Abstract
Objectives
This study aimed to compare growth, development, quality of life, and attention abilities between Chinese children with and without obstructive sleep apnea.
Methods
A cross-sectional study was conducted at the 980th Hospital, Joint Logistics Support Force. Validated scales, including the Pupil Rating Scale, Inventory of Subjective Life Quality, Self-Rating Anxiety Scale, Parent Symptom Questionnaire, and Continuous Performance Task, were used to assess children with obstructive sleep apnea and healthy controls.
Results
Children with obstructive sleep apnea (n = 67) showed significantly poorer outcomes than controls (n = 60). They exhibited lower Pupil Rating Scale and Inventory of Subjective Life Quality scores (p < 0.001), higher anxiety levels (p < 0.001), abnormal learning (p = 0.028), increased impulsivity and hyperactivity (p = 0.003), and an abnormal hyperactivity index (p = 0.004). Sustained visual (p = 0.010) and auditory attention (p < 0.001) were also impaired.
Discussion
This study highlighted the significant negative impact of obstructive sleep apnea on children’s growth, development, and cognitive functions. Further research is warranted to examine the broader implications of obstructive sleep apnea on children’s well-being across diverse populations.
Conclusions
Children with obstructive sleep apnea demonstrate significant impairments in growth, quality of life, and cognitive functions compared with controls.
Introduction
Obstructive sleep apnea (OSA) is a breathing disorder that manifests as recurrent complete and/or partial upper airway obstruction during sleep. 1 Common complications of OSA include chronic snoring symptoms, disturbed sleep patterns, and daytime neurobehavioral problems. 1 In children, the prevalence of snoring ranges from 3% to 12%, 1 and the prevalence of OSA (diagnosed with an obstructive apnea–hypopnea index (OAHI) ≥1/h)1,2 ranges from 1% to 6%.1,3–7 The primary risk factor for OSA in children is adenotonsillar hypertrophy,8–10 whereas additional risk factors include snoring for >3 months,11,12 male sex,11,13 obesity,10,13 and lack of breastfeeding.14,15 Therefore, the management of pediatric OSA primarily involves adenotonsillectomy based on the most common etiology and also includes intranasal steroids, montelukast, and continuous positive airway pressure. 1 The main complication of OSA in children is growth restriction. 16
From a developmental perspective, OSA in 5-year-old children can lead to poor executive functions, reduced memory skills, and low intelligence levels. 17 These changes result in poor academic performance in school-age children. 18 OSA can also lead to anxiety, depression, and decreased quality of life.19–21 Systematic and comprehensive observational studies evaluating multiple parameters in children with OSA remain limited. Most existing studies have focused on one or more of these aspects, such as growth and development,22–24 quality of life,25–27 emotional being,19–21 and cognitive functions.17,18,28–31
Chinese (and more broadly, Asian) children tend to experience more severe OSA for a given degree of obesity compared with Caucasian (Western) children, although the overall prevalence may be lower in some Asian populations. 32 In Western children, obesity is a predominant risk factor, and the prevalence of obesity-related OSA is correspondingly higher. In Chinese children, OSA can be severe even in the absence of significant obesity, suggesting the involvement of other contributing factors.33,34 Therefore, this study was based on the hypothesis that OSA contributes to developmental impairments in Chinese children, thereby impacting their overall quality of life. This study aimed to compare the differences in growth and development, quality of life, and cognitive and attention abilities between Chinese children with OSA and healthy controls.
Materials and methods
The reporting of this study conforms to the Strengthening the Reporting of Observational studies in Epidemiology (STROBE) guidelines. 35
Study design
The original cross-sectional study included consecutive children with OSA and healthy controls who visited the Department of Otolaryngology of the 980th Hospital, Joint Logistics Support Force, China, between February 2017 and February 2018. 36 The current investigation represents a post hoc analysis of patients who underwent adenotonsillectomy in the original 2017–2018 study. Healthy children undergoing routine physical check-ups between February and August 2017 at the same hospital served as the control group. The recruitment of children in the outpatient department was entirely voluntary, with no refusals observed, as the trial was noninvasive, required no additional visits or procedures, and was funded by the project.
