Abstract
The surgical management of mitral valve disease is undergoing rapid development, with various treatment options available. Increasing evidence has emerged on the optimal strategy for the management of secondary mitral regurgitation, a condition characterised by ventricular pathology. This review discusses surgical treatments for secondary mitral regurgitation. Currently, there is no consensus on the optimal strategy for the treatment of patients with secondary mitral regurgitation. Management of this condition requires a dedicated heart team to ensure the most favourable outcome by tailoring interventions to individual patients. The aim of this narrative review is to compare available interventions for secondary mitral regurgitation.
Keywords
Introduction
Recent studies have demonstrated increasing adoption of new transcatheter heart valve approaches, particularly following randomised controlled trials (RCTs) that compared these less invasive techniques with established surgical methods.1–6 These RCTs have contributed to a marked shift in treatment guidelines for secondary mitral regurgitation (SMR). The European Society of Cardiology (ESC) 7 and the American College of Cardiology/American Heart Association (ACC/AHA) 8 guidelines now diverge from earlier recommendations. Despite efforts to identify the most suitable patients for transcatheter edge-to-edge repair (TEER), international guidelines need to clarify the specific role of each intervention in low-risk patients.7–9 It is hypothesised that an erosion of international guidelines will result in a shift towards younger patients with a lower-risk profile. This is considered a plausible outcome of this transition from high-risk patients to those with intermediate- or low-risk characteristics, despite the existence of substantial evidence supporting the utilisation of TEER. In these patients, recommendations for the use of MitraClip must be substantiated by more robust scientific data.
In the COAPT (Cardiovascular Outcomes Assessment of the MitraClip Percutaneous Therapy for Heart Failure Patients With Functional Mitral Regurgitation) trial, patients with heart failure (HF) and severe SMR who remained symptomatic despite maximal doses of medical therapy and other indicated treatments experienced significantly better outcomes with TEER. TEER was associated with reduced hospitalisation rates for HF and lower all-cause mortality at 5-year follow-up. 10 This benefit was observed even though protocol-permitted crossover therapy for severe mitral regurgitation (MR) was administered to control patients after 2 years. TEER showed favourable results across all predefined subgroups, with consistent reductions in mortality and hospitalisation rates regardless of patient characteristics. Factors considered included age, sex, severity of MR, left ventricular (LV) function and volume, aetiology of cardiomyopathy and surgical risk. Notably, the symptoms exhibited by patients in terms of New York Heart Association (NYHA) functional class also improved at 5 years following TEER for MR. 10
The safety of MitraClip therapy was demonstrated by the fact that only four patients (1.4%) in the device group experienced device-specific complications within 5 years, all occurring within 30 days of the procedure. Furthermore, there was a reduction in the number of unplanned mitral valve (MV) surgeries and percutaneous interventions over the course of follow-up in the device cohort compared with that in controls. Despite the favourable risk–benefit profile of TEER, adverse outcomes still occurred. At 5 years, 73.6% of patients in the device cohort had died or been hospitalised for HF, compared with 91.5% in the control group. 10
These findings underscore the need for therapeutic interventions that are designed to address the underlying LV dysfunction in this high-risk population.
Methods
This review was conducted in accordance with the Scale for the Assessment of Narrative Review Articles (SANRA). 11
A thorough review of the extant literature was conducted in accordance with a pre-designed protocol established before data collection. The objective of this study was to identify the existing empirical research on SMR, focusing on the following areas: pathogenesis, manifestations, imaging, treatment, surgery and devices. The scope was expanded to include echocardiographic findings related to TEER and comparative analyses with standard surgical procedures. A comprehensive search was conducted in Embase, Ovid Medline and PubMed for studies published between January 2004 and December 2024, using the following terms: ‘secondary mitral regurgitation’, ‘secondary ischemic mitral regurgitation’ and ‘functional mitral regurgitation’. These were combined with ‘mitral valve replacement’, ‘mitral valve repair’, ‘restrictive mitral annuloplasty’ and ‘transcatheter edge-to-edge repair’. Although most publications were from the past two decades, older, highly cited studies were also included. Reference lists of the identified articles were screened to determine additional relevant sources, and review articles were cited where appropriate to provide further context.
Results
Knowing the pathophysiology to drive the best treatment
The management of secondary or functional MR presents considerable clinical and procedural challenges. It is asserted that this manifestation of secondary MR is predominantly associated with the progression of a pathology involving the left ventricle, as opposed to a pathology involving the MV. The principal objective of therapeutic intervention is to address the inherent LV dysfunction. Judicious use of neurohormonal therapy in combination with resynchronisation devices has been demonstrated to promote reversible remodelling.12,13 Conversely, studies have demonstrated that a decrease in LV volume has the potential to ameliorate the severity of MR in a substantial number of patients.14,15 Nonetheless, endeavours to perform surgical interventions on the MV have not yielded favourable results. Two RCTs conducted in patients with moderate-to-severe MR due to ischaemic disease, with relatively preserved LV function (left ventricular ejection fraction (LVEF) of 40%), revealed that MV repair with undersized mitral annuloplasty (UMA) or chordal-sparing MV replacement did not result in the prespecified enhancement in cardiac geometry or long-term clinical outcomes.16,17 In addition, patients undergoing UMA experienced higher recurrence rates of MR compared with those treated using alternative approaches.16–19 Consequently, instead of relying solely on direct mechanical interventions to mitigate MR, current management strategies emphasise restoring LV structure and function through pharmacological and device-based therapies.3,4,10
Despite the consistency of the reported outcomes, it can be hypothesised that patients with SMR are classified into two distinct clinical subgroups. The first subgroup may comprise patients in whom MR arises primarily from substantial geometric alterations in MV function caused by marked dilatation of the LV chamber. The second subgroup comprises patients in whom LV pathology disproportionately affects the ventricular muscle components that stabilise the MV leaflets. In this latter subgroup, MR enhancement may not be adequately achieved by pharmaceutical treatments targeting the reduction of LV dimensions. Consequently, mechanical interventions that directly reduce MR—such as TEER—may provide greater benefit. These observations have prompted ongoing debate among cardiologists centred on two key questions: (a) whether MV interventions remain valuable when SMR is fundamentally a ventricular disease and (b) how to accurately define the subgroup of patients most likely to benefit from MV repair.
