Abstract
Pregnant women are more likely to face cardiovascular disease than non-pregnant women. However, abdominal aortic aneurysm is an extremely rare complication of pregnancy. Abdominal aortic aneurysms in pregnancy are difficult to identify in the early stage and are often diagnosed only when the symptoms have manifested. We report the case of a multiparous 35-year-old patient with a history of abnormal ascending aorta and ruled-out Marfan syndrome by genetic testing. After a multidisciplinary medical team evaluation, she delivered a live baby by cesarean section at 37 weeks of gestation, and the abdominal aortic aneurysm was repaired simultaneously. This case offers evidence-based recommendations for obstetricians to carry out preventive imaging examination for pregnant women with similar risk factors and provide successful experience in prenatal examination for similar diseases.
Introduction
Abdominal aortic aneurysm (AAA) refers to an aortic disease in which arterial diameter is ⩾30 mm through Doppler or computed tomography (CT).1,2 The symptom of AAA is insidious and it is often found by accidental inspections. The registered mortality rate is not less than 80% once the AAA is ruptured. 3 In order to meet the needs of fetal growth, pregnant women can increase blood volume by 35%–45% and stroke output by 30% until it returns to normal, 1 month after delivery. In our case, at this stage, the level of estrogen and progesterone increased significantly, resulting in the damage of elastic fibers and collagen in the aortic wall. AAA in pregnancy is often associated with diseases such as Marfan syndrome, neurofibromatosis, and Takayasu arteritis. This report is a successful delivery case of pregnancy complicated with AAA, hoping to provide a relevant reference for the healthcare management of delivery.
Case description
A 35-year-old pregnant woman, who had delivered two girls through vaginal delivery and miscarried twice, was diagnosed with true infrarenal AAA at the 25th week of gestation, accidentally. She had the aorta dilated but had ruled-out Marfan syndrome by genetic testing in 2015. AAA through B-ultrasound showed about 10 cm long and 4.8 cm wide. At the 34th week, she suffered the mild anemia in 104 g/L. Oral iron supplements failed to correct the anemia. Hypertension was not detected during the whole pregnancy period. After admission, physical examination and the laboratory examinations were normal, except hemoglobin 94 g/L. The computed tomography angiography (CTA) of thoracoabdominal aorta of the woman suggested that the true fusiform aneurysm from the end of abdominal aorta to the proximal end of left common iliac artery was accompanied by wall thrombosis. The length of AAA was about 80 mm, and the section is 5.9 cm × 4.3 cm (Figure 1). Cardiac ultrasound presented aorta was 42 mm, which had changed little in the past 7 years. After an emergency multidisciplinary treatment, doctors decided to perform cesarean section and endovascular repair of AAA. Inapparent changes of aorta were found compared with before, as the patient needed no additional aortic treatment. The local anesthesia turned to general anesthesia when the puncture was finished. Two venous channels were established for fluid infusion. Continuous arterial monitoring was performed by puncturing the radial artery. After successful puncture of the right side and femoral artery, a 6F catheter sheath (Avanti + Catheter Sheath Introducer) was placed, respectively, in order to handle emergencies of all kinds. A 6F catheter sheath is a guiding catheter (Terumo F is a unit of circumference, i.e. an outer circumference of 6 mm, created in France by French experts and here abbreviated in English).
