Abstract
Objective
Elevated cerebrospinal fluid red blood cell counts in patients on antiplatelet therapy during lumbar puncture raise concerns about increased bleeding risk. This study investigated whether antiplatelet therapy potentiates bleeding from inadvertent small-vessel injury during lumbar puncture.
Methods
This retrospective, real-world study analyzed data from hospitalized patients who underwent lumbar puncture between January 2013 and December 2022. After applying exclusion criteria and data preprocessing, univariate and multivariate logistic regression analyses were conducted to identify factors associated with cerebrospinal fluid red blood cell counts, focusing on perioperative antiplatelet therapy. Additional adverse events were recorded and analyzed.
Results
Among 1677 patients included (55.22% male; median age, 53 (interquartile range, 39–67) years; median body mass index, 22.65 (interquartile range, 20.41–24.97) kg/m2; 368 (21.94%) patients receiving antiplatelet therapy), red blood cells were observed in 728 (43.4%) patients. Multivariate analysis revealed that older age (odds ratio, 1.01; 95% confidence interval, 1.00–1.01; p = 0.005) was associated with a higher likelihood of red blood cell presence, whereas antiplatelet therapy (p > 0.05) was not. The incidence of adverse events was comparable between patients receiving and those not receiving antiplatelet therapy (5.57% vs. 4.22%).
Conclusions
Perioperative antiplatelet therapy was not independently associated with an increased risk of red blood cell presence following lumbar puncture, and adverse event rates were comparable regardless of antiplatelet therapy, suggesting a favorable safety profile in real-world practice.
Keywords
Introduction
Antiplatelet therapy (APT) plays a central role in the primary and secondary prevention of atherosclerotic cardiovascular diseases by significantly reducing the risk of thrombotic events such as myocardial infarction and ischemic stroke through the inhibition of platelet aggregation. 1 However, despite their established efficacy in preventing thrombosis, antiplatelet agents are associated with an increased risk of bleeding, particularly during invasive procedures. 2
Lumbar puncture (LP), a fundamental diagnostic procedure in neurology, has been widely used to evaluate central nervous system (CNS) disorders and differentiate infectious, inflammatory, hemorrhagic, and demyelinating conditions. 3 Although LP is contraindicated in individuals with coagulopathy or those receiving oral anticoagulants because of an increased bleeding risk, data on safety profile and complication rates associated with APT remain limited.4–6 Clinical guidelines generally do not recommend withholding aspirin prior to LP, given the lack of sufficient evidence for a clinically significant increase in bleeding risk. For elective, nonurgent procedures, de-escalation from dual antiplatelet therapy (DAPT) to single antiplatelet therapy (SAPT) is commonly advised to reduce the potential risk of bleeding.7–11 However, discontinuation of antiplatelet agents is associated with a risk of ischemic events, including myocardial infarction, stroke, and venous thromboembolism. Consequently, clinicians are required to carefully weigh the diagnostic benefits of LP against the potential hazards of temporarily withholding APT. In acute clinical scenarios, such as suspected CNS infection or subarachnoid hemorrhage, LP is often deemed necessary even in patients receiving DAPT, to prevent diagnostic delays or missed opportunities for timely intervention.7–11 Nevertheless, concerns persist among clinicians regarding the safety of performing LP in patients on APT, particularly in those receiving DAPT, such as aspirin in combination with clopidogrel.
Clinically, cerebrospinal fluid (CSF) samples from patients on DAPT contain an elevated number of red blood cells (RBCs) compared with those from patients not on such therapies. Elevated RBC counts in CSF typically indicate CNS hemorrhage, potentially caused by traumatic injury, subarachnoid hemorrhage, intraventricular hemorrhage, or other intracranial bleeding events but could also result from iatrogenic vascular injury during LP. 12 This raises a critical question: does APT exacerbate bleeding from accidental small-vessel puncture during LP, thereby increasing the risk of spinal hematoma? Addressing this issue is crucial for guiding clinical decisions and optimizing patient management strategies. Consequently, investigating the impact of antiplatelet drugs on CSF RBC counts following LP holds substantial clinical significance and warrants focused research efforts.
