Abstract
Central compartment atopic disease is a recently delineated inflammatory endotype of chronic rhinosinusitis. It is characterized by involvement of the central intranasal compartment—the middle turbinate, superior turbinate, and posterior superior nasal septum—and is associated with a predominantly polypoid phenotype. This review synthesizes current evidence on local allergic pathophysiology, diagnostic frameworks, and stratified management of central compartment atopic disease to facilitate accurate diagnosis and individualized treatment.
Keywords
Introduction
Chronic rhinosinusitis (CRS) is a prevalent rhinologic disorder comprising diverse clinical phenotypes and immunologic endotypes. 1 Its global prevalence is estimated at 5%–28%.2,3 In the United States alone, CRS accounts for direct medical expenditures of approximately US$13 billion and indirect costs of approximately US$20 billion annually, imposing a substantial public health burden and markedly diminishing quality of life. Although the pathogenesis of CRS remains incompletely defined, several subtypes, notably allergic fungal rhinosinusitis (AFRS), aspirin-exacerbated respiratory disease (AERD), and the more recently delineated central compartment atopic disease (CCAD), exhibit strong links to aeroallergen exposure.4,5 CCAD, in particular, appears tightly coupled to inhalant allergens 6 and likely represents a localized, type 2 (T2) skewed allergic condition. 7 Endotypic heterogeneity has also been observed across regions and populations. 8
By framing CCAD within an “airflow–particulate deposition–mucosal barrier disruption” paradigm, this review explains its central and focal involvement and the rationale for local allergic rhinitis (LAR). Moreover, it outlines practical diagnostic pathways and stratified management. We also proposed the hypothesis that postoperative airflow remodeling modulates the efficiency of topical drug delivery, providing an additional perspective for recurrence prevention and long-term disease control.
Definition and current advances in CCAD
CCAD has emerged as a distinct subtype of CRS defined by a characteristic anatomic distribution and immune profile. 9 Disease involvement is confined to the central intranasal compartment—encompassing the middle turbinate (MT), superior turbinate (ST), and posterior superior nasal septum (PSNS). It typically presents with a polypoid phenotype, 10 whereas the inferior turbinate is generally spared, plausibly reflecting differences in embryologic origins (Figure 1). 11 The entity was first described by White et al. in 2014, who associated polypoid edema or polyp formation of the MT with inhalant allergen–mediated responses, thereby implicating this region as a focal site of local hypersensitivity. 12 In a cross-sectional study involving 303 patients, Hamizan et al. demonstrated strong associations between diffuse edema (positive predictive value (PPV): 91.7%; likelihood ratio (LR): 8) or polypoid edema (PPV: 88.9%; LR: 6.2) and aeroallergen sensitization. 13 Brunner et al. further showed that polypoid change of the MT was correlated more strongly with allergic rhinitis (AR) than with CRS (AR: 83% vs. 34%, p < 0.001; CRS: 10% vs. 100%, p < 0.0001) and was associated with broader polysensitization compared with paranasal sinus polyposis. 14 Synthesizing these findings, CCAD is characterized by allergic inflammation predominantly involving the central nasal compartment, with relative sparing of the lateral paranasal sinus mucosa. The European Position Paper on Rhinosinusitis and Nasal Polyps 2020 (EPOS 2020) subsequently recognized CCAD as a distinct CRS subtype. 4 Pediatric data (n = 82) from Lee et al. revealed higher sensitization to inhalant allergens in CCAD compared with non-CCAD (87.1% vs. 62.4%, p = 0.008), increased mite sensitization (74.2% vs. 53.4%, p = 0.035), and more frequent elevation of total serum IgE (51.6% vs. 30.1%, p = 0.023). 1

CCAD: representative nasal endoscopic and CT findings. CCAD: central compartment allergic disease; CT: computed tomography; R: right nasal cavity; MT: middle turbinate; S: nasal septum; P: polypoid changes.