Over the past decade, technological advances have greatly improved our understanding of MR, enabling quantitative evaluation of regurgitant volume and effective regurgitant orifice area (EROA). 20 The assessment of factors contributing to MR is indeed feasible, encompassing leaflet geometry and the geometric or functional distortions induced by LV enlargement. 21 RCTs of MV repair in patients with systolic dysfunction have provided critical information on the range of responses to therapeutic interventions aimed at reducing EROA in different groups of individuals.2,5,6,22 Emerging evidence also suggests that patients with SMR may benefit from transcatheter procedures directed at the MV. For clinicians evaluating SMR patients in the pre- and perioperative periods, a new conceptual framework has emerged, distinguishing those with SMR attributable primarily to LV disease. According to recent studies, patient selection for specialised interventions must be based on a solid physiological foundation. 3 The need for this redefined evaluation arises from the conflicting results of previous RCTs. Rigorous validation of this framework will require a thorough analysis of existing databases, supported by further research.
A new step in characterising SMR for optimal management
Traditionally, the distinction between primary MR and SMR has been based either on identifying the specific structural defect responsible or on the identifying the sequence of events triggering the disease. Clinicians have also attempted to quantify MR using echocardiography or by inferring its impact on the patient’s clinical course and prognosis. Both approaches, however, have notable limitations.
Problems with identification of MR by anatomy or timing
Treatment options may include repair or replacement if the valve is identified as structurally compromised. Although it seems reasonable to assume that precise anatomical knowledge would guide clinical decision-making, reliance on structural analysis alone can be misleading. For example, acute ischaemic papillary muscle (PM) rupture is not caused by intrinsic mitral leaflet pathology but by local myocardial damage. In such cases, surgical replacement or repair of the MV can lead to dramatic clinical improvement. 23 Thus, a rigid classification based solely on anatomical site is insufficient for selecting the best management strategy, particularly when MV replacement improves outcomes in the context of LV dysfunction.
To overcome the theoretical and conceptual issues raised by acute ischaemic PM rupture, the classification of patients with MR may be based on the temporal sequence of events rather than on anatomy alone. If abnormal regurgitant flow is the initial event leading to the patient’s clinical presentation, the pathophysiology of MR can be attributed to valvular rather than ventricular causes. Conversely, if LV dysfunction with marked chamber dilation precedes the onset of MR, the regurgitant injury may not be considered the primary driver of the clinical course. In patients with acute PM rupture, MR is regarded as the main cause, even though the disease process never involved the leaflets or chordae tendineae.
However, similar to anatomy-based classification, a framework centred on the time course of events has important limitations. Many patients present with both severe MR and advanced LV dysfunction at the same time, making it difficult to determine the exact progression of the course of events. More importantly, LV abnormalities that cause chamber dilation of the heart may also produce disproportionate damage to the muscle that maintains functional coaptation of the MV. These patients may experience significant MR after ventricular insult and early remodelling but before a substantial increase in LV size. Mitral leaflet coaptation may be compromised by this alone.
Difficulties in assessing the severity of MR
Quantification of MR volume is of paramount importance, regardless of whether classification is based on anatomical features or temporal sequence of events. Mild MR does not require specific treatment, whereas severe MR can substantially reduce stroke volume and worsen outcomes, particularly when reduced systolic function is present.
A regurgitant fraction of ≥50%, i.e. at least half of the total stroke volume flowing back into the left atrium instead of forward into the aorta, is widely accepted as indicative of severe MR.24,25 Based on this, careful consideration must be given to the parameters of two-dimensional Doppler echocardiography that can reliably identify patients with a regurgitant fraction above this threshold.