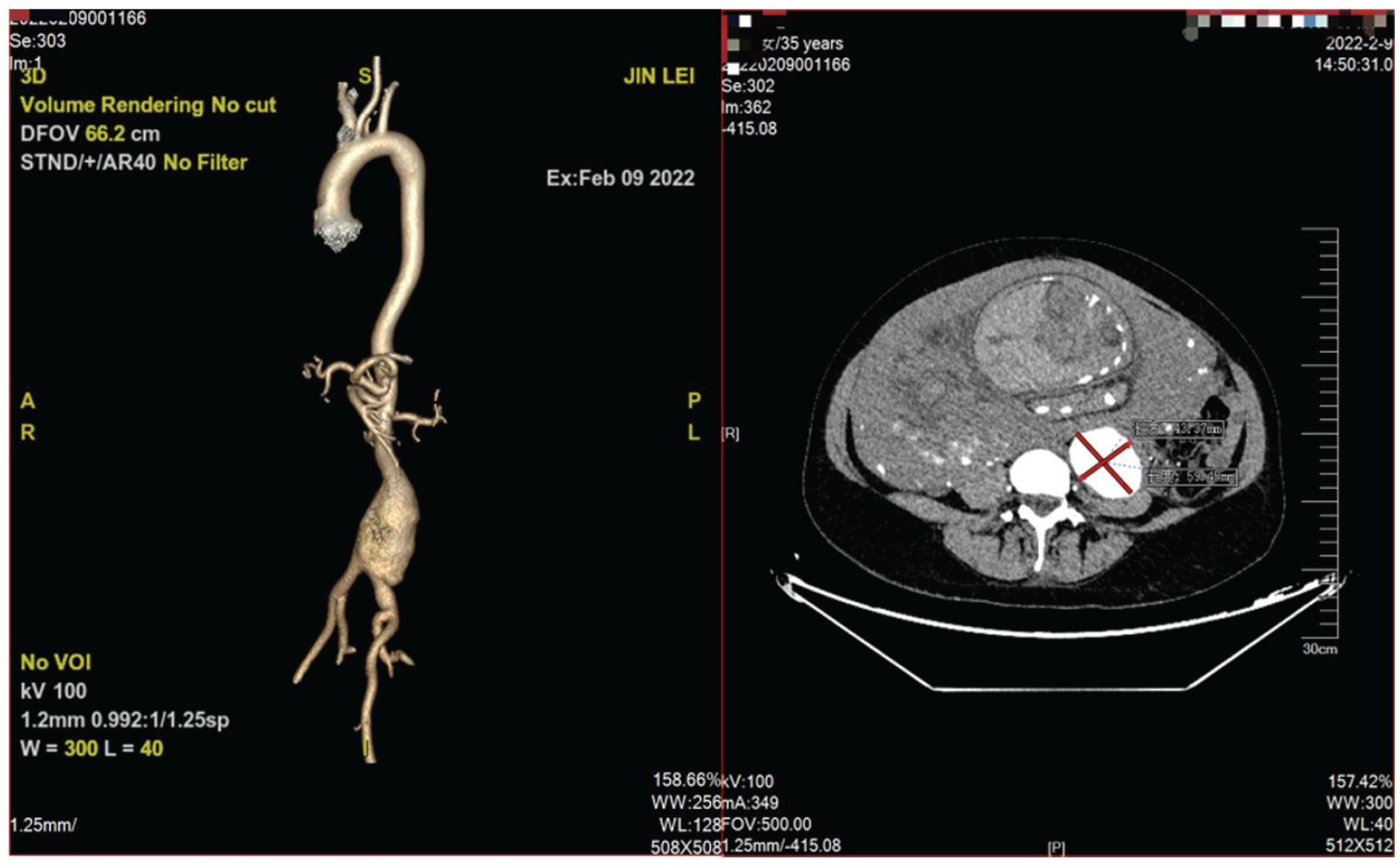
CTA of abdominal aortic aneurysm with the largest transverse section (the diameter is 59 mm × 43 mm).
After conversion to general anesthesia, the obstetricians went to the stage and delivered a newborn with the application of vacuum extractor. Apgar score: 1/5/10 min are 10 points. After complete delivery of placental membranes, the uterus contracted well. The ligation of bilateral ascending uterine arteries was given to prevent postpartum hemorrhage. The arterial condition was not explored to avoid the change of blood tension caused by stimulating aneurysm. Then, the intraoperative bleeding was approximately 400 mL. Next, vascular surgeons participated in the surgery. The vertebral artery catheter was placed at the branches of the anterior and posterior trunk of the internal iliac artery and introduced into two detachable coils (Interlock Boston Scientific, 12 cm × 40 cm). CTA showed that the blood flow of internal iliac artery was slow and the staining became light. The diameter of AAA was 58 mm, the maximum left common iliac artery aneurysm expanded 19 mm, the proximal aneurysm neck was 18 mm, and the length was 35 mm. The main body of covered stent delivery system (GORE PXC161416) was placed on the lower edge of the right renal artery opening, and the catheter was selected to the left iliac branch. The covered stent delivery system (GORE PXC161400) was introduced and then the left iliac branch was connected to the left common iliac artery. Another system was introduced (GORE PXC121000) and the left iliac branch was connected to the left external iliac artery. The main body was released on the right side to the end of the right common iliac artery in the same way and the balloon was placed to expand and make it fit well (Figure 2). After the operation, the patient’s vital signs were stable and transferred to the surgical intensive care unit to continue the intensive care of fluid replacement anti-infection, anticoagulation, acid suppression, and so on. On the first day after operation, she returned to the obstetric ward with a stable condition and discharged smoothly on the fifth day after operation. After half a year of follow-up, the patient and the newborn had no complications.