Despite growing interest, the pathophysiology, diagnostic thresholds, and nosologic position of CCAD remain incompletely defined. Key priorities for future research include clarifying its distinction from AR and other chronic rhinosinusitis with nasal polyps (CRSwNP) subtypes and determining whether CCAD represents a localized extension of AR or an independent CRS entity—evidence that will be critical for precise diagnosis and individualized, stratified management.
Pathobiology and immune features
Anatomy and airflow dynamics
The nasal cavity constitutes the anterior segment of the upper airway, opening anteriorly to the vestibule and posteriorly to the nasopharynx through the choanae. The nasal septum divides the cavity bilaterally, while the inferior, middle, and superior turbinates define their corresponding meatuses. The central compartment—comprising the middle and superior turbinates, olfactory cleft, and adjacent posterosuperior septum—lies adjacent to the ethmoidal ostia, functions as an aerodynamic convergence zone, and represents a principal site of aeroallergen deposition.7,15–18 Embryologically derived from the ethmoid and lacking the venous sinusoids and fibrous stroma characteristic of the inferior turbinate, the MT is particularly susceptible to allergen-driven structural remodeling.7,12
In AR and CRS, mucosal congestion and edema increase nasal resistance, and under congested conditions, unilateral airway resistance rises substantially in obstructed patients. 19 Particles smaller than 50 μm preferentially deposit at the junction of the anterior septum and MT, creating focal regions of high antigen load that perpetuate inflammation. Septal deviation and other anatomic variants further redistribute intranasal airflow, generating heterogeneous velocities and localized pressure elevations that prolong allergen residence time and amplify mucosal inflammation. 20 Following partial middle turbinectomy, airflow accelerates within the middle–superior meatus, 21 increasing deposition of inhaled particles (e.g. pollen) along the septum, maxillary sinus, and olfactory cleft and potentially precipitating focal allergic inflammation. 22 Patient-specific computed tomography (CT) models after functional endoscopic sinus surgery (FESS) demonstrate that wider surgical openings enhance nasal–sinus coupling, with marked increases in maxillary and ethmoid ventilation. 23 This structural and functional remodeling likely influences the efficiency of topical drug delivery, regulation of local inflammation, and mucosal repair. 24
Environmental context further modulates risk: cold, dry conditions intensify airflow–mucosa interactions, promote epithelial desiccation and barrier disruption, and facilitate allergen penetration with subsequent immune activation. 25 In the central compartment of AR, edema commonly coexists with dysbiosis—characterized by reduced bacterial diversity and enrichment of potential pathobionts—compromising the local barrier and altering airflow sensing, thereby amplifying local hypersensitivity. 26
Taken together, CCAD appears to arise from the interplay of central compartment anatomy, airway aerodynamics, and the mucosal immune barrier. These insights support airflow-informed optimization of intranasal corticosteroids and other topical delivery strategies, an approach that warrants verification through multicenter, large-scale controlled studies using multidisciplinary tools such as three-dimensional printed models and computational fluid dynamics (CFD).
Immune inflammation and cytokine profile
EPOS 2020 and the EPOS/EUFOREA 2023 update on biologics in CRS with nasal polyps classify CCAD as a primary, diffuse T2 endotype of CRS, with diagnostic thresholds of tissue eosinophils ≥10 per high-power field (HPF) or serum eosinophils ≥150.4,27 Owing to its unique anatomic location, the central compartment exhibits distinct airflow dynamics that promote aeroallergen aggregation and deposition, thereby triggering localized immune activation. 28 Current models indicate that environmental allergens, viruses, or bacteria stimulate nasal epithelial cells to release epithelial-derived cytokines—notably thymic stromal lymphopoietin (TSLP), interleukin (IL)-25, and IL-33—which drive naïve CD4+ Th0 cells toward a Th2 phenotype.29,30 Activated Th2 cells secrete IL-4, IL-5, and IL-13; among these, IL-5 is pivotal for eosinophil recruitment and activation, underpinning the characteristic eosinophilic infiltration of the nasal mucosa. 29 Th2 cytokines further induce IgE synthesis, which activates mast cells and basophils, resulting in mediator release, mucus hypersecretion, and tissue remodeling. 31 Furthermore, innate lymphoid cells type 2 (ILC2s) amplify this process, enhancing eosinophilia and mucus production. 32
Although CCAD shares features with AR and eosinophilic CRS (eCRS), it exhibits a distinct immune signature. Eosinophil counts in CCAD mucosa are often lower than those observed in prototypical eCRS; nevertheless, the inflammatory program remains T2-dominant. 8 A defining feature is the presence of local IgE: even when serum total IgE and skin prick testing (SPT) are negative, appreciable IgE can be detected within nasal tissue, indicating localized atopy.33,34 Bacterial superantigens, such as staphylococcal enterotoxins, may further modulate the local immune response by recruiting B cells and promoting a cytokine milieu that includes interferon (IFN)-γ, tumor necrosis factor (TNF)-α, and macrophage-activating factors targeting intracellular pathogens. Concurrently, Th2 cytokines (IL-4, IL-5, IL-10, and IL-13) sustain humoral and allergic inflammation, while Th17 (IL-17) contributes to barrier disruption, impaired host defense, and tissue inflammation with potential autoimmunity (Figure 2). 8

T2 immune response pathway underlying CCAD. CCAD: 2 central compartment allergic disease; T2: type.