Doppler echocardiography is a frequently employed technique for the quantification of MR, with EROA typically determined using the proximal isovelocity surface area (PISA) method. 25 According to the American Society of Echocardiography, 24 an EROA of 0.4 cm2 is typically indicative of a regurgitant fraction of ≥50%. However, the estimation of EROA can be a complex process because of multiple interrelated factors. The PISA method assumes a circular orifice through a flat surface; however, in SMR, the regurgitant orifice is often crescent-shaped, the jets are eccentric, the proximal convergence area is asymmetric and the regurgitant flow exhibits fluctuations during systole, typically following a biphasic pattern. 26
EROA assessment is further complicated in mild MR. It is imperative to acknowledge the potential divergence between EROA measurements ascertained through two-dimensional and three-dimensional echocardiography methodologies. It has been demonstrated that even minor variations in the observer’s measurement of the PISA radius21,27,28 can lead to substantial alterations in the calculated EROA. The quantitative assessment of MR severity can result in substantial interobserver disagreement, even among experienced operators. 29
Due to the ambiguities inherent within the guidelines, between 2012 and 2014, it was recommended26,30 that the most efficacious method for determining patients suffering from severe MR was not to accurately quantify the magnitude of the regurgitant jet visualised on imaging. Instead, it was proposed that severity should be judged by the potential impact of the lesion on clinical outcomes. In cases exhibiting substantial MR, it was reasonable to conclude that the regurgitant defect must be haemodynamically significant even when EROA values suggested only mild-to-moderate MR. Clinical studies showed that patients with an EROA ranging from 0.2 to 0.4 cm2 demonstrated similarly poor clinical follow-up outcomes.31,32 Consequently, the European 30 and US 26 guidelines were revised in 2012 and 2014, respectively, with the threshold for detecting severe SMR being lowered from the previous EROA value of 0.4 cm2 to a new cut-off value of 0.2 cm2. This new threshold was used, for example, in the inclusion criteria of the MITRA-FR trial of transcatheter MV repair. 5 Nevertheless, a patient-specific evaluation of the estimated EROA, whether derived from echocardiography or from its predicted relationship with clinical endpoints, is subject to the same inherent limitations. Each method tends to overestimate the role of MV pathology while underestimating the influence of LV volume, pressure and function in determining haemodynamic impact and long-term prognosis of patients afflicted with SMR.20,21,31,32
As demonstrated by Grayburn et al.,20,21 the EROA is dependent on both LV end-diastolic volume (LVEDV) and LVEF for a given regurgitant fraction. With an LVEF of 30% and normal LVEDV, a regurgitant fraction >50% is associated with an EROA as small as 0.2 cm2. However, most patients with chronic HF and an LVEF of 30% also present with marked LV dilatation (LVEDV: 200–250 mL). In this setting, an EROA of 0.2 cm2 more likely represents moderate MR. In patients with systolic dysfunction and normal LV size, a regurgitant fraction of >50% is generally achieved when the EROA is >0.3 cm2. When LVEF falls below 30% or LVEDV exceeds 300 mL, elevated EROA thresholds are needed to indicate severe MR. The mean systolic pressure gradient between the left ventricle and left atrium further modifies this relationship. In chronic HF, where systolic blood pressure is reduced but left atrial pressure is elevated, an EROA of 0.4 cm2 may be necessary to reliably identify severe MR (regurgitant fraction ≥50%).
Grayburn et al.20,21 showed that in patients with reduced LVEF, marked LV dilatation and systolic blood pressure <120 mmHg, an EROA of 0.2 cm2 was indicative of the absence of haemodynamically significant MR. In chronic HF with reduced LVEF, impaired leaflet coaptation due to LV dilatation alone can yield an EROA of 0.2 cm2. The clinical significance of this degree of MR is limited, as transcatheter MV repair has been shown to favourably remodel the LV—similar to MV replacement—even when the residual grade of MR following a successful procedure is 1+ or 2+. 33 Failure to account for LV size and function may lead to misinterpretation of studies that rely on clinical progression alone to estimate MR severity. 24 In patients with significant MR but preserved LV dimensions (normal LVEDV), outcomes are likely driven primarily by the severity of the valvular disorder. In contrast, in patients with chronic HF, reduced LVEF and LVEDV of 200–250 mL, the resultant outcome is significantly influenced by the underlying LV disease. 3 In this group, an EROA of 0.2 cm2, indicative of non-severe MR, has been demonstrated to be associated with high morbidity and mortality, but the poor outcome reflects LV dysfunction rather than MR severity. Hence, in patients with advanced LV impairment, it is inappropriate to assume that MR alone drives an adverse prognosis.
A new conceptual framework to guide treatment selection
In the management of SMR, clinical decision-making should not be confined to determining whether disease progression originates in the MV, identifying the sequence of events or correlating MR with adverse outcomes. Instead, the fundamental aim is to achieve a comprehensive, quantitative understanding of the contributing factors to SMR to assess whether MV intervention can alter the clinical course. In cases of SMR, the leaflets are structurally normal, and the regurgitant jet may emerge gradually during the evolution of the MV disorder. For interventions to be meaningful, MR must reach a clinically relevant threshold, as reductions in regurgitant volume through repair or replacement have been shown to lower mortality and hospitalisation. Conversely, if interventions prove unsuccessful in resolving the MV pathology, any subsequent arguments regarding the clinical significance of the MR—irrespective of its anatomical origin or temporal development—would be unpersuasive.
In this context, the detailed evidence that led to a patient’s presentation is not pertinent. If an MV intervention can alter the natural course of a disease, then MR should be considered a therapeutic target, even if it is secondary to LV dysfunction. Clear and precise nomenclature is therefore essential to identify such patients and ensure timely referral for mechanical intervention.
