Abstract
Objective
To assess the diagnostic accuracy of sputum smear microscopy compared with culture in patients who were already confirmed positive for tuberculosis by Xpert Mycobacterium tuberculosis/rifampicin assay in Tanzania.
Methods
A cross-sectional study was conducted across five tuberculosis clinics in Dar es Salaam. Adults (aged 18–80 years) with symptoms of tuberculosis and a positive Xpert Mycobacterium tuberculosis/rifampicin assay result provided additional sputum specimens for smear microscopy using Ziehl–Neelsen staining and culture on Löwenstein–Jensen medium, processed at the Central Tuberculosis Reference Laboratory, Muhimbili National Hospital.
Results
We enrolled 200 participants, with a median age of 35 (interquartile range: 27–46) years; 74.5% of them were male. Using culture as the reference, Ziehl–Neelsen smear microscopy showed a sensitivity of 86.6% and a specificity of 40.7%. The Xpert Mycobacterium tuberculosis/rifampicin assay showed higher diagnostic accuracy. Notably, 43% of smear-negative cases were culture-positive, indicating that smear microscopy alone led to a substantial proportion of missed diagnoses. The Ziehl–Neelsen smear’s receiver operating characteristic curve had an area under the curve of 0.64, indicating moderate performance.
Conclusion
This study underscores the limitations of smear microscopy and highlights the operational utility of Xpert Mycobacterium tuberculosis/rifampicin assay. In regions where Xpert Ultra remains inaccessible, combining Xpert with culture selectively may enhance detection in low-resource settings.
Background
Tuberculosis (TB) remains the leading cause of death worldwide among diseases caused by a single infectious pathogen. In 2022, approximately 7.5 million individuals globally received a TB diagnosis, with an estimated 1.3 million fatalities resulting from the disease. 1 Tanzania is listed among the 30 nations bearing the most substantial TB burden. Nevertheless, significant advancements have been made; by 2021, the incidence of TB in the country had decreased by 32% relative to 2015, positioning Tanzania as one of the only three high-burden nations progressing toward the End TB Strategy targets. 2 In the same year, 87,415 TB cases were reported nationwide, with Dar es Salaam, Tanzania’s largest commercial hub, accounting for approximately 17% of the total cases. 3
For decades, sputum smear microscopy with acid-fast bacillus (AFB) staining was the standard method for diagnosing TB. Although this method offers rapid results and is inexpensive, its ability to detect TB, especially when bacterial counts are low, is limited.4,5 Culture testing for Mycobacterium tuberculosis (MTB) offers higher accuracy, but the trade-off is extensive time and resources; results can take weeks, and the process requires well-equipped laboratories. 6 The Xpert® Mycobacterium tuberculosis/rifampicin assay (Xpert MTB/RIF; Cepheid, Sunnyvale, USA), introduced in Tanzania in 2012 as part of the national diagnostic algorithm, has significantly improved TB diagnostics. It delivers faster, more sensitive results and allows for earlier treatment decisions.7–10
Despite its limitations, smear microscopy plays a key role, especially where resources are limited. It can detect TB in 58%–68% of cases and provides results within an hour.11,12 Acknowledging the demand for improved diagnostic tools, the World Health Organization (WHO) endorsed the Xpert MTB/RIF assay in 2010.13,14 This molecular test can detect MTB and assess RIF resistance within 2 h. Studies show that the assay has a sensitivity of 83%–92% in culture-confirmed patients and approximately 67% in those who test negative by smear, with a specificity of 97%–99%. 14 For all presumptive TB cases in Tanzania, the Xpert MTB/RIF assay is recommended as the primary diagnostic tool. 15 However, not all health facilities have the means to use it consistently, facing ongoing challenges such as equipment breakdowns, maintenance gaps, and intermittent cartridge supply, especially in rural or underfunded areas. Because of these limitations, smear microscopy remains widely used. Understanding the practical performance of these diagnostic tools in real-world Tanzanian settings is essential. Evaluating their advantages and limitations can help shape evidence-based, effective TB control strategies. 16 The objective of this study was to assess the diagnostic accuracy of sputum smear microscopy compared with culture in patients with TB confirmed by the Xpert MTB/RIF assay in Tanzania.