Collectively, these findings support CCAD as a distinct, chronic, localized inflammatory subtype originating in the central nasal compartment, centered on T2 immunity with eosinophilic infiltration and local IgE production. Incorporating nasal airflow into this framework refines the pathogenic model and provides a mechanistic basis for precise diagnosis and individualized therapy.
Local versus systemic atopy
The International Consensus Statement on Allergy and Rhinology: Allergic Rhinitis 2023 (ICAR:AR 2023) defines LAR as the presence of allergic nasal symptoms in patients with negative systemic testing but positive results on the nasal provocation test (NPT), nasal specific IgE (sIgE), or the basophil activation test (BAT), despite negative systemic allergen assays. 34 As a newly recognized subtype of CRS, CCAD exhibits a mixed atopic pattern in which local atopy is dominant and systemic atopy is secondary. 33 Within the central compartment, concentrated allergen deposition—particularly along the anterior margin of the MT—creates an “allergen filter.” This filter facilitates antigen presentation, activates Th2 cells and ILC2, and triggers the release of IL-4, IL-5, and IL-13. Together, these events drive eosinophilic infiltration and IgE synthesis, establishing a localized allergic microenvironment. 8 In some patients, elevated sIgE can be detected in MT tissue even when serum sIgE and SPT results are negative, highlighting the primary role of the local allergic response in the pathogenesis of CCAD. 33
The deposition of inhaled allergens on central nasal structures may trigger an IgE-mediated type 2 inflammatory cascade, leading to obstructive mucosal edema and the development of CRS, particularly the CCAD phenotype.4,35 Research from the United States indicates that the prevalence of asthma in patients with CCAD is relatively low, ranging from approximately 17.1% to 50%, which is broadly comparable to the prevalence reported in Asian cohorts (6.3%–33%).14,36–38 Many authors have suggested that the lower asthma burden in CCAD may be related to a “filtering effect” of the central nasal compartment. This region partially limits the penetration and distal deposition of inhaled aeroallergens in the lower airways, thereby reducing lower respiratory tract involvement. 8 However, findings from Lee et al. challenge this assumption, showing a higher prevalence of asthma in the CCAD group than in the non-CCAD group (16.1% vs. 3.8%, p = 0.010). 1 These discrepant results may be attributable, at least in part, to differences in disease stage or duration among the studied populations. Elevated levels of local sIgE have been detected in MT tissue from patients with CCAD, 33 and persistently increased local sIgE over the course of the disease may contribute to the development or exacerbation of asthma.26,35,39 Whether variation in asthma comorbidity across studies is primarily driven by geographic and ethnic differences or instead reflects heterogeneity in study design, inclusion criteria, and control group selection remains unclear. Large, multicenter studies across different regions and populations, incorporating appropriately matched control groups, are urgently needed to clarify these associations.