Methods
Study design
This was a real-world, observational, retrospective study based on electronic medical records, reflecting routine clinical practice and patients’ real-world health conditions. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 13 In this study, a systematic three-step methodology was implemented, consisting of patient enrollment, data collection and preprocessing, and data analyses. A schematic representation of this approach is provided in Figure 1.
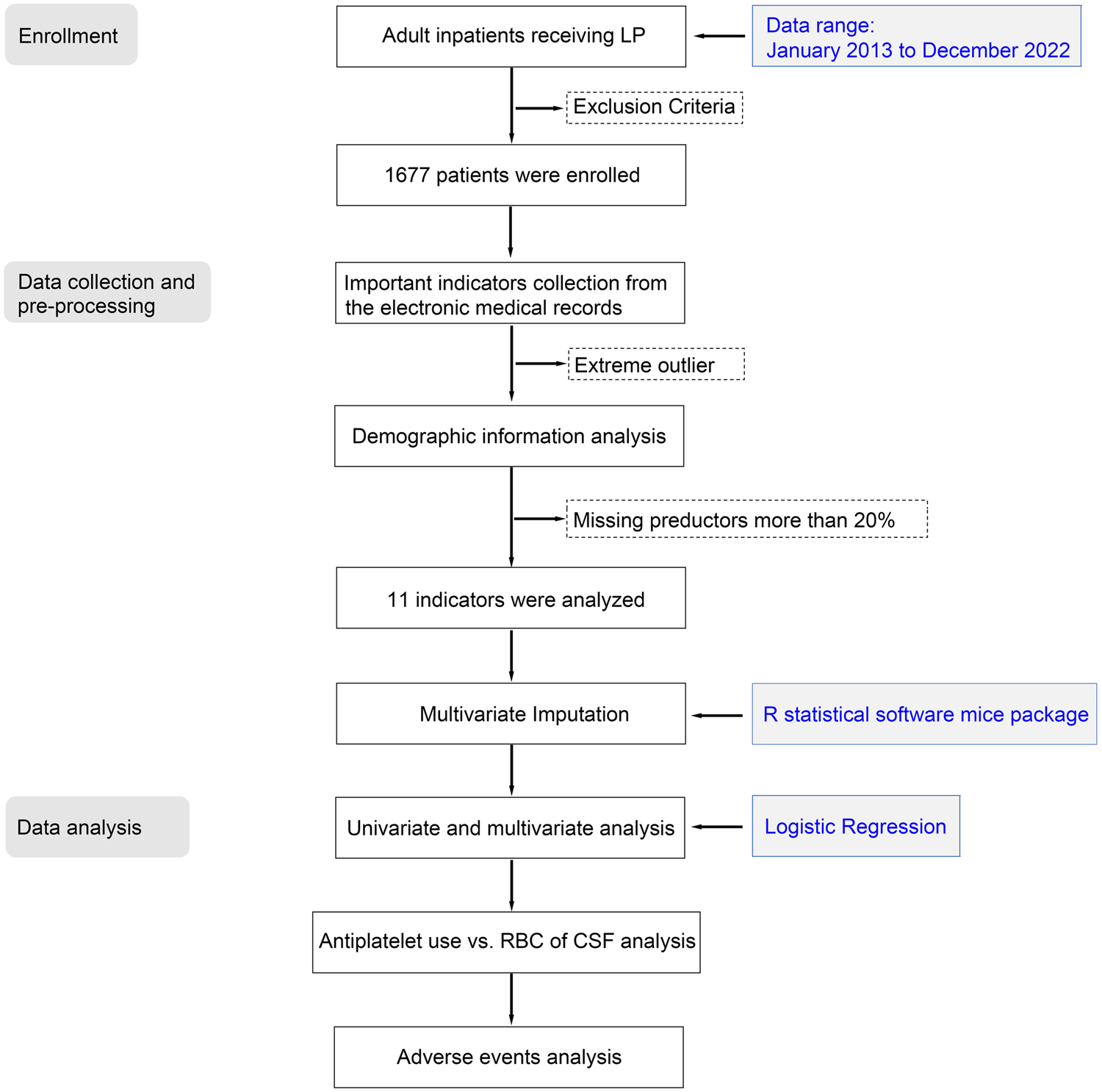
Overall workflow chart. CSF: cerebrospinal fluid; LP: lumbar puncture; RBC: red blood cell.