Ethical considerations
This post hoc analysis adhered to the principles of the Declaration of Helsinki (1975, as revised in 2013) and was approved by the Ethics Committee of the 980th Hospital, Joint Logistics Support Force, China (No. 2021-KY-2; approved on 27 January 2021). In the original 2017–2018 study, the legal guardians of all children provided written informed consent before the study. The present post hoc analysis used only aggregated data, and the participants could not be identified in any way. All data were analyzed using a deidentified database; only the original principal investigator has access to the ID key.
Diagnosis of OSA
The presence or absence of snoring in the experimental and control groups was recorded as the chief complaint during the medical history interview with the legal guardian. The examinations and anamnesis procedures were identical for all children. The diagnostic criteria for OSA in children were based on the Third Edition of the International Classification of Sleep Disorders developed by the American Academy of Sleep Medicine. 37 At the study hospital, all children scheduled for adenotonsillectomy underwent polysomnography (PSG).
Patient eligibility criteria
The inclusion criteria for the OSA group included children aged 6–12 years with an indication for adenotonsillar surgery due to sleep snoring and mouth breathing lasting for more than 3 months. The inclusion criteria for the control group were children aged 6–12 years with no indications for adenotonsillar surgery and no snoring or mouth breathing. In both groups, children with OSA-related syndromes (such as central sleep apnea or hypopnea syndrome, craniomaxillofacial deformities, and Down syndrome), neuromuscular dysfunction, genetic diseases (such as sickle cell disease and laryngomalacia), other chronic diseases (such as nervous system diseases, cardiopulmonary dysfunction, and metabolic diseases), or mental illness were excluded from the study. When analyzing the PSG results, we referred to the American Academy of Sleep Medicine (2014), European Respiratory Society (2017), and Guidelines for the Diagnosis and Treatment of Obstructive Sleep Apnea in Children in China (2020), using the following diagnostic thresholds for OSA in children: mild, 1 time/h ≤ OAHI ≤ 5 times/h; moderate, 5 times/h < OAHI ≤ 10 times/h; and severe, OAHI > 10 times/h.
Eligibility for adenotonsillectomy
Eligibility for adenotonsillectomy was evaluated through medical history collection and physical examination before surgery. The criteria included the following: (a) recurrent acute episodes of chronic tonsillitis; (b) excessive hypertrophy of the tonsils interfering with swallowing, breathing, and vocal functions; (c) chronic tonsillitis causing injury to other organs or associated with lesions in neighboring organs; (d) tonsil hypertrophy leading to OSA; (e) adenoid hypertrophy not responding to medication; (f) severe adenoid hypertrophy; (g) complications such as otitis media, sinusitis, and posterior nostril drip; (h) occlusive nasal sound; (i) adenoid visage; and (j) wasting and developmental disorder.
Outcome measures
The collected data included age, sex, height, weight, body mass index (BMI), OAHI, learning disabilities, quality of life, anxiety, behavioral problems, and attention abilities. BMI values were converted into Z-scores to account for age, based on recent data from China. 38 Children with OSA were evaluated using questionnaires and tests by the inpatient department before surgery, while the healthy controls were assessed by the outpatient department.