Methods
Study design and area
This cross-sectional study was conducted among individuals with presumptive TB attending five clinics in Dar es Salaam: Mwananyamala Regional Referral Hospital (MRRH), Buguruni Hospital, Tandale Hospital, Sinza Palestina Hospital, and Mbagala Rangi-Tatu. These clinics, representing diverse districts within Dar es Salaam, are major contributors to Tanzania’s national TB caseload, with Dar es Salaam accounting for 15% of all cases.
Study population
Eligibility for study enrollment included adult patients (≥18 years of age) reporting a minimum 2-week history of symptoms such as fever, cough, or weight loss.
Sample size estimation
Using Leslie Kish’s formula and considering a 15% prevalence of TB in the Dar es Salaam region, the minimum required sample size was determined to be 195 patients. 3
Study enrollment and inclusion/exclusion criteria
For study participation, patients were required to provide informed written consent and test positive for active TB using the Xpert MTB/RIF assay. Participants meeting the eligibility criteria were consecutively enrolled as they presented to the clinics and were asked to provide sputum samples for smear microscopy and culture. Individuals were excluded if they had commenced anti-TB treatment for more than 14 days or were unable to produce a sputum specimen. This study was conducted in accordance with the Declaration of Helsinki (1975, as revised in 2024).
Sample collection and data collection
After obtaining signed informed consent, participants who tested positive for TB using the Xpert MTB/RIF assay were enrolled. Sociodemographic details, including age, sex, level of education, occupation, and marital status, were collected through structured interviews. Clinical data were also recorded, covering symptoms at presentation, body measurements, human immunodeficiency virus (HIV) infection status, other existing health conditions, and any prior history of TB. Each participant provided a sputum sample in a sterile specimen container. Specimens were maintained at 28°C and transported on the day of collection to the Central Tuberculosis Reference Laboratory (CTRL) for culture using Löwenstein–Jensen (LJ) solid media.
At CTRL, samples were first prepared for Ziehl–Neelsen (ZN) staining to detect AFB. After completing all aseptic procedures, one aliquot of each sputum sample was smeared, air-dried, and heat-fixed. Slides were covered with 1% carbol fuchsin, gently heated to steaming as per protocol, rinsed, decolorized with 25% sulfuric acid, counterstained with 0.1% methylene blue, and viewed under oil immersion (100×). Bacillary density was categorized as scanty (1–9 AFB per 100 fields), 1+, 2+, or 3+, following the World Health Organization (WHO) guidelines.
A second aliquot of each sputum sample was decontaminated and inoculated onto glycerol-enriched LJ slants. The inoculated media were incubated at 37°C for up to 8 weeks, with weekly inspections for growth. The presence of typical rough, buff colonies was recorded as positive growth.
Data analysis
All statistical analyses were performed using Stata version 15 (StataCorp LLC, College Station, TX, USA). Graphical representations were created using GraphPad Prism version 9.0 (GraphPad Software, San Diego, CA, USA). Descriptive statistics were used to summarize the sociodemographic and clinical characteristics of the study participants. Continuous variables were reported as medians with interquartile ranges (IQR), while categorical variables were presented as frequencies and percentages.
The diagnostic performance of AFB smear microscopy was evaluated against sputum culture, which served as the gold standard reference. Diagnostic accuracy was assessed using key parameters, including sensitivity, specificity, positive predictive value (PPV), and negative predictive value (NPV). Overall diagnostic accuracy was calculated as the proportion of correctly identified true positives and true negatives relative to the total number of cases. Each of these indicators was accompanied by a 95% confidence interval (CI) to estimate precision.
To examine the association between the proportion of culture-positive cases and the AFB smear grading (zero, scanty, 1+, 2+, or 3+), a chi-squared trend test was performed. Subsequently, a linear regression model was used to describe the quantitative nature of this trend. The level of agreement between AFB smear microscopy and sputum culture results was assessed using Cohen’s kappa coefficient (κ), with values interpreted according to standard guidelines (e.g. Landis and Koch). For all inferential analyses, a p-value of <0.05 was considered statistically significant. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines, as outlined by von Elm et al. 17 To minimize potential sources of bias, participants were enrolled consecutively, standardized laboratory protocols were applied for specimen handling and analysis, and data were reported in accordance with the STROBE guidelines to enhance transparency.