Step 1: We identified all hospitalized patients who underwent LP between 1 January 2013 and 31 December 2022. The initial cohort was meticulously screened using predefined exclusion criteria to obtain the final study population.
Step 2: Detailed clinical data were collected from the electronic medical records and crosschecked to ensure accuracy and completeness. Data preprocessing was performed to construct a clean and analyzable final dataset.
Step 3: Univariate and multivariate analyses were conducted to explore factors associated with RBC counts in CSF. Particular attention was given to the effect of perioperative antiplatelet medication use on RBC counts in CSF. Additionally, other adverse events (AEs) were recorded and analyzed statistically.
Patient enrollment
Records from patients who underwent LP in the inpatient departments of Dongyang People’s Hospital, affiliated with Wenzhou Medical University, between January 2013 and December 2022, were extracted using a medical record information mining software program (Shanghai Le9 Health Technology Co., Ltd., Shanghai, China). Subsequently, the information was crosschecked in the electronic medical record system using the medical record number. The exclusion criteria were as follows: (a) age <18 years; (b) duplicate data and incorrect medical record number; (c) follow-up time of <3 days; (d) failed procedure; (e) bleeding complications before LP, such as epidural hematoma, subdural hematoma, subarachnoid hemorrhage, intracranial hemorrhage, and extramedullary hemorrhage; (f) a history of cranial surgery within the past year; (g) simultaneous use of anticoagulants; (h) a tendency to bleed, including activated partial thromboplastin time (APTT) >65.25 s (1.5 times the upper limit of the normal range) and/or international normalized ratio (INR) > 1.5; (i) undergoing intrathecal chemotherapy; (j) refusal to undergo LP or multiple LPs with intervals greater than 30 days; and (k) death due to other medical conditions.
Data collection
We collected data from hospitalized patients who underwent LP via the electronic medical record system, which were clinically organized and recorded by professional doctors and nurses in a timely manner. This included demographic information (such as age, sex, height, and weight), concomitant illnesses (such as hypertension, diabetes, history of gastrointestinal bleeding, and recent major bleeding events), test results (such as hemoglobin concentration, platelet count, CSF analysis, APTT, and INR), medication use (such as antiplatelet agents), and AEs related to LP (such as headache, dizziness, pain in the lumbar region, nausea, and vomiting). Perioperative APT was defined as the administration of antiplatelet aggregation medications within 1 week before and/or after the LP. CSF RBC counts were determined by preparing a CSF smear and examining 10–20 high-power fields under a microscope at 400× magnification (10× ocular lens and 40× objective lens). The average number of RBCs across these fields was calculated. Other AEs were documented according to the records of doctors or nurses. Two professionals collected the required data in the electronic medical record system using the medical record number, while two professionals conducted data verification.
Endpoints and follow-up
The follow-up for all enrolled patients continued until discharge. The primary outcome was the risk of RBC presence in the CSF of patients after LP. The secondary outcomes were other AEs related to LP, such as post-LP headache, dizziness, pain in the lumbar region, nausea, and vomiting.
Preprocessing
After removing all personally identifiable information, data were assessed for normality, and extreme oultiers were excluded. Patients were then classified into two groups based on CSF routine test results: those with no RBCs and those with RBC presence. In demographic information and logistic regression analyses, the APT regimens were divided into three categories: no antiplatelet drug use group, SAPT group, and DAPT group. Patients who used aspirin or clopidogrel alone or those who switched from aspirin to clopidogrel or from clopidogrel to aspirin within 7 days prior to the LP but did not use both drugs concurrently were classified into the pre-LP SAPT group. Those who received both aspirin and clopidogrel during the same period were included in the pre-LP DAPT group. Similarly, post-LP antiplatelet drug treatment regimens were categorized according to the same criteria. Continuous variables were presented as mean ± SD or median, and categorical variables were presented as percentages. Continuous variables with a normal distribution were analyzed using the unpaired and independent t-test, those with a non-normal distribution were analyzed using the Wilcoxon rank-sum test, and categorical variables were analyzed using χ2 test or Fisher exact test. Variables with more than 20% missing values were excluded. Missing values in the remaining variables were imputed using the ‘mice’ package in R statistical software, and a complete analytic dataset was constructed. To further clarify the specific impact of APT regimens on CSF RBC levels, patients were reclassified into five groups based on APT regimens and specific medication details prior to the initial LP: no APT, aspirin-only, clopidogrel-only, medication switch (within 7 days), and DAPT groups.