Systemic atopy plays a less prominent but still relevant role in CCAD. Studies from North America and Australia report SPT positivity rates of 73%–100% in CCAD, with frequent comorbid AR.39,40 In contrast, Asian cohorts show lower systemic sensitization, including 36.4% systemic allergen positivity in Taiwan 36 and approximately 23.1% comorbid AR in South China. 37 These differences likely reflect variations in lifestyle, exposure patterns, and regional aeroallergen spectra. Systemic markers, such as total serum IgE and peripheral eosinophils, are elevated in CCAD but remain below levels typically observed in eosinophilic CRSwNP, consistent with a moderate T2 phenotype. 38 Systemic sensitization may synergize with local immune circuits to amplify focal inflammation and promote disease recurrence or progression to lower airway conditions such as asthma. 26
In summary, CCAD follows a dual atopic model dominated by local mechanisms, with systemic contributions observed in selected populations. Central compartment allergen deposition, local sIgE production, and activation of T2 cytokine pathways form the core pathology, while systemic atopy may act as an amplifier in specific contexts. Regional variations in aeroallergen exposure may account for differences in CCAD prevalence and could influence responses to LAR-targeted desensitization or biologics aimed at systemic T2 inflammation. These hypotheses warrant validation in large, multicenter studies.
Clinical features and diagnostic pathway
Phenotype
CCAD resembles AR clinically, 41 presenting with paroxysmal sneezing, watery rhinorrhea, ocular itch, and hyposmia; asthma and atopic dermatitis may occur in a subset of patients.1,13,14,42 Reports on olfactory function are divergent—some describe milder loss, while others report more severe impairment than that in non-CCAD CRS—likely reflecting differences in diagnostic criteria, with environmental and population factors also acting as plausible contributors.36,38 We speculate that the particularly pronounced olfactory loss in CCAD results from a combination of its anatomical predilection and its T2 inflammatory endotype. Central compartment lesions lie in close proximity to the olfactory cleft, where mucosal edema and polypoid changes markedly impair the delivery of odorant-laden airflow.7,43 In addition, a T2-driven inflammatory milieu—with elevated IL-5 and IL-13 levels in olfactory cleft mucus reported in CRSwNP patients with hyposmia—may further damage the olfactory epithelium.44,45 Consequently, olfactory dysfunction in CCAD is likely more severe than that in other CRS subtypes, and formal olfactory testing should be considered as part of CRS assessment to facilitate earlier recognition and phenotypic differentiation of CCAD. Compared with other CRS subtypes, CCAD shows higher comorbidity with perennial AR but a lower asthma prevalence. 39
Endoscopy and imaging
Early recognition of CCAD relies on nasal endoscopy. The disease is typically confined to the middle and superior turbinates and the posterosuperior septum, presenting as polypoid edema or mucosal thickening, usually associated with a low Lund–Kennedy score. 42 CT often demonstrates the “black halo sign,” which is considered a characteristic feature of CCAD.1,46
Immunologic testing and histology
Work-up should include total serum IgE, sIgE, and SPT. In suspected LAR, additional tests such as NPT and tissue local IgE assays 34 should be considered. Biopsy usually shows moderate eosinophilia without mucus impaction. 1
Differential diagnosis
CCAD is distinguished from central structure–predominant entities—respiratory epithelial adenomatoid hamartoma (REAH), AFRS, and AERD—by integrating anatomic localization, inflammatory distribution, histopathology, and asthma comorbidity. 8
Diagnostic anchor and re-assessment
When endoscopy confirms that central compartment disease and allergic symptoms are present but systemic indices are negative—or when histology fails to meet T2 CRSwNP thresholds—the diagnosis of CCAD should remain grounded in endoscopic findings. If both systemic and local allergen tests are negative and histology indicates non-T2 inflammation, recent therapies (e.g. corticosteroids and antihistamines) should be reviewed, a washout should be introduced, and the case should be re-evaluated (Figure 3).

Diagnostic algorithm for CCAD. CCAD: central compartment allergic disease.