Results
Sociodemographic characteristics
The study successfully enrolled 200 participants. The median age was 35 (IQR: 25–46) years. Nearly half of the participants (93) were single. Overall, 60% of the patients reported having at least primary education, while 7% had no formal education. The majority of the participants (58%) had a normal body mass index (BMI), with a median BMI of 19.9 (IQR: 17.3–22.0) kg/m2 (Table 1).
Sociodemographic and clinical attributes of Xpert MTB/RIF assay–confirmed TB patients in Dar es Salaam clinics (n = 200).

IQR: interquartile range; BMI: body mass index; HIV: human immunodeficiency virus; TB: tuberculosis; Xpert MTB/RIF: Xpert Mycobacterium tuberculosis/rifampicin.
Regarding sociodemographic characteristics, 28% of the participants reported consuming alcohol, and 23% reported smoking. Given that TB is an airborne infectious disease, participants were asked whether they resided with other individuals. Only 20% reported living alone. Among those living with others, a slightly more than half (52.5%) of the participants resided with more than three household members. Fourteen 14 participants reported that their household members exhibited symptoms similar to those of the index patient, with the most common symptoms being cough (100%), fever (36.7%), and night sweats (28.6%). The TB clinic team was notified to conduct contact tracing, particularly for symptomatic household members.
Sputum smear microscopy, Xpert MTB/RIF, and culture
Laboratory analysis of sputum smear showed that 22% of the participants tested negative for AFB on microscopy (Table 2). The majority of sputum smear results (45%) showed 1+ AFB per 100 fields. All samples were inoculated onto LJ medium; a total of 134 samples (67%) were culture-positive, and 29.5% were culture-negative after up to 8 weeks of incubation. The remaining 3.5% of the samples were contaminated, and no growth was detected. Among 23 patients coinfected with HIV, 16 (69.5%) were smear-positive, whereas 19 (82.6%) were culture-positive. Four 4 HIV coinfected participants whose sputum smears were negative yielded positive culture results.
Laboratory results of sputum sample analysis (n = 200).
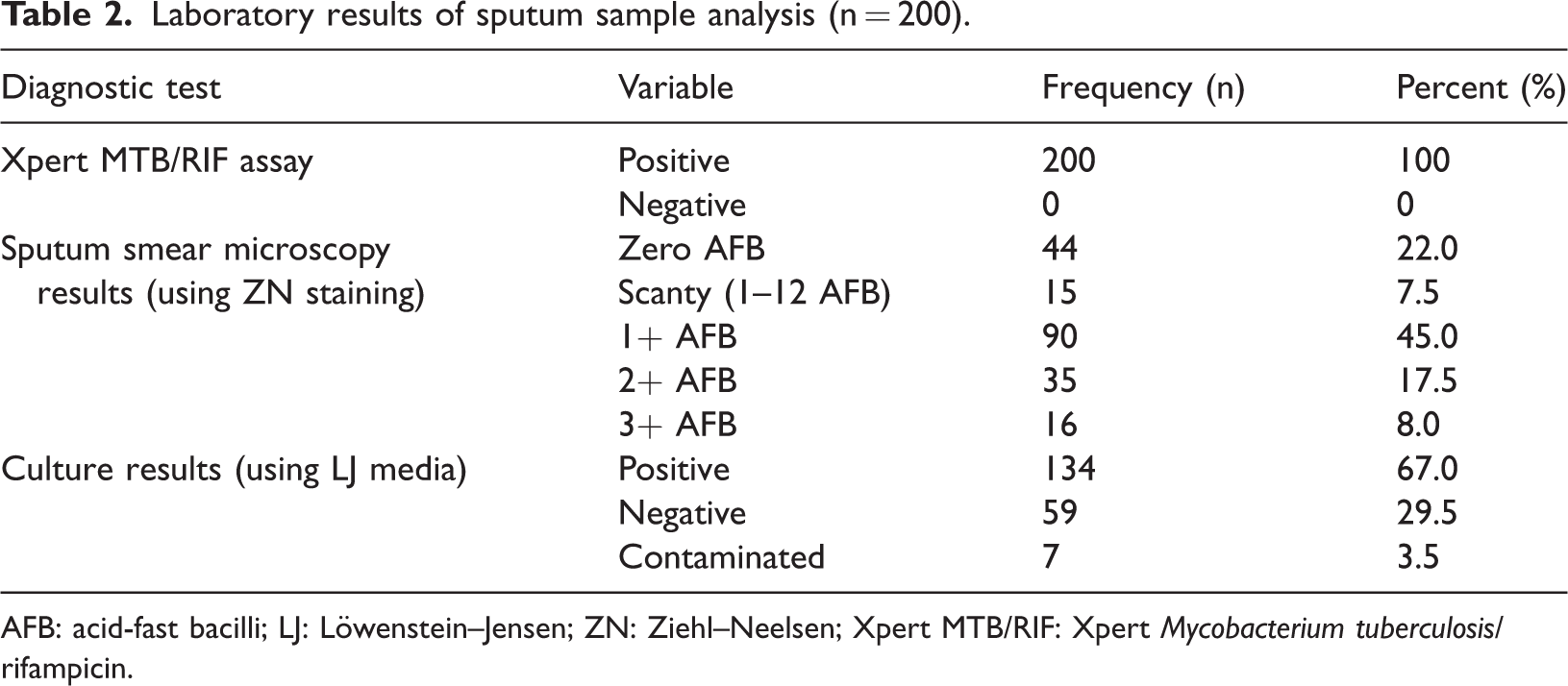
AFB: acid-fast bacilli; LJ: Löwenstein–Jensen; ZN: Ziehl–Neelsen; Xpert MTB/RIF: Xpert Mycobacterium tuberculosis/rifampicin.