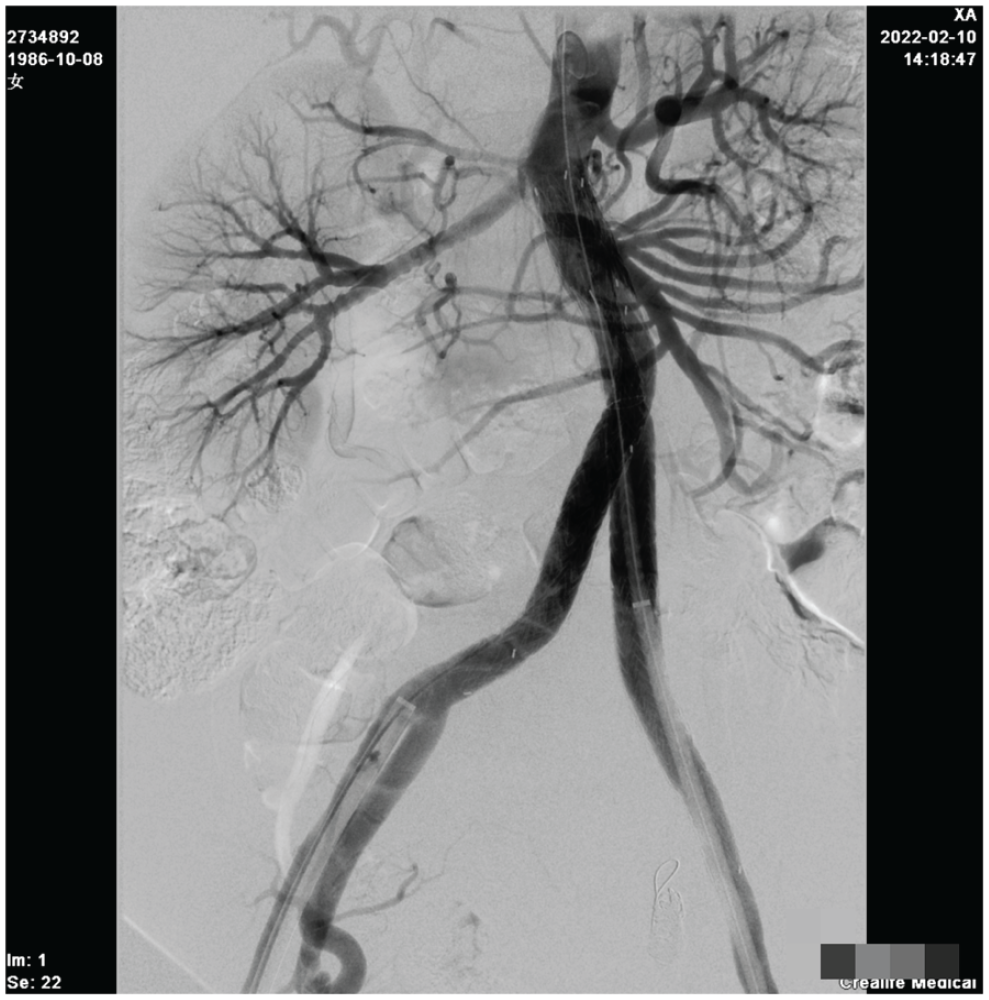
The stent position is fixed, the morphology is good, and the abdominal aortic body is well isolated.
Discussion
AAA is an uncommon pregnancy complication. Currently, there is scarce international literature on statistics concerning the incidence rate of AAA during pregnancy, and only a few cases have been reported. A list of previously published relevant case reports is presented in Table 1.4–10 Understanding the risk factors of abdominal aortic dilatation and making predictive diagnosis is the key point. The risk factors of AAA include age (over 40), male sex, smoking, family history of AAA, presence of other cardiovascular disease, hypertension, hyperlipemia, and inherited disorders.1,11–13 Pregnancy is also a known risk factor for aortic disease. 14
Descriptive statistics of AAA associated with pregnancy.
