Abstract
Objectives
Numerous studies have demonstrated the safety and efficacy of percutaneous MitraClip transcatheter interventional therapy in treating mitral regurgitation. This study reports the largest series of outcomes in Vietnamese patients who underwent the MitraClip procedure.
Methods and results
Between September 2014 and December 2024, 23 patients (20 males and 3 females; mean age, 66.3 ± 10.2 years) underwent the MitraClip procedure at five hospitals across Vietnam. Key findings included a high procedural success rate, with 91.3% of patients receiving successful implantations. Postprocedural complications were observed in 33.3% of patients. During the 30-day follow-up, two patients (9.5%) were readmitted; however, no mortality was reported. All patients achieved a mitral regurgitation grade of ≤2+, and 92.9% showed improvement to New York Heart Association Class I/II within 30 days postprocedure.
Conclusions
Multicenter data from hospitals across Vietnam indicate that the MitraClip procedure is effective in reducing mitral regurgitation and demonstrate favorable short-term safety outcomes.
Introduction
Mitral regurgitation (MR) is an abnormal retrograde flow of blood from the left ventricle through the mitral valve into the left atrium, most commonly occurring during ventricular systole. 1 This condition results from anatomical or functional abnormalities in any component of the mitral valve apparatus, including the left ventricle, papillary muscles, chordae tendineae, leaflets, and annulus.2,3 MR is the most common valvular disorder in the United States, affecting more than 2 million individuals, and its prevalence sharply increases with advanced age. 4
Although conventional surgical mitral valve repair (replacement if repair is unsuitable) remains the standard of care for patients with severe MR, only 20% with significant disease receive it. 3 Many patients remain untreated due to the unavailability of surgical treatment or concerns regarding surgical risks associated with full sternotomy and cardiopulmonary bypass. Particularly, patients of advanced age and those with comorbidities, impaired left ventricular function, or high surgical risk face additional limitations and a higher 30-day mortality rate following surgery.3,5,6
The MitraClip (Abbott Laboratories, Abbott Park) (Figure 1) has evolved as the first transvenous, transseptal, edge-to-edge repair system for the treatment of severe functional and degenerative MR. Its development has been driven by anticipated benefits for patients, particularly those ineligible for surgery, including comparable clinical outcomes with reduced perioperative mortality and morbidity, decreased pain, shorter hospital stays, earlier return to full activities, superior preservation of lung function, and improved cosmetic outcomes,

Fluoroscopic steps of the procedure. (a) Atrial septal puncture; (b) guide catheter advanced into the left atrium; (c) clip with open arms advanced into the left atrium toward the mitral valve; (d) MitraClip with opened arms advanced into the left ventricle; (e) leaflets are grasped and clip is closed; and (f) clip is released.
Research on the MitraClip for treating MR demonstrates promising outcomes, particularly from landmark trials such as EVEREST7,8 and COAPT. 9 Although EVEREST II suggested potential limitations in MR reduction compared with surgery, it highlighted the MitraClip’s safety advantages and positive effects on New York Heart Association (NYHA) functional class and quality of life. 8 COAPT further established the clinical value of the device by demonstrating reduced mortality, improved NYHA functional status, and lower hospitalization rates in patients with heart failure and moderate-to-severe secondary MR. Importantly, device-related complications were minimal. 9
In Vietnam, on 21 September 2014, the Vietnam Heart Institute, Bach Mai Hospital, in collaboration with Singaporean experts, successfully performed the first transcatheter mitral valve repair using the MitraClip system. As of December 2024, 30 transcatheter mitral valve repair procedures using the MitraClip device have been performed across six major hospitals and cardiovascular centers in Vietnam. However, published clinical evidence on the safety and effectiveness of the MitraClip in Vietnam remains limited. Therefore, we conducted this study with two objectives: (a) to characterize the clinical and paraclinical profiles and procedural indications of Vietnamese patients with MR undergoing the MitraClip procedure and (b) to evaluate the safety and short-term outcomes of this technique.
