Abstract
Background
This study aimed to investigate the association between age at menarche and adult obesity among adults in the United States.
Methods
Data were obtained from the 1999–2018 National Health and Nutrition Examination Survey. A total of 19,170 female participants were included. Adult obesity was assessed using body mass index, weight-adjusted waist index, waist circumference, and waist-to-height ratio. To examine the association between age at menarche and adult obesity, weighted multivariable linear regression analyses were performed using continuous or categorical variables, and age at menarche was classified as early, normal, or late. Subgroup analyses stratified by age, race, and economic status were also conducted. Potential nonlinear associations between age at menarche and adult obesity were evaluated using restricted cubic spline analysis.
Results
Data from 19,170 females were analyzed. Multivariable linear regression analyses demonstrated a negative association between age at menarche and obesity indicators in adulthood. Restricted cubic spline analysis indicated that the associations between age at menarche and body mass index as well as waist-to-height ratio were nonlinear.
Conclusions
Among the 19,170 participants, age at menarche was negatively associated with the risk of obesity in adulthood. Increased attention should be given to the physical condition of girls with early menarche, and preventive interventions should be implemented as early as possible to reduce the risk of obesity.
Keywords
Background
Obesity is characterized by the accumulation of excess fat. This chronic condition can lead to health problems 1 and arises from an imbalance between energy intake and expenditure.2,3 Over the past few decades, the global prevalence of obesity has increased significantly. 4 The causes of obesity are multifactorial and include social, economic, genetic, and environmental factors.5,6 Obesity is linked to multiple noncommunicable diseases, including cardiovascular disease (CVD), 7 type 2 diabetes, musculoskeletal disorders, and endometrial, 8 breast, ovarian, prostate, liver, gallbladder, kidney, and colon cancers. 9 Consequently, addressing the obesity epidemic to prevent chronic illnesses is especially challenging. The primary measures used to assess obesity include body mass index (BMI), waist circumference (WC), weight-adjusted waist index (WWI), and waist-to-height ratio (WHtR).
The association between age at menarche and the risk of obesity among women remains controversial. A study reported that adult Chinese women were more likely to have a higher BMI and a larger WC when menarche occurred at a later versus earlier age. 10 In contrast, a meta-analysis of 30 studies demonstrated that pubertal timing was inversely correlated with adult BMI, and 10 cohort studies showed that early (<12 vs. ≥12 years of age) and late (≥15 vs. <15 years of age) menarche were correlated with higher and lower adult BMI, respectively. 11 A study conducted in China found no significant association between age at menarche and BMI or abdominal obesity. 12 Other studies have reported a potential association between obesity and earlier menarche as well as delayed menarche among young girls with low BMI and body fat content.13,14 To clarify the relationship between age at menarche and adult obesity, we examined these parameters using the National Health and Nutrition Examination Survey (NHANES) database.
Methods
All data for this study were obtained from the NHANES database (https://www.cdc.gov/nchs/nhanes/index.htm). Female participants from 1999 to 2018 (n = 28,608) were screened. Participants who were pregnant or breastfeeding (n = 1660), had missing information on age at menarche (n = 4413) or body measurements (n = 896), or were <8 or >20 years of age (n = 39) were excluded. Finally, 19,170 females were included in the analysis (Figure 1).

Flow chart of the participant selection process in the study.
Height and weight were measured by trained health technicians using standardized stadiometers and digital scales in accordance with NHANES protocols. WC was measured midway between the lowest rib and the iliac crest using a flexible steel tape. BMI, WWI, and WHtR were calculated as weight (kg)/height (m2), WC/√weight, and WC/height, respectively. All obesity indicators were analyzed as continuous variables in the regression and spline models and were additionally categorized for the description of sample characteristics.
Age at menarche
Based on responses to the question “How old were you when you had your first menstrual period,” age at menarche was categorized as early (<12 years), normal (12–13 years), or late (≥14 years). Early menarche is generally defined as menarche occurring before 12 years of age. 15 Age at menarche was also considered as a continuous variable in the sensitivity analysis.