Treatment advances and therapeutic targets
Endotype-driven insights are steering CRS care toward precision, subtype-specific management. As a newly delineated CRS subtype, CCAD lacks fully standardized pathways and remains under active investigation. Reflecting CCAD’s LAR-driven, T2-skewed biology, emerging interventions are showing early promise in clinical studies and may shape future diagnostic and therapeutic algorithms.
Symptom control and pharmacotherapy
Baseline management of CCAD mirrors standard CRS care, prioritizing local anti-inflammatory therapy. Intranasal corticosteroid sprays and high-volume corticosteroid nasal irrigation are first-line measures 47 that reduce mucosal edema and dampen local inflammation. 48 Antihistamines (oral or intranasal) and leukotriene receptor antagonists (LTRAs) are appropriate for AR comorbidity. 49 Short oral corticosteroid (OCS) tapers may be considered for acute flares or preoperative optimization.
For early CCAD, allergen management and topical steroids are reasonable initial strategies 50 ; however, patients with central compartment obstruction or recurrent disease often require surgical intervention with FESS. 41 Postoperative regimens—particularly steroid nasal irrigation combined with brief adjunctive medication—are integral to ongoing care.
Overall, pharmacotherapy—including intranasal corticosteroids, antihistamines, and LTRAs—remains the first-line foundation in both early and postoperative settings. Given interindividual variability and disease severity, some moderate-to-severe patients achieve only partial relief with medication alone. Therefore, management should be individualized based on symptom burden and adherence while also considering cost and access.
Surgical intervention and postoperative management
When medical therapy is inadequate or the disease is prominently centered in the central compartment, FESS is the principal intervention. 41 Surgical objectives include removing polypoid disease, re-establishing central compartment airflow pathways, and improving the efficiency of topical drug delivery. Intraoperatively, key anatomic structures—particularly the MT—should be preserved whenever feasible to maintain native antigen-filtering and mucosal barrier functions. As the MT appears to be an important component of the nasal allergen filter and likely contributes to limiting allergen access to the lower airway, MT management should be as conservative as possible. When polypoid changes are present, MT sculpting can be performed to remove diseased tissue while preserving a substantial portion of the turbinate and its protective function. In contrast, total MT resection reduces the mucosal surface area available for allergen filtration and increases allergen exposure of the nasal septum. In the setting of central compartment disease, the extent of surgery should be tailored to the degree of septal involvement. If septal polypoid disease extends anteriorly to the leading edge of the MT, the affected tissue should be addressed by carefully trimming the anterior MT and sculpting the adjacent septal mucosa to allow postoperative topical steroid irrigations to more effectively reach the upper nasal septum. Excessive MT resection may, however, predispose to progression of polypoid changes involving the nasal septum. 40 Sinuses should only be opened if they are secondarily involved. 51 Long-term postoperative high-volume corticosteroid nasal irrigation can attenuate polyp recurrence and improve mucociliary clearance. 52
CT-based airflow simulations in eight postoperative FESS patients showed that as the surgical extent increased and middle-meatus anatomy was altered, nasal–sinus airflow coupling was enhanced, with ventilation notably improved in the maxillary and ethmoid sinuses. This structural–functional remodeling may substantially influence postoperative topical delivery, local inflammatory control, and mucosal repair mechanisms. 53 Clinically, outcomes are favorable: in a cohort of 51 CCAD patients, 84.3% (43/51) achieved a “well-controlled” or “partly controlled” disease state within 1 year, with SNOT-22 scores declining significantly and particularly marked improvements in olfaction and nasal obstruction.54,55 Comparative analyses across CRSwNP subtypes further indicate greater SNOT-22 gains in CCAD—especially in the olfaction and allergy domains—and no 1-year revisions, compared with approximately 10% requiring revision in control subtypes. 40 These data suggest that an appropriately tailored surgical approach yields enhanced responsiveness and superior long-term effectiveness in CCAD.
