Abstract
Ectopic splenosis is a disorder caused by the overflow of splenic myeloid cells or intravascular migration after splenic trauma or splenectomy. It is often asymptomatic and detected incidentally during imaging evaluation. Splenosis lacks specificity on clinical imaging examinations such as ultrasound, computed tomography, and magnetic resonance imaging and is often confused with tumors. We report the case of a primigravida (G1P0) in her early 30s with multiple hypoechoic masses in her upper abdomen that were detected during routine prenatal ultrasound at 23 + 1 weeks of gestation, with a recommendation for follow-up monitoring. Tumor marker analysis showed normal results. The patient remained asymptomatic without abdominal pain or fever, and monitoring showed normal fetal heart rate. The patient was admitted to our hospital, and we conducted a multidisciplinary consultation. We used misoprostol and a Cook balloon to induce labor; the patient delivered vaginally successfully. In this case report, we aimed to highlight the significance of taking detailed medical history and using the “minimal intervention” approach for managing pregnancy-complicated splenosis, highlighting the pivotal role of multimodal imaging evaluation.
Introduction
Ectopic splenosis (ES) refers to the heterotopic transplantation of splenic tissue fragments caused by splenic trauma, abdominal trauma, or splenectomy. 1 It was first described by Buchbinder and Lipkoff in 1939. 2 ES can occur at any location in the body. The absence of typical radiological features makes it difficult to establish an accurate diagnosis. 3 In general, ES is usually asymptomatic but can cause site-specific discomfort mimicking endometriosis or peritoneal metastases. Furthermore, several studies have reported other complications, including gastrointestinal bleeding, bowel obstruction, and hydronephrosis. 4
Case presentation
A primigravida (G1P0) in her early 30s, with a medical history of splenectomy that was performed 21 years ago following a traumatic fall, was found to have multiple hypoechoic masses in the upper abdomen (the largest measuring 5.4 × 2.6 cm) during a routine prenatal ultrasound at 23 + 1 weeks of gestation, and follow-up monitoring was recommended. At 34 + 3 weeks of gestation, a repeat ultrasound revealed persistent hypoechoic lesions in the abdominal cavity (the largest measuring 5.8 × 2.4 ×4.5 cm, Figure 1(a)), demonstrating ill-defined borders and irregular morphology. Subsequent magnetic resonance imaging (MRI) demonstrated multiple nodular lesions and masses in the anterior renal fascia of the left kidney, anterior to the uterus in the greater omentum region, and along the left colon, with unclear demarcation from adjacent abdominal wall structures (Figure 1(b)). Tumor marker analysis results were as follows: cancer antigen (CA)125, 12.9 U/mL; CA199, 14.2 U/mL; CA153, 21.2 U/mL; and alpha-fetoprotein (AFP), 119.7 ng/mL. The patient remained asymptomatic without abdominal pain or fever, and monitoring showed normal fetal heart rate. The patient was admitted to the West China Second University Hospital of Sichuan University, Chengdu, in mid-2024. A multidisciplinary consultation suggested a gastrointestinal stromal tumor (GIST). However, the patient had a clear history of splenic trauma and splenectomy, which is a typical cause of splenic ectopic implantation, while GIST is not associated with such a specific medical history. Furthermore, imaging examinations indicated that the lesion was located on the greater omentum without any connection to the gastrointestinal wall, whereas GIST originates from the gastrointestinal wall. Postoperative pathology confirmed splenic tissue. After obtaining signed consent for treatment from the patient, we administered misoprostol (25 µg) and used a Cook balloon to induce labor; the patient delivered vaginally successfully at 38 + 5 weeks of gestation; she gave birth to a neonate with a birth weight of 3120 g and Apgar scores of 10-10-10. The 6-month postnatal follow-up showed normal development of the infant. The patient underwent computed tomography (CT)–guided fine needle aspiration biopsy of a mesenteric mass in the left lower abdomen after delivery. The biopsy confirmed splenic ectopic implantation, which was managed with regular monitoring (Figure 1(c)).