Of the 156 patients who tested positive on smear microscopy, 116 (76.8%) were also confirmed positive by culture (Table 3). Among the 42 participants with negative smear results, 18 (42.9%) yielded positive culture outcomes. Considering the Xpert MTB/RIF assay results, 78% of those who tested positive were also smear-positive, and 69.4% had culture-confirmed TB. Overall, 60.1% of the Xpert MTB/RIF–positive samples were both smear- and culture-positive. Interestingly, 12.4% of the Xpert MTB/RIF–positive cases were negative by both smear and culture. Notably, culture was positive in approximately 43% of the participants who had negative smear results, while 23% of smear-positive individuals did not undergo culture confirmation.
Comparison of sputum AFB smear microscopy and sputum culture using Löwenstein–Jensen media.

AFB: acid-fast bacilli; TB: tuberculosis.
Figure 1 shows the relationship between smear grading for AFB and TB culture results. The proportion of samples yielding positive TB culture increased with higher smear grades for AFB. When the smear grade for AFB was zero, the proportion of culture-positive samples was 42.9%, increasing to 60.0% when the smear grade was scanty. Further increases to 78.2% and 75.8% were observed for smear grades of +1 and +2, respectively. The highest proportion of culture-positive samples (87.5%) was observed when the smear grade for AFB was +3. Assessment of these variations using a chi-squared trend test demonstrated a significant linear trend (χ2trend(1) =17.18, p < 0.001).

Frequency of the relationship between sputum smear grading for AFB and sputum culture. AFB: acid-fast bacilli.
Figure 2 illustrates the fitted trend line for the proportion of TB culture-positive samples according to AFB smear grade, using a linear regression model. On an average, each increment in smear grade for AFB was associated with a 10.5% increase in the proportion of culture-positive samples, as described by the following equation: % positive culture = 37.34 + 10.5 × smear grade for AFB.

Fitted trend line for the variation in proportions of TB culture-positive samples with the smear grade for AFB using a linear regression model. AFB: acid-fast bacilli; TB: tuberculosis.
Diagnostic performance of MTB smear microscopy
The diagnostic performance of the MTB smear microscopy was evaluated against sputum culture, which served as the gold standard. The relationship between sensitivity and specificity was examined, and a receiver operating characteristic (ROC) curve was plotted. This analysis yielded an area under the curve (AUC) of 0.636 (95% CI: 0.547–0.726; p = 0.003), as shown in Figure 3.

ROC curve assessing the diagnostic accuracy of MTB smear microscopy and sputum culture for the diagnosis of TB. ROC: receiver operating characteristic; MTB: Mycobacterium tuberculosis; TB: tuberculosis.
