Abstract
Objective
To examine the effects of antibiotic administration on radiation-induced oral and oropharyngeal mucositis, and on the prognosis of patients with nasopharyngeal carcinoma (NPC).
Methods
We retrospectively analyzed data for patients with NPC with grade 1/2 or 3/4 mucositis. Forty-two patients with grade 3/4 mucositis received antibiotics. Univariate survival analysis was assessed by Kaplan–Meier survival curves, survival curves were compared using log-rank tests, and multivariate analysis was carried out by Cox regression.
Results
A total of 463 patients with NPC were included in the study (194 grade 1/2 mucositis, 269 grade 3/4 mucositis). Univariate analyses identified T-stage, N-stage, clinical stage, type of treatment, and antibiotic use as factors affecting overall and disease-free survival. Multivariate analysis also determined that T-stage, N-stage stage, type of treatment, and antibiotic usage were independent factors affecting overall and disease-free survival. Mucositis improved in 32 of the 42 patients who received antibiotics (76.19%). However, red blood cell count and hemoglobin levels decreased in all patients after antibiotic treatment.
Conclusions
Antibiotics may be effective for the treatment of severe radiation-induced mucositis (grade 3/4) during chemoradiotherapy, but may potentially adversely affect the prognosis of patients with NPC.
Keywords
Introduction
Nasopharyngeal carcinoma (NPC) is a common malignant tumor in southern China, 1 with World Health Organisation type III being the most common pathological type in high-incidence regions. This type of NPC is sensitive to radiotherapy and is therefore often treated accordingly. However, due to the insidious onset of the disease, most patients are at a middle-late stage when they are diagnosed. Since publication of the significant results from the Intergroup Study 0099, 2 radiotherapy with concurrent chemotherapy has become established as the standard care for patients with locoregionally advanced NPC. Chemoradiotherapy is commonly used in patients with middle-late stage NPC, and intensity-modulated radiation therapy (IMRT) is the preferred technique to improve local control in these patients in clinical practice. 3 However, the mucosa is an early-response tissue and increasing the local radiation dose will thus inevitably lead to radiation-induced mucosal damage. According to the literature, the incidence of radiation-induced mucositis in NPC patients treated with radiotherapy is 100%, of which 30%–66.7% of cases are severe mucositis.4,5 Chemotherapy is thought to exacerbate mucositis 6 by reducing immunity and damaging mucosal integrity, making patients prone to comorbid infections. Antibiotics are usually used for severe mucositis in clinical practice, particularly in patients with suspected or diagnosed bacterial infections. Antibiotics were previously reported to alleviate mucositis in patients in one study, 7 while another study showed that antibiotics decreased overall survival (OS) in patients with chronic myeloid leukemia and relapsed lymphoma. 8 However, reports on the effect of antibiotic usage during chemoradiotherapy on the prognosis of patients with NPC are currently lacking. We therefore conducted a preliminary retrospective assessment of the effects of antibiotics on radiation-induced mucositis and on the clinical prognosis of patients with NPC to determine if antibiotics might benefit these patients. The results of this study will provide the rationale for using antibiotics in patients with NPC and severe comorbid mucositis during chemoradiotherapy.
Materials and methods
Patients
The inclusion criteria for this study were: (1) pathological diagnosis of nasopharyngeal biopsies; (2) Karnofsky score > 80 points; (3) completed standard radiotherapy or concurrent radiochemotherapy; and (4) informed consent obtained. The exclusion criteria were: (1) distant metastases; (2) discontinued treatment; and (3) anti-inflammatory treatment due to respiratory tract inflammation prior to this study. Patients were enrolled at the Nasopharyngeal Carcinoma Research Institute of the People’s Hospital of Guangxi Zhuang Autonomous Region from January 2013 to December 2014. Disease was staged in all patients based on the Union for International Cancer Control (UICC 2010) TNM staging system for NPC.
This retrospective study was approved by the ethics committee of The People’s Hospital of Guangxi Zhuang Autonomous Region. All the patients recruited in this study provided written informed consent, in which they agreed to the use of their clinical data for non-commercial scientific research.
