Abstract
Objectives
This study aimed to comprehensively investigate the possible associations between neutrophil- and high-density lipoprotein cholesterol (HDL-C)–related inflammatory markers and the incidence of sarcopenia among adults in the United States.
Methods
This cross-sectional study analyzed data from 8878 participants in the National Health and Nutrition Examination Survey. The potential associations between neutrophil- and HDL-C–related inflammatory markers and sarcopenia were evaluated using multiple statistical methods, including weighted logistic regression analysis, sensitivity analyses, restricted cubic spline models, and subgroup analyses. A receiver operating characteristic curve was employed to thoroughly analyze the predictive accuracy of various inflammatory markers for sarcopenia risk.
Results
All examined inflammatory markers were significantly associated with sarcopenia. The association between the neutrophil-to-HDL-C ratio and sarcopenia was nonlinear, with risk increasing notably beyond a certain threshold (breakpoint = 2.791), whereas the neutrophil percentage-to-albumin ratio demonstrated a linear association. Further threshold effect analyses identified specific breakpoints for neutrophil-to-HDL-C ratio, platelet-to-HDL-C ratio, lymphocyte-to-HDL-C ratio, and monocyte-to-HDL-C ratio. Notably, the area under the curve value for neutrophil-to-HDL-C ratio exceeded those for other inflammatory markers in receiver operating characteristic analysis (area under the curve = 0.632, 95% confidence interval: 0.612–0.652), suggesting its potential as a promising predictor of sarcopenia prevalence.
Conclusions
The findings suggest positive associations between inflammatory biomarkers and sarcopenia, with the neutrophil-to-HDL-C ratio demonstrating superior predictive performance.
Keywords
Introduction
Sarcopenia is a severe and prevalent systemic condition associated with the aging process, characterized by a decline in skeletal muscle strength and physiological function.1,2 With the acceleration of global population aging, the prevalence of sarcopenia is increasing. Studies have projected that the prevalence of sarcopenia is anticipated to increase 4-fold, rising from 50 million to more than 200 million cases over the next 40 years. 3 Notably, sarcopenia significantly increases the likelihood of functional impairments, falls, and fractures and may ultimately lead to mortality, thereby potentially imposing a considerable burden on patients, families, society, and healthcare systems.4–6
As research into the etiology of sarcopenia advances, oxidative stress, inflammatory response, and lipid metabolism disequilibrium have emerged as central factors in understanding this condition.7–9 Studies indicate that oxidative stress may elevate the levels of various inflammatory substances and proinflammatory factors, thereby promoting protein degradation while suppressing protein synthesis, which, in conjunction with aging, could be a significant contributor to sarcopenia.10,11 Neutrophils play a critical role in inflammatory responses; they typically increase during acute inflammation and release cytokines to activate lymphocytes and recruit macrophages, potentially leading to chronic inflammation.12,13 Current evidence indicates a strong association between high neutrophil-to-lymphocyte ratio (NLR) and an increased prevalence of sarcopenia. 14 Furthermore, the systemic immune inflammation index (SII), calculated as platelet counts multiplied by neutrophil counts divided by lymphocyte counts, has been identified as a novel predictor of sarcopenia. 15 However, a single inflammatory indicator often captures only one aspect of the body’s inflammatory state, limiting its sensitivity and specificity. To more comprehensively capture the dynamic imbalance between inflammatory status and metabolic function, researchers have proposed ratios combining inflammatory cells with metabolic indicators.
High-density lipoprotein cholesterol (HDL-C), the smallest and most compact class of lipoproteins, exerts multiple beneficial effects, including anti-inflammatory, antioxidant, and immunomodulatory functions. HDL-C levels reflect the body’s metabolic reserve and protective capacity; thus, combining HDL-C with inflammatory markers may better quantify the imbalance between proinflammatory stress and anti-inflammatory/metabolic reserve, theoretically offering greater sensitivity and clinical translational value than individual indicators.16–18 For example, Yanming et al. proposed that the non-HDL-C to HDL-C ratio (NHHR) may serve as a valuable predictor of metabolic dysfunction–related diseases, with a higher NHHR potentially indicating an increased risk of sarcopenia. 19 Moreover, recent research suggests that the monocyte-to-HDL-C ratio (MHR) outperforms SII, NLR, and NHHR in predicting sarcopenia. 2
The neutrophil-to-HDL-C ratio (NHR), a novel HDL-related inflammatory biomarker, simultaneously reflects systemic inflammatory response and lipid metabolism status. NHR has been reported as an early predictor of numerous systemic diseases, including cardiovascular disease, cognitive disorders, and depression, due to its ease of acquisition and calculation.20–22 Albumin, a well-established marker of systemic nutritional status, plays a pivotal role in transporting biomacromolecules and exhibits antioxidant activity. 23 The neutrophil percentage-to-albumin ratio (NPAR), which concurrently reflects systemic inflammation and nutritional status, has demonstrated favorable predictive performance for stroke, metabolic syndrome, rheumatoid arthritis, and periodontitis.23–26 Unlike conventional indices such as SII or NHHR, which focus solely on immune cell ratios or lipid metabolism, our proposed biomarkers (e.g. NHR and NPAR) integrate these two dimensions by combining neutrophils (proinflammatory drivers) with HDL-C/albumin (anti-inflammatory/metabolic regulators).22,24 This dual-target strategy enhances sensitivity to the oxidative–inflammatory–metabolic imbalance characteristic of sarcopenia.
To address the limitations of prior unidimensional biomarkers, we systematically evaluated the first-time applications of NPAR and NHR in sarcopenia, alongside comparative analyses with established ratios (NLR, platelet-to-HDL-C ratio (PHR), lymphocyte-to-HDL-C ratio (LHR), and MHR). This approach leverages multidimensional interactions among inflammation, nutrition, and lipid metabolism to establish a novel predictive framework.