The diagnostic performance characteristics of ZN smear microscopy were determined using sputum culture as the reference standard. Results showed a sensitivity of 86.6% and a specificity of 40.7%, with an overall diagnostic accuracy of 72.5% (Table 4). The test yielded a PPV of 76.8% and an NPV of 57.1%. The calculated positive likelihood ratio (LR) was 1.46 (95% CI: 1.17–1.82), and the negative LR was 0.33 (95% CI: 0.19–0.56). To assess the level of agreement between smear microscopy and culture results, a comparative analysis was performed. The observed agreement between the two methods showed a statistically significant difference (p < 0.0001), as evidenced by a Cohen’s kappa coefficient of 0.296 (95% CI: 0.15–0.44).
Diagnostic performance of MTB sputum smear microscopy compared with culture.
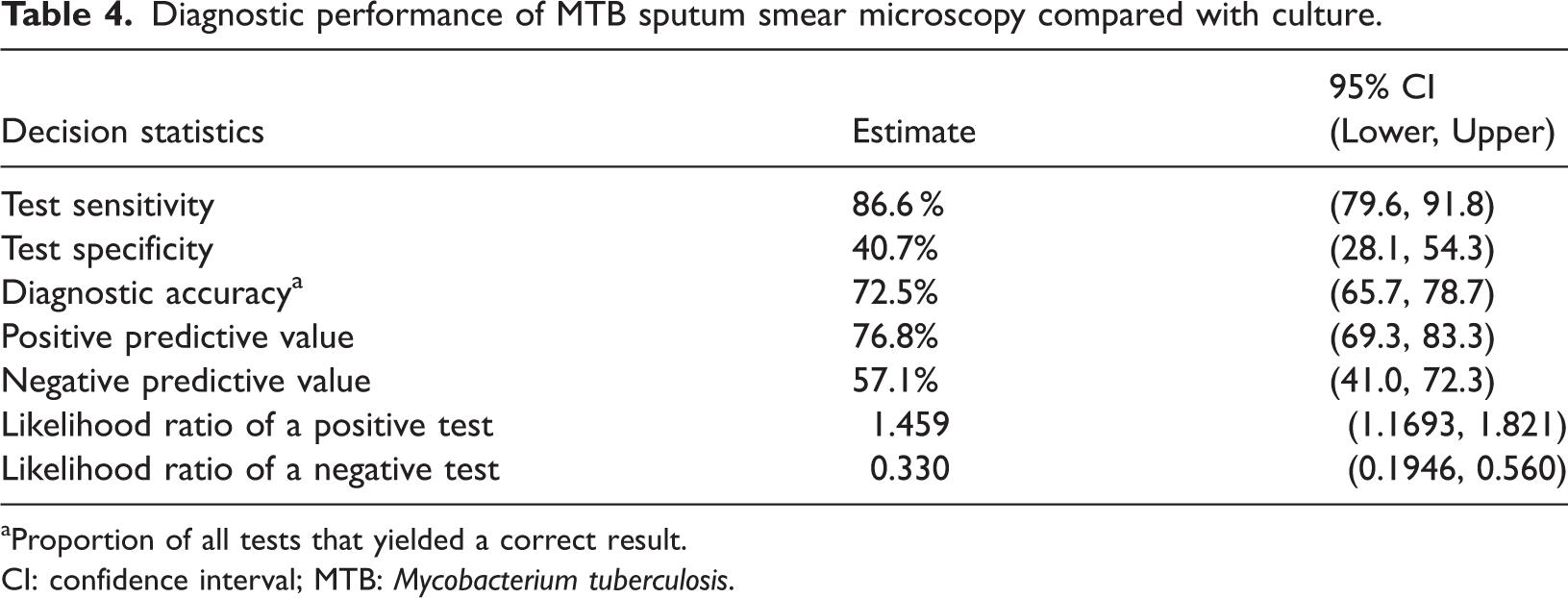
Proportion of all tests that yielded a correct result.
CI: confidence interval; MTB: Mycobacterium tuberculosis.