In this novel conceptualisation, cardiologists are tasked with ascertaining whether the estimated magnitude of MR is commensurate with the extent of LV dilatation or whether the severity of MR is either unanticipated or disproportionate to the degree of LV enlargement. Notably, non-severe MR (EROA ≤0.2 cm2) is commonly observed in patients with chronic HF and markedly enlarged LVEDVs (>220 mL or 120 mL/m2).34,35 In these cases, particularly when LV systolic volumes are also high, MV repair 36 has been shown to be ineffective.34,35 The misclassification of an EROA of 0.2 cm2 as ‘severe’ in patients with significant LV enlargement in the 2012–2014 European and US guidelines was later corrected in the 2017 update. 37
Grayburn et al. worked towards standardising the relationship between EROA and LVEDV20,21,33 to help determine whether MR in a patient is proportionate or disproportionate to ventricular dilatation. Because EROA estimation is imprecise and influenced by multiple haemodynamic and echocardiographic variables, defining appropriate thresholds must be undertaken empirically. Patients whose MR severity is proportionate to the degree of LV enlargement are expected to gain limited benefit from MV intervention, as they would lie close to the linear relationship between EROA and LVEDV. In contrast, patients whose MR is disproportionate to LV dilatation may be a suitable candidate for MV repair.
A patient with non-severe MR—as indicated by an EROA/LVEDV ratio significantly lower than the cut-off for proportionality—is unlikely to benefit from any MV procedure. This framework therefore provides a strategy to refine the evaluation of MR and identify patients with chronic HF, systolic dysfunction and SMR who are most likely to benefit from MV repair.
Recommendations from international heart disease management guidelines
Medical therapy
According to the guidelines for the management of HF, determining the optimal medical therapy is the first line of care in patients with SMR. 38 The ESC guidelines recommend switching from an angiotensin-converting enzyme inhibitor (ACEI) or angiotensin receptor blocker (ARB) to sacubitril/valsartan, with the addition of sodium-glucose transport protein 2 (SGLT2) inhibitors and/or ivabradine where indicated.39,40 The ACC/AHA guidelines recommend (class of recommendation (COR) 1/level of evidence (LOE) A) guideline-directed medical therapy (GDMT) for patients with chronic severe SMR and reduced LVEF in stages C and D, consisting of ACEIs, ARBs, beta-blockers, aldosterone antagonists and/or sacubitril/valsartan.38–47 In addition, the ACC/AHA guideline advise (COR 1/LOE C-EO) that a cardiologist with expertise in the management of patients with severe SMR predisposing to HF and LV systolic dysfunction should be the lead member of the multidisciplinary team (MDT), supervising the implementation and monitoring of optimal GDMT.3,44 Cardiac resynchronisation therapy (CRT) should also be considered for eligible patients in line with the current CRT guidelines.48,49
If patients remain symptomatic despite optimal GDMT, mechanical intervention on the MV—either conventional surgery or TEER—should be considered to prevent further deterioration of LV systolic function or cardiac remodelling.
MV procedures
The available evidence consistently shows that chronic SMR is associated with an adverse prognosis.48–52 Consequently, determining the optimal interventional strategy remains challenging, as illustrated in Figure 1. Given the complexity of this clinical context, decision-making should involve a multidisciplinary heart team (HT), whose role is central to individualised patient management.

(a) Multidisciplinary decision-making in the surgical management of moderate-to-severe SIMR, with or without CABG. CAD: coronary artery disease; CABG: coronary artery bypass grafting; MR: mitral regurgitation; EROA: effective regurgitant orifice area; Rvol: regurgitant volume; RF: regurgitant fraction; ESC: European Society of Cardiology; RA: right atrium; LV: left ventricle; PMA: papillary muscle approximation.3–8,10,16–19,53–56,82
The HT at work
The HT, including a dedicated HF specialist, should ensure optimisation of GDMT and assess all treatment options. This involves deciding whether to pursue electrophysiological intervention, catheterisation or standard surgery while carefully weighing risks, benefits and the order of treatment modalities. Robust evidence supporting standard surgery remains limited, owing to the scarcity of multicentre RCTs providing high-level evidence.
The ESC guidelines recommend MV surgery for patients with severe MR who are deemed eligible for coronary artery bypass graft (CABG) surgery or other cardiac interventions. The HT is responsible for tailoring the surgical strategy to the clinical profile of each patient.48–52 In patients without evidence of progression of LV remodelling, restrictive mitral annuloplasty with a full rigid ring is advised, as this promotes restoration of valvular function, improvement in symptoms and reverse LV remodelling.47,51,52 Techniques such as subvalvular repair or chordae-sparing valve replacement may be considered suitable for patients with echocardiographic predictors of elevated repair risk.53–63
Valve replacement is associated with a lower risk of recurrent MR. However, this technique has not been shown to improve reverse LV remodelling or survival.16,59,61 The limited number of multicentre RCTs underscores the narrow indications for isolated MV surgery in patients with severe MR. Given the high risk of adverse events, the frequent recurrence of MR and the lack of a proven survival benefit with standard surgery in this population,16,54,57,59–61,64 this strategy warrants cautious consideration.
A distinct subgroup comprises patients with persistent SMR due to atrial fibrillation (AF). In these cases, the LV chamber size is smaller, with less pronounced dilatation. Because mitral annular dilatation is the primary anatomical mechanism of MR in this population, this UMA—often combined with AF ablation—can be effective. Nevertheless, robust evidence supporting this approach remains limited.49,65–67
TEER with the MitraClip system has become an established minimally invasive procedure for SMR, providing an alternative to surgical repair in patients deemed ineligible or unsuitable for standard surgery (Figure 2). Two pivotal RCTs—COAPT and MITRA-FR—3–6,10 evaluated its safety and efficacy in patients with symptomatic HF and severe, persistent SMR despite optimal medical therapy.