Questionnaires and tests
The revised version of the Myklebust Pupil Rating Scale (PRS) was used to screen for learning disabilities. 39 The PRS is a 24-item, teacher-reported tool for assessing academic performance. 39 Scores ranged from 24 to 120 (including verbal measures (9–45) and nonverbal measures (15–75)). Learning disabilities were determined as verbal scores <20 and nonverbal scores <40. 40 The PRS was used to evaluate auditory comprehension (understanding word meaning, following instructions, comprehending class instructions, and retaining information), spoken language (vocabulary, grammar, word recall, storytelling-relating experience, and formulating ideas), orientation (judging time, spatial orientation, judging relationships, and recognizing directions), motor coordination (general coordination, balance, and manual dexterity), and personal–social behavior (cooperation, attention, organization, new situations, social acceptance, responsibility, completion of assignments, and tactfulness). The PRS scores were summarized as verbal subtotal, nonverbal subtotal, and total scores. The Chinese version of the PRS has demonstrated reliability coefficients >0.95 for both subscores. 41
The Inventory of Subjective Life Quality (ISLQ) is a 52-item self-report scale that assesses multiple domains, including family life, peer relationships, school life, living environment, self-knowledge, cognitive component, experience of depression, anxiety, physical emotions, and emotional component. Children can complete the ISLQ. Higher scores indicate a better quality of life. It has been validated in the Chinese population, demonstrating a Cronbach’s α of 0.89. 42
The Zung self-rating anxiety scale (SAS) is a widely used tool for screening anxiety based on manifestations in the cognitive, autonomic, motor, and central nervous system dimensions. It has been validated in the Chinese population, showing a Cronbach’s α of 0.931. 43 A standard cutoff of 50 was used to indicate anxiety. 44
The Conners Parent Symptom Questionnaire (PSQ) evaluates behavioral characteristics such as hyperactivity, compulsive behavior, perfectionism, playing up in class, violence and aggressiveness, mathematics difficulties, language difficulties, separation anxiety, social issues, and emotional distress. Scores ≥60 indicate potential issues (e.g. attention deficit and hyperactivity disorder). 45 It has been validated in the Chinese population, with a Cronbach’s α > 0.85. 46
Attention abilities (visual and/or auditory) were assessed using the Continuous Performance Task (CPT), 47 which evaluates sustained attention abnormalities of visual sense, auditory sense, and both visual and auditory senses.47,48 The CPT task was administered to children on computers within their classrooms. A score >60 indicates potential attention-related difficulties. 49 Each child completed a practice session until the examiner confirmed that the child could adequately understand. The task involved sequential presentation of 540 2-cm tall numbers in the center of the screen for 200 ms each, with an interstimulus interval of 1000 ms. The task was divided into three stages (4 min each). Children were instructed to press the space bar whenever the number 9 was preceded by the number 1. The event rate was 10%, maintained consistently across all three stages. The CPT generated multiple dependent measures, including reaction time for correct responses, omission error rate, commission error rate, and signal detection parameters (d’ and β).
Statistical analysis
Statistical analyses were conducted using SPSS version 22.0 (IBM, Armonk, NY, USA). The Kolmogorov–Smirnov normality test was performed on all continuous variables with a sample size >50, and the Shapiro–Wilk test was used for sample sizes of <50. Normally distributed continuous variables were expressed as mean ± SD and analyzed using Student’s t-test. Non-normally distributed continuous variables were expressed as median (interquartile range) and analyzed using the Mann–Whitney U test. Categorical data were presented as n (%) and analyzed using the chi-squared test. All statistical tests were two-tailed, and p values <0.05 were considered statistically different. The ordered logistic regression analysis model was used for further statistical analysis.
Results
Characteristics of children with OSA and healthy controls
Tables 1 and 2 summarize the characteristics of children with OSA (n = 67) and healthy controls (n = 60). No differences were observed in age, sex, BMI, and BMI Z-scores between the two groups (p > 0.05). Compared with the control group, the proportion of children with height below the 25th percentile was higher in the OSA group (14.9% vs. 0%). Similarly, the proportion of obesity was greater (22.4% vs. 11.7%) in the OSA group compared with that in the control group. Overall, children with OSA exhibited growth restriction in terms of percentile height (p = 0.036) and weight (p = 0.698) compared with healthy controls.
Comparison of different degrees of obstructive sleep apnea in children.

The “mild” group was used as the reference group.
BMI: body mass index; OAHI: obstructive apnea–hypopnea index.
The OAHI was not compared among groups because it was the grouping criterion.
Characteristics of growth in children with OSA and healthy controls.