Participants and methods
Study participants
Participants were drawn from the National Health and Nutrition Examination Survey (NHANES) conducted between 2011 and 2018. Since 1998, the NHANES has conducted extensive surveys using a multistage, multistratum sampling technique to assess the prevalent body status of individuals in the United States, with each participant signing a written informed consent form. Leveraging the large, nationally representative NHANES sample, this study had adequate statistical power to detect small-to-moderate association effects. This study was reported in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 27
Initially, this study incorporated 39,156 individuals from the NHANES database across 4 cycles. The study’s reliability and representativeness are ensured by implementing the following stringent exclusion criteria: (a) participants under the age of 20 years (n = 16,830); (b) individuals with incomplete sarcopenia assessment data (n = 11,502); (c) participants with missing data on NPAR, NLR, NHR, PHR, LHR, MHR, or other pertinent blood cell metrics and biochemical indicators (n = 543); and (d) individuals lacking other relevant covariates (n = 1043). Ultimately, the research encompassed 8878 individuals to investigate the potential association between the neutrophil- and HDL-C–related inflammatory indicators and sarcopenia (Figure 1).
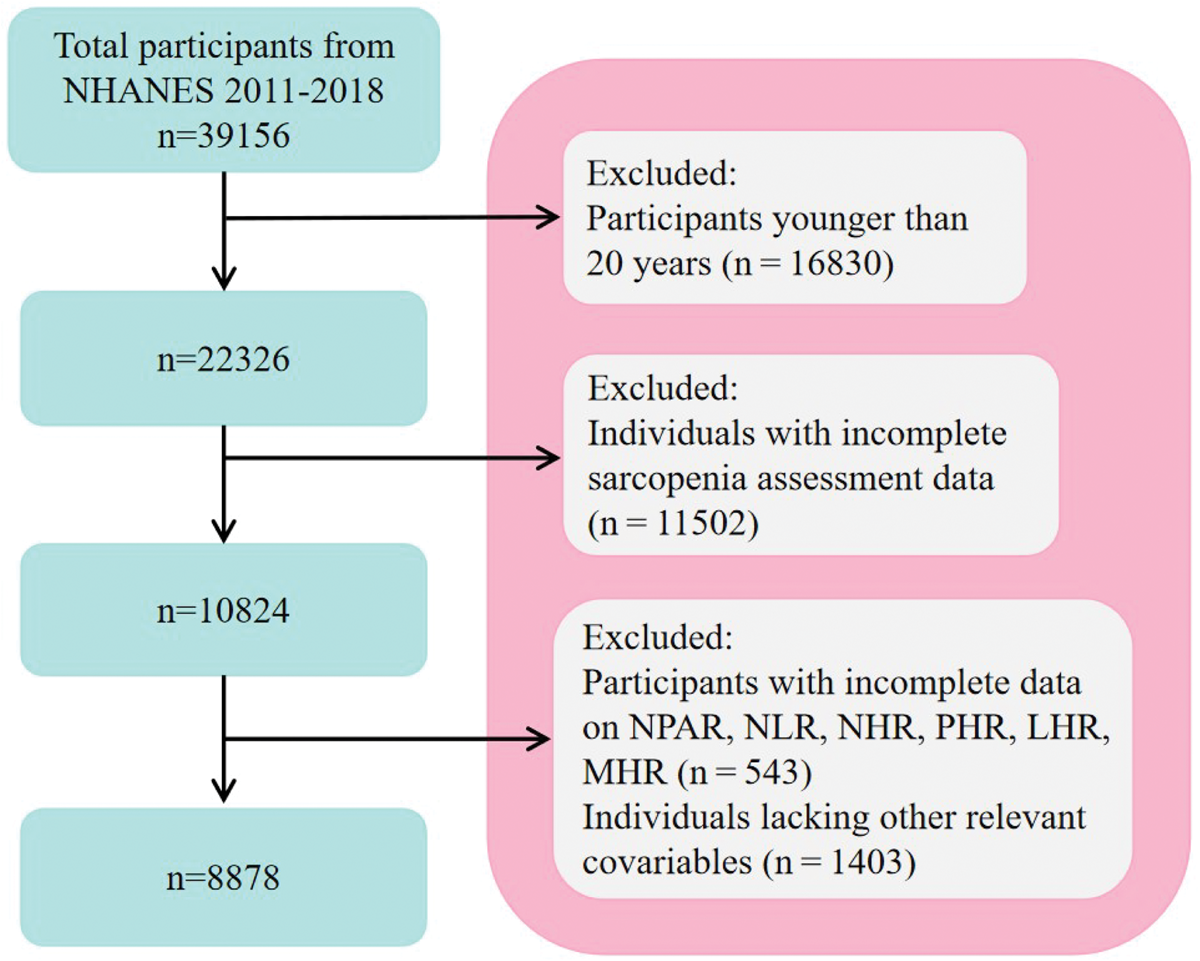
Flowchart of participant inclusion.
Exposure variable
Fasting venous blood samples were collected from all participants to measure complete blood cell count parameters and biochemical markers using specialized laboratory equipment. Neutrophil- and HDL-C–related inflammatory markers were calculated for all individuals using specific blood cell metrics and biochemical indicators. The formulas used for these calculations are listed below:
Assessment of sarcopenia
Appendicular skeletal muscle mass (ASM) was precisely measured using dual-energy X-ray absorptiometry (DXA), which quantifies the lean mass of all limbs, excluding bone. Sarcopenia was defined using a specific index, calculated as ASM divided by body mass index (BMI). Index values below 0.512 for females and below 0.789 for males indicate sarcopenia.28,29
Assessment of covariates
To ensure the comprehensiveness and representativeness of the study, we incorporated multiple confounders such as demographic characteristics, health status, lifestyle factors, and laboratory data. Demographic variables included age, sex, race, educational level, marital status, and poverty–index ratio (PIR). Race was categorized into five primary categories, and education was divided into three distinct levels. Health status was assessed based on the presence of various conditions, including diabetes, chronic kidney disease (CKD), hypertension, and other cardiovascular diseases, using pertinent data obtained from the NHANES database questionnaire module. The diagnostic criteria for CKD were consistent with those established in prior research. 30 Lifestyle factors included smoking status, alcohol intake, sedentary behavior, and BMI. Based on the questionnaire results, smoking status was defined as having smoked more than 100 cigarettes in a lifetime, while alcohol consumption was defined as having consumed at least 12 alcoholic beverages. Comprehensive laboratory data were collected, including measurements for white blood cells, platelets, lymphocytes, monocytes, and neutrophils, along with percentages of neutrophils, albumin, HDL-C, triglycerides, and total cholesterol.
