Abstract
Objective
To examine the association between serum 25-hydroxyvitamin D levels and stress urinary incontinence among women in the United States.
Methods
This cross-sectional analysis included data from 13,768 female participants of the National Health and Nutrition Examination Survey (2005–2018). Weighted multivariable logistic regression, subgroup analyses, and restricted cubic spline analysis were used to evaluate associations.
Results
Overall, 42% of the participants had stress urinary incontinence. In fully adjusted models, continuous serum 25-hydroxyvitamin D level was inversely associated with stress urinary incontinence (odds ratio = 0.98, 95% confidence interval: 0.96–1.00, p = 0.040). The highest quartile (fourth quartile, Q4) of 25-hydroxyvitamin D level also showed a significant inverse association with stress urinary incontinence (odds ratio = 0.81, 95% confidence interval: 0.68–0.96, p = 0.016). A significant interaction with smoking status was detected (p < 0.001). Restricted cubic splines revealed a nonlinear relationship (p-nonlinear = 0.012) with a threshold level of 73.8 nmol/L, and 25-hydroxyvitamin D levels beyond this value were associated with a reduced risk of stress urinary incontinence.
Conclusion
Higher 25-hydroxyvitamin D levels (≥73.8 nmol/L) are associated with a lower risk of stress urinary incontinence among women in the United States, indicating a nonlinear relationship. Further prospective studies are needed to confirm these findings.
Keywords
Introduction
Stress urinary incontinence (SUI) is characterized by involuntary urine leakage precipitated by physical movement, exertion, coughing, or sneezing. 1 SUI is more prevalent in women, particularly among middle-aged and older women. 2 Epidemiological studies indicate that >25% of women worldwide are affected by SUI, and its incidence continues to rise. 3 SUI profoundly affects patients’ quality of life, physical health, psychological well-being, and socioeconomic status. 4
Vitamin D plays a crucial role in the human body. It exists in two forms, vitamin D2 and vitamin D3, both of which are fat-soluble steroids. 5 Insufficient levels of 25-hydroxyvitamin D (25(OH)D) have been associated with various conditions, including immune system disorders, skeletal diseases, diabetes, renal impairment, and heart failure.6–8
Serum 25(OH)D levels can be determined via the measurement of classical vitamin D metabolites. However, the optimal serum 25(OH)D level remains debatable. 9 Although substantial research has suggested a potential association between serum 25(OH)D levels and SUI,10–12 this relationship has not been specifically investigated in adult women in the United States. Based on the existing evidence, there is a critical need for a comprehensive, population-based study to determine whether serum 25(OH)D levels are correlated with SUI risk in this demographic. Such studies would provide valuable insights into this potential relationship and guide clinical decisions.
Objective
SUI, a prevalent urological issue that primarily affects women, can be significantly disruptive to daily life and substantially diminish patients’ quality of life. This study aimed to investigate the correlation of SUI risk with serum 25(OH)D levels.
Methods
This cross-sectional study utilized data from the National Health and Nutrition Examination Survey (NHANES, 2005–2018). The study sample comprised 13,768 women aged ≥18 years. A weighted multivariate regression model was used to investigate the relationship between serum 25(OH)D levels and SUI risk. Additional subgroup analyses were performed, stratified by age, ethnicity, marital status, smoking status, and alcohol consumption. Restricted cubic spline (RCS) models were subsequently utilized to delineate any nonlinear relationships.
Materials and methods
25(OH)D quartile cutoff values
A quartile-based analysis was performed to assess the potential nonlinear dose–response relationship between serum 25(OH)D levels and SUI risk because the initial continuous analysis did not demonstrate a significant association. The quartile cutoff values were determined as follows: first quartile (Q1), 5.49–51.30 nmol/L; second quartile (Q2), 51.30–68.80 nmol/L; third quartile (Q3), 68.80–90.50 nmol/L; and fourth quartile (Q4), 90.50–422.00 nmol/L. Missing values for the poverty-to-income ratio (PIR) were addressed using weighted median imputation. Individuals with missing values for any other variables were excluded from the analysis. The weighted median imputation method adopted for PIR preserved the sample size and was more robust than simple median imputation; however, it failed to account for the uncertainty of the imputed values. For the excluded data, if the missingness is classified as “missing not at random,” it may oversimplify the true underlying relationships. Although these methods were employed to maximize the use of available data, residual confounding bias may exist due to the missing data.