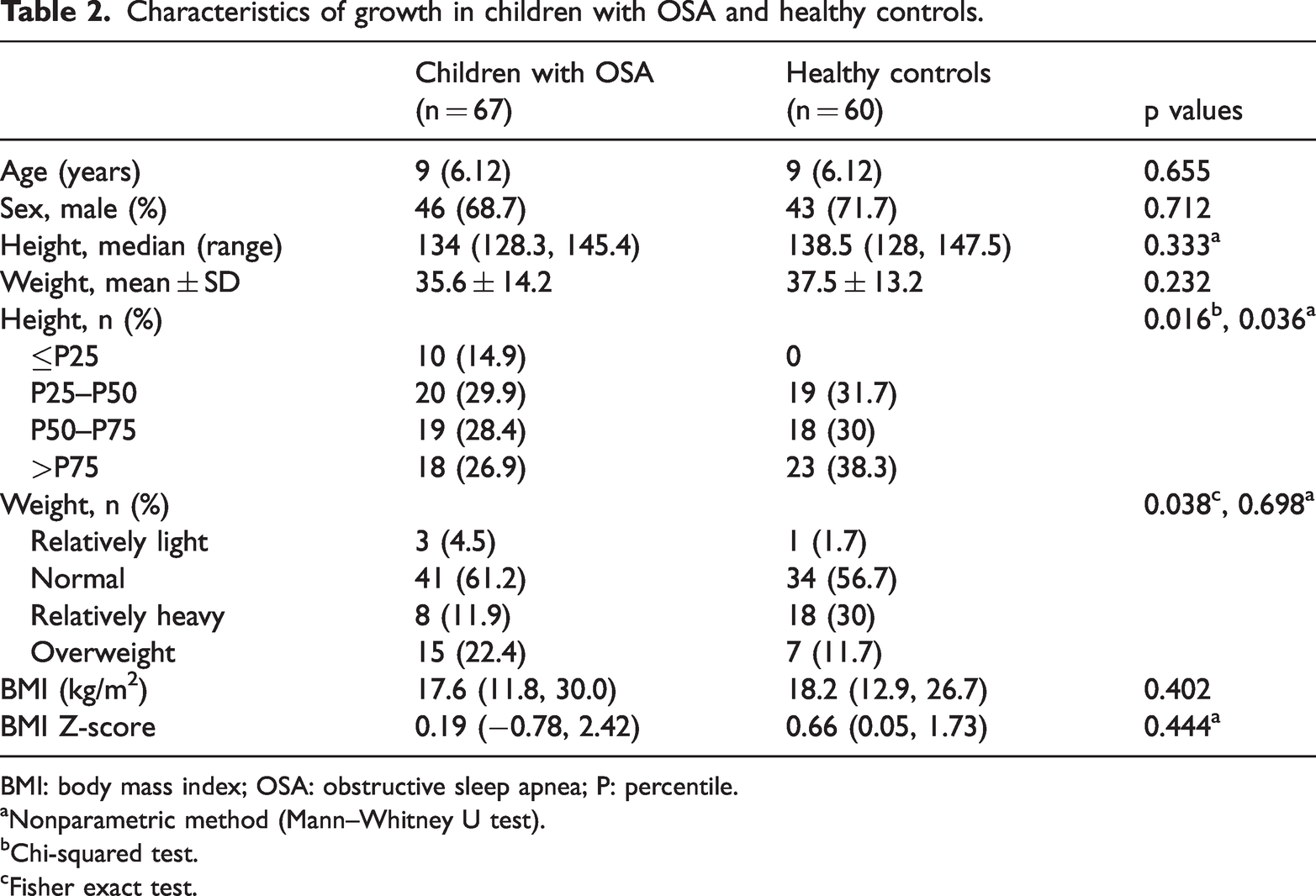
BMI: body mass index; OSA: obstructive sleep apnea; P: percentile.
aNonparametric method (Mann–Whitney U test).
bChi-squared test.
cFisher exact test.
Differences in PRS scores between children with OSA and healthy controls
Compared with the control group, children with OSA had lower total PRS scores (74.9 ± 13.1 vs. 87.9 ± 15.3, p < 0.001) as well as reduced verbal (29.2 ± 11.7 vs. 39.7 ± 13.1, p < 0.001) and nonverbal (45.7 ± 4.0 vs. 47.9 ± 5.2, p = 0.028) subscores (Table 3).