Patients and methods
Study design
This uncontrolled clinical study was conducted at five hospitals across Vietnam from September 2014 to December 2024. The study was performed in accordance with the Declaration of Helsinki (1975, as revised in 2024). Reporting of the study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 10
Patient selection
A convenience sampling approach was used due to the limited number of patients undergoing transcatheter mitral valve repair with the MitraClip device in Vietnam. All eligible MitraClip procedures with available data from participating centers during the study period were included to minimize selection bias. Figure 2 illustrates the clinical workflow of patients in our study. Table 1 details the inclusion and exclusion criteria. All criteria had to be met for participation in the clinical investigation. Patient information was processed anonymously, ensuring that the data could not be linked to individual patients, in compliance with national regulations on personal data protection.

Clinical workflow of patients.
Inclusion and exclusion criteria.

ASE: American Society of Echocardiography; MR: mitral regurgitation; NYHA: New York Heart Association; LVEF: left ventricular ejection fraction; LVESD: left ventricular end-systolic dimension; CK-MB: creatine kinase–muscle/brain isoenzyme; CABG: coronary artery bypass grafting; PCI: percutaneous coronary intervention; TAVR: transcatheter aortic valve replacement; COPD: chronic obstructive pulmonary disease; ACC/AHA: American College of Cardiology/American Heart Association; PAP: pulmonary artery pressure; PVR: pulmonary vascular resistance; mPCAP: mean pulmonary capillary pressure; CRT/CRT-D: cardiac resynchronization therapy/cardiac resynchronization therapy with defibrillator; TEE: transesophageal echocardiography.
Echocardiographic parameters
Echocardiography plays a pivotal role in the MitraClip procedure, serving as a diagnostic, guiding, monitoring, and evaluation tool throughout the process. Figure 3 presents echocardiographic images illustrating each step of transcatheter mitral valve repair using the MitraClip system. MR severity was graded using an integrative approach according to the American Society of Echocardiography guidelines. Severe MR was defined by a combination of various echocardiographic parameters, including