Survey description
We used data obtained from the NHANES (2005–2018). The NHANES is a biennial survey that comprehensively assesses the health and dietary habits of diverse US populations across age, ethnicity, and sex. The Research Ethics Review Board of the National Center for Health Statistics has approved all the techniques used in the survey. Each participant was comprehensively informed about the study and provided signed consent. The NHANES employs a complex stratified multi-stage probability sampling design to recruit a nationally representative sample of participants, and we utilized de-identified, publicly available data from the database. The analysis of this anonymized data was conducted in accordance with the ethical principles outlined in the Declaration of Helsinki (as revised in 2024). This study adhered to the relevant Enhancing the QUAlity and Transparency Of health Research (EQUATOR) Network guidelines, and the reporting of this study is in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 13 Detailed information regarding the study design and datasets is available at https://www.cdc.gov/nchs/nhanes/.
Study sample
This research evaluated data from seven NHANES cycles conducted between 2005 and 2018, which focused on participants reporting urinary incontinence associated with activities that increase pressure inside their abdomen, including exercise, weight training, coughing, and comparable physical activities (n = 70,190). We excluded male participants (n = 34,709), those aged <18 years (n = 13,796), those with incomplete SUI data (n = 4169), and those with incomplete vitamin D data (n = 1083). Those with missing data for covariates were also excluded (marital status, n = 8; smoking status, n = 9; history of cesarean section, n = 2; alcohol consumption, n = 1842; history of hysterectomy, n = 765; and history of hormone therapy, n = 39). Finally, a total of 13,768 participants were included in our study; the patient selection process is presented in Figure 1.

Flow diagram depicting the patient selection process.
Definitions of exposure and outcome
SUI assessment
SUI occurrence was assessed by asking participants if they had experienced loss of bladder control or involuntary urine leakage, even in minimal quantities, during the past year, specifically during activities that increase intra-abdominal pressure such as coughing, sneezing, laughing, weightlifting, and exercise. Individuals who reported such incidents were categorized as having SUI, while those who did not were classified as not having SUI.
Vitamin D assessment
The serum levels of 25-hydroxyvitamin D3 (25(OH)D3), 3-epi-25-hydroxyvitamin D3 (epi-25(OH)D3), and 25-hydroxyvitamin D2 (25(OH)D2) were measured using high-performance liquid chromatography–tandem mass spectrometry. Total serum 25(OH)D level, calculated as the sum of 25(OH)D3 and 25(OH)D2 (in nmol/L), was used as the exposure variable in this study. For any analyte with a concentration below the limit of detection (LOD), the LOD/√2 value was imputed.
Covariates
This study focused on several parameters, including age (in years), race, educational level, marital status, PIR, alcohol consumption, diabetes, cigarette use, physical activity, and histories of cesarean section, hysterectomy, and hormone therapy. The analyzed variables included age (18–44, 45–64, or ≥65 years), race (Mexican American, other Hispanic, non-Hispanic White, non-Hispanic Black, or other races), educational level (<high school or ≥high school), PIR (<1.3, 1.3–1.5, or >1.5), alcohol consumption (yes or no), diabetes (yes or no), smoking status (yes or no), and histories of cesarean section (yes or no), hysterectomy (yes or no), and hormone therapy (yes or no). Alcohol consumption was determined by estimating the alcohol content from the total dietary intake on the first and second days; participants were classified as nondrinkers if both values were 0 and as alcohol consumers if otherwise. The smoking status was ascertained from participants’ responses to the question, “Have you smoked a minimum of 100 cigarettes in your lifetime?” The presence of diabetes was determined based on self-reported physician diagnosis, fasting blood glucose level ≥126 mg/dL, glycated hemoglobin ≥6.5%, or current use of antidiabetic medication.