Allergen-specific immunotherapy (AIT)
ICAR:AR 2023 strongly recommends subcutaneous immunotherapy and sublingual immunotherapy (SLIT) for AR. 34 Given the close association between CCAD and inhalant aeroallergens, AIT represents a plausible disease-modifying intervention, particularly in patients with comorbid AR. 56 Evidence indicates that AIT significantly improves nasal symptoms and quality of life and may, through induction of immune tolerance, reduce reliance on corticosteroids. 57 In CCAD phenotypes predominantly driven by allergic inflammation, AIT may even be considered a first-line therapeutic option. 33
In a retrospective cohort, Meerwein et al. evaluated AIT in postoperative CCAD; among 86 house dust-mite–sensitized patients, 32 received SLIT. At 12-month follow-up, the rate of recurrence of MT edema was 15.6% in the AIT group—significantly lower than that in patients not receiving AIT. The AIT group also demonstrated superior mucosal recovery and treatment adherence. 58 These findings suggest that introducing AIT in the early postoperative period can delay or reduce the recurrence of allergic tissue remodeling and may improve long-term outcomes. Management in children remains challenging and should be individualized.
At present, the optimal candidacy and timing of AIT in CCAD (preoperative versus postoperative) are not well defined due to the paucity of high-quality prospective data. Large, multicenter studies are needed to establish evidence-based indications and sequencing for therapy.56,58
Biologic-targeted therapy
EPOS 2020 and EPOS/EUFOREA recommend considering biologics in the early management of CRSwNP.4,27 In the analysis by Mullol et al., biologic therapy achieved an olfactory improvement rate of approximately 60%. 59 In CCAD, MT tissue often shows upregulation of IL-4, IL-5, and IL-13 with eosinophilic infiltration, indicating a prototypical T2 inflammatory profile and suggesting suitability for biologic therapy; however, CCAD-specific guidance on biologic use is not yet standardized. 60