Differences in PRS scores between children with OSA and healthy controls.

OSA: obstructive sleep apnea.
Differences in ISLQ scores between children with OSA and healthy controls
As shown in Table 4, children with OSA had lower total ISLQ scores than healthy controls (45.0 ± 15.5 vs. 60.7 ±11.4, p < 0.001), along with low values for all components of ISLQ (p < 0.05). In addition, children with OSA exhibited higher levels of anxiety than healthy controls (20.5 ± 9.4 vs. 10.9 ± 7.3, p < 0.001).
Differences in the quality of life between children with OSA and healthy controls.

ISLQ: Inventory Of Subjective Life Quality; OSA: obstructive sleep apnea; SAS: self-rating anxiety scale.
Differences in Conners PSQ scores between children with OSA and healthy controls
No significant differences were observed in the total PSQ scores or in any of the six separate dimensions between the two groups (p > 0.05). Each indicator was evaluated qualitatively according to age-specific standard thresholds, with any value exceeding the standard considered abnormal. Therefore, considering the proportions of abnormality, children with OSA had a higher proportion of abnormal learning performance (35.8% vs. 18.3%, p = 0.028), impulsivity, hyperactivity (50.7% vs. 25.0%, p = 0.003), and abnormal hyperactivity index (53.7% vs. 28.3%, p = 0.004) than healthy controls (Table 5).
Differences in the PSQ between children with OSA and healthy controls.

OSA: obstructive sleep apnea.
Differences in attention abilities between children with OSA and healthy controls
As shown by the CPT values in Table 6, the OSA group demonstrated worse performance scores in visual attention ability (p = 0.010), auditory attention ability (p < 0.001), and both visual and auditory attention abilities (p = 0.002). Compared with healthy controls, a higher number of children with OSA had severe sustained visual attention abnormality (31.3% vs. 8.3%), severe sustained auditory attention abnormality (62.7% vs. 23.3%), and severe abnormality of both sustained visual and auditory attention (38.8% vs. 11.7%) (Table 6).
Differences in attention abilities between children with OSA and healthy controls.

OSA: obstructive sleep apnea.
Discussion
Most previous studies on pediatric OSA have focused on specific aspects rather than examining the comprehensive picture. In addition, evidence about OSA among Chinese children remains limited. The present study suggested significant differences in growth and development, quality of life, and attention abilities between children with OSA and healthy controls. This research is novel in its comprehensive evaluation of these interrelated parameters within a single cohort, emphasizing the multifaceted negative effects of OSA in children.
OSA is known to induce physical effects in children, particularly involving the cardiovascular system. 50 However, these effects were not examined in the present study. Nevertheless, this study demonstrated that children with OSA were shorter than healthy controls, considering the age- and sex-standardized height percentiles, as supported by the literature. 16 The American Academy of Pediatrics recognizes growth failure as a serious complication of OSA in children. 51 Furthermore, adenotonsillar hypertrophy remains a common cause of OSA in children. Enlarged adenoids and tonsils can obstruct the upper airway during sleep, leading to repeated episodes of partial or complete airway obstruction.9,11,32 Hence, adenotonsillectomy often results in catch-up growth. 23 The leading theory for growth failure in children with OSA is the increased energy expenditure during sleep. 52 However, nocturnal hypoxemia, metabolic alkalosis, 22 and systemic inflammation 24 can also result in abnormal growth. Consistent with prior studies, the present study demonstrated that OSA is more prevalent among obese children, underscoring obesity as a risk factor for the disorder.1,6,23
The PRS is a well-known tool for evaluating auditory comprehension, spoken language, orientation, motor coordination, and personal–social behavior, generating total, verbal, and nonverbal scores. 39 In the present study, children with OSA exhibited lower total, verbal, and nonverbal PRS scores, accompanied by poorer physical development. These findings are consistent with those of previous studies reporting delays in verbal skill acquisition and comprehension among children with OSA.53,54 A study showed improvements in all PRS scores after adenotonsillectomy in children with OSA. 36