Statistical analysis
To evaluate the predictive performance of inflammatory markers for sarcopenia, the dataset was randomly divided into a training set (70%) and a test set (30%), stratified by age, sex, and sarcopenia status to ensure balanced distribution. This partitioning was conducted prior to any statistical modeling to prevent data leakage. All preprocessing steps (e.g. normalization and handling missing values) were performed separately within each dataset using the parameters derived from the training set. The test set was used exclusively for final validation and remained entirely concealed during model development.
Data extraction and statistical analyses were performed using Empower Stats and R software. For multiple comparisons, statistical significance was determined using a Bonferroni-corrected p value threshold (0.05/6 = 0.0083), with p < 0.0083 considered statistically significant. For all other analyses, p < 0.05 was considered statistically significant. Survey sampling weights were incorporated in accordance with the NHANES statistical guidelines to ensure population representativeness. In the descriptive analysis of baseline data, the normality of continuous variables was evaluated using the Kolmogorov–Smirnov test, which indicated that all variables were non-normally distributed. Consequently, the Wilcoxon rank-sum test was used to analyze these non-normally distributed continuous variables between the groups, with results presented as median (interquartile range (IQR)), while categorical variables were assessed using the chi-square test and expressed as percentages.
We constructed three weighted bivariate logistic regression models, adjusting for various confounders, to investigate the potential association between neutrophil- and HDL-C–related inflammatory markers and sarcopenia. Model 1 served as a baseline model, investigating the association between these biomarkers and sarcopenia without any covariate adjustments. In contrast, Model 2 was adjusted for four crucial demographic factors—sex, age, educational level, and race—to account for demographic discrepancies. Model 3 further expanded these adjustments to include a comprehensive range of confounding factors, including additional demographic variables (PIR and marital status), health conditions (diabetes, CKD, hypertension, and cardiovascular diseases), lifestyle factors (smoking status, alcohol consumption, and sedentary duration), and partial laboratory data (triglycerides and total cholesterol). To evaluate the stability of the observed associations, we excluded extreme values beyond ±3 standard deviations for each biomarker and re-estimated Model 3 in the restricted sample. Furthermore, to enhance the generalizability of the findings and reduce the risk of overfitting, these markers were transformed from continuous to categorical variables to assess the consistency of these associations.
Restricted cubic spline (RCS) analysis was utilized to more accurately characterize the linear or nonlinear correlation between neutrophil- and HDL-C–related inflammatory markers and the incidence of sarcopenia. In addition, weighted subgroup analyses were conducted to evaluate the potential differences in the association between sarcopenia and neutrophil- and HDL-C–related inflammatory markers across different demographic variables, BMI categories, and health conditions; we also performed formal tests for interaction by introducing a cross-product term into the fully adjusted Model 3 to statistically assess whether the effect of the marker significantly differed between the subgroups. Finally, the optimal inflammatory marker for predicting sarcopenia was identified using the area under the receiver operating characteristic curve (AUC-ROC), with AUC values below 0.6 generally considered indicative of poor predictive performance. 31
It should be noted that, due to the cross-sectional nature of this study, the observed associations between inflammatory markers and sarcopenia represent correlations rather than causal relationships.
Ethics statement
All study procedures involving human data were conducted in accordance with the ethical standards of the responsible committee on human experimentation and the Declaration of Helsinki.
The NHANES study was approved by the National Center for Health Statistics (NCHS) Research Ethics Review Board, and all participants provided written informed consent. As the present analysis is a retrospective cross-sectional study based on publicly available, deidentified data, it was deemed exempt from institutional review board approval. All data were extracted from the publicly available NHANES database and were fully deidentified; thus, participant anonymity was maintained throughout the study.
Results
Baseline characteristics
After applying stringent exclusion criteria, the study ultimately included 8878 participants, of whom 765 were diagnosed with sarcopenia, showing an overall prevalence of 9.43% (Table 1). Individuals with sarcopenia were typically older, predominantly of Mexican American descent, and exhibited lower PIR, lower educational level, and higher levels of sedentary time and BMI compared with those without sarcopenia. In addition, individuals with sarcopenia exhibited higher levels of white blood cells, platelets, lymphocytes, monocytes, and neutrophils, along with elevated concentrations of albumin, HDL-C, triglycerides, and total cholesterol and high neutrophil percentages. Figure 2 depicts a sex–age distribution pyramid diagram, illustrating that participants with sarcopenia were predominantly older.
Baseline features of the sarcopenia group versus the nonsarcopenia group.