Statistical analyses
For our analysis, we applied the NHANES-provided weights, calculating the 14-year projections from 2005 to 2018 by dividing the 2-year weights by 4. Continuous variables were analyzed using weighted t-tests, and the results were presented as means and their standard errors. For categorical variables, we performed analyses using weighted chi-square tests and reported the results as counts and percentages. We performed weighted multivariate analysis to investigate the relationship between serum 25(OH)D level and SUI risk. A weighted multivariable logistic regression model was employed to assess the collective effect of serum 25(OH)D level and SUI, with 25(OH)D categorized into quartiles. Finally, we performed multivariable logistic regression to explore potential dose–response correlations. Three models were developed: model 1, unadjusted; model 2, adjusted for age, race, and marital status; and model 3, fully adjusted for all aforementioned factors as well as alcohol consumption, diabetes, smoking status, PIR, and histories of cesarean section, hysterectomy, and hormone therapy.
Additionally, to explore how serum 25(OH)D levels influence the likelihood of SUI development within different demographic segments, subgroup analyses were performed considering factors such as race, educational level, marital status, and smoking status, alongside interaction assessments. Techniques such as threshold effect analysis and RCS curve fitting were employed to identify nonlinear patterns and possible dose–response correlations. Statistical calculations were conducted using R software (version 4.4.2), with the significance threshold set at p <0.05.
Results
Baseline characteristics
We analyzed the data of 13,768 participants; Table 1 provides a breakdown of the key demographics of the study population. In total, 5791 participants had a history of SUI, while 7977 did not. In the group that had not experienced SUI, the serum 25(OH)D level was 72.22 (41.66, 102.72) nmol/L, while in the group that had experienced SUI, it was 73.91 (42.15, 103.29) nmol/L, representing a significant difference (p = 0.040). The analysis revealed no meaningful variations in family PIR (p = 0.540), alcohol consumption (p = 0.111), history of cesarean section (p = 0.812), or diabetes prevalence (p = 0.816). However, all remaining variables showed statistically significant associations (p < 0.05).
Clinical characteristics of the study population.

N not missing (unweighted).
n (unweighted) (%); mean ± SD.
Pearson’s X2: Rao & Scott adjustment; Design-based t-test.
PIR: poverty-to-income ratio; 25(OH)D: 25-hydroxyvitamin D.
Correlation of serum 25(OH)D level with SUI risk
We performed complete multivariate analysis with weighted adjustments to investigate the potential relationship of serum 25(OH)D level with SUI risk. The study accounted for key demographic and health factors, including participants’ age, ethnicity, educational level, marital status, PIR, alcohol consumption, diabetes, smoking status, and histories of cesarean section, hysterectomy, and hormone therapy. Significant associations were observed in both unadjusted and partially adjusted models that accounted for confounding variables. Table 2 presents the findings from an analysis examining the association of serum 25(OH)D level with SUI risk. The initial unadjusted model (model 1) indicated a statistically significant positive association (odds ratio (OR) = 1.02, 95% confidence interval (CI): 1.00–1.03, p = 0.038). Upon controlling for demographic variables such as ethnicity, age, and marital status in model 2, the significance of the association persisted, but the direction of the effect was reversed, indicating an inverse association (OR = 0.98, 95% CI: 0.96–1.00, p = 0.026).
Weighted logistic regression analysis of the association between 25(OH) D level and SUI risk.