a central and large color flow jet (covering >40% of the left atrium (LA)) or a wall-impinging jet of any size swirling in the LA, large flow convergence, systolic flow reversal in pulmonary veins, vena contracta width >0.7 cm, effective regurgitant orifice area ≥0.4 cm2, regurgitant fraction ≥50%, and regurgitant volume ≥60 mL. Moderate-to-severe MR included a vena contracta ≥0.3 cm, effective regurgitant orifice area 0.30–0.39 cm2, regurgitant fraction 40%–49%, and regurgitant volume 45–59 mL. Postprocedural MR severity was assessed according to published recommendations. For these reasons, echocardiographic parameters were central to this study, serving as the primary method to evaluate MitraClip outcomes and grade the severity of MR.
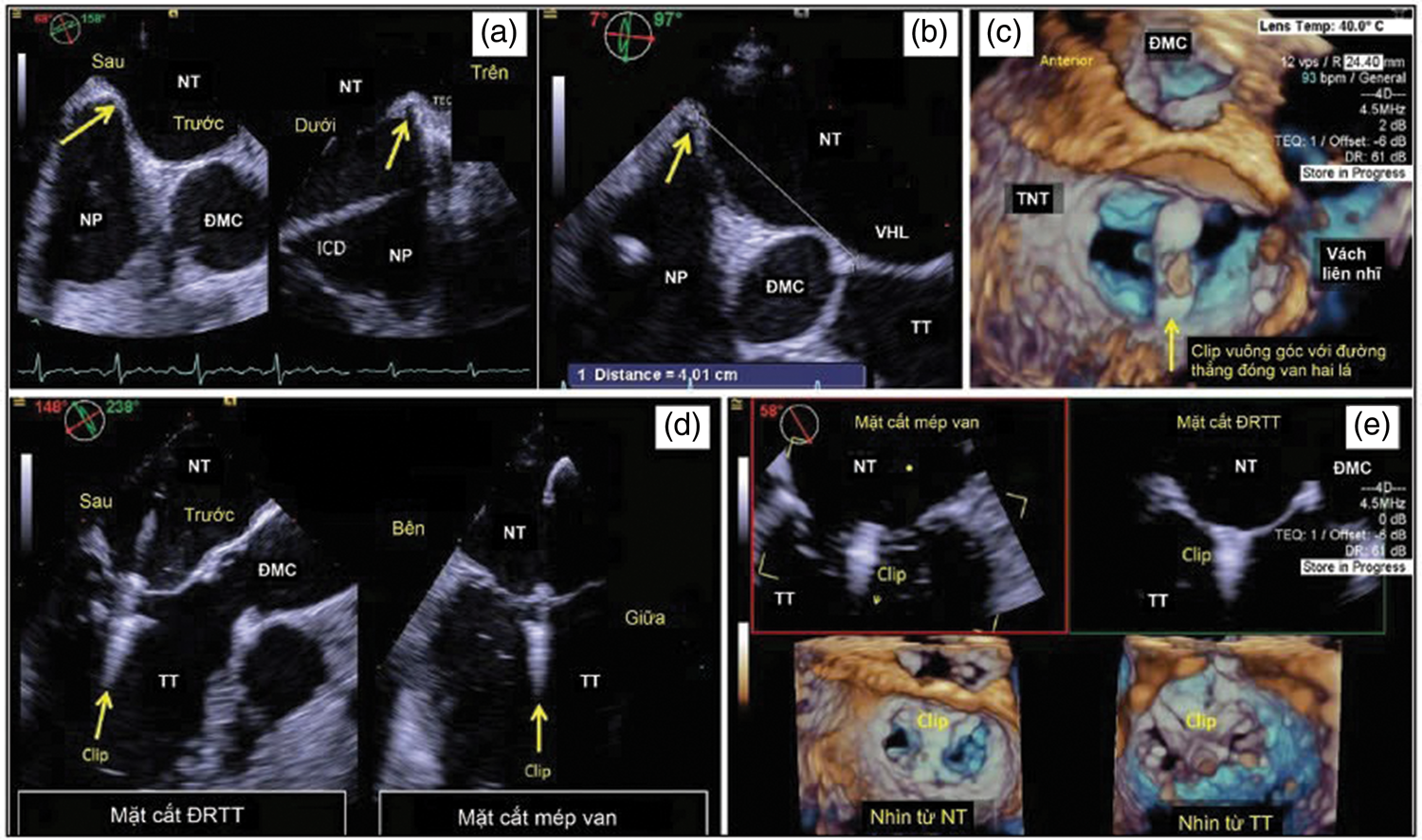
Echocardiographic images of each step in transcatheter mitral valve repair using the MitraClip system. TEE guided septal puncture above and posterior to the interatrial septum (a, b). (a) Biplane 3 D-TEE images showing septal puncture site; (b) distance from the septal puncture site to the mitral valve plane; (c) 3 D-TEE guides clip orientation perpendicular to a line through the mitral valve closure area viewed from the atrium; (d) biplane 3 D-TEE guides leaflet clamping viewed from the ventricular aspect of the valve; and (e) the final result shows a clip released in the closed area of A2-P2. TEE: transesophageal echocardiography; 3D-TEE: three-dimensional transesophageal echocardiography.
Study outcomes
Acute procedural success was defined as the successful implantation of one or more clips with immediate reduction of MR to ≤2+. The primary efficacy outcome of the MitraClip procedure was defined as a reduction in heart failure-related readmissions over 2 years. Secondary efficacy outcomes included improvements in quality of life, exercise capacity, and NYHA functional class at 1 year. Safety outcomes were assessed for short-term (30 days) and mid-term (12 months) complications. Major adverse events included stroke, myocardial infarction, bleeding requiring transfusion of more than 1 unit of blood, septicemia, reoperation for a failed mitral valve procedure, nonelective cardiac surgery for adverse events, renal failure, gastrointestinal complications requiring surgery, ventilation for >48 h, and new-onset atrial fibrillation.