Treatment
Radiotherapy consisted of IMRT at a prescribed dose of 69–72 Gy delivered to the planning target volume (PTV) of the nasopharynx and cervical metastatic lymph node, respectively, and 60–65 Gy delivered to the PTV encompassing all macroscopic disease with a border of 1 cm (PTV1). The PTV2 was treated with 50–56 Gy. All patients were given one fraction daily for 5 days a week. Chemotherapy included 30 mg/m2 cisplatin by intravenous infusion on days 1–3 and 2000 mg/m2 fluorouracil continuously for 120 hours by intravenous pump. One cycle lasted for 28 days and two to three cycles were administered consecutively. Alternatively, patients received 80 mg/m2 nedaplatin by intravenous infusion on day 1 and 2000 mg/m2 fluorouracil continuously for 120 hours by intravenous pump. As above, one cycle lasted for 28 days and two to three cycles were administered consecutively.
Grading of radiation-induced mucositis and general treatment
Acute oral and oropharyngeal mucosal reactions were classified into five grades according to the Common Terminology Criteria for Adverse Events (CTCAE, Version 4.03). 9 A mucosal reaction of ≥ grade 3 was considered to be severe mucositis and grade 1 or 2 was mild mucositis. Oral care, pain-control medication, and parenteral nutrition were applied routinely in patients with severe mucositis.
Antibiotic administration
Treatment for severe mucositis (grade 3/4) was based on the results of a routine blood test suggesting comorbid bacterial infection (white blood cell count (WBC) >10.0 × 109/L, neutrophil percentage >75.0%), in which case 1.5 g cefuroxime sodium was administered by intravenous infusion, twice a day for at least 7 days.
Blood tests
A 2 mL sample of venous blood was drawn on admission and again before and after antibiotic use. The blood samples were used to test for absolute lymphocyte count (ALC), hemoglobin (HGB), neutrophil count (NEUT), platelet count (PLT), red blood cell count (RBC), and WBC. Analyses were carried out using an automated hematology analyzer (Sysmex, Kobe, Japan) according to the manufacturer’s instructions. Neutrophil-lymphocyte ratio (NLR) was determined by dividing NEUT by ALC.
Imaging evaluation
All patients underwent routine magnetic resonance scans (T1W1 and T2W1 sequences) before and after radiotherapy to assess tumor regression, based on the Response Evaluation Criteria In Solid Tumors (RECIST) 1.1. 10
Follow-up
After discharge, patients were followed-up once every 3 months in the first year, once every 4–6 months in the second year, and once every 12 months in the third year. Telephone or written follow-up was used for patients who did not return for scheduled follow-ups.
Statistical analysis
The data were analyzed using SPSS for Windows, Version 24.0 (IBM Corp., Armonk, NY, USA). Qualitative data were compared between groups using χ2 tests. Normally distributed quantitative data were compared by
Results
General information
A total of 463 patients met the criteria and were enrolled in the study. Of these, 41.9% (194/463) had mild oral or oropharyngeal mucositis and 58.1% (269/463) had severe mucositis. Among the patients with severe mucositis, 15.6% (42/269) were given antibiotics. There were no significant differences in sex, age, clinical stage, and type of treatment between the patients with severe mucositis who were given antibiotics and those who were not given antibiotics (Table 1).
Baseline characteristics of patients.
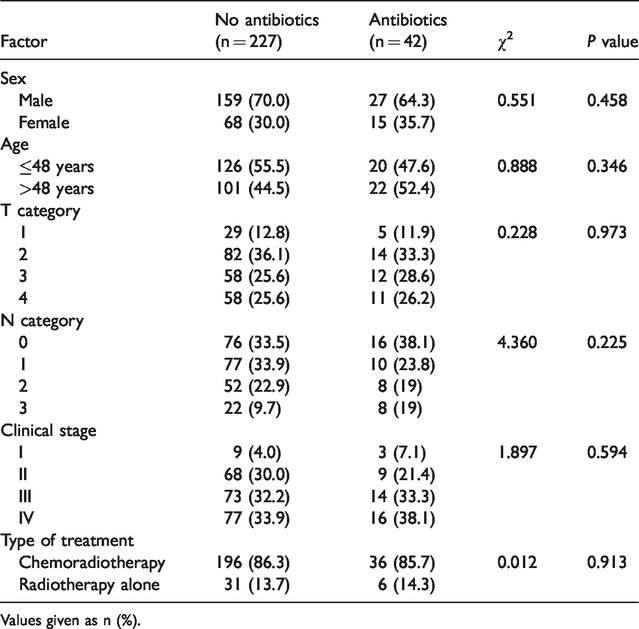
Values given as n (%).