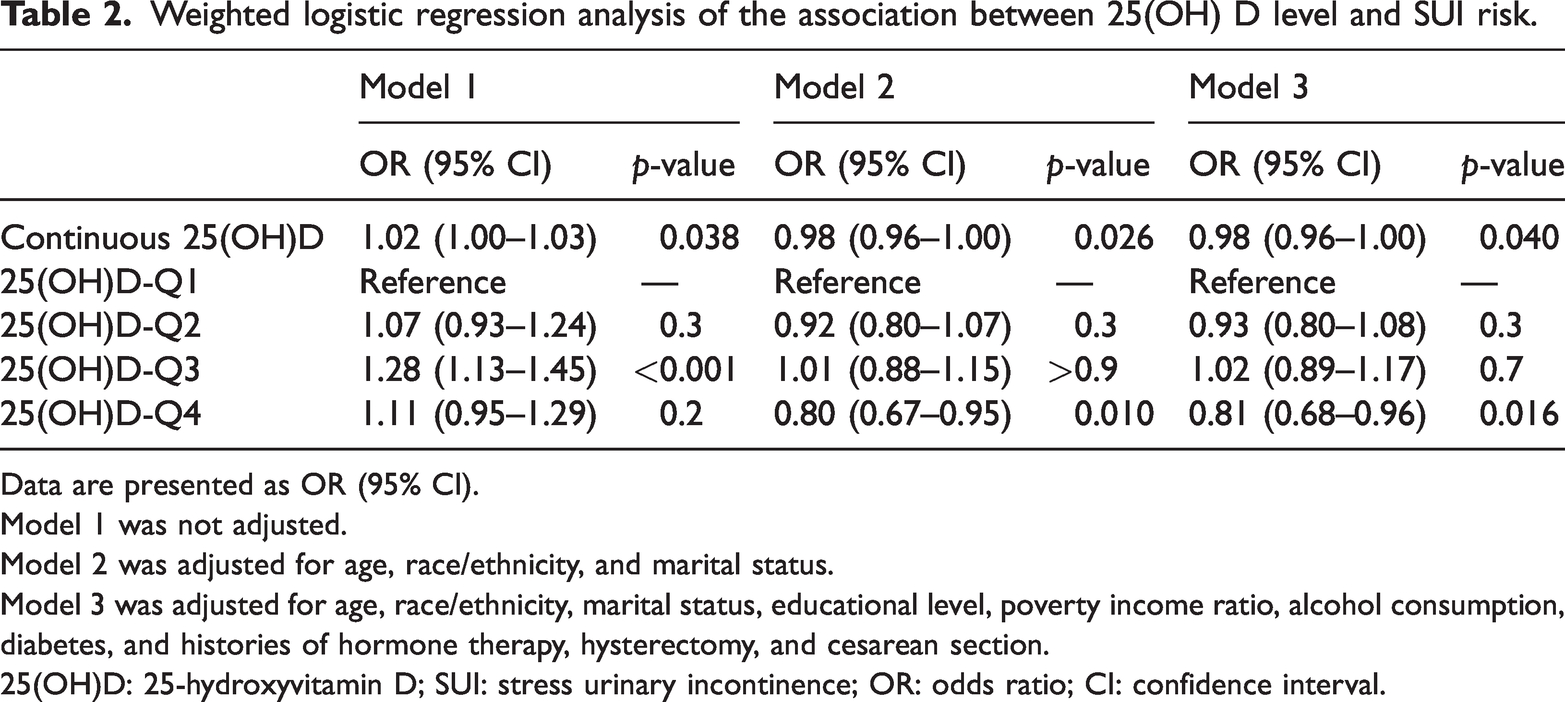
Data are presented as OR (95% CI).
Model 1 was not adjusted.
Model 2 was adjusted for age, race/ethnicity, and marital status.
Model 3 was adjusted for age, race/ethnicity, marital status, educational level, poverty income ratio, alcohol consumption, diabetes, and histories of hormone therapy, hysterectomy, and cesarean section.
25(OH)D: 25-hydroxyvitamin D; SUI: stress urinary incontinence; OR: odds ratio; CI: confidence interval.
Model 3, the most thorough model, which accounted for all relevant confounders—including socioeconomic status, lifestyle characteristics, and medical history—demonstrated a significant correlation between serum 25(OH)D level and SUI risk (OR = 0.98, 95% CI: 0.96–1.00, p = 0.040).
Analysis of vitamin D levels by quartiles in model 1 indicated a positive association between Q3 and SUI risk (OR = 1.28, 95% CI: 1.13–1.45, p < 0.001). However, after partial adjustment in model 2, the highest quartile (Q4) showed an inverse relationship (OR = 0.80, 95% CI: 0.67–0.95, p = 0.010). The fully adjusted model 3 indicated that higher serum 25(OH)D levels may protect against SUI as the negative correlation persisted after adjusting for all confounders (OR = 0.81, 95% CI: 0.68–0.96, p = 0.016).