Statistical analysis
The clinical characteristics of the study population were analyzed using descriptive statistics. Continuous data were presented as mean ± SD, and categorical data were presented as frequency and percentage. The χ2 test was used to compare categorical variables. For continuous variables, between-group comparisons were performed using the independent t-test. Echocardiographic data pre- and post-clip deployment were compared using the paired t-test or the Wilcoxon signed-rank test, as appropriate. A two-tailed p-value of <0.05 was considered statistically significant. All analyses were conducted using IBM Statistical Package for the Social Sciences (IBM SPSS) version 25 for Windows.
Results
Baseline demographic characteristics
Over 10 years, from September 2014 to December 2024, 23 patients underwent the MitraClip procedure at five hospitals across Vietnam (one center declined to share patient-level data for inclusion in the analysis). Table 2 presents the baseline clinical characteristics of patients included in our study. Of these patients, 21 (91.3%) had one or more clips successfully deployed. The mean age of these patients was 66.8
Baseline clinical characteristics of patients.

BMI: body mass index; BSA: body surface area; NYHA: New York Heart Association; MR: mitral regurgitation; STS: Society of Thoracic Surgeons; IQR: interquartile range.
Moreover, 85.7% (n = 18) of patients presented with dyspnea, whereas 14.3% (n = 3) experienced acute pulmonary edema. Regarding clinical symptoms, 66.7% of patients reported dyspnea on exertion and 38.1% experienced dyspnea at rest. Additionally, lower extremity edema was observed in 23.8% (n = 5) of the study population.
The majority of study participants (95.2%) presented with severe MR at baseline, categorized as grade 4+, and had multiple comorbidities, including hypertension (42.9%), chronic kidney disease (28.6%), dyslipidemia (23.8%), atrial fibrillation (23.8%), type 2 diabetes mellitus (9.5%), and chronic lung disease (9.5%). Additionally, 23.8% of patients had a history of percutaneous coronary intervention, and 4.8% had a history of coronary artery bypass grafting. Degenerative MR was present in 81% of patients (n = 17), whereas functional MR was observed in 19% (n = 4). Twelve patients (57.1%) were classified as NYHA functional Class III/IV, whereas nine (42.9%) were in Class II. The median Society of Thoracic Surgeons (STS) score was 1.6 (interquartile range (IQR), 7.8%), with 61.9% of patients classified as low risk. Remaining baseline characteristics are summarized in Table 3.
Complication during MitraClip procedure.

IQR: interquartile range; MR: mitral regurgitation.
Safety of the MitraClip procedure
Table 3 presents complications during the MitraClip procedure. The median procedure duration was 80.0 min (IQR, 96.5). One-clip implantation was performed in 5 patients (23.8%), whereas 15 patients (71.4%) received two clips. Only one patient (4.8%) had three clips implanted. Notably, a single patient (4.8%) experienced a procedural complication, which was a vascular disruption. No mortalities were reported.
No mortality occurred after the MitraClip procedure or during the 30-day follow-up. However, 33.3% of patients (n = 7) experienced postprocedural complications, and 9.5% (n = 2) were readmitted due to heart failure within 30 days. Postprocedure complications included major or severe hemorrhage (n = 3), hematoma at the puncture site (n = 2), and atrial fibrillation (n = 2) (Table 4).
Complications after MitraClip procedure.

AV: atrioventricular.
Effectiveness of the MitraClip procedure
MR severity significantly improved 30 days postprocedure (p < 0.001). At baseline, majority of patients (95.2%) presented with severe MR, classified as grade 4+. Following the intervention, all patients (n = 21) showed improvement, with 90.5% classified as having grade 2+ or 1+ MR (Figure 4(a)).
(a) Comparison of mitral regurgitation grade at baseline and 30 days post-MitraClip procedure and (b) comparison of NYHA functional class status at baseline, immediately after MitraClip procedure, and 30 days postprocedure. NYHA: New York Heart Association.
Similarly, NYHA functional class demonstrated significant improvement at 30 days postprocedure (p < 0.001). At baseline, 57.1% of patients (n = 12) were classified as NYHA Class III/IV. Following the intervention, all participants (n = 21) exhibited improvement to NYHA Class I/II (Figure 4(b)).