Follow-up and clinical prognosis
The follow-up period ended on 1st October 2017. The median follow-up was 44 months (range 12–58 months). Four subjects were lost to follow-up.
The 3-year OS and 3-year disease-free survival (DFS) rates for the entire cohort were 87.71% and 78.16%, respectively. The 3-year OS (86.34% vs 89.61%) and DFS rates (77.07% vs 79.67%) were similar in patients with severe and mild mucositis. The 3-year OS rates for patients with severe mucositis with and without antibiotics were 78.17% and 87.85%, respectively, and the equivalent 3-year DFS rates were 64.15% and 79.47%, respectively. These differences were not significant. Among all the enrolled patients, 38 patients had recurrence, 71 had distant metastasis, and seven had both recurrence and distant metastasis. Among the patients with severe mucositis, 19 patients had recurrence, 46 had distant metastasis, and four had both. Among the patients with severe mucositis, the rates of recurrence and metastasis were both significantly higher in patients who received antibiotics compared with those who did not (38.1% vs 23.3%,
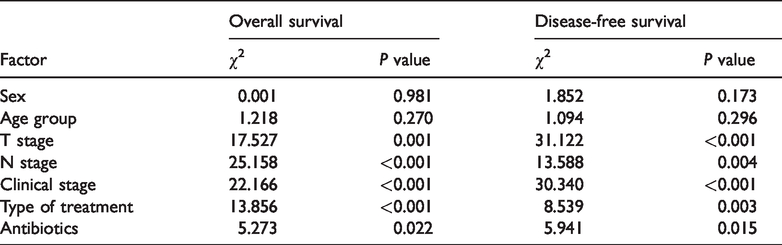
Univariate analysis identified T-stage, N-stage, clinical stage, type of treatment, and antibiotic use as factors affecting OS (all
Univariate analysis of factors affecting prognosis in patients with nasopharyngeal carcinoma.

Overall survival curves for patients with nasopharyngeal carcinoma with different clinicopathological features. (a) T stage; (b) N stage; (c) clinical stage; (d) treatment; and (e) antibiotic use. CRT, chemoradiotherapy; RT, radiotherapy.

Disease-free survival curves for patients with nasopharyngeal carcinoma with different clinicopathological features. (a) T stage; (b) N stage; (c) clinical stage; (d) treatment; and (e) antibiotic use. CRT, chemoradiotherapy; RT, radiotherapy.
Multivariate analysis of factors affecting prognosis in patients with nasopharyngeal carcinoma.
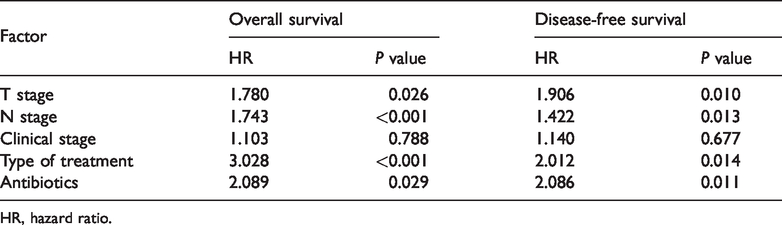
HR, hazard ratio.
Bacterial culture
Among the 42 patients with severe mucositis who were treated with antibiotics, bacterial cultures were carried out for 32 patients, with a positivity rate of 34.38% (11/32). The positive cases included six cases of
Changes in mucositis after antibiotic treatment
Of the 42 patients with severe mucositis who received antibiotics, 16 had a fever before antibiotic administration. After antibiotic treatment, mucositis improved in 32 patients (76.19%) but there was no apparent improvement in the other 10 (23.81%), and five of these patients required nasogastric feeding or intravenous nutrition, with temporary discontinuation of radiotherapy until their condition improved (Tables 4 and 5; Figure 3).