Subgroup analyses
To evaluate potential variations in the association between serum 25(OH)D levels and SUI risk, subgroup analyses were conducted across strata defined by age, race, educational level, marital status, smoking status, and alcohol consumption, as summarized in Table 3. The findings revealed a significant interaction between serum 25(OH)D level and smoking status (p for interaction <0.001). Notably, among participants with less than a high school diploma, the correlation between serum 25(OH)D level and SUI risk was not statistically significant (OR: 1.02, 95% CI: 0.99–1.04). Participants who had experienced widowhood, divorce, or separation (OR: 1.03, 95% CI: 1.00–1.06), along with those in the age group of 18–44 years (OR: 1.05, 95% CI: 1.02–1.08), showed a stronger link between serum 25(OH)D levels and SUI risk.
Subgroup analyses of the association between 25(OH) D levels and SUI risk.

25(OH)D: 25-hydroxyvitamin D; SUI: stress urinary incontinence; OR: odds ratio; CI: confidence interval.
RCS
Figure 2 depicts the correlation between serum 25(OH)D levels and SUI risk, as examined using an RCS model. A nonlinear relationship was identified, with both overall association and the nonlinear component reaching statistical significance (p < 0.001 and p = 0.012, respectively). An inflection point was observed at a serum 25(OH)D level of 73.8 nmol/L (OR = 0.95, 95% CI: 0.92–0.99). This indicated a nonlinear association between serum 25(OH)D level and SUI risk, which reflects an underlying dose–response pattern. When serum 25(OH)D levels were <73.8 nmol/L, increases in the serum 25(OH)D level were associated with only a marginal SUI risk reduction. At serum 25(OH)D levels higher than the threshold of 73.8 nmol/L, the SUI risk decreased by approximately 5% for every unit rise in the serum 25(OH)D level (OR = 0.95).

RCS plot showing the correlation between serum 25(OH)D levels and SUI risk. RCS: restricted cubic spline; 25(OH)D: 25-hydroxyvitamin D; SUI: stress urinary incontinence.
Discussion
This study analyzed data obtained from the NHANES 2005–2018 database, which included serum 25(OH)D measurements for 13,768 adult women in the US. The study also incorporated comprehensive data from participants who reported experiencing SUI during physical exertion. Initial unadjusted results (model 1) suggested a positive association between higher serum 25(OH)D levels and SUI risk. However, after adjustment for potential confounders in models 2 and 3, this relationship was reversed, indicating that higher serum 25(OH)D levels were associated with a reduced SUI risk.
For categorical analysis, serum 25(OH)D levels were stratified into quartiles. Weighted multivariate logistic regression revealed that after adjustment for all confounders, the highest vitamin D quartile demonstrated a significant inverse association with SUI risk. On further evaluation of potential effect modifiers, smoking status was found to significantly modify this association (p for interaction <0.01), while other factors—including race, educational level, marital status, and alcohol consumption—did not exhibit significant effect modification (p > 0.05). RCS modeling confirmed a nonlinear dose–response relationship (p = 0.012), suggesting a biologically plausible association.