Among available agents, dupilumab, an interleukin-4 receptor-α (IL-4Rα) antagonist, blocks IL-4 and IL-13 signaling simultaneously, thereby inhibiting Th2 differentiation, eosinophil chemotaxis, and IgE production (Figure 4). By improving the local inflammatory milieu and reducing thymus and activation-regulated chemokine (TARC/CCL17), monocyte chemoattractant protein-4 (MCP-4/CCL13), and mucus secretion, dupilumab has been shown to significantly relieve nasal obstruction and hyposmia in CRSwNP. 61 The IL-5 axis is central to eosinophilic infiltration in CRSwNP and CCAD. Mepolizumab neutralizes IL-5, whereas benralizumab targets IL-5Rα and induces antibody-dependent cellular cytotoxicity to deplete eosinophils (Figure 5), thereby attenuating nasal mucosal inflammation and reducing polyp burden and recurrence. 62

IL-4/IL-13 signaling via the interleukin-4 receptor alpha chain (IL-4Rα): shared and cytokine-specific pathways and the ensuing immunomodulatory effects of IL-4Rα-targeting antagonists (e.g. dupilumab and stapokibart). IL: interleukin.
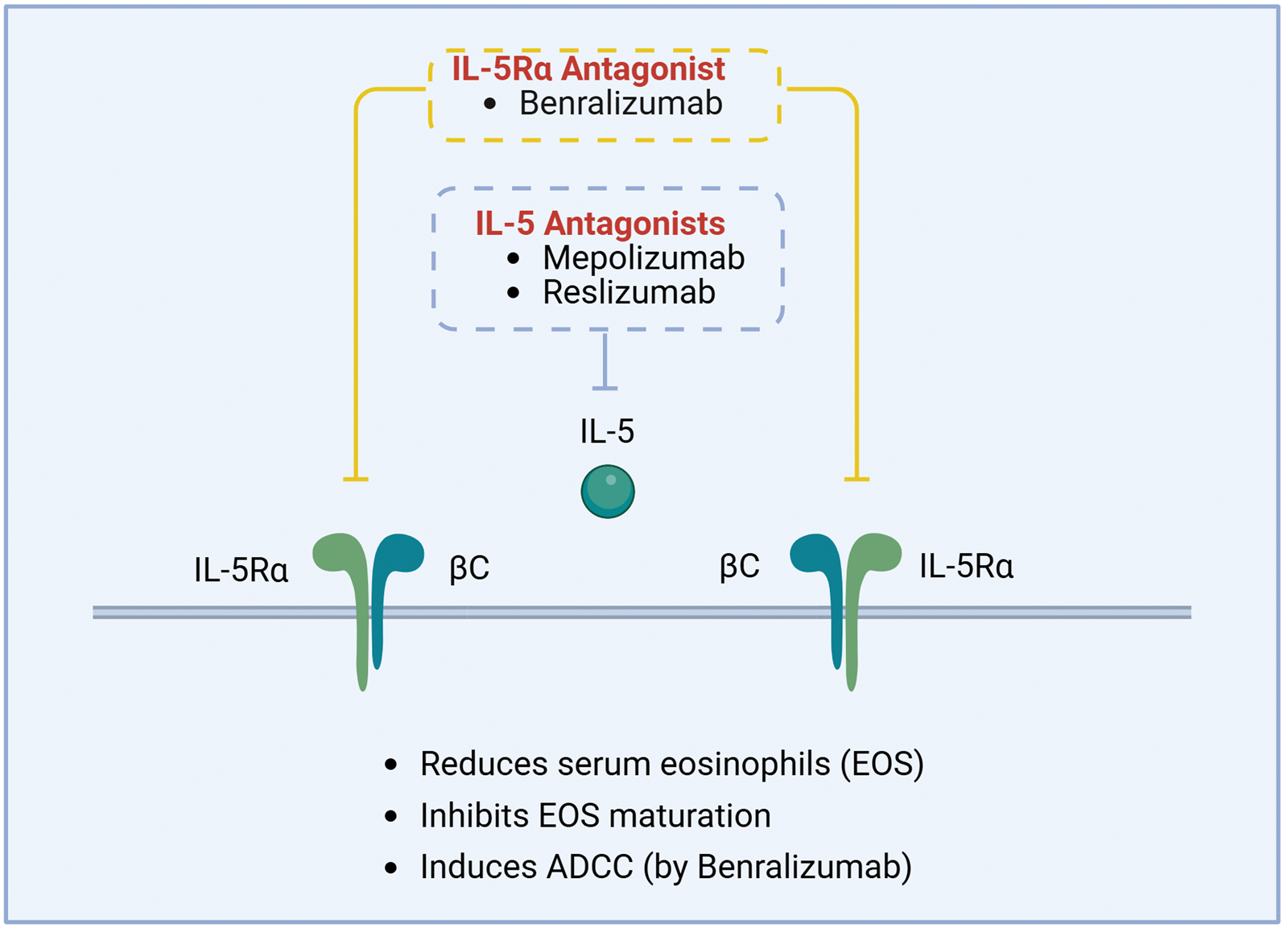
IL-5 signaling and therapeutic targets: anti-IL-5 and anti-IL-5Rα monoclonal antibodies attenuate eosinophil-driven inflammation by disrupting ligand–receptor signaling. IL-5 signals through a heterodimeric receptor comprising IL-5Rα and the common β-chain (βc), which is principally expressed on eosinophils and basophils, and promotes their activation, proliferation, and survival. IL: interleukin; IL-5Rα: interleukin-5 receptor alpha; βc: common beta chain.
Current evidence specific to CCAD remains limited. Given the localized T2 inflammation, anti-IgE strategies, such as omalizumab, may be considered for patients with markedly elevated IgE or for those unable to tolerate AIT. For early CCAD confined to the central compartment, whether biologic monotherapy can obviate surgery or is best used as an adjunct to surgery requires confirmation in large multicenter prospective studies.