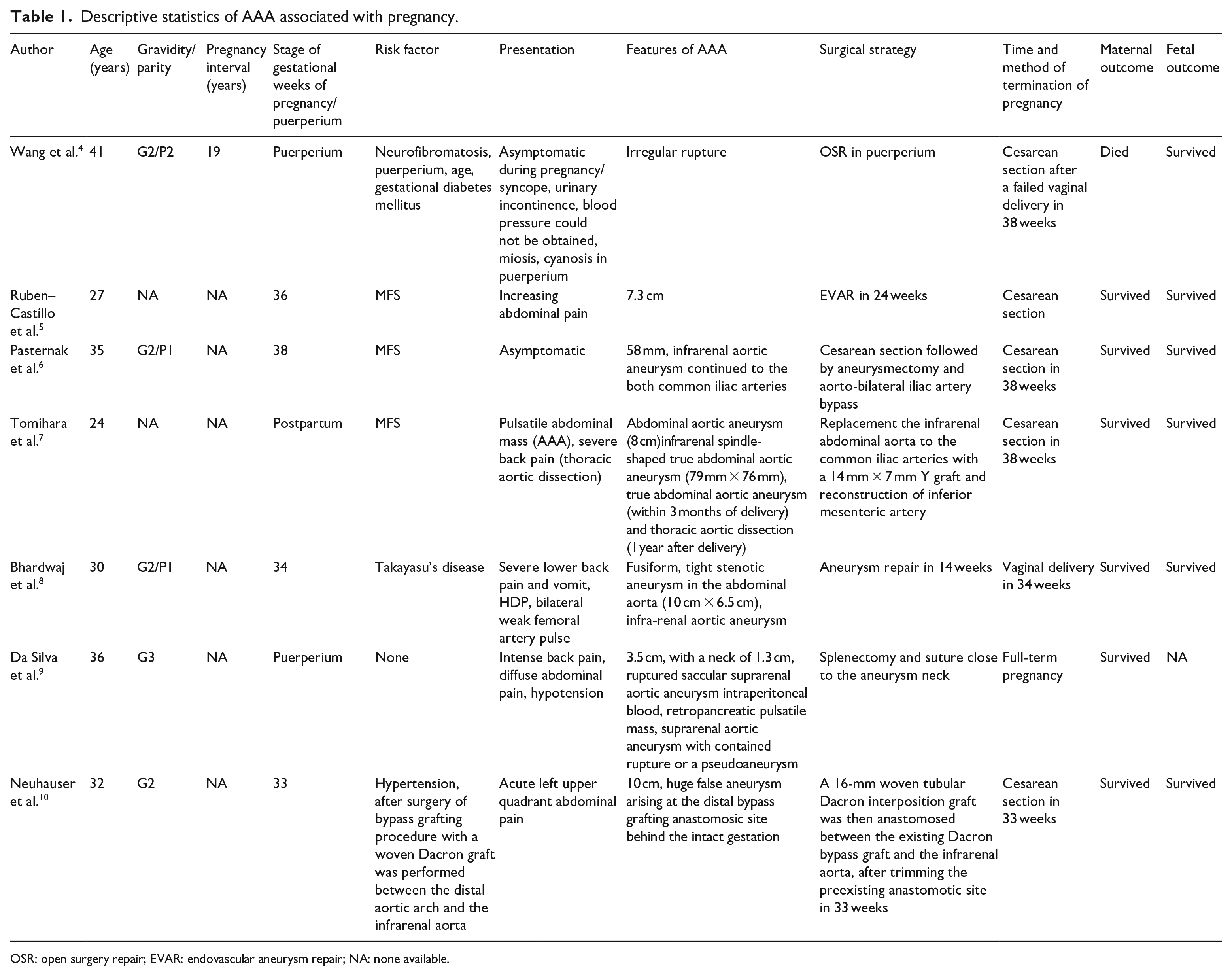
OSR: open surgery repair; EVAR: endovascular aneurysm repair; NA: none available.