Data analysis
Univariate and multivariate logistic regression analyses were conducted to identify potential predictors using ‘autoReg’ package. Variables with p < 0.20 in the univariate analysis were retained for inclusion in the multivariate analysis to evaluate their independent association with the RBC count in CSF. 14 A dedicated analysis was performed to assess the impact of perioperative APT using the χ2 test, with p values adjusted via Bonferroni correction for multiple comparisons. To eliminate the interference from multiple LPs, our analysis was restricted to patients who underwent their first LP. RBC counts in the CSF were categorized based on manual microscopic evaluation of CSF smears at 400× magnification. The grading is defined as follows: 0 RBCs/HPF is classified as grade 0, 1–5 RBCs/HPF as grade 1, 6–10 RBCs/HPF as grade 2, 11–15 RBCs/HPF as grade 3, 16–20 RBCs/HPF as grade 4, 21–25 RBCs/HPF as grade 5, and more than 25 RBCs/HPF as grade 6. The effects of the type, duration of use, and time since last dose of antiplatelet agents on CSF RBC counts were examined. Other AEs were also documented and analyzed. All statistical analyses were performed using R statistical software (version 4.4.1), with the statistical significance set at a p value of <0.05.
Ethics statement
This study was conducted in accordance with the revised Declaration of Helsinki (1975, as updated in 2024). The study was approved by the Ethics Committee of Dongyang People’s Hospital (Approval No. Dong Ren Yi 2023-YX-369, October 2023). As this was a retrospective observational study using deidentified patient data with no risk of harm, the requirement for informed consent was waived in accordance with the Ethical Guidelines for Epidemiological Research by the Ministry of Health, Labor, and Welfare.
Results
Patient characteristics
From January 2013 to December 2022, adult inpatients undergoing LP at Dongyang People’s Hospital (affiliated with Wenzhou Medical University) were retrospectively reviewed. Based on the exclusion criteria, 1677 patients were finally included in the study. The median age at the time of the procedure was 53 (interquartile range (IQR), 39–67) years, and 55.22% of the patients were male. The distribution of missing data is summarized in Table 1 and Supplementary Figure 1. Variables including CSF routine examination results, APT regimens before and after LP, number of LPs, sex, and age were fully recorded with no missing values. Hemoglobin concentration data were missing for 135 patients (8.05%), and platelet count data were missing for 136 patients (8.11%). Pre-LP laboratory values for INR and APTT were unavailable for 265 patients (15.80%). Anthropometric data showed substantial missingness: height data were missing in 25.4% of the cases and weight data in 20.75%, resulting in a body mass index (BMI) missing rate of 26.06%. BMI was excluded from subsequent univariate and multivariate regression analyses to minimize potential bias.
Demographic information of patients.

APT: antiplatelet therapy; APTT: activated partial thromboplastin time; BMI: body mass index; DAPT: dual antiplatelet therapy; INR: international normalized ratio; LP: lumbar puncture; Q1: first quartile; Q3: third quartile; RBC: red blood cell; SAPT: single antiplatelet therapy
Patients were divided into two groups based on CSF routine examination results: those with RBCs and those without RBCs. Their detailed characteristic comparisons are presented in Table 1. Overall, RBCs were detected in 728 patients. Only age (p = 0.025) demonstrated a statistically significant difference between the two groups. However, there were no statistically significant differences between the two groups with regard to sex (p = 0.587), BMI (p = 0.127), hemoglobin concentration (p = 0.805), platelet count (p = 0.316), INR (p = 0.862), APTT (p = 0.592), perioperative use of antiplatelet drugs (pre-LP APT, p = 0.270 and post-LP APT, p = 0.146), and the number of LPs (p = 0.209).