Discussion
This study evaluated the diagnostic performance of sputum smear microscopy compared with conventional culture among 200 patients in Tanzania, all of whom were confirmed positive for MTB using the Xpert MTB/RIF assay; the accuracy of the Xpert assay itself was not assessed. When benchmarked against sputum culture, sputum smear microscopy exhibited moderate diagnostic performance, with a sensitivity of 86.6% but a low specificity of 40.7%. Nearly half (43%) of the smear-negative samples were culture-positive, indicating that smear microscopy frequently misses active TB cases. The overall agreement between the Xpert MTB/RIF assay and sputum culture was 69%, suggesting that although the Xpert assay is highly sensitive, it may yield false-positive results in certain cases. Furthermore, a Cohen’s kappa coefficient of 0.296 between smear microscopy and culture results indicates weak concordance, supporting the need for a multi-modality approach in TB diagnosis. The ROC curve for smear microscopy yielded an AUC of 0.636, reflecting only moderate discriminative ability.
Although smear microscopy demonstrated acceptable sensitivity, its specificity and predictive values were relatively limited. These results are contradictory to the findings of Umair et al., who reported moderate sensitivity and higher specificity when the Xpert MTB/RIF assay was used as the comparator. 18 Similarly, Chadha et al. reported lower sensitivity (46.2%) but much higher specificity (99.3%). 19 A meta-analysis by Rahmati et al. estimated the combined sensitivity and specificity of smear microscopy versus culture at 75.1% and 93.9%, respectively, with studies involving fewer than 200 samples reporting a specificity of approximately 88.9%, substantially higher than that (40.7%) observed in our study. 20 Despite this, our observed sensitivity of 86.6% exceeded the meta-analytic estimate, suggesting variations due to sample handling, patient selection, or study context.
The negative LR of 0.33 in our study indicates that a negative smear result reduces, but does not eliminate, the likelihood of TB. This is consistent with the results of Umair et al., who reported a similar negative LR of 0.4. 18 The ROC AUC of 0.64 also aligns with their findings, reflecting that smear microscopy can moderately differentiate between TB-positive and TB-negative cases. 18 Even this moderate performance can be clinically useful when followed by confirmatory testing, particularly in settings where access to advanced diagnostics is limited.
Among participants who tested positive using the Xpert MTB/RIF assay, 78% also had positive smear results. This is comparable to the 74.6% overlap observed by Mavenyengwa et al. 21 Our study found that 69% of the participants who tested positive on the Xpert MTB/RIF assay were also confirmed by culture, indicating reasonable but imperfect alignment between the molecular test and gold standard culture. Importantly, unlike sputum culture, which can be compromised by contamination, the Xpert MTB/RIF assay remains unaffected and therefore provided reliable diagnostic confirmation in our cohort. This represents a particular advantage in resource-limited settings, where maintaining optimal culture conditions can be challenging. According to Boehme et al., the Xpert MTB/RIF assay demonstrated a sensitivity of 98.2% in participants who were smear-positive and culture-confirmed for MTB, whereas a lower sensitivity of 72.5% was observed in smear-negative but culture-positive cases. 7 Notably, repeating the Xpert MTB/RIF assay can improve detection rates, particularly among smear-negative individuals.7,22
In our cohort, 43% of the smear-negative patients were culture-positive, exceeding the 21% reported by Rimal et al., and the 34% reported by Rasool et al.23,24 This finding may reflect the reliance of smear microscopy on a high bacillary load (typically ≥5000 bacilli/mL) for detection. 25 Factors such as sputum quality also influence microscopy results, whereas molecular tests such as the Xpert MTB/RIF assay are less affected by these variables.26,27 Although smear-positive cases are often considered more infectious, evidence suggests that 10%–20% of TB transmission originates from smear-negative individuals, highlighting the importance of detecting low-burden cases to curb community spread.28,29
Unlike culture and smear microscopy, which detect only viable bacilli, the Xpert MTB/RIF assay identifies both viable and nonviable MTB by amplifying bacterial DNA. 30 This molecular detection capability contributes to its high reported sensitivity and specificity (often exceeding 90%), although it may yield false positives in patients with recent or inactive infection.31–33 For the detection of viable TB organisms, culture remains the reference method, requiring a minimum concentration of 10 live bacilli/mL to produce growth. 34 As suggested by Theron et al., high cycle threshold values in the Xpert MTB/RIF assay indicate low bacterial loads, increasing the likelihood of discordant positive Xpert MTB/RIF and negative culture results. 35 This phenomenon is particularly common among newly diagnosed individuals with low bacillary burden or minimal lung involvement. 36
These findings have important implications for TB diagnostic strategies in resource-limited settings. Although the introduction of the Xpert MTB/RIF assay has improved TB detection, particularly among smear-negative individuals, its implementation in Tanzania continues to face practical challenges, including reliance on the earlier MTB/RIF platform due to financial and logistical constraints. This study did not evaluate the diagnostic accuracy of the Xpert MTB/RIF assay, as all participants were positive by design; therefore, the results should not be interpreted as an assessment of Xpert MTB/RIF performance. The observed 69% concordance between the Xpert MTB/RIF assay and culture underscores the need for diagnostic strategies that consider test performance, patient characteristics, and operational realities. Rather than proposing new diagnostic tools, this study emphasizes the importance of addressing implementation gaps and aligning recommendations with what is feasible in routine clinical practice. Our findings support a pragmatic, tiered diagnostic approach that enhances case detection while remaining adaptable to local resource constraints.