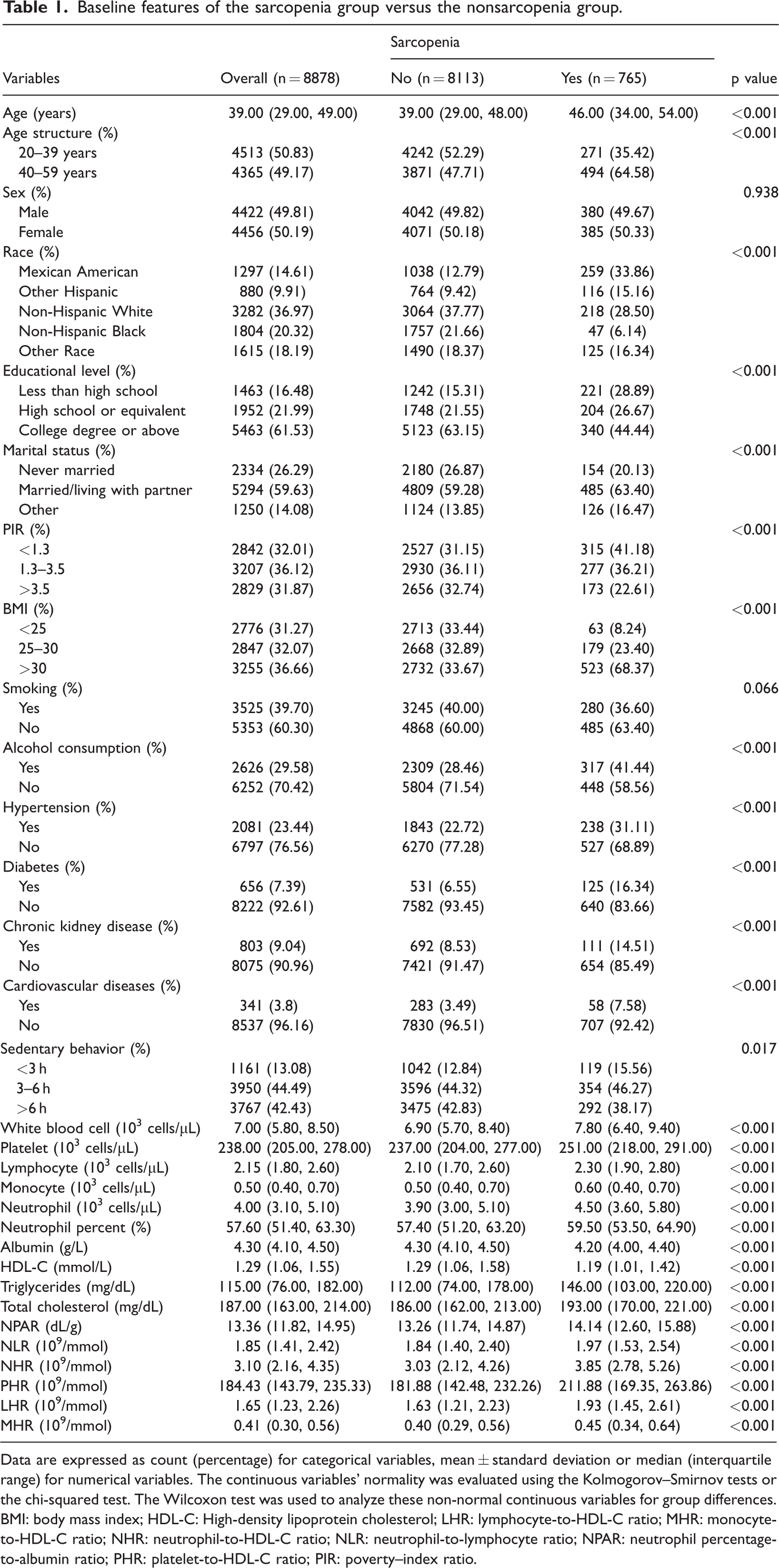
Data are expressed as count (percentage) for categorical variables, mean ± standard deviation or median (interquartile range) for numerical variables. The continuous variables’ normality was evaluated using the Kolmogorov–Smirnov tests or the chi-squared test. The Wilcoxon test was used to analyze these non-normal continuous variables for group differences.
BMI: body mass index; HDL-C: High-density lipoprotein cholesterol; LHR: lymphocyte-to-HDL-C ratio; MHR: monocyte-to-HDL-C ratio; NHR: neutrophil-to-HDL-C ratio; NLR: neutrophil-to-lymphocyte ratio; NPAR: neutrophil percentage-to-albumin ratio; PHR: platelet-to-HDL-C ratio; PIR: poverty–index ratio.

Sex–age distribution pyramid for sarcopenia.
Association between neutrophil- and HDL-C–related inflammatory indicators and sarcopenia
The associations between neutrophil- and HDL-C–related inflammatory indicators and sarcopenia are displayed in Table 2, which summarizes the results of three weighted regression models. In both Model 1 (unadjusted) and Model 2 (partially adjusted), all indicators exhibited a clear positive association with sarcopenia risk (p < 0.0083). For most of these biomarkers, the associations remained significant in Model 3 after further adjustment for additional variables (NPAR: odds ratio (OR) = 1.143 (1.081, 1.208); NLR: OR = 1.087 (1.008, 1.172); NHR: OR = 1.188 (1.131, 1.248); PHR: OR = 1.003 (1.001, 1.006); LHR: OR = 1.272 (1.134, 1.427); MHR: OR = 2.398 (1.538, 3.738); all p < 0.0083). However, the association between NLR and sarcopenia was not statistically significant. These findings indicate that the prevalence of sarcopenia increased by 14.3%, 18.8%, 0.3%, 27.2%, and 139.8% for each unit increase in NPAR, NHR, PHR, LHR, and MHR, respectively.
The correlation between neutrophil- and HDL-C–related indicators and sarcopenia in different models.

Model 1 serves as a baseline, investigating the link between these biomarkers and sarcopenia without any covariate adjustments. In contrast, Model 2 incorporates adjustments for four crucial demographic factors—sex, age, educational level, and race—to address demographic discrepancies. Model 3 further extends these adjustments to cover a broad spectrum of confounding factors, including additional demographic variables (PIR, marital status), health conditions (diabetes, CKD, hypertension, cardiovascular diseases), lifestyle factors (smoking status, alcohol consumption, sedentary duration), and partial laboratory data (triglycerides, total cholesterol).