Paraclinical assessments, including hematology, biochemistry, and electrocardiogram (ECG), revealed no significant changes postprocedure or during 30-day follow-up period (Table 5).
Changes in paraclinical results.

Hb: hemoglobin; NT-proBNP: N-terminal pro–B-type natriuretic peptide; GFR: glomerular filtration rate; HAS-BLED: hypertension, abnormal renal/liver function, stroke, bleeding history or predisposition, labile INR, elderly, drugs/alcohol concomitantly; N/A: not applicable; AV: atrioventricular; ECG: electrocardiogram.
The mean values for left ventricular ejection fraction (LVEF), left ventricular end-diastolic diameter (LVEDD), left ventricular end-systolic diameter (LVESD), left ventricular end-diastolic volume (LVEDV), and pulmonary artery systolic pressure (PASP) were 56.5% ± 17.0%, 59.2 ± 7.7 mm, 40.4 ± 10.1 mm, 172.8 ± 51.3 mL, and 43.1 ± 18.0 mmHg, respectively, whereas the median left ventricular end-systolic volume (LVESV) was 65 (IQR, 64.7). At 30-day follow-up, significant reductions were observed in LVEDD, LVESD, LVEDV, and PASP compared with baseline, indicating improvements in ventricular dimensions and reduced pulmonary pressure. No significant changes were detected in aortic valve regurgitation or other measured parameters (Table 6).
Change in echocardiographic parameters.

IQR: interquartile range; LVEF: left ventricular ejection fraction; LVEDD: left ventricle end-diastolic dimension; LVESD: left ventricle end-systolic dimension; LVEDV: left ventricular end‐diastolic volume; LVESV: left ventricle end-systolic volume; AVR: aortic valve regurgitation; PASP: pulmonary artery systolic pressure; N/A: not applicable.
Bold text indicates p-values less than 0.05.
Discussion
This study provides a snapshot of the current population of patients undergoing transcatheter treatment for MR in Vietnam and represents the largest report of MitraClip procedures in the country up to December 2024. The mean age of study participants was 66.8 ± 10.4 years, with the majority being male (85.7%). Most patients had degenerative MR (81.0%) and multiple comorbidities, including hypertension (42.9%), chronic kidney disease (28.6%), dyslipidemia (23.8%), atrial fibrillation (23.8%), type 2 diabetes mellitus (9.5%), and chronic lung disease (9.5%). Furthermore, 23.8% of patients had previously undergone percutaneous coronary intervention, and 4.8% had a history of coronary artery bypass grafting. Most patients were highly symptomatic, with 95.2% presenting with grade 4+ MR and 57.1% classified as NYHA functional Class III or IV at baseline, and had mostly preserved LVEF (56.5% ± 17.0%).
When compared to the population in the EVEREST II trial, 8 our cohort exhibited similarities in variables such as mean age (67.3 ± 12.8 years), predominantly preserved LVEF (60.0% ± 10.1%), and history of percutaneous coronary intervention (24%). However, certain characteristics in our population were higher than those in EVEREST II, including the proportion of patients with grade 4+ MR (25.0%), NYHA functional Class III or IV (51.1%), and degenerative etiology (73.4%). By contrast, our population differed from the cohort treated in the MitraClip Asia–Pacific Registry (MARS) study. 11 The MARS population was slightly older (71.4 ± 11.9 years), had a lower prevalence of grade 4+ MR (81%), and included a higher proportion of patients in NYHA functional Class III or IV (68.3%). Moreover, the MARS cohort demonstrated a higher prevalence of comorbidities, including diabetes mellitus (28.9%), hypertension (64.1%), atrial fibrillation (45.1%), and a lower prevalence of degenerative MR (45.8%) and LVEF (47% ± 17%).
As reported in previous studies on the MitraClip procedure,8,11–13 the therapy is associated with improvements in MR severity, echocardiographic parameters, and functional status. Our key findings are as follows. First, percutaneous edge-to-edge mitral valve repair was successfully performed in 21 cases, achieving a procedural success rate of 91.3% and an in-hospital complication rate of 0%. Second, 33.3% of patients experienced postprocedural complications, with two cases (9.5%) requiring hospital readmission due to heart failure; no mortality was reported within 30 days. Third, the MitraClip procedure resulted in significant improvements in MR grade, echocardiographic parameters, and functional status, with 90.5% of patients achieving ≤2+ MR and 100% demonstrating improvement to NYHA Class I/II at 30 days postprocedure.