This study has several limitations. By excluding participants who tested negative on the Xpert MTB/RIF assay, we could not evaluate its false-negative rate. Prior studies suggest that 4%–5% of smear-positive patients may test negative using the Xpert MTB/RIF assay. 37 Additionally, we did not perform MTB drug-susceptibility testing, which limits the assay’s potential for identifying multidrug-resistant TB. The relatively short study period, modest sample size (n = 200), and potential selection bias may also limit generalizability. Furthermore, culture-negative results do not necessarily invalidate Xpert MTB/RIF positivity, particularly in cases with low bacillary load or recent TB exposure.
Conclusion
This study reaffirms existing knowledge on the diagnostic performance of sputum smear microscopy, Xpert MTB/RIF assay, and culture. Its value lies in providing context-specific, operational data from routine healthcare settings in Tanzania, which may guide implementation strategies under resource constraints. Although a tiered diagnostic approach may enhance case detection, considerations of cost-effectiveness and feasibility remain critical. Importantly, the study highlights persistent gaps in diagnostic access despite WHO’s recommendation of the Xpert MTB/RIF Ultra assay, reinforcing the need for pragmatic deployment strategies tailored to available infrastructure.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605251390703 - Supplemental material for Diagnostic accuracy of sputum smear microscopy compared with culture in patients with tuberculosis confirmed by Xpert Mycobacterium tuberculosis/rifampicin assay in Tanzania: A cross-sectional study
Supplemental material, sj-pdf-1-imr-10.1177_03000605251390703 for Diagnostic accuracy of sputum smear microscopy compared with culture in patients with tuberculosis confirmed by Xpert Mycobacterium tuberculosis/rifampicin assay in Tanzania: A cross-sectional study by Sabina Mugusi, Jacob Lema, Peter Ponsian, Doreen Kamori, Happiness Kimambo, Paul Masanja, Avitus Anthony, Mwemezi Ngemera and Amos Kahwa in Journal of International Medical Research
Footnotes
Acknowledgments
We extend our sincere gratitude to all study participants for their contributions. We also appreciate the dedication and support of the healthcare personnel at the participating study sites, particularly for their assistance with data collection and sample processing. Additionally, we acknowledge the use of AI-assisted language editing to enhance the clarity and readability of this manuscript.
Authors’ contributions
SM contributed to the conceptualization and study design, performed data analysis, interpreted the findings, and drafted the initial manuscript. PM, HK, and AA coordinated activities at the study sites and collected data. PP contributed to the statistical analysis and interpretation of the findings. JL, DK, MN, and AK contributed to data interpretation and provided substantive feedback to strengthen the manuscript. All authors participated in the final review and approved the submission.
Availability of data and materials
The datasets generated and analyzed during the current study are available from the corresponding author on reasonable request.
Consent for publication
Not applicable.
Declaration of conflicting interests
The authors declare that there are no conflicts of interest, financial or otherwise, related to this study.
Ethics approval and consent to participate
This study was conducted in accordance with the Declaration of Helsinki (1975, as revised in 2024). Ethical approval was obtained from the Institutional Review Board of the National Institute for Medical Research (NIMR), reference number NIMR/HQ/R.8a/Vol.IX/3888. All participants provided written informed consent prior to enrollment. All patient data were de-identified to ensure confidentiality.
Funding
This work was funded by Cerba Xpert.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