Diagnosis of the patient with SMR and TEER treatment. (a) 3D TEE showing central SMR. (b) 3D TEE colour en-face view demonstrating central SMR. (c) 3D TEE en-face view following successful implantation of two central MitraClips (Abbott Vascular, Menlo Park, CA) and (d) three-chamber TEE view at 1-year follow-up showing residual mild MR with a mean gradient of 4 mmHg. 3D: three-dimensional; TEE: transesophageal echocardiography; MR: mitral regurgitation.
In COAPT, 3-year follow-up demonstrated that MitraClip was both safe and effective, significantly reducing SMR and lowering HF-related rehospitalisations.3,4,10,68 The trial als showed improvements in secondary endpoints, including all-cause mortality at 2 years. In contrast, MITRA-FR reported no significant benefit of MitraClip over GDMT alone with respect to all-cause mortality of HF rehospitalisation at 12 months and 2 years.5,6 Figure 2(a) to (d) illustrates TEER in a patient with ischaemic SMR. Baseline imaging (Figure 2(a)) shows three-dimensional transesophageal echocardiography, while follow-up (Figure 2(d)) demonstrates residual mild MR and no haemodynamic stenosis at 1 year.
The two RCTs yielded conflicting results, prompting a sustained debate within the cardiovascular community. These inconsistencies are partly attributable to differences in study design, which led to heterogeneous patient populations. Key factors under discussion included the magnitude of treatment effect, the echocardiographic evaluation of SMR severity and the implementation of optimised medical therapy.
In COAPT, enrolled patients had more severe SMR (EROA: 41 ± 15 mm2 vs 31 ± 10 mm2) but less LV dilatation (indexed LVEDV: 101 ± 34 mL/m2 vs 135 ± 35 mL/m2) compared with MITRA-FR.3,5 This imbalance suggested that the COAPT cohort exhibited SMR disproportionate to LV size and was therefore more likely to benefit from TEER in terms of reduced mortality and HF hospitalisation. 69 These findings underscore the need for further analyses and better stratification criteria.
Accordingly, the COAPT trial implies that patients with severe SMR considered for TEER should closely match its inclusion criteria (Table 1). They should receive maximised GDMT, be regularly monitored by an HF specialist and demonstrate clinical and echocardiographic features comparable to those of the COAPT population in order to optimise outcomes and strengthen the rationale for TEER.
TEER: transcatheter edge-to-edge repair; SMR: secondary mitral regurgitation; LVEF: left ventricular ejection fraction; HF: heart failure; NYHA: New York Heart Association; GDMT: guideline-directed medical therapy; LVESD: left ventricular end-systolic diameter; SPAP: systolic pulmonary artery pressure; RV: right ventricle; TTE: transthoracic echocardiography.
The recommendation for TEER is limited to patients who do not meet COAPT criteria and for whom the primary goal is symptom relief and improved quality of life. The clinical benefit of MitraClip in patients with less severe SMR (EROA <30 mm2) and progressive LV dilatation or failure remains uncertain. Patients with end-stage LV or right ventricular failure, in whom percutaneous coronary intervention (PCI) or CABG is not feasible, are not suitable candidates for MitraClip. In this subgroup, the most favourable outcomes are achieved with heart transplantation or use of an LV assist device as a bridge to transplant. MV surgery is generally considered unethical in patients with LVEF <15%.41,51,70
The management of moderate ischaemic SMR in post-CABG patients remains a topic of ongoing debate.19,71 For patients with preserved myocardial viability and low comorbidity burden, surgical intervention may represent the most appropriate strategy. In individuals with exercise-induced dyspnoea and a marked increase in MR severity and systolic pulmonary arterial pressure, a combined surgical approach is typically recommended. In parallel, technological advances have led to the development of novel transcatheter MV repair systems beyond TEER, with the aim of enhancing transcatheter MV replacement devices. It is evident that these devices are currently subject to rigorous evaluation, with their reliability potentially contingent on the availability of limited clinical data. Emerging differences between ACC/AHA guidelines and other recommendations are summarised in Table 2, Table 3 and Figure 1.
As outlined in the referenced recommendation, the ACC/AHA guidelines provide a robust foundation for this advice.
Factors to consider when predicting surgical risk: LVEF, myocardial viability, coronary anatomy and target vessels, type of concomitant procedure, TEER eligibility, likelihood of durable surgical repair, need for MV replacement and local expertise.
LVEF: left ventricular ejection fraction; TEER: transcatheter edge-to-edge repair; MV: mitral valve; COR: class of recommendation; LOE: level of evidence; MR: mitral regurgitation; NYHA: New York Heart Association; GDMT: guideline-directed medical therapy; HF: heart failure; TEE: transthoracic echocardiography; LV: left ventricle; SMR: secondary mitral regurgitation; CABG: coronary artery bypass graft; CAD: coronary artery disease.
As outlined in the referenced recommendation, the ESC guidelines provide a robust foundation for this advice.
Factors to consider when predicting surgical risk: LVEF, myocardial viability, coronary anatomy and target vessels, type of concomitant procedure, TEER eligibility, likelihood of durable surgical repair, need for surgical MV replacement and local expertise.
COAPT criteria (Cardiovascular Outcomes Assessment of the MitraClip Percutaneous Therapy for Heart Failure Patients With Functional Mitral Regurgitation)
LVEF: left ventricular ejection fraction; TEER: transcatheter edge-to-edge repair; MV: mitral valve; COR: class of recommendation; LOE: level of evidence; SMR: secondary mitral regurgitation; GDMT: guideline-directed medical therapy; HT: heart team; CABG: coronary artery bypass grafting; PCI: percutaneous coronary intervention; TAVR: transcatheter aortic valve replacement; MR: mitral regurgitation.