Therapeutic efficacy of antibiotics in 42 patients with radiation-induced mucositis.

Values give as n (%).
Data for patients with radiation-induced mucositis and comorbid bacterial infection.

M, male; F, female.
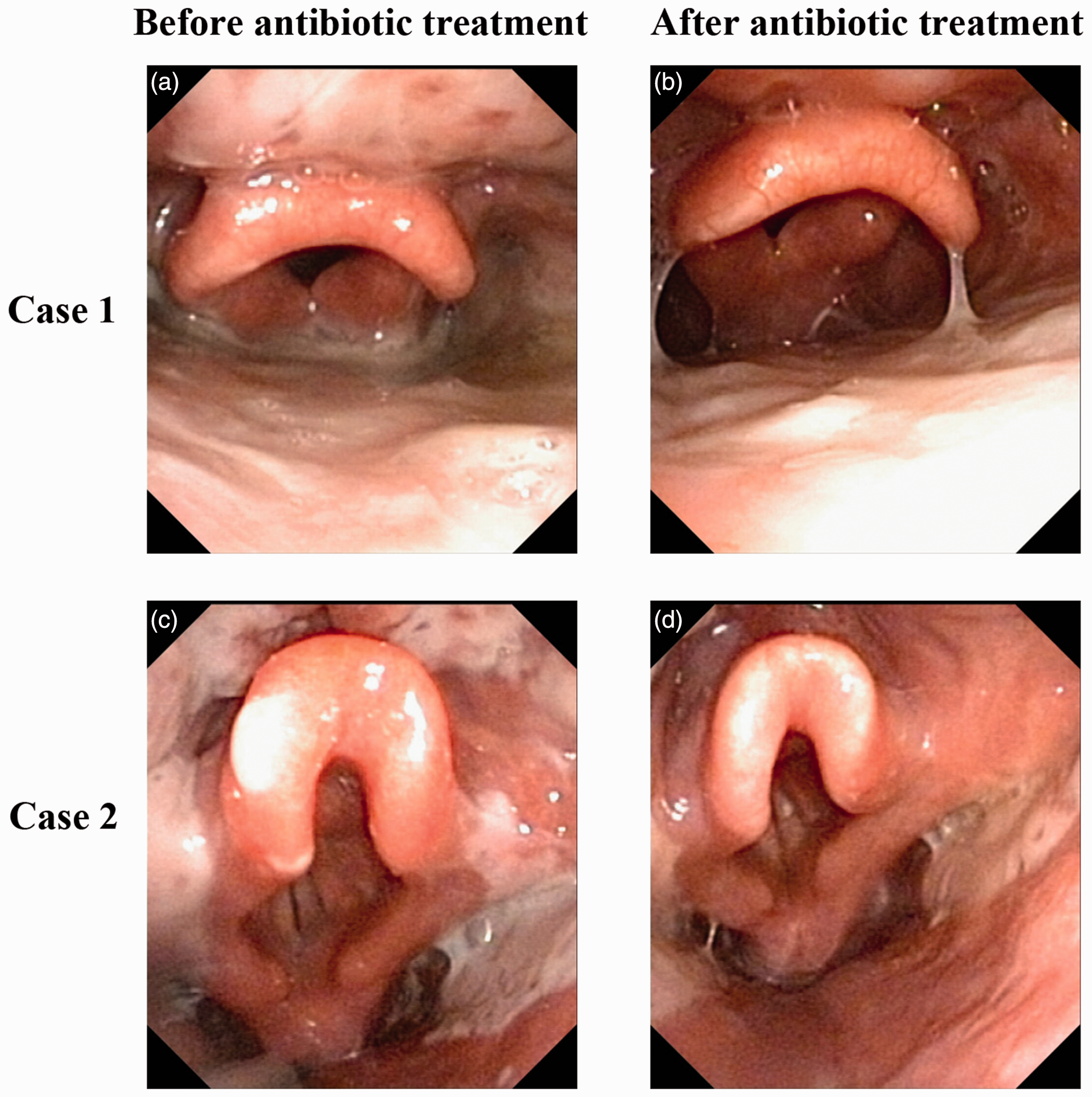
High-definition endoscopic images pre- and post-antibiotic treatment. Mucositis with
Changes in hematology markers after antibiotic use
A total of 42 patients were given cefuroxime sodium. RBC and HGB decreased significantly after antibiotic administration (both
Hematology before and after antibiotic therapy.