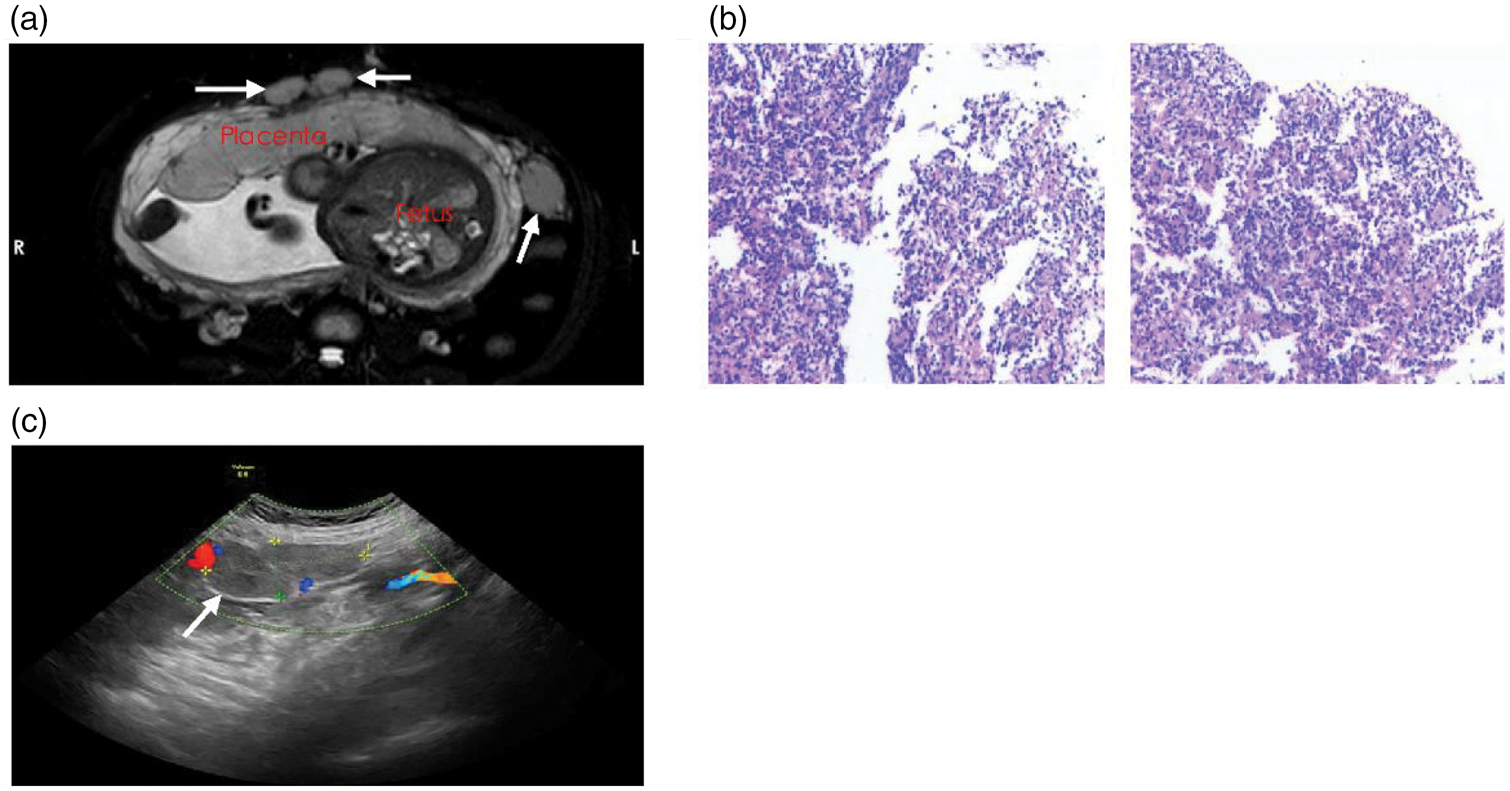
MRI, ultrasound manifestations, and histopathological characteristics of abdominal splenosis. (a) A hypoechoic mass located in the lower left abdomen of the pregnant woman, with blood flow signals detected in the periphery and within the mass. The arrow indicates the lesion location. (b) MRI demonstrated multiple nodular masses in the anterior greater omentum area and left paracolic region of the uterus and (c) histopathological examination indicates a lymphoproliferative disorder identified within a background of adipose tissue and proliferative blood vessels. MRI: magnetic resonance imaging.