Covariates
The covariates included age, race/ethnicity (Mexican American, non-Hispanic Black, non-Hispanic White, other Hispanic, and other race), marital status, educational level (<high school, high school, and >high school), economic status (indicated by poverty–income ratio (PIR)), smoking status (current, former, and nonsmoker), diabetes, hypertension, and CVD. Drinking status among females was categorized as never (<12 drinks/lifetime), former (≥12 drinks/year but none in the past year), mild (one drink/day), moderate (two drinks/day), or heavy (three drinks/day). 16 The diagnostic criteria for diabetes were as follows: (a) physician diagnosis of diabetes; (b) glycated hemoglobin >6.5%; (c) fasting blood glucose >7.0 mmol/L; (d) random blood glucose ≥11.1 mmol/L; (e) blood glucose ≥11.1 mmol/L during a 2-h glucose tolerance test; and (f) use of diabetes medications such as insulin. The diagnostic criteria for hypertension were as follows: (a) physician diagnosis of high blood pressure; (b) use of antihypertensive medications; and (c) abnormal blood pressure readings recorded ≥3 times. The presence of CVD was determined based on response to the question “Has a doctor or other health professional ever told you that you have congestive heart failure/coronary heart disease/cardiac disease/cardiopulmonary gastritis/stroke?” as well as menopausal status.
Statistical analysis
Continuous variables were expressed as mean (standard deviation), and categorical variables were expressed as frequency (percentage). Continuous variables were compared across the three menarche groups using survey-weighted analysis of variance (ANOVA), and categorical variables were compared using survey-adjusted χ² tests, incorporating strata, clusters, and sample weights. Linear regression models were used to assess the association between age at menarche and obesity. Model 1 was adjusted for age, race/ethnicity, marital status, economic status, and educational level. Model 2 was further adjusted for smoking status, drinking status, diabetes, hypertension, CVD, menopausal status, and the covariates included in Model 1. Restricted cubic spline (RCS) curves based on logistic regression were used to explore potential nonlinear relationships between age at menarche and obesity indices, with all analyses weighted. Statistical analyses were performed using R software version 4.1.3 (The R Foundation) and Empower software (X&Y Solutions, Inc., Boston, MA, USA). p-values <0.05 were considered statistically significant. The reporting of this study conforms to The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 17
Results
Our study included 19,170 females. In the analytic sample, age at menarche was categorized as early (<12 years), normal (12–13 years), or late (≥14 years), with weighted proportions of 21.2%, 50.4%, and 28.4%, respectively. The mean (SD) age at menarche was 13.3 (2.1) years. Sample characteristics stratified by early, normal, and late menarche are presented in Table 1, and the study selection criteria are shown in Figure 1. Increased age at menarche was significantly associated with increased height (standing height in cm measured at the Mobile Examination Center according to NHANES protocol; it represents adult height) (early: 161.093 ± 0.170 cm; normal: 162.304 ± 0.110 cm; and late: 162.503 ± 0.165 cm) and decreased weight (early: 80.709 ± 0.479 kg; normal: 76.387 ± 0.326 kg; and late: 72.785 ± 0.422 kg), WC (early: 99.804 ± 0.395 cm; normal: 96.328 ± 0.284 cm; and late: 93.569 ± 0.378 cm), BMI (early: 31.059 ± 0.179; normal: 28.970 ± 0.123; and late: 27.541 ± 0.152), WWI (early: 11.165 ± 0.020; normal: 11.071 ± 0.015; and late: 11.014 ± 0.020), and WHtR (early: 0.500 ± 0.003; normal: 0.470 ± 0.002; and late: 0.447 ± 0.002) (Table 1). Significant differences were also observed in race/ethnicity, smoking status, drinking status, diabetes, CVD, and menopausal status among the study participants.
Sample characteristics of the participants.