CI: confidence interval; LHR: lymphocyte-to-HDL-C ratio; MHR: monocyte-to-HDL-C ratio; NHR: neutrophil-to-HDL-C ratio; NLR: neutrophil-to-lymphocyte ratio; NPAR: neutrophil percentage-to-albumin ratio; OR: odds ratio; PHR: platelet-to-HDL-C ratio.
To further assess the associations between these inflammatory biomarkers and sarcopenia, the markers were converted into categorical variables based on tertiles. The results from Model 3, after comprehensively adjusting for confounding variables, demonstrated that the top tertile displayed an obviously greater OR for sarcopenia than the bottom tertile across all inflammatory markers. Notably, the highest OR was observed for NHR (OR: 2.505; 95% confidence interval (CI): 1.737, 3.613). Additionally, the associations of NPAR, NLR, and PHR with sarcopenia were not significant in the middle tertile (NPAR: OR = 1.216 (0.881, 1.679); NLR: OR = 1.256 (0.926, 1.704); and PHR: OR = 1.212 (0.868, 1.692)).
Sensitivity analysis, nonlinear relationship, and threshold effect analysis
Sensitivity analysis (Table S1) demonstrated that the associations of NHR (OR = 1.128, p = 0.002) and NPAR (OR = 1.105, p = 0.003) with sarcopenia remained highly robust in the fully adjusted Model 3 after the exclusion of outliers for all six markers. Notably, the associations of NLR, LHR, and MHR lost statistical significance in Model 3 after the exclusion of outliers, with MHR being the most affected, as its OR value declined from 2.398 to 1.045 (p = 0.900).
Based on the RCS curves, a pronounced nonlinear dose–response association was identified between NLR, NHR, PHR, LHR, and MHR and the incidence of sarcopenia, whereas NPAR demonstrated a linear association (p for nonlinear = 0.741) (Figure 3). Furthermore, we observed that the risk of sarcopenia obviously increased with rising scores of these indicators (p for overall <0.05). Additional threshold effect analyses indicated that NPAR and NLR did not exhibit any notable turning points. In contrast, the infection points for NHR, PHR, LHR, and MHR were identified as 2.791, 259.664, 1.892, and 0.367, respectively. Specifically, the likelihood of sarcopenia markedly increased when PHR levels were below 259.664 (OR = 1.007 (1.005, 1.009)) and LHR levels were below 1.892 (OR = 2.532 (1.784, 3.594)). Interestingly, there was a persistent positive relationship between the prevalence of sarcopenia and the levels of NHR and MHR, with the association being more obvious when the values were below 2.791 and 0.367, respectively (NHR: OR = 1.860 (1.318, 2.626) and MHR: OR = 15.747 (1.270, 195.207)) (Table 3).

RCS curve depicts the connection between neutrophil- and HDL-C–related inflammatory indicators and sarcopenia. LHR: lymphocyte-to-HDL-C ratio; MHR: monocyte-to-HDL-C ratio; NHR: neutrophil-to-HDL-C ratio; NLR: neutrophil-to-lymphocyte ratio; NPAR: neutrophil percentage-to-albumin ratio; PHR: platelet-to-HDL-C ratio.
Analysis of threshold effects of different biomarkers on sarcopenia in Model 3.
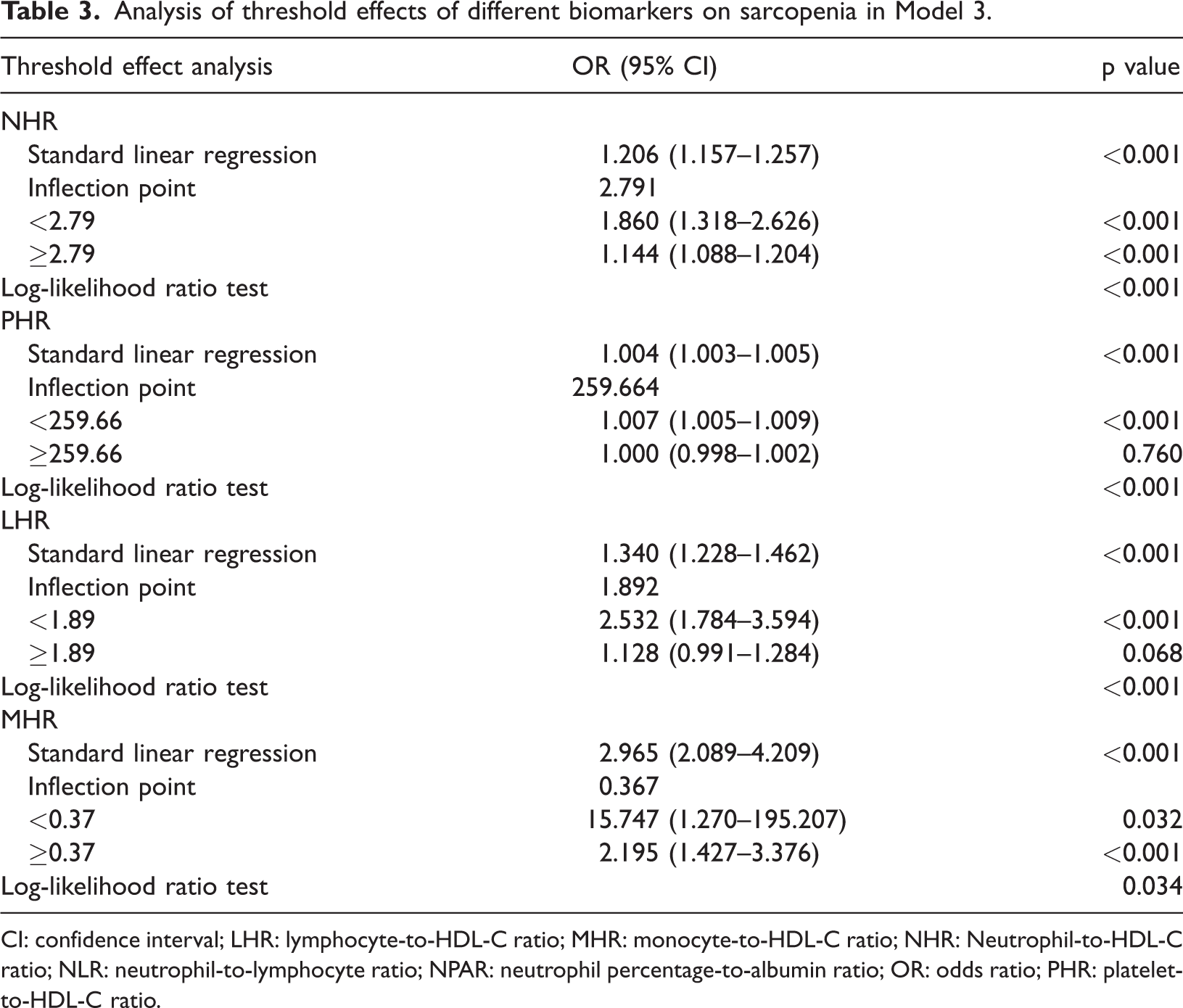
CI: confidence interval; LHR: lymphocyte-to-HDL-C ratio; MHR: monocyte-to-HDL-C ratio; NHR: Neutrophil-to-HDL-C ratio; NLR: neutrophil-to-lymphocyte ratio; NPAR: neutrophil percentage-to-albumin ratio; OR: odds ratio; PHR: platelet-to-HDL-C ratio.