Discussion
Pregnancy complicated with ES represents a rare clinical phenomenon. The incidence of splenosis in the general population remains unknown; however, given that its development is preceded by splenic injury, its prevalence is expected to be much lower. 5 The diagnostic and therapeutic processes in this case offer valuable clinical insights. Given the anatomical alterations induced by gravid uterine enlargement, particular attention should be paid to differential diagnosis from ovarian neoplasms and gastrointestinal malignancies to prevent misdiagnosis or unnecessary surgical interventions.
Studies have reported the mechanism of splenosis. Ruptured splenic tissue, particularly the red pulp rich in highly proliferative and multipotent reticuloendothelial cells, detaches from the rupture site as “seeds” and disseminates within the peritoneal cavity via the blood, lymph, or direct spreading. The scattered splenic tissue fragments are very small (typically only a few millimeters in size); this facilitates their short-term survival without reliance on the original arterial supply, sustained only by diffusion of tissue fluid. The splenic tissue fragments attached to the host tissue release a series of angiogenic factors, stimulating the proliferation and migration of capillary endothelial cells in the surrounding host tissue, which gradually grow into the splenic tissue. The newly formed capillary network progressively extends into the internal splenic tissue, connecting with its original sinusoid structures, ultimately forming a functional microcirculation system.
During pregnancy, the maternal blood volume increases by approximately 40%–50%. This may boost the blood flow to ectopic splenic nodules, making them more visible during imaging.5–7 In addition, the uterus’s expansion can push and compress the abdominal organs, changing the position of ectopic splenic nodules, making them more difficult to detect and track on ultrasound.
Although biopsy remains the gold standard for diagnosis, the use of imaging in combination with a history of splenic trauma or splenectomy can prevent unnecessary intervention. Currently, the preferred imaging method to identify splenosis is the use of Technetium Tc 99m heat-damaged red blood cells (RBCs). 8 MRI may also identify splenic masses; in particular, superparamagnetic iron oxide particles, which localize to the sites of phagocytic reticuloendothelial cells, have been used to identify splenosis on MRI. However, these imaging techniques may harm the fetus.9,10 Therefore, there is an urgent need to develop specific imaging methods for diagnosing splenosis in pregnant women.
For asymptomatic splenosis during gestation, expectant management remains the preferred approach, with surgical intervention reserved for high-risk scenarios such as implants adjacent to the uterine vasculature. Notably, splenosis may retain immunomodulatory functions, necessitating long-term follow-up to monitor immune development in the offspring. Complications of splenosis, such as acute abdomen, are the main indications for intervention. Management strategies vary across different stages of pregnancy. During early pregnancy, for nonemergencies, expectant management with close observation is recommended, deferring intervention to the second trimester. During the second trimester, if complications requiring surgical intervention (e.g. torsion) are diagnosed, surgery should be actively scheduled during this period. During the third trimester, for stable patients, conservative treatment can be attempted until term, with deferred management postpartum. For emergencies, decisive surgery is required. Preoperative consultations with obstetrics and neonatology experts should be arranged to prepare for potential emergency cesarean section and premature infant rescue. Surgery may be performed concurrently with elective cesarean section. During the perinatal period, if the splenosis nodule is asymptomatic, vaginal delivery can be attempted. If the nodule is large enough to obstruct the birth canal or poses a rupture risk, cesarean section is a reasonable choice, and simultaneous resection of the splenic tissue nodule can be considered.11,12
Conclusion
This case exemplifies the “minimal intervention” principle for managing pregnancy-complicated splenosis, highlighting the pivotal role of multimodal imaging evaluation and dynamic surveillance in optimizing maternal–fetal outcomes. This comprehensive approach underscores the importance of maintaining physiological pregnancy progression while ensuring timely intervention when indicated.
Footnotes
Acknowledgments
We are grateful to the colleagues of the Radiology Department and Pathology Department.
Author contributions
Zheng Liu was involved in the conception, design, and interpretation of the data. Weiwei Sun revised the manuscript critically for intellectual content, and gave the final approval of the version to be published.
Data availability statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
The authors declare that they have no competing interests.
Funding
None.
Informed consent statement
Informed consent was obtained from the patient.
Statement
The reporting of this study conforms to Case Report (CARE) guidelines. 13