This case is a 35-year-old pregnant woman who suffered from ascending aorta dilatation for 6 years. Ascending aortic dilation may be related to the occurrence of AAA, but the specific mechanism is not clear, and the development may be related to genetic factors. 15 What is more, she has a history of repeated pregnancy. A meta-analysis of cohort studies suggests that parity is related to cardiovascular disease risk. 16 It means that the repetitive interaction of altered hemodynamics, varied hormonal levels, abdominal fat, endothelial dysfunction, and systemic inflammation may result in an accumulated burden and generate more opportunities to severe cardiovascular complications.1,2 These cardiometabolic changes may permanently impact the whole life, particularly dangerous in late pregnancy and the postpartum period. Continuous anemia is another adverse point. Low levels of hemoglobin can cause erythropoietin (EPO) production in the kidney. EPO stimulates vascular endothelial cells, selectively activates the erythropoietin receptor (EPOR) homodimer to activate JAK2/STAT5 pathway, and promotes the secretion of vascular endothelial matrix metalloproteinases (MMPs) and the apoptosis of smooth muscle cells, leading to the occurrence of AAA. 17
The surgical indication of asymptomatic AAA is 5.5-cm wide, which is recommended to be confirmed by CTA and followed up by B-ultrasound. 18 In our case, and due to the smaller diameter of abdominal aorta in Asian women, surgical repair should be more active. 19 Pregnant women should weigh the advantages and disadvantages of pregnancy loss when they are less than 28 weeks old. They can choose B-ultrasound diagnosis and follow-up, and if necessary, choose CTA to diagnosis and determine the operation. In this case, the imaging examination showed that the aneurysm diameter grew more than 11 cm in 3 months. It conforms to the definition of AAA growing too fast. The Chinese guideline proposes a monitoring interval <6 months for abdominal aortic >4.5 cm. 18 According to our case and experience, it should be recommended to shorten the follow-up interval of AAA during pregnancy to be at least 3 months or even shorter. 11 The mortality rate of ruptured AAA is reported to be as high as 81%. 3 The way to terminate pregnancy should take ensuring safety as the primary goal and solve two problems at once as far as possible. To minimize AAA pressure changes, our experience in this case suggests and confirms the need to perform cesarean section immediately followed by endovascular aneurysm repair (EVAR). 20 If the above pregnant women have symptoms related to AAA rupture, doctors should promote multidisciplinary treatment to ensure the maternal safety. EVAR is a commonly recommended surgical option for AAA repair but it is not ruled out that there may be better evidence that open surgery may achieve the same effect. Therefore, it is also worth considering endovascular treatment as a bridge to open surgery in young patients with AAA. This approach may provide a temporary solution during pregnancy and could be followed by surgical intervention to achieve a more definitive treatment.
Hence, during the literature review undertaken to support this case, it was found that more sample studies are needed to explore the methods of early identification of AAA to improve the rate of success treatment and reduce the maternal and infant mortality. This finding also confirms recent recommendations for healthcare research.21,22
In addition, recent research on this topic reinforces the claim that acute-type aortic dissection (AD) during pregnancy is considered a fatal complication for both mother and fetus. The most recommended treatment for an acute-type AD is open-heart surgery. However, the decision to perform such a surgery during pregnancy carries significant risk and requires a multidisciplinary approach. Performing a cardiac magnetic resonance (CMR) study without contrast in stable patients is recommended to evaluate the extension of the flap. Open-heart surgery can be a lifesaving potential intervention while also maximizing the likelihood of preserving the fetus. 23 Other recent research has clarified further these principles.24,25
One-year follow-up
The patient was followed upon on 10 April 2023. No discomfort symptoms such as chest tightness or abdomen pain occurred within 1 year of EVAR. All laboratory tests were normal and CTA examination showed that the position of the stent did not move, and no endoleak took place (Figure 3).