Risk factors for LP-related CSF RBC counts
Univariate analysis revealed that the p values for the risk factors age (p = 0.003), INR (p = 0.057), pre-LP APT (SAPT, p = 0.112), post-LP APT (SAPT, p = 0.140; DAPT, p = 0.179), and the number of LPs (n = 2; p = 0.192) were below 0.2, warranting their inclusion in the multivariate analysis (Table 2). However, multivariate analysis demonstrated that CSF RBC counts were exclusively correlated with age (odds ratio, 1.01; 95% confidence interval (CI), 1.00–1.01; p = 0.005). Other factors, including sex, hemoglobin concentration, platelet count, coagulation function, perioperative use of antiplatelet drugs, and the number of LPs, were not statistically correlated with the presence of RBCs (p > 0.05).
Univariate and multivariate logistic regression analyses.

APT: antiplatelet therapy; APTT: activated partial thromboplastin time; CI: confidence interval; DAPT: dual antiplatelet therapy; INR: international normalized ratio; LP: lumbar puncture; OR: odds ratio; RBC: red blood cell; SAPT: single antiplatelet therapy.
Impact of pre-LP antiplatelet drug usage on CSF RBC counts
The distribution of RBC counts in the CSF is illustrated in Figure 2 and Supplementary Table 1. In the CSF, the RBC counts were predominantly concentrated in grade 0 and grade 1. Before LP, among the 1372 patients who had not been using antiplatelet drugs, the CSF RBC counts were mainly concentrated in grade 0 (57.29%), followed by grade 1 (37.46%). Additionally, 110 patients had only taken aspirin and 143 patients had only taken clopidogrel prior to LP, and the predominant CSF RBC count grading in these patients was grade 0 (52.73% and 52.45%, respectively). However, this proportion was slightly lower than that in the group that did not use antiplatelet drugs. Regarding patients who changed medications midway or were on DAPT, the CSF RBC count grading may be less accurate due to the smaller sample size (13 and 39 patients, respectively).