Subgroup analysis
Subgroup analyses were conducted according to age, sex, PIR, BMI, and health status, including diabetes, CKD, hypertension, and other cardiovascular diseases, to evaluate the strength and potential variations in the association of NHR and NPAR with sarcopenia across diverse populations (Figure 4). Notably, no significant interactions were observed between NPAR and sarcopenia, suggesting that NPAR may independently influence the risk of sarcopenia (Figure 4(a)). Conversely, interaction analysis confirmed that the association between NHR was significantly modified by sex (p for interaction = 0.012). Specifically, the association between NHR and sarcopenia was stronger in males (OR = 0.147, 95% CI: 1.062–1.239) (Figure 4(b)).
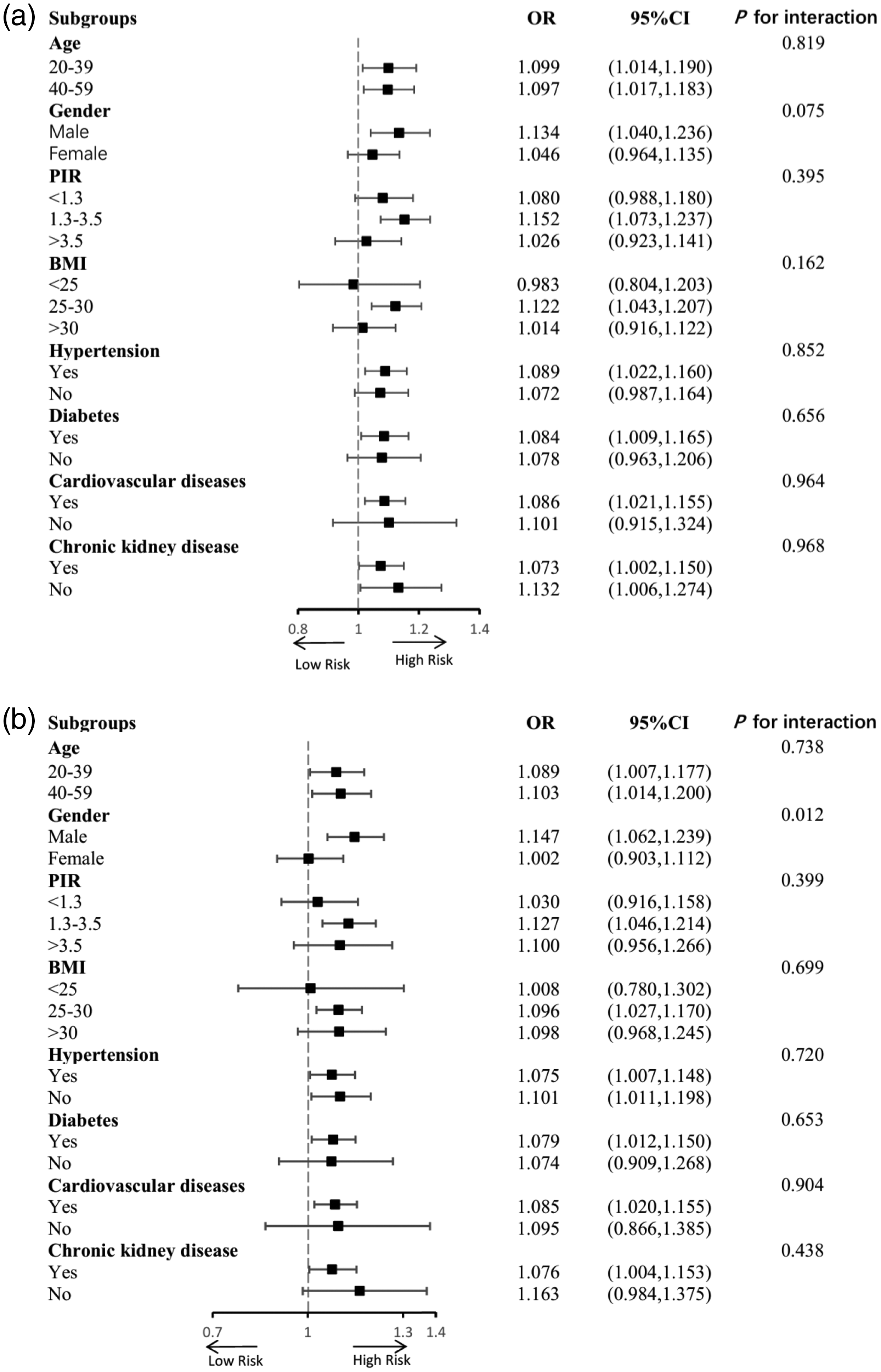
Subgroup analysis of the correlation of NPAR (a) and NHR (b) with sarcopenia. NHR: neutrophil-to-HDL-C ratio; NPAR: neutrophil percentage-to-albumin ratio.