These findings are consistent with those of previous research.10,12,14 Stafne et al. reported an elevated SUI risk at serum 25(OH)D levels <50 nmol/L. This was supported by a 60-participant randomized controlled trial (RCT) demonstrating the therapeutic benefits of vitamin D supplementation, particularly in premenopausal women with SUI. Moreover, Sharma et al. observed that 75% of women with SUI had vitamin D insufficiency. 14
The two forms of vitamin D—ergocalciferol (vitamin D2) and cholecalciferol (vitamin D3)—are metabolized to form 25(OH)D. Vitamin D2 is derived from ergosterol in fungi and plants exposed to ultraviolet B (UVB) radiation.15,16 Vitamin D3 originates from two sources: cutaneous synthesis via solar UVB-induced thermal photochemical conversion17,18 and dietary intake from fish. 19 The hepatic 25-hydroxylase pathway metabolizes vitamin D, with enzyme activity present in both mitochondria and microsomes. The mitochondrial isoform includes CYP27A1, a critical regulator of circulating serum 25(OH)D levels. 20 As a mediator, the vitamin D receptor (VDR) acts as a nuclear transcription factor that mediates the genomic effects of vitamin D. 21 VDR activation promotes neural stem cell proliferation while suppressing the mitogen-activated protein kinase signaling pathway, thereby attenuating neuroinflammatory responses.22,23
The underlying mechanism linking serum 25(OH)D level and SUI pathogenesis in women remains unclear. SUI pathogenesis involves multiple factors, including urethral anatomy, periurethral musculature, and neural structures.24–28
Urethral integrity, surrounding muscle function, and pelvic floor innervation constitute the key elements of SUI pathophysiology. 2 Its active metabolite, 1,25-dihydroxyvitamin D (1,25(OH)2D), activates VDR to modulate intracellular signaling pathways to regulate transcriptional programs governing myocyte differentiation and proliferation. 29 The VDR binds to calcitriol with high affinity, leading to increased intramuscular calcium content. 30 Through nongenomic pathways, it promotes calcium ion influx into the cells, which is essential for proper muscle contraction and strength. 31 Moreover, 1,25(OH)2D-VDR–mediated signaling mitigates muscle wasting through inhibition of the renin–angiotensin cascade. 32
Our study makes three key contributions to the existing literature. First, we investigated the correlation between 25(OH)D level and SUI risk in women aged ≥18 years, a demographic inadequately represented in previous studies. Second, by analyzing nationally representative data from seven NHANES cycles, we provided robust evidence regarding this association in US adult women. Third, we employed advanced statistical approaches—including multivariable regression, weighted logistic regression, subgroup analyses, and RCS—to rigorously control for potential confounding variables.
These findings suggest a plausible association between serum 25(OH)D levels and SUI risk; however, further studies are required to establish causality.
Potential clinical implications of vitamin D screening or supplementation for SUI prevention include the following: 1. Vitamin D level could serve as a potential indicator associated with SUI risk in women; 2. This study may have paved the way for future large-scale RCTs to further investigate the potential role of vitamin D on SUI; and 3. It provides a new potential strategy for preventing SUI among adult women in the US. Recommendations are as follows: 1. Female patients should ensure sufficient daily vitamin D intake; 2. The vitamin D intake should be optimized with the aim of achieving serum levels above the identified threshold to maximize potential benefits.
Conclusion
A crucial role of 25(OH)D level in predicting SUI risk was demonstrated in the current study; a 25(OH)D level ≥73.8 nmol/L corresponds to a decreased incidence of SUI in women. Nevertheless, large-scale prospective studies are warranted to validate these findings.
Footnotes
Acknowledgements
We acknowledge the use of the DeepSeek AI tool for assistance in English editing and refining of the manuscript text. This AI tool was used solely for language enhancement purposes, and all scientific content as well as intellectual insights remain the sole responsibility of the authors.
Author contributions
Feng Wu: Writing–original draft, project administration, investigation, formal analysis, validation, software, methodology, and data curation. Mingguo Li: Investigation, validation, and writing–review & editing. Qing Zhao: Investigation and validation. Tingjiang Guo: Validation and formal analysis. Zhenliang Pan: Conceptualization, methodology, writing–review & editing, supervision, writing–review & editing, project administration, and funding acquisition. All authors have read and agreed to the submitted version of the manuscript.
Consent for publication
Not applicable.
Data availability statement
Declaration of conflicting interests
The authors declare that there are no conflicts of interest regarding the publication of this paper.
Ethical approval and consent to participate
This study used publicly available data from the National Health and Nutrition Examination Survey (NHANES), conducted by the National Center for Health Statistics (NCHS). NHANES protocols were approved by the Research Ethics Review Board of the NCHS. This analysis was based on de-identified, publicly available data; therefore, additional institutional ethical approval was not required. All patients/participants provided written informed consent to participate in this study.
Funding
The authors received no financial support for the research, authorship, and/or publication of this study.