Discussion
The 5-year results of the COAPT trial demonstrated that TEER was associated with sustained reductions in hospitalisation for cardiovascular and HF causes, with most of the benefit observed during the first 3 years of follow-up. 10 Similarly, TEER was correlated with reduced all-cause mortality, cardiovascular mortality and HF-related mortality within a 5-year time frame, with the greatest effect observed within the first 2 years after randomisation. By 2 years, approximately 50% of patients in the control group had died—the prespecified cut-off for crossover—resulting in 49% of patients ultimately undergoing crossover as permitted by the protocol. 10
The haemodynamic benefit of TEER is largely attributable to reductions in volume and pressure overload. Multiple studies have shown that TEER improves symptoms and prognosis in patients with HF and SMR. However, the primary underlying disease in most cases—LV cardiomyopathy—is not directly influenced by MV repair procedures.59,72,73 Consequently, cardiovascular and non-cardiovascular adverse events persisted during follow-up, reflecting the advanced age and comorbidity burden of the COAPT population.
During the COAPT trial, the standard of care provided to patients with HF had evolved,74,75 particularly with the increasing use of angiotensin receptor–neprilysin inhibitors. The uptake of sacubitril/valsartan rose progressively during follow-up, and compared with controls, a higher proportion of patients in the device group showed favourable outcomes. This is likely attributable to improved haemodynamics after mitral TEER. The extent to which sacubitril/valsartan contributed independently to the observed benefit remains uncertain. More widespread adoption of neprilysin inhibitors may have reduced the cohort of patients with refractory symptoms and severe MR eligible for TEER.76,77 However, it is unlikely that this has eliminated the benefits of correcting MR in suitable candidates.
Concerns have also been raised regarding surgical options in patients who previously underwent TEER. In such cases, MV replacement—rather than repair—is generally preferred, particularly when transcatheter intervention has failed to achieve the desired outcome. 78 In the COAPT trial, MV surgery (comprising replacement procedures) was performed less frequently in the device group than in the control group during the 5-year follow-up. 10
Determining the superior intervention for SMR remains challenging because no large-scale RCTs, such as the PARTNER trial, have included patients eligible for TEER, MV replacement or MV repair with or without subvalvular procedures. Current evidence for TEER is primarily derived from two RCTs, MITRA-FR5,6 and COAPT.3,4,10 Comparative analyses of these trials,69,78 together with observational studies of the MitraClip procedure, 79 a small observational comparison of TEER and standard surgery 80 and evaluations of the proportionate/disproportionate concept in SMR, 69 have shaped current perspectives. The most recent recommendations endorse the use of TEER in patients with characteristics similar to those enrolled in COAPT trial. It is important to note that the study reporting the MITRA-FR result at 2 years of follow-up was excluded from these analyses.
There is a paucity of research on this topic, with only one study published in 2015 that directly compared TEER with standard surgical procedures in high-risk patients. 1 Current guidelines recommend TEER (COR 2a LOE B-R) but do not provide specific guidance regarding concomitant treatment of myocardial ischaemia. 8 In COAPT trial, although 60.9% of patients had ischaemic cardiomyopathy, only 43% underwent PCI and 40% underwent CABG.3,4,10
LV remodelling is a major concern in patients with SMR of Carpentier type IIIb, particularly when there is extensive scarring after myocardial infarction or prolonged heart muscle damage with abnormal movement. Untreated diffuse coronary heart disease can lead to SMR from ischaemic cardiomyopathy, resembling the pattern seen in non-ischaemic cardiomyopathy.62,81 In such cases, SMR may also result in central MR, with global wall motion abnormalities due to multivessel coronary disease causing equal lateral displacement of both PMs, similar to that observed in non-ischaemic cardiomyopathy.54–57,82
Okuno et al. 83 compared the 2-year outcomes of standard MV surgery with UMA versus TEER in 202 propensity-matched patients with SMR. Their findings highlighted inconsistencies in the 2020 ACC/AHA guidelines regarding SMR management. Although overall survival did not differ significantly between UMA and TEER, UMA combined with coronary revascularisation was associated with greater reductions in MR, improved LVEF and decreased rates of NYHA class III–IV symptoms. 83 At discharge, none or mild MR was more frequent after UMA than TEER (90.8% vs 72.0%; P < 0.001), and this advantage persisted at 2 years (86.5% vs 59.6%; P < 0.001). Among patients with none or mild MR at discharge, only 10.1% in the UMA group progressed to moderate or greater MR compared with 34.9% in the TEER group (P = 0.003). Similarly, LVEF improved considerably after UMA (45.7% ± 12.8%) compared with that after TEER (−1.3% ± 8.9%; P = 0.260).
In the sub-analysis of the Cardiothoracic Surgical Trials Network (CTSN), 75% of patients underwent concomitant CABG, which limited the potential for improvement in regional wall motion in the remaining 25% of patients. 18 Several studies have demonstrated that subvalvular MV procedures in combination with valvular restrictive repair procedures are both a safe and effective alternative to UMA alone, in both ischaemic and non-ischaemic cohorts.53–57,82
The Papillary Muscle Approximation Randomized Clinical Trial (PMA-RCT) further investigated a dual-level repair strategy targeting both valvular and subvalvular components of the MV apparatus. This study included 96 patients with severe secondary ischaemic MR who underwent revascularisation. At 5-year follow-up, both isolated restrictive mitral repair and PMA combined with UMA resulted in significant improvements in LV end-diastolic dimension (LVEDD) (−5.8 ± 4.1 and −0.2 ± 2.3 mm, respectively; P < 0.001).