BMI: body mass index; WWI: weight-adjusted-waist index; WHtR: waist-height ratio; SD: standard deviation; CV: cardiovascular disease; PIR: poverty–income ratio.
Table 2 shows the association between age at menarche and current obesity status. We performed partially (Model 1) and fully (Model 2) adjusted multivariate linear regression analyses to account for potential confounders. In both models, later age at menarche was associated with lower obesity indices. In the fully adjusted model, BMI decreased by 0.661 for each unit increase in age at menarche (β = −0.661, 95% confidence interval (CI) = −0.749, −0.574). The decrease in BMI was greater by 1.703 in the normal versus early group (95% CI = −2.068, −1.339); in the late group, the decrease was greater by 3.079 (95% CI = −3.515, −2.643; p for trend <0.0001). For each unit increase in age at menarche, WWI decreased by 0.037 (β = −0.037, 95% CI = −0.046, −0.029). The decrease in WWI was greater by 0.083 in the normal versus early group (95% CI = −0.122, −0.045); in the late group, it was greater by 0.179 (95% CI = −0.222, −0.137; p for trend <0.0001). Similarly, for each unit increase in age at menarche, WHtR decreased by 0.010 (β = −0.010, 95% CI = −0.011, −0.008). The decrease in WHtR was greater by 0.024 in the normal versus early group (95% CI = −0.030, −0.018); in the late group, it was greater by 0.045 (95% CI = −0.052, −0.038; p for trend <0.0001).
The association between age at menarche and obesity.

BMI: body mass index; WHtR: waist-to-height ratio; WWI: weight-adjusted waist index; CI: confidence interval; Ref.: reference.
Crude model: No covariates were adjusted for.
Model 1: Adjusted for age, race, marital status, educational level, and poverty-to-income ratio.
Model 2: Adjusted for all covariates presented in Table 1.
Nonlinear relationship
In the RCS analysis, BMI and WHtR demonstrated nonlinear relationships with age at menarche (Figure 2). Specifically, obesity risk—as reflected by BMI and WHtR—increased more steeply among women who experienced menarche before approximately 13 years of age and then plateaued, indicating a higher risk of obesity in those experiencing early versus late menarche.

Survey-weighted restricted cubic spline models showing the association between age at menarche and adult adiposity. (a) BMI (p for trend <0.0001); (b) WHtR (p for trend <0.0001) and (c) WWI (p for trend = 0.3067). Shaded bands indicate survey-weighted 95% confidence intervals. BMI: body mass index; WHtR: waist-to-height ratio; WWI: weight-adjusted waist index.
Subgroup analysis
To further evaluate the relationship between age at menarche and obesity indicators, we performed subgroup analyses stratified by age, race/ethnicity, and economic status. The results indicated that these associations were inconsistent. BMI and age at menarche showed a significant (p < 0.05) and negative relationship across all PIR categories and in subgroups regardless of menopausal status (Table 3), suggesting that this relationship is influenced by both economic status and menopausal status. Similarly, the negative correlation between WHtR and age at menarche depended on age, economic status, and menopausal status, with the strongest association observed in the early group and among non-menopausal females.
Subgroup analysis of the association between age at menarche and obesity.