ROC curve
We performed ROC analyses and calculated the AUC values to evaluate the predictive capacity of neutrophil- and HDL-C–related inflammatory indicators for sarcopenia. The findings showed that the AUC values for NLR and MHR were merely 0.555 and 0.583, respectively, suggesting that both have a poor predictive value for sarcopenia. Although the AUC was modest (0.632), NHR demonstrated relatively better performance compared with other markers (Figure 5). Table 4 summarizes the AUC values and 95% CIs for NPAR, NLR, NHR, PHR, LHR, and MHR.

The ROC curves of NPAR, NLR, NHR, PHR, LHR, and MHR. AUC: area under the curve; LHR: lymphocyte-to-HDL-C ratio; MHR: monocyte-to-HDL-C ratio; NHR: neutrophil-to-HDL-C ratio; NLR: neutrophil-to-lymphocyte ratio; NPAR: neutrophil percentage-to-albumin ratio; PHR: platelet-to-HDL-C ratio; ROC: receiver operating characteristic curve.
The AUC and its 95% CI of inflammatory indicators associated with neutrophils and HDL-C.

AUC: area under the curve; CI: confidence interval; LHR, lymphocyte-to-HDL-C ratio; MHR, monocyte-to-HDL-C ratio; NHR, neutrophil-to-HDL-C ratio; NLR, neutrophil-to-lymphocyte ratio; NPAR, neutrophil percentage–to-albumin ratio; PHR, platelet-to-HDL-C ratio.
Discussion
This study is the first to demonstrate significant positive associations between NPAR, NHR, and other neutrophil- and HDL-C–related inflammatory markers and sarcopenia. Among these biomarkers, NHR exhibited the most prominent predictive capacity. The observed nonlinear relationships between these indices further suggest that inflammatory and metabolic dysregulation may exert threshold-dependent effects on muscle health. Collectively, these findings highlight the potential of NHR and NPAR as integrative indicators reflecting the imbalance between proinflammatory burden and metabolic reserve, providing new insights into the pathophysiological mechanisms linking between systemic inflammation and sarcopenia.
Research suggests that sarcopenia is a persistent chronic inflammatory disorder driven by oxidative stress and immune responses, with notable elevations in tumor necrosis factor α and interleukin-6.10,32,33 Although these cytokines could potentially regulate the inflammatory responses, their continuous elevation is detrimental to muscle mass.34,35 These inflammatory mediators could affect skeletal muscle through various signaling pathways, such as nuclear factor κB, disrupting the equilibrium between protein synthesis and breakdown in muscle tissue, thereby accelerating the progression of sarcopenia.10,36,37 Moreover, chronic inflammation can indirectly decrease growth hormone and insulin-like growth factor 1 concentrations, negatively impacting skeletal muscle mass and strength and potentially leading to the development of sarcopenia.11,38 Notably, oxidative stress resulting from mitochondrial dysfunction and the excessive accumulation of reactive oxygen species (ROS) can damage the biological macromolecules, particularly lipids and proteins, ultimately contributing to a decline in muscle mass.39–41 The accumulation of ROS, combined with the inflammatory response, may trigger various types of programmed cell death within skeletal muscle cells, including autophagy, apoptosis, ferroptosis, and pyroptosis, further compromising muscle mass and strength.42–45 To conclude, oxidative stress and inflammatory responses are pivotal in the development and progression of sarcopenia, implying that therapies targeting anti-inflammatory and antioxidant mechanisms could be a promising strategy for early intervention.
Neutrophils, essential components of the immune system in humans, play a crucial role in orchestrating the inflammatory process. These cells might alter muscle fiber homeostasis, leading to muscle weakness and reductions in muscle strength and mass.46,47 Dysregulation of the phosphoinositide 3-kinase–Akt pathway within neutrophils might contribute to the pathogenesis of sarcopenia. 48 Albumin exhibits anti-inflammatory and antioxidant properties, which are influenced by inflammatory conditions, resulting in variations in its concentration.23,49 Furthermore, reduced levels of albumin have been associated with an increased risk of sarcopenia. 50 Our research suggests that NPAR, an inflammatory marker related to neutrophil percentages and albumin levels, demonstrated a moderate yet consistent association with sarcopenia. In contrast, NLR exhibited a poor predictive capacity for the disease prevalence, despite previous research suggesting a strong connection between NLR levels and sarcopenia prevalence.14,31 Overall, the interaction between neutrophils and albumin in relation to sarcopenia may impact its progression to some degree. Meanwhile, NPAR, representing the association between proinflammatory and anti-inflammatory processes, may serve as a potential biomarker for predicting the disease.
HDL-C is known for its anti-inflammatory, antioxidant, and immune-regulating properties. First, HDL-C can inhibit the production of inflammatory mediators and chemokines, thereby improving immune cell function. 51 Second, HDL-C suppresses the function of neutrophils by reducing their activation, proliferation, and migration while also preventing the formation of oxidized low-density lipoprotein.13,52 Additionally, HDL-C neutralizes free radicals and mitigates oxidative stress, thereby indirectly reducing inflammation. 22 These favorable features have prompted extensive research into HDL-C-related biomarkers, as they may reflect underlying systemic inflammation and oxidative stress equilibrium.17,18 Interestingly, emerging novel markers associated with HDL-C, such as NHR, LHR, PHR, and MHR, have been identified as valuable predictors for various diseases, including cardiovascular diseases, CKD, frailty, psoriasis, carcinoma, and metabolic diseases.49,53–56 Among these, NHR integrates neutrophil-driven inflammation with HDL-C-mediated protective effects, offering a unique perspective on the inflammatory–metabolic crosstalk relevant to muscle health.