The study demonstrated that preserving the benefits achieved in the immediate post-operative period led to freedom from major adverse cardiac and cerebrovascular events (P = 0.004). 79 In contrast, the COAPT trial showed no enhancement in LV remodelling with TEER. Specifically, LVEDV exhibited no significant difference before and after the procedure (194.4 ± 69.2 vs 192.2 ± 76.5 mL). 4 However, the 3-year follow-up data from TEER recipients revealed sustained improvements in MR severity and functional capacity. Importantly, TEER has been shown to provide greater improvement in terms of both symptomatic relief and quality-of-life measures compared with GDMT alone. 4 This advantage was also evident in a subset of 58 patients who transitioned from GDMT to TEER, among whom the composite rate of mortality or hospitalisation for HF was significantly lower than that with GDMT alone (P = 0.006). 4
In RCTs, UMA has been demonstrated to be associated with a high incidence of recurrent MR at both 2-year and 5-year follow-up, with recurrence rates of 58.8% and 55.9%, respectively.18,55 UMA yields the most favourable outcomes in patients with smaller preoperative LV end-systolic dimension and reduced apical leaflet tethering. In the CTSN trial, among 74 patients with severe ischaemic MR, those without MR recurrence after UMA demonstrated a significantly greater reduction in LV size at 2 years compared with those who experienced recurrence (43 ± 26 vs 63 ± 27 mL/m2). 18 A comparative analysis of LV end-systolic volume (LVESV) showed that the measurements in the UMA group remained higher than those in the MV replacement group, despite the use of complete chord-sparing techniques that preserve LV architecture (61 ± 39 mL/m2). 18
The PMA-RCT demonstrated that double-level repair can normalise three geometric measures, thereby achieving ventricular–valvular recovery. The objective of this approach is to restore the altered spatial relationship among the components of the MV apparatus by addressing both the valve and ventricle in patients with Carpentier type IIIb SMR.55–57,60 The Osaka group also examined the role of the PMs. Kainuma et al. 84 reported that UMA alone does not fully resolve leaflet tethering, although it facilitates PM realignment by reducing the interpapillary muscle distance (IPMD) through improvement in LVEDD. IPMD was identified as the main predictor of MR recurrence. These benefits were further augmented by reverse LV remodelling after UMA, with IPMD decreasing from 31 ± 6 mm to 25 ± 5 mm, potentially offsetting the increase in posterior leaflet angle. 84 Accordingly, two-stage repair strategies incorporating surgical manipulation of the PMs may be more appropriate than TEER for patients with SMR secondary to non-ischaemic cardiomyopathy. These patients typically exhibit MV annular dilatation, lateral displacement of both PMs and symmetric leaflet tethering with apical anterior leaflet involvement, resulting in a central regurgitant jet that cannot be corrected with UMA alone.18,55,56,82,85 Such cases resemble the proportionate cohort that did not respond favourably to TEER3–6,10,69,85 in the COAPT3,4,10 and MITRA-FR5,6 trials. Notably, patients with non-ischaemic cardiomyopathy, severe LV dilatation and moderate-to-severe MR experienced poorer outcomes in both trials (Figure 3).
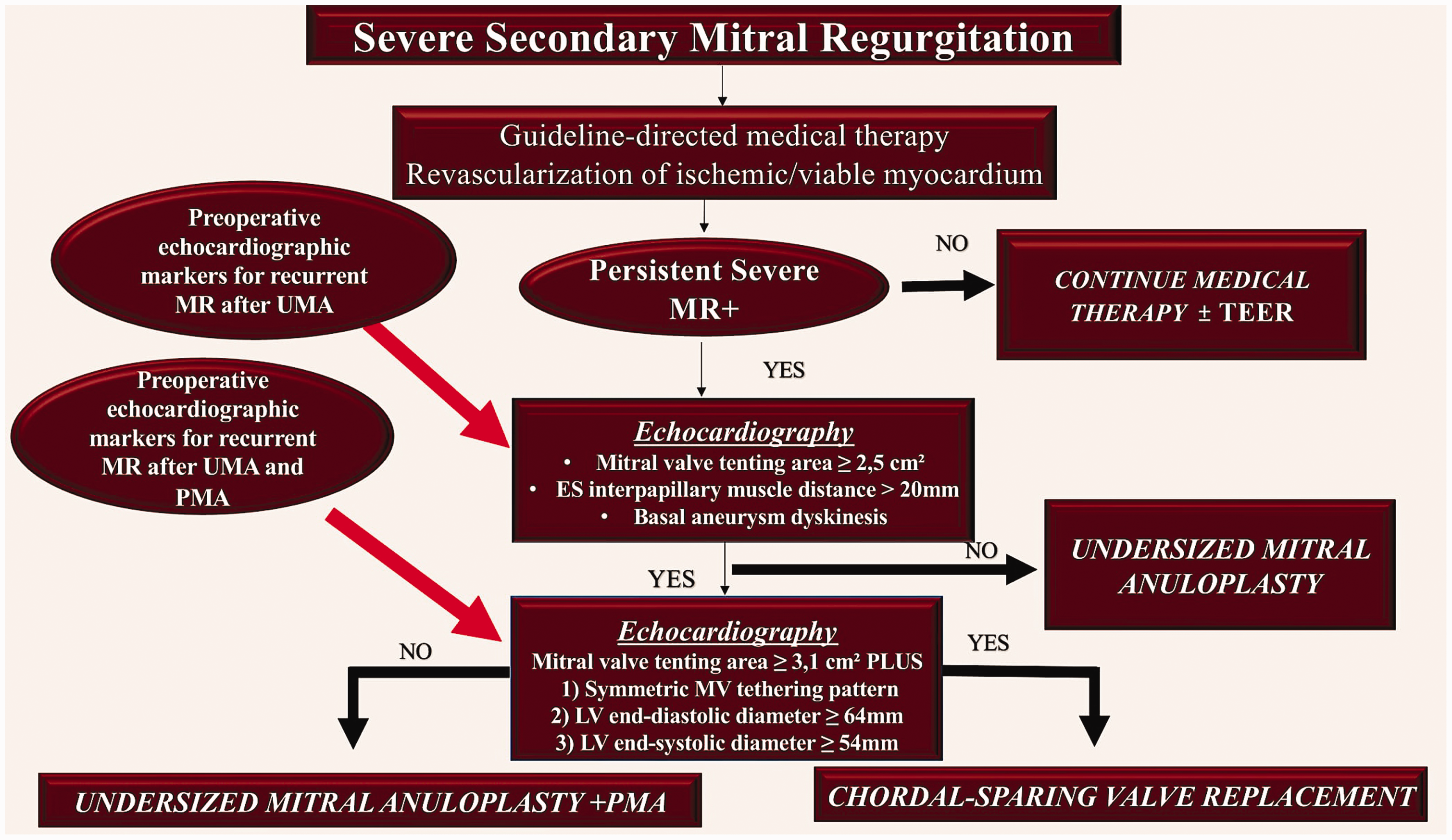
Treatment algorithm for patients with severe SMR undergoing MV surgery. Patients are treated with either isolated RMA or RMA combined with subannular repair and CABG. Preoperative echocardiographic markers are used to predict recurrent MR after undersized RMA (red arrow) or after combined RMA and PMA (red arrow).3–8,10,16–19,53–56,82 SMR: secondary mitral regurgitation; MV: mitral valve; RMA: restrictive mitral annuloplasty; CABG: coronary artery bypass grafting; MR: mitral regurgitation; PMA: papillary muscle approximation.