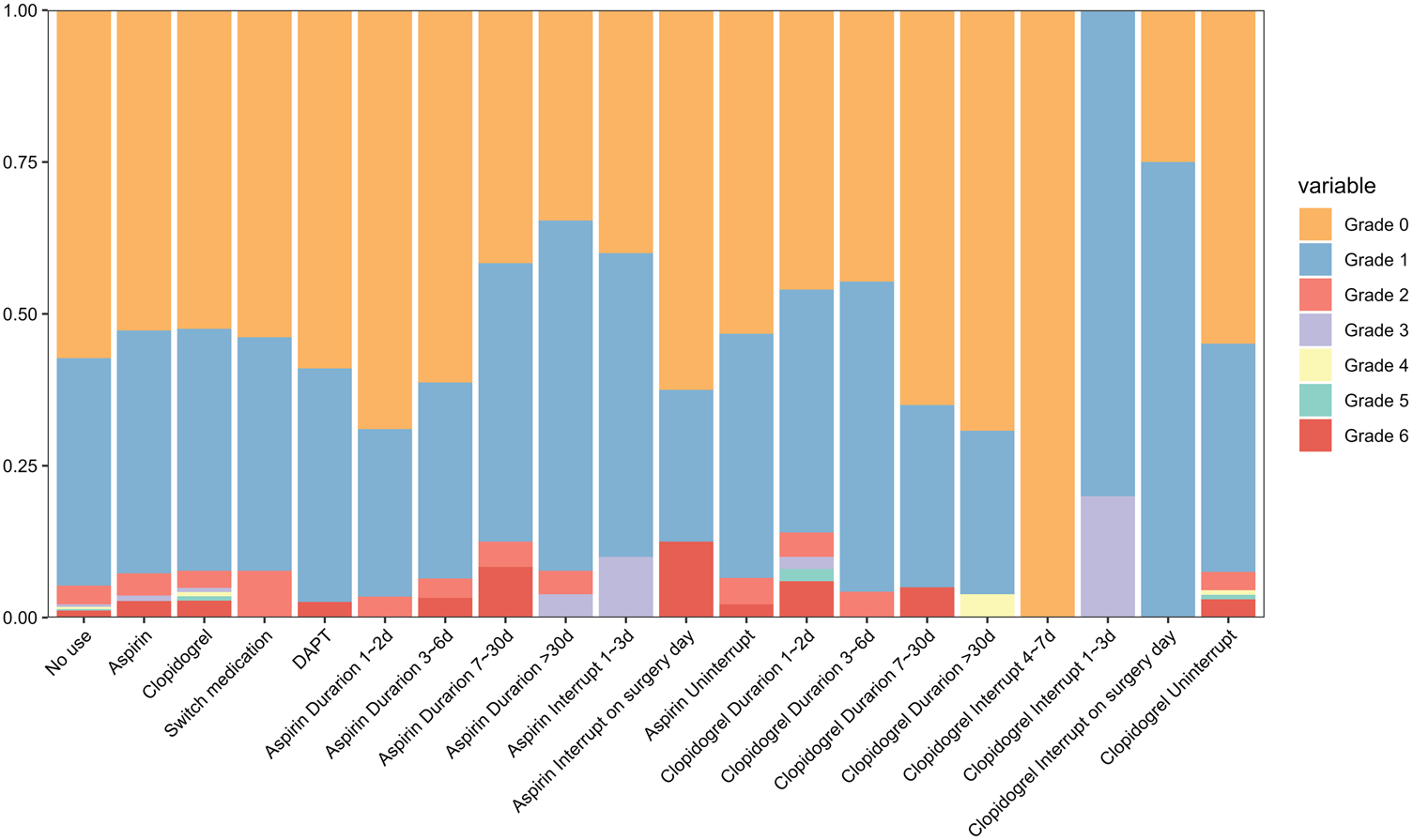
The distribution of red blood cell counts in the cerebrospinal fluid. DAPT: dual antiplatelet therapy.
A further analysis was conducted on patients who used only aspirin or clopidogrel, examining the impact based on the duration of medication use and the time since interruption. In the aspirin group, patients who used aspirin for 1–2 days had a grade 0 rate of 68.96% and grade 1 rate of 27.59%; those who used it for 3–6 days had a grade 0 rate of 61.29% and grade 1 rate of 32.25%; patients who used it for 7–30 days had a grade 0 rate of 41.67% and grade 1 rate of 45.83%; and patients who used aspirin for more than 30 days had a grade 0 rate of 34.61% and grade 1 rate of 57.69%. With the prolonged use of aspirin, a trend of decreasing grade 0 and a corresponding increase in grade 1 was observed. In the clopidogrel group, patients who used clopidogrel for 1–2 days had a grade 0 rate of 46.00% and grade 1 rate of 40.00%; those who used it for 3–6 days had a grade 0 rate of 44.68% and grade 1 rate of 51.06%; patients who used it for 7–30 days had a grade 0 rate of 65.00% and grade 1 rate of 30.00%; and patients who used clopidogrel for more than 30 days had a grade 0 rate of 69.23% and grade 1 rate of 26.92%. Regarding the timing of APT discontinuation, the distribution of RBC grades in CSF was similar between patients who did not interrupt their APT and those who did not receive antiplatelet drugs. Moreover, no statistically significant differences were observed in CSF RBC grading with respect to pre-LP APT, duration of APT usage, or interruption of therapy (Figure 3).

Heatmap of intergroup differences in the graded distribution of red blood cell count in the cerebrospinal fluid. Gray squares indicate that no comparison was performed between the two groups.