Comparative analysis with other studies revealed a procedural success rate of 91.3%, slightly lower than that reported in the MARS study 11 (93.7%) and the German Transcatheter Mitral Valve Interventions (TRAMI) study 13 (94%). The larger and more established ACCESS-EU study 12 reported an even higher success rate of 99.6%. Regarding complication rates, the figure in our study (33.3%) was higher than those reported in MARS 11 (12.7%) and EVEREST II 8 (15%). These differences may be influenced by the relatively small sample size and limited infrastructure in Vietnamese hospitals. Moreover, our study observed a lower 30-day mortality rate compared with MARS 11 (5.6%) and EVEREST II 8 (6%). This outcome may be attributable to the majority of our patients (61.9%) having low STS scores (median (IQR), 1.6% (7.8%)), in contrast to the higher STS scores reported in the MARS study 11 (7.4% ± 8.1%).
Furthermore, the MitraClip procedure was associated with significant improvements in MR grade and NYHA functional class, as demonstrated in Figure 4. These findings are consistent with observed improvements in echocardiographic parameters, including LVEDD, LVESD, LVEDV, and PASP, in line with results reported in other studies.7,8,11,12 These enhancements in echocardiographic parameters are likely attributable to the reduction in volume overload caused by decreased MR severity and the potential for positive early remodeling of the heart chambers.
Limitations
Although this study represents a pivotal investigation into the efficacy of the MitraClip procedure within the Vietnamese population, several limitations should be acknowledged. First, the sample size was relatively small, and longer-term follow-up is necessary to evaluate the durability of these improvements and identify potential late complications. Second, the absence of a control group limits the ability to definitively establish a causal relationship between the MitraClip procedure and the observed outcomes. Finally, data from one center could not be included in the analysis due to institutional restrictions on data sharing, which may have introduced a degree of selection bias.
Future directions
Future research should include larger, randomized controlled trials to compare MitraClip with other treatment modalities and assess its long-term efficacy and safety. Additionally, investigating patient selection criteria can help optimize outcomes and identify those who would benefit most from this procedure.
Conclusion
This study provides valuable real-world data on the characteristics of Vietnamese patients with MR undergoing the MitraClip procedure. The findings demonstrate the procedure’s efficacy and safety in this population, contributing to the growing evidence base supporting the MitraClip procedure as a viable therapeutic option.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605251396350 - Supplemental material for Ten-year experience with transcatheter mitral valve repair using the MitraClip system in Vietnam: A multicenter observational study
Supplemental material, sj-pdf-1-imr-10.1177_03000605251396350 for Ten-year experience with transcatheter mitral valve repair using the MitraClip system in Vietnam: A multicenter observational study by Nhat Minh Pham, Quang Ngoc Nguyen, Manh Hung Pham, Hoai Nguyen Thi Thu, Huynh Linh Dinh, Tuan Dat Phan, Lan Anh Nguyen and Van Thang Le in Journal of International Medical Research
Footnotes
Acknowledgments
Artificial intelligence tools were used for language improvement.
Author contribution statement
Conception and design: NMP, QNN
Collection and assembly of data: NMP, QNN
Data analysis and interpretation: H NMP, QNN
Manuscript writing: All authors
Final approval of manuscript: All authors
Data availability statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
The authors have no conflicts of interest to report.
Ethics statement
This retrospective study was approved by the Institutional Ethics Review Board of Hanoi Medical University, Vietnam (decision No. 655/GCN-HĐĐĐ NCYSH-ĐHYHN, dated 4 July 2022), which granted a waiver of written informed consent due to the retrospective design and de-identification of data. This study complied with the Declaration of Helsinki (1975, as revised in 2024).
Funding
No funding was obtained for this study.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