In both the PSQ and CPT, a higher proportion of children in the OSA group exhibited abnormal learning, impulsivity, and hyperactivity, along with lower scores in visual and auditory attention, compared with the control group. Several studies using similar instruments have documented an association between OSA and hyperactivity55–60 as well as improvements in behavior after adenotonsillectomy, especially in terms of the mean total problem scores; the internalizing and individual syndrome scales of withdrawal, somatic complaints, anxiety/depression, attention problems, and thought problems 61 ; and oppositional behavior, cognitive problems or inattention, hyperactivity, and attention-deficit hyperactivity disorder. 62 A study revealed impaired sustained attention in children with OSA, consistent with the findings of the present study. 63 Another study revealed impaired selective and sustained attention among children with OSA. 64 Caporale et al. 28 attributed cognitive impairments in children with OSA to decreased daytime vigilance and nocturnal hypoxemia. Shpirer et al. 30 found that attention was the most affected cognitive parameter in children with OSA and correlated this deficit with hypoxemia. Conversely, a systematic review concluded that the cognitive ability driving cognitive impairment in OSA remains unclear. 65 Vaessen et al. 66 suggested that cognitive impairments in children with OSA were primarily associated with excessive sleepiness. Adenotonsillectomy has also been shown to improve behavior in children, 31 supporting the hypothesis that correcting hypoxemia may reverse cognitive impairments, as reported by Caporale et al. 28
Quality of life is a comprehensive dimension that encompasses all events and factors that influence life experiences. Our previous study, which included a relatively small sample size, focused solely on comparing quality of life and anxiety between children with OSA and healthy controls, revealing a lower quality of life and higher anxiety among children with OSA. 67 In the present study, children with OSA demonstrated lower quality of life in all aspects of the ISLQ, which is a comprehensive questionnaire examining aspects such as family life, peer relationships, school life, living environment, self-knowledge, the cognitive component, experience of depression and anxiety, physical emotion, and emotional well-being in children. 42 Studies have consistently shown a decreased quality of life in children with OSA using different assessment tools,27,68–71 as supported by a meta-analysis. 25 Factors such as poor academic performance, language impairment, lower attention, hyperactivity, and impulsivity can negatively affect the quality of life in children.25,27,68–70 Given that all these factors were present among children with OSA in this study, the finding of an unsatisfactory quality of life is unsurprising. Future studies should examine how the parent–child relationship impacts the quality of life among children with OSA. Adenotonsillectomy has been shown to reverse the impairments in OSA-specific quality of life metrics. 26 The ISLQ indicated that children with OSA had higher anxiety levels than the healthy controls, whereas the SAS indicated that the two groups had similar anxiety levels. This difference may be due to the limitations of the tools (ISLQ and SAS). The ISLQ examines experiences related to anxiety, 42 while the SAS functions as a diagnostic tool for anxiety.43,44 Therefore, the two tools assess anxiety from different perspectives using different questions. Given the emphasis on academic performance in China, pressure from legal guardians and schools can lead to performance anxiety,72,73 irrespective of the OSA status. Additional studies are needed to examine and characterize anxiety in Chinese children with OSA. Nevertheless, several studies have demonstrated an association between pediatric OSA and anxiety. 74
The impact of OSA on children has been well-documented in previous studies.17–19,21–31 Furthermore, the negative consequences of OSA on growth, quality of life, and cognitive and attention abilities in children remain underrecognized. More importantly, most studies focus only on one or a limited number of the OSA-related impacts in children. In contrast, the present study comprehensively evaluated the differences in growth and development, quality of life, and cognitive and attention abilities of Chinese children with OSA vs. healthy controls, all within the same study sample, enabling the direct observation of interrelationships among these factors. This study contributes to the existing knowledge of OSA in Chinese children and holds potential value beyond China and Asia. Naturally, there are considerable cultural, economic, genetic, medical, lifestyle, and demographic differences between Asian and Western countries (even among Asian countries),75–77 and the present study focused exclusively on Chinese children. Cultural differences in the criteria used to assess quality of life and cognitive and attentional functions exist between Western and Asian countries.78–80 Therefore, the present study’s conclusions should be validated in Western populations using the same study design, and ideally, in comparative studies involving both Asians and Western participants.