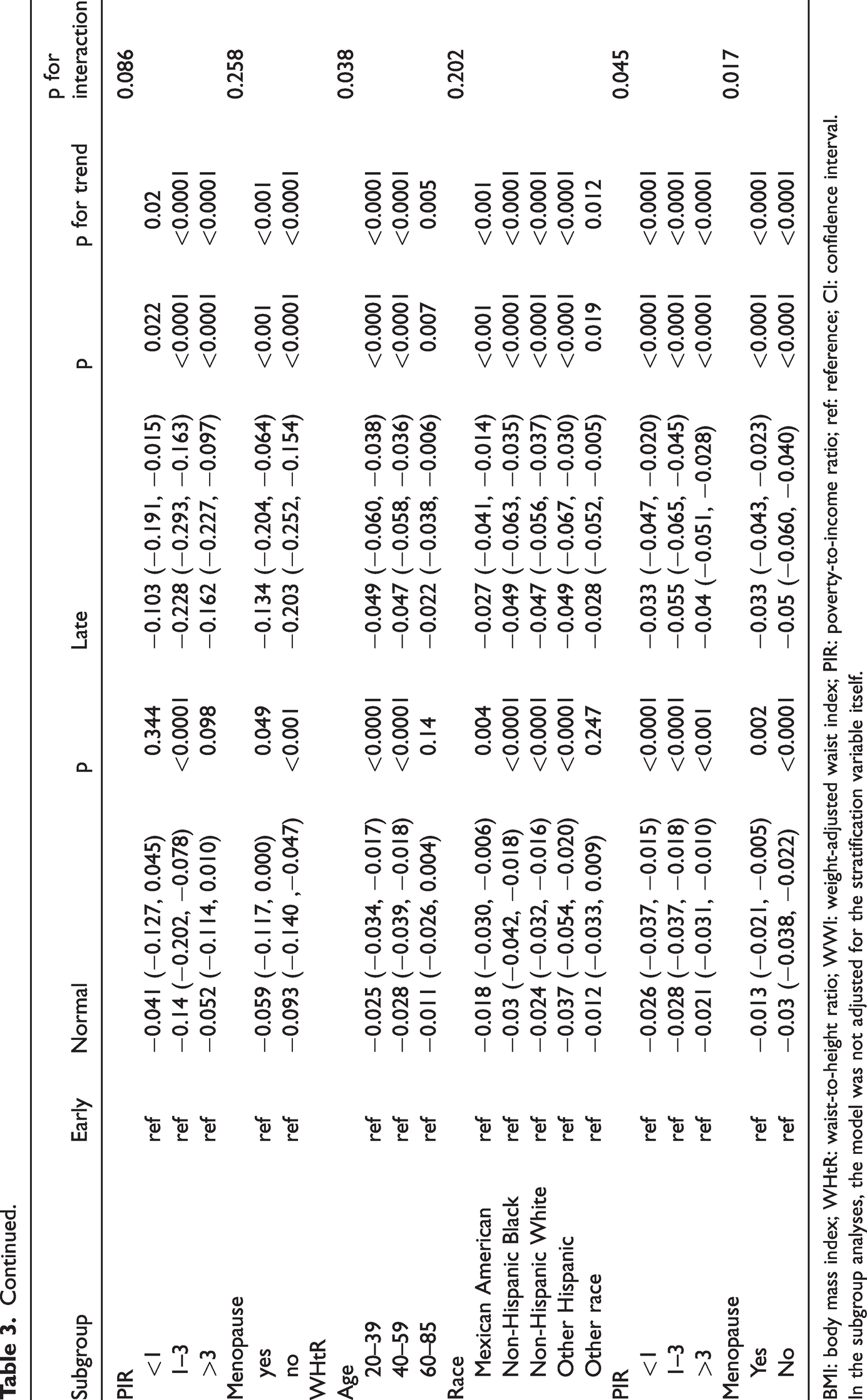
BMI: body mass index; WHtR: waist-to-height ratio; WWI: weight-adjusted waist index; PIR: poverty-to-income ratio; ref: reference; CI: confidence interval.
In the subgroup analyses, the model was not adjusted for the stratification variable itself.
Discussion
This study used the NHANES database (1999–2018) to investigate the association between age at menarche and adult obesity. A total of 19,170 American women were included. The results showed a negative correlation between age at menarche and BMI, WHtR, and WWI in adulthood. After adjusting for all covariates, BMI, WWI, and WHtR decreased by 0.661, 0.037, and 0.01, respectively, for each additional year of menarche. The association between age at menarche and both BMI and WWI was nonlinear. The observed difference of approximately 3 BMI units between early- and late-menarche groups is clinically meaningful, corresponding to approximately 8–9 kg of body weight in an average adult woman (165 cm). This magnitude of difference has been linked to elevated risks of metabolic syndrome and cardiovascular morbidity, highlighting the long-term health implications of early menarche.