In the MATTERHORN clinical trial, 86 a total of 210 patients were randomised in a 1:1 ratio to receive either TEER (intervention group) or surgical MV repair or replacement (surgery group). Among patients with HF and SMR, TEER was demonstrated to be non-inferior to MV surgery with respect to a composite endpoint comprising death, rehospitalisation for HF, stroke, reintervention or implantation of an LV assist device at 1 year.
In the CLAPS RCT (n = 124; CLASP Study, Edwards PASCAL Transcatheter Mitral Valve Repair System Study), the PASCAL system was evaluated in patients with SMR. The trial demonstrated favourable outcomes, including improved survival and reduced HF-related hospitalisations at both 1- and 2-year follow-up. Furthermore, patients treated with the PASCAL system exhibited a substantial reduction in MR, positive reverse LV remodelling and improved functional status and exercise capacity.87–90 The CLASP IID RCT further demonstrated that TEER using the PASCAL system is non-inferior to MitraClip for major adverse events in patients with degenerative MR at prohibitive surgical risk. Echocardiographic outcomes at 6 months were comparable between the two devices. The durability of these results extends up to 2 years, although there was a relatively higher attrition rate in the PASCAL cohort compared with the MitraClip group. These findings are clinically significant, as they supported Food and Drug Administration approval for the PASCAL device in the United States for this indication. Long-term outcomes and potential cost-effectiveness differences remain to be determined, and data collection for patients with functional MR is ongoing. 90
Conclusion
TEER should be considered for patients with severe SMR who meet the COAPT inclusion criteria and are receiving optimal medical therapy.91,92 These patients should closely resemble the population enrolled in the COAPT trial to ensure optimal procedural outcomes. TEER may also be considered for individuals who do not meet the COAPT criteria, with the primary goal of improving symptoms and quality of life (Table 1).93,94 For patients with less severe SMR (EROA <30 mm2) and advanced LV dilatation or dysfunction, the benefits of MitraClip remain unproven.9,95,96 In cases of end-stage LV or right ventricular dysfunction where revascularisation is not an option, heart transplantation or LV assist device implantation may be the most appropriate management strategy. Valve interventions are generally contraindicated in patients with an LVEF ≤15%. 97
The management of moderate-to-severe ischaemic SMR in patients undergoing CABG remains controversial.19,71,81 Surgical intervention is most likely to be beneficial when myocardial viability is present and comorbidity burden is low, often in combination with other surgical procedures. Emerging transcatheter MV repair and replacement systems, beyond the TEER model, offer potential therapeutic options; however, clinical data remain limited.
Multicentre RCTs with a minimum follow-up period of 5 years are recommended to evaluate the long-term efficacy and safety of TEER and double-level repair strategies.
Footnotes
Acknowledgements
Not applicable.
Author contribution
Conceptualisation, methodology, validation, investigation, data curation, writing—original draft preparation, writing—review and editing, visualisation, supervision: F.N.
Data availability statement
Not applicable.
Declaration of conflicting interest
The author declares that there are no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.