Incidence of other AEs related to LP
The distribution of AEs is shown in Figure 4 and Supplementary Table 2. Patients were divided into three groups based on the pre-LP APT: no antiplatelet drug use group (none), SAPT group, and DAPT group. Among the 1677 patients, 75 AEs were recorded (4.47%), including 37 patients with headache (2.21%); 4 with back pain (0.24%); 3 with nausea and vomiting (0.18%); 20 with dizziness (1.19%); 5 with headaches and nausea and vomiting (0.30%); 5 with both headaches and dizziness (0.30%); and 1 with dizziness, nausea, and vomiting (0.06%). In the group that did not receive antiplatelet drugs before LP, there were 29 cases of headache (2.13%); 3 cases of back pain (0.22%); 2 cases of nausea and vomiting (0.15%); 17 cases of dizziness (1.25%); 4 cases of headache, nausea, and vomiting (0.29%); and 3 cases of headache and dizzy (0.22%). In the SAPT group, there were eight cases of headache (3.01%); one case of back pain (0.37%); three cases of dizziness (1.13%); one case of headache, nausea, and vomiting (0.37%); two cases of headache and dizzy (0.75%); and one case of dizziness, nausea, and vomiting (0.37%). In the DAPT group, there was one case of nausea and vomiting–related AEs (2.56%). No statistically significant differences were observed.

Distribution of other adverse events. SAPT: single antiplatelet therapy; DAPT: dual antiplatelet therapy.
Discussion
This study aimed to investigate whether APT potentiates bleeding from inadvertent small-vessel injury during LP in a real-world clinical cohort. Two principal findings emerged: 1. perioperative APT was not independently associated with an increased risk of RBC presence in CSF following LP; 2. other AE rates were comparable regardless of APT use.
An elevated CSF RBC count typically indicated CNS hemorrhage due to conditions such as traumatic injury, subarachnoid hemorrhage, intrawentricular hemorrhage, or other intracranial bleeding events. However, it may also reflect iatrogenic vascular injury during LP. 12 To analyze the impact of APT regimes on procedural trauma, patients with preexisting hemorrhagic conditions (including epidural, subdural, subarachnoid, intracranial, and extramedullary hemorrhage) or a bleeding diathesis (APTT > 65.25 s and/or INR > 1.5) were excluded. Therefore, in this cohort, the CSF RBC count served as a reliable marker of iatrogenic vascular injury during the procedure.
Current clinical guidelines generally do not recommend discontinuing aspirin before LP, due to insufficient evidence regarding a clinically significant increase in bleeding risk. For elective, nonurgent procedures, de-escalation from DAPT to SAPT was often advised to reduce potential bleeding complications.7–11 Our findings supported this approach, as no significant association was found between APT and elevated CSF RBC counts. However, the limited number of patients on DAPT in our cohort precluded definitive conclusions regarding this subgroup. Nevertheless, the overall evidence suggested that LP in patients receiving APT did not substantially increase the bleeding risk of procedural vascular injury.5,6 Notably, no spinal hematomas were observed in this study, although rare cases of subarachnoid hemorrhage and subdural hematoma have been reported in the literature.15–18
In this retrospective study, both univariate and multivariate analyses demonstrated a significant association between CSF RBC counts and age, with RBC presence group being older than the RBC absence group. The increased risk of vascular injury during LP in older adults is likely attributable to age-related physiological changes in the vasculature and spine as well as the burden of comorbidities.19–27 With aging, blood vessels become more fragile and less elastic, increasing susceptibility to mechanical injury.19,20 Atherosclerosis, which is more prevalent in the older population, affects both large and small vessels, compromising their structural integrity. 21 Additionally, degenerative spinal changes, such as osteophyte formation and disc degeneration, alter local anatomy, increasing technical difficulty and the risk of inadvertent vascular puncture.22,23 Common comorbidities such as hypertension and diabetes further impair endothelial function and promote vascular fragility.24–27 A limitation of this study was the lack of data on the use of traumatic spinal needles, which may have introduced bias and limited our ability to fully assess procedural risk factors.