A previous study on 17-β-estradiol and its relationship with age at menarche and adult obesity in premenopausal women revealed that early menarche was linked to greater WC, 18 as observed in our study. The subgroup analyses in our study revealed that the negative correlation between age at menarche and obesity indicators was strongest in the younger, non-menopausal population, indicating a link between obesity and estrogen levels. Factors that confound these association emerge in postmenopausal women and include shifts in metabolic cardiovascular risks that may affect obesity, consistent with findings from a previous Chinese study. 19
The timing of estrogen exposure may explain the negative correlation between age at menarche and obesity. Greater cumulative exposure to ovarian hormones in earlier versus later menarche may alter energy balance and metabolism, leading to fat accumulation.20,21 The nonlinear relationship of age at menarche with BMI and WHtR (as shown via RCS analysis) indicates that earlier menarche is more strongly associated with adult obesity than later menarche. The nonlinear association between age at menarche and obesity risk may be explained by biological and developmental factors. Early menarche reflects prolonged lifetime estrogen exposure, earlier adiposity rebound, and altered neuroendocrine regulation, all of which promote fat accumulation. After approximately 13 years, these effects may plateau as hormonal maturation and metabolic set points stabilize, resulting in the observed flattening of the risk curve. Preliminary research suggests that early overnutrition is associated with insulin sensitivity mediated by LIN28B through the mTOR pathway as well with the prevention of oxidative stress-related β-cell apoptosis via GABAB receptor signaling. 22 These events typically correlate with earlier onset of menstruation.
Potential effect modification was explored by examining interaction terms between age at menarche and age group, race/ethnicity, PIR, and menopausal status. Significant p-values for interaction indicated heterogeneity in the associations across subgroups. The subgroup analyses revealed similar inverse associations across most strata. p-values for interaction indicate whether these associations vary by age, race/ethnicity, socioeconomic status, or menopausal status. Modest heterogeneity was observed primarily by menopausal status and PIR.
Economic status was included in the subgroup analysis because obesity is prevalent in low-income populations in the US. Our results indicated that the obesity rate was lower in high-income populations than in low-income populations, even though menarche occurred earlier in the high-income population. To obtain more precise results, we used economic status rather than general living conditions as the metric in our analysis. Obesity prevention can be achieved through acquired control measures that reduce reliance on subjective evaluation.
Unlike previous reports on obesity and reproductive history, 23 our obesity indices included not only the BMI, which reflects central obesity, but also WWI. According to some studies, WWI is a more standardized measure of fat mass than WC and represents centripetal obesity.24,25 The RCS analysis in our study showed no nonlinear relationship between age at menarche and WWI.
This study has several limitations. First, because it was a cross-sectional study, we could not determine causality. Second, the age at menarche reported in the questionnaire may have been affected by recall bias. Third, confounding factors other than those listed in Table 1 were not examined; additional factors include contraceptive use, which can also influence estrogen levels.
Conclusion
Age at menarche is negatively correlated with the risk of obesity in adulthood. This correlation is particularly pronounced in non-menopausal females, and early menarche has a greater impact on obesity than late menarche. Therefore, greater attention should be paid to the physical health of girls who experience early menarche, and interventions should be implemented as early as possible to prevent obesity. However, because of the cross-sectional design, causal inference is not possible. Further research, including basic studies and cohort investigations, is needed to clarify the mechanisms underlying the association between age at menarche and obesity.
Footnotes
Acknowledgments
None.
Authors’ contributions
MJ wrote the main manuscript text.
Availability of data and materials
Consent for publication
Not applicable.
Declaration of conflicting interests
The authors declare that they have no competing interests.
Ethics approval and consent to participate
All participants provided written informed consent, and the study was approved by the NCHS Research Ethics Review Board.
Funding
None.