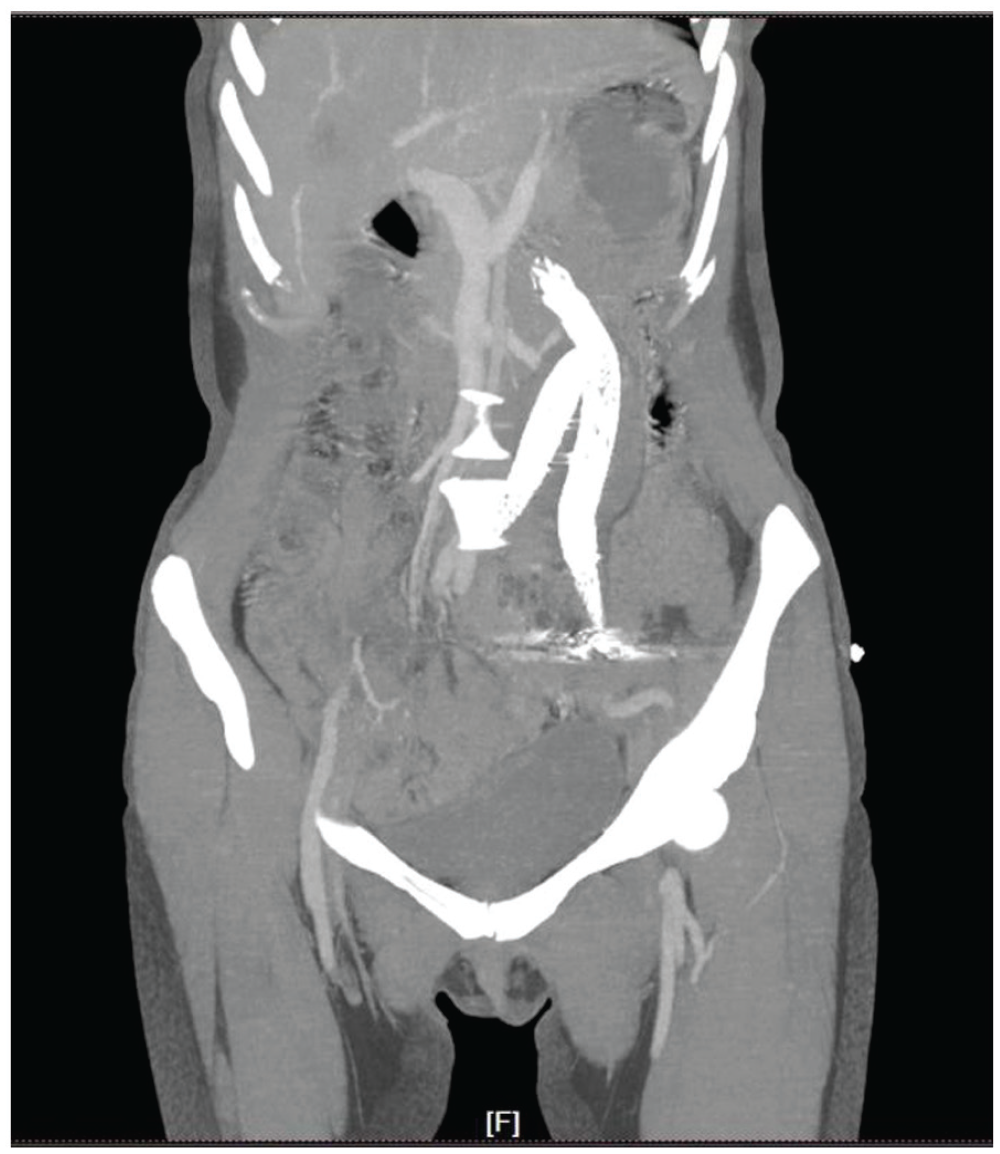
No discomfort symptoms such as chest tightness or abdomen pain occurred within 1 year of EVAR. After 1 year, all laboratory tests were normal and CTA examination showed that the position of the stent did not move, and no endoleak took place.
Conclusion
AAA of insidious nature can go easily misdiagnosed by obstetricians due to its low prevalence. Based on our experience in this case, a few recommendations can be put forward: (1) at least one comprehensive vascular screening when pregnant women who have met one of the risk factors; (2) the follow-up time should be as short as 3 months to reduce the possibility of pregnancy loss on the basis of ensuring maternal safety, and (3) at least 1 year of follow-up with tomography to determine the absence of endoleakage. More international sample studies are required to explore the methods of early identification of AAA.
Footnotes
Acknowledgements
Our deepest gratitude goes first and foremost to Professor Guo as thesis supervisor. Our thanks to the obstetrics department of the Shandong Provincial Qianfoshan Hospital.
Author contributions
P.S. and Z.L. wrote and revised the article together, have contributed equally to this article and share first authorship. W.G. contributed to conceptualizing and methodology application and P.M. revised for methodology quality and reviewed the whole text.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Our institution does not require ethical approval for reporting individual cases or case series.
Informed consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