This study was a post hoc analysis of data from patients who underwent tonsillectomy in the original study. 36 All children selected as surgical candidates for adenoid and/or tonsillar hypertrophy need not fulfill the diagnostic criteria for OSA. Although adenoid and/or tonsillar hypertrophy is the primary cause of OSA in children, it is not the only contributing factor, and all children diagnosed with OSA do not meet the surgical indications. The original study compared preoperative and postoperative outcomes related to adenoids and tonsils within the same cohort of children. 36 Therefore, the children included in the present study were required to meet both the diagnostic criteria for OSA and indications for surgery.
This study has certain limitations. First, considering the relatively high prevalence of OSA, the study sample size was small and derived from a single center. Therefore, subgroup analyses of OSA based on disease severity or course could not be performed. Second, as all patients were children who met surgical indications for adenotonsillar surgery, selection bias may have been introduced. Nevertheless, PSG test parameters indicated that most of the children with OSA had mild-to-moderate illness, with only a small proportion exhibiting severe OSA. Of note, due to limited medical resources, PSG could not be conducted for children in the control group. Furthermore, because children with OSA were evaluated in the inpatient department before surgery, whereas healthy controls were assessed in the outpatient department, setting/contextual differences may have confounded the anxiety assessment results. In addition, this study used Zung’s SAS, which, despite being a widely used and well-validated tool for assessing anxiety, is not specifically designed for children. Third, OSA has been associated with depression,19–21,67 metabolic imbalances, 50 and reduced academic performance,50,64,65 but these dimensions were not assessed in the present study. Finally, due to its cross-sectional design, no cause-and-effect relationships could be established.
Conclusions
Chinese children with OSA exhibited significantly impaired growth and development, quality of life, and cognitive and attention abilities compared with healthy controls. Moreover, this study adds to the growing body of literature on OSA in Chinese children, and the results of this study can help draw attention to the adverse impacts of OSA on children in a range of aspects. Further research is required to comprehensively investigate the growth and neurodevelopmental outcomes of children with OSA in China and to determine whether these conclusions are generalizable to other populations and ethnic groups.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605251389357 - Supplemental material for Differences in attention abilities, growth, development, and quality of life between Chinese children with obstructive sleep apnea and healthy controls
Supplemental material, sj-pdf-1-imr-10.1177_03000605251389357 for Differences in attention abilities, growth, development, and quality of life between Chinese children with obstructive sleep apnea and healthy controls by Shan Shan, Li Zhang, Shuyu Wang, Shufen Xiao, Xiaoming Li, Linying Xiu, Fan Liu and Cong Xu in Journal of International Medical Research
Footnotes
Acknowledgments
None.
Authors’ contributions
Shan Shan and Shufen Xiao conducted the studies. Linying Xiu and Fan Liu participated in collecting data. Shan Shan drafted the manuscript. Linying Xiu, Fan Liu, and Cong Xu performed the statistical analysis. Shuyu Wang and Xiaoming Li participated in its design. All authors read and approved the final manuscript.
Consent to participate
Not applicable.
Consent to publish
Not applicable.
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
The authors declare that they have no competing interests.
Ethical approval
The study was based on the Declaration of Helsinki. This study was approved by the Ethics Committee of The No. 980 Hospital, Joint Logistics Support Force, PLA, China (No. 2021-KY-2). The legal guardians of all children provided written informed consent before the study.
Funding
This work was supported by the 2018 Key Project of Medical Science Research of the Provincial Health Commission of Hebei [grant number: 20180908] and the Shijiazhuang Science and Technology Bureau, Hebei Province (211200663).
Patient consent
The legal guardians of all children included in the study have provided written informed consent.
Statement of informed consent
Verbal informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