It is noteworthy that among the evaluated inflammatory indices, NHR exhibited the highest AUC value; however, its discriminative ability remained modest (AUC = 0.632), indicating its limited standalone predictive power. Although Xue et al. 2 demonstrated that MHR possesses favorable predictive capacity for sarcopenia prevalence, as evidenced by ROC analysis, the current findings suggest that NHR may represent a more efficient biomarker. Our analysis identified a critical inflection point for NHR at 2.791, beyond which sarcopenia risk increased nonlinearly. This threshold has important clinical implications. We propose that NHR could serve as a readily available, cost-effective triage indicator. Patients with sustained NHR values above this threshold could be prioritized for comprehensive diagnostic evaluation using tools such as DXA, grip strength measurement, and gait speed assessment. This strategy would allow clinicians to efficiently allocate resources and initiate timely interventions (e.g. resistance training and nutritional support) before irreversible muscle loss and functional impairment occur, potentially improving patient outcomes and reducing the healthcare burden.
It is important to highlight that our analyses revealed a positive association between NPAR and sarcopenia, whereas the relationship between NHR and sarcopenia appears to be influenced by sex. This discrepancy may be attributed to both biological and behavioral factors. From a hormonal perspective, estrogen in females exerts protective effects by enhancing HDL-C levels, reducing oxidative stress, and attenuating systemic inflammation, which could mitigate the impact of NHR on muscle decline. In contrast, men tend to exhibit higher inflammatory activity and less favorable lipid profiles, partly due to differences in sex hormones and lifestyle behaviors such as smoking and alcohol consumption.57,58 These factors may collectively amplify the association between NHR and sarcopenia in males. Further studies incorporating hormonal profiles and detailed lifestyle data are needed to elucidate the mechanisms underlying these sex-specific differences. In summary, considering the associations between NHR and sarcopenia described above, we propose that NHR could serve as a simple, cost-effective laboratory index to assist in early risk stratification, facilitating timely assessment and intervention in individuals at elevated risk for sarcopenia. Nonetheless, external validation in diverse populations and in prospective studies remains essential to confirm its predictive value and clinical applicability.
This study includes the following strengths. First, the utilization of NHR and NPAR—derived from routine hematological and biochemical parameters—offers a practical advantage for large-scale screening, as these measures require no specialized equipment. Second, to enhance the robustness and accuracy of the findings, a comprehensive range of potential confounders was incorporated into the analysis through multivariable adjustment and sensitivity analyses. Third, through the application of nationally representative NHANES data and proper sampling weights, the findings of this research are broadly applicable and generalizable to the US adult population. Importantly, to address potential concerns about multiple testing, we implemented Bonferroni correction (p < 0.0083 considered significant) when examining the six inflammatory indicators. Finally, by analyzing a nationally representative and diverse sample of 8878 individuals, this study is the first to thoroughly investigate the potential links between NPAR, NHR, and other neutrophil- and HDL-C–related inflammatory indicators (NLR, PHR, LHR, and MHR) and sarcopenia among Americans. Although the cross-sectional design precludes causal inference, our findings provide valuable preliminary insights into potential pathophysiological mechanisms and may inform future longitudinal studies aimed at developing integrated screening strategies combining these biomarkers with functional assessments.
However, certain limitations of this study should be acknowledged. First, despite employing complex sampling techniques and accounting for various potential confounders (including demographic characteristics, lifestyle factors, and comorbidities listed in Table 1), there remains the possibility of residual confounding due to unmeasured variables such as genetic predisposition or detailed dietary patterns, potentially introducing bias. Second, the multiple tests in logistic regression analyses introduce several drawbacks, including increased Type I error rates, reduced statistical power, and practical limitations, all of which may affect the accuracy and robustness of the findings. Third, most importantly, the cross-sectional design fundamentally limits causal interpretation of the observed associations between inflammatory markers and sarcopenia. Although our findings are biologically plausible given existing evidence on neutrophil-mediated inflammation and HDL-C metabolism in muscle aging,15,19 longitudinal studies are needed to establish temporal relationships. To enhance the translational relevance of these findings, future studies should consider integrating inflammatory biomarkers such as NHR and NPAR with established clinical screening tools for sarcopenia, including the SARC-F (Strength, Assistance with walking, Rising from a chair, Climbing stairs, and Falls) questionnaire, grip strength, and gait speed assessments, to improve early identification of at-risk individuals and enable more comprehensive risk stratification by capturing both physiological and functional dimensions of muscle health.
Conclusion
This study conducted a comprehensive examination of the associations between NPAR, NHR, and other neutrophil- and HDL-C–related inflammatory indicators (NLR, PHR, LHR, and MHR) and sarcopenia in a representative cohort of US adults. The results indicated that these biomarkers were positively correlated with sarcopenia, with NHR demonstrating the strongest predictive value. Consequently, monitoring NHR levels may be critical for the early identification of individuals at elevated risk for sarcopenia and for implementing targeted interventions to mitigate its progression.
Footnotes
Acknowledgments
The authors have no acknowledgments to declare.
Author contributions
Daoran Xu: Conceptualization, Investigation, Writing–original draft, Writing–review & editing. Hanke Xu and Jiachen Wang: Methodology, Investigation, Writing–original draft. Kai Ban and Xiangzhong Huang: Validation, Data curation. Wu Yang and Chao Liu: Supervision, Writing–review & editing. Yunrong Zhu: Methodology, Supervision, Validation, Writing–review & editing. All authors have read and agreed to the current version of the manuscript.
Data availability statement
Declaration of conflicting interests
The authors declare no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
