We therefore advocate for further case series and analyses to better elucidate the risk factors involved.
Conclusions
ARDS is an exceedingly rare and severe complication following laparoscopic appendectomy in patients without underlying comorbidities. In this case, although the patient initially presented with an acute abdomen without shock, he subsequently developed ARDS. Management of ARDS focuses on effective control of the infection source combined with supportive care measures. With timely and appropriate intervention, the patient achieved complete recovery.
Footnotes
Acknowledgments
The authors thank the patient for consenting to the publication of this case. We also acknowledge the clinical staff involved in the patient’s care.
Author contributions
HZ: Conceptualization, writing–original draft, and data curation.
YJ: Supervision and writing–review & editing.
ZW: Resources and investigation.
Data availability statement
The data supporting this case report are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
The authors declare no potential conflicts of interest with respect to the research, authorship, and publication of this article.
Ethical statement
This case report was prepared in accordance with the CARE guidelines. 20 Ethical approval for publication was obtained from the Ningbo Beilun Third People’s Hospital Ethics Committee. Written informed consent for both treatment and publication of this case report was obtained from the patient. All patient details have been deidentified to protect privacy.
Funding
The authors received no financial support for the research, authorship, and publication of this article.