Population differences and research frontiers
CCAD, as an emerging subtype within the spectrum of CRS phenotypes, exhibits considerable variation in clinical presentation and immunologic profile across ethnicities and geographic regions. Studies from the United States and Australia have reported allergen positivity rates of 73%–100% among CCAD patients.7,39,40,63 In contrast, studies from Asia have shown substantially lower allergen positivity rates and a lower prevalence of comorbid AR compared with Western studies.14,36–38
The predominant aeroallergens also differ by region. In a retrospective study from Taiwan, Lin et al. found that 90% of CCAD patients were sensitized to house dust mite. 38 Similarly, in Guangdong, house dust mite was the most frequent airborne allergen, affecting 71.43% of patients. 64 In Philadelphia, Tripathi et al. reported that the most common sensitizing aeroallergen was weeds (66.7%), followed by house dust mite (62.5%). 65
Currently, differences in allergy positivity rates and allergen profiles are primarily attributed to geographic and ethnic variation; however, heterogeneity in diagnostic criteria and allergen-testing methods likely plays a role. In most US-based studies, both serum sIgE panels and SPT with multiple aeroallergens are routinely performed, whereas many Asian cohorts use only a single modality—either SPT or serum sIgE. Given the close association of CCAD with local allergic responses, it remains unclear whether NPT, nasal sIgE, or BAT should be routinely incorporated into the diagnostic work-up. The establishment of standardized testing protocols would help harmonize case definitions and enhance the accuracy and methodological robustness of future CCAD research.
The prevalence of comorbid asthma also varies considerably across populations. In a US cohort, Brunner et al. found no significant difference in asthma prevalence between CCAD and other CRSwNP subtypes (34% vs. 22%, p = 0.21). 14 In contrast, a Korean pediatric study (<20 years) by Lee et al. reported a higher prevalence of asthma in CCAD patients than in non-CCAD patients (16.1% vs. 3.8%, p = 0.01). 1 Studies from Taiwan and New York further highlight regional differences in asthma comorbidity.36,39 These discrepancies may be attributed to multiple factors, including genetic background, environmental exposure patterns, regional allergen profiles, and variations in study design, particularly the selection of control groups.
Immune response mechanisms may also exhibit heterogeneity across populations. Unlike Western CRSwNP, which typically presents with a dominant T2 inflammatory pattern, Chinese CRSwNP often displays a mixed Th1/Th2/Th17 inflammatory endotype. 66 Although most scholars consider CCAD as an independent CRS subtype, 9 others suggest that it may represent an early manifestation of eosinophilic CRS (eCRS). 67 This hypothesis is supported by clinical features such as lower eosinophil counts, lower endoscopic and Lund–Mackay scores, and disease localization to the central compartment.
Future research should focus on multicenter, large-sample, and population-diverse prospective cohort studies to clarify the interrelationships among CCAD, AR, and CRSwNP. Simultaneously, we advocate that otolaryngologists worldwide adopt nasal endoscopy–based diagnostic criteria and further standardize allergen testing methods to guide precision diagnosis and individualized treatment strategies for CCAD.
Conclusion
CCAD is a localized CRS phenotype driven by central compartment airflow–particle deposition and sustained by the interaction between LAR and T2 inflammation.
Regarding diagnosis, nasal endoscopy should be initially performed to confirm central lesions confined to the MT, ST, and PSNS. Second, AR should be established based on clinical history or positive allergen testing. Third, T2 inflammation should be verified through histopathology.
Regarding treatment, the therapeutic backbone comprises topical corticosteroids combined with FESS. For refractory or high-risk cases, management should be individualized according to endotype and clinical indications, with consideration of AIT or T2-targeted biologics.
Regarding research agenda, multicenter prospective studies are needed to validate long-term outcomes in stratified populations and define the optimal timing and sequencing of AIT and biologics. Given the airflow-driven pathogenesis of CCAD, future studies should investigate the causal chain “postoperative airflow remodeling → local drug deposition → recurrence risk” to refine prevention and treatment strategies in CRS.
Footnotes
Acknowledgments
None.
Author contributions
Conceptualization: Shaokun Huang; Formal analysis: Chaofan Li and Chun Zhang; Writing—original draft: Shaokun Huang; Writing—review & editing: Juan Ye and Xuan Shan. All authors have read and approved the final manuscript.
Data availability statement
Data sharing is not applicable to this article, as no new datasets were generated or analyzed for this narrative review.
Declaration of conflicting interests
The authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Not applicable. This article is a narrative review based on previously published studies; no new research involving human participants or animals was conducted by the authors.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Figure originality statement
All figures in this article are original works created by the authors and have not been previously published.