Additionally, both univariate and multivariate analyses indicated that perioperative APT was not independently associated with an increased risk of RBC elevation in CSF following LP. However, in previous logistic regression models, pre-LP APT regimens were divided into three categories. This approach was insufficient to distinguish the independent effects of aspirin and clopidogrel on CSF RBC counts. Therefore, to further clarify the specific impact of APT regimens on CSF RBC counts, patients were reclassified into five groups based on APT agents and specific medication details prior to the initial LP. Additionally, the previous analysis did not account for whether antiplatelet agents had reached steady-state concentrations or the precise timing of drug discontinuation before the procedure. In subsequent analyses, the aspirin-only and clopidogrel-only groups were stratified by duration of therapy and time from last dose to LP, and their effects on CSF RBC counts were compared. The results showed that the use of APT, the specific agent (aspirin, clopidogrel or both), the duration of preprocedural therapy, or the timing of preprocedural discontinuation were not significantly associated with CSF RBC counts. These findings further suggested that APT did not increase the patient’s risk of microvascular injury during LP. Although there were no specific studies examining the impact of APT on CSF RBC count, previous studies, primarily focused on hematoma formation, have consistently affirmed the safety of antiplatelet drugs during the perioperative period for LPs.5,16,28 Lee and Levy 16 retrospectively analyzed bleeding risks associated with aspirin, clopidogrel, or DAPT in patients undergoing LP at Johns Hopkins Hospital between 2004 and 2018. Patients were stratified by time from drug discontinuation to LP: <1 week, 1–4 weeks, and >4 weeks. Their results demonstrated that neither aspirin nor clopidogrel, alone nor in combination, significantly increased the risk of bleeding complications, regardless of the timing of discontinuation relative to the procedure.
Moreover, other AEs occurred in 75 patients, representing a lower incidence than that reported previously. 29 Postdural puncture headache (PDPH) was the most common AE, consistent with prior studies.29,30 However, many studies have used stricter definitions, requiring a positional headache accompanied with symptoms such as nausea, photophobia, phonophobia, tinnitus, or neck stiffness. 6 In contrast, our definition included any headache documented in the electronic medical record following LP, which depended on the accuracy of documentation and may have led to underestimation of AEs. Population heterogeneity across studies may also have contributed to the variability in the reported PDPH incidence.31–33 The lack of data on needle type, particularly the use of cutting versus atraumatic needles, which is known to affect complication rates,31–33 may have partially accounted for the lower AE rate observed in this study. Importantly, no severe AEs were recorded, consistent with the existing literature indicating that serious complications from LP are exceedingly rare. 29
Limitations
Our study has several limitations. First, as a single-center study, the findings may be subject to selection bias and limited generalizability. Second, variability in documentation quality within the electronic medical record system potentially results in an underestimation of AEs. Third, several important procedural and operator-dependent variables, including the experience of the physician, type of needle used (e.g. traumatic versus atraumatic needles and needle size), and patient positioning were not analyzed in the current study. Fourth, the study included only cases with successful CSF collection, excluding those with failed LP attempts or those deemed ineligible due to high bleeding risk, potentially introducing selection bias. Future research should include multicenter data to further validate the safety of administering antiplatelet medications during LP.
Conclusion
Older age is associated with a higher likelihood of RBC presence following LP. Perioperative APT was not independently linked to an increased risk of RBC presence, and AE rates were comparable regardless of APT use, suggesting a favorable safety profile of LP in real-world clinical practice.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605251386563 - Supplemental material for Safety of lumbar puncture in patients on antiplatelet therapy: A real-world study assessing red blood cell counts in cerebrospinal fluid
Supplemental material, sj-pdf-1-imr-10.1177_03000605251386563 for Safety of lumbar puncture in patients on antiplatelet therapy: A real-world study assessing red blood cell counts in cerebrospinal fluid by Qiwei Ran, Jvping He, Xiang Zheng and Haiyan Chen in Journal of International Medical Research
Footnotes
Acknowledgments
None.
Author contributions
All authors discussed the results and provided comments. Haiyan Chen designed the study. Haiyan Chen, Qiwei Ran, Jvping He, and Xiang Zheng conducted the study and collected the data. Xiang Zheng analyzed and interpreted the data. Haiyan Chen created the figures and tables and drafted the manuscript. Xiang Zheng and Jvping He contributed to manuscript revisions. All authors approved the final version for publication.
Data availability statement
The original research data reported in the current study are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
The authors have no direct or indirect financial or nonfinancial interests related to the work submitted for publication.
Funding
This manuscript was funded by the Jinhua Municipal Science and Technology Bureau (grant number 2023-4-232).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