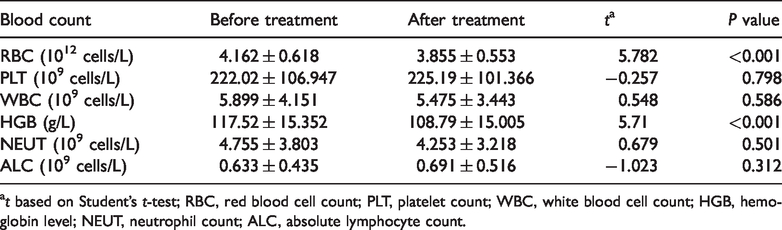
a
Tumor regression after treatment
Among the 42 patients with severe mucositis who were treated with antibiotics, 26 had a complete response (CR), 15 had a partial response (PR), and one had stable disease (SD). Twenty patients showed a reduction in HGB of at least grade 2 following radiotherapy, including nine CR, 10 PR, and one SD cases. The 22 patients with a grade 0–1 reduction in HGB included 17 CR and five PR cases. Tumor regression rates (complete or partial response) were significantly higher in patients with a reduction in HGB of grade 0–1 compared with a reduction of grade 2–4 (χ2 = 4.627,
Comparison of tumor regression rates by hemoglobin reduction grade.

HGB, hemoglobin; CR, complete response; PR, partial response; SD, stable disease.

High-definition endoscopic images pre- and post- chemoradiotherapy (CRT) treatment. (a) Pre-treatment endoscopic image showing the tumor located in the posterior wall (T3). (b) Post-treatment endoscopic examination image showing no visible tumor. (c) Pre-treatment endoscopic image showing the tumor located in the upper posterior wall (T3). (d) Post-treatment endoscopic examination image showing the residual tumor.
Discussion
NPC is usually considered to be sensitive to radiation and radiotherapy is therefore recommended. However, radiation inevitably irradiates the oral cavity and oropharynx, leading to an accepted incidence of radiation-induced oral and oropharyngeal mucositis of 100%. In addition, the National Comprehensive Cancer Network (NCCN) guidelines recommend chemoradiotherapy for patients with stage II–IVa NPC, and chemotherapy can also cause and exacerbate mucositis. 11 Patients who receive radiotherapy and develop radiation-induced mucositis may also have comorbid bacterial infection. 12 Broad-spectrum antibiotics (such as cephalosporins) are currently the most frequently used treatment. Gram-positive bacteria are generally thought to be the most common type of bacteria responsible for these infections, 13 as demonstrated in the current study. However, other studies have reported more Gram-negative than Gram-positive bacteria. 12 Some previous research found that antibiotics were effective against radiation-induced mucositis, 7 while others found no such efficacy. 14 In addition, some researchers advocate the use of quinolone antibiotics 15 to take account of the culture of Gram-negative rods. They administered levofloxacin and compound matrine injection to patients with head and neck cancer who developed mucositis after chemoradiotherapy, and found that pain was alleviated and mucositis was significantly improved in these patients. These studies suggested that quinolone antibiotics could not only effectively control mucositis caused by oropharyngeal flora but could also effectively control the nasopharyngeal flora. 15 In contrast, however, the current data showed that Gram-negative bacteria accounted for only 36.36% (4/11) of infections. We therefore used cefuroxime sodium to treat most patients, resulting in improvements in mucositis in 76.19% of cases. Among the 11 patients with mucositis with positive bacterial cultures, 10 improved after antibiotic treatment, suggesting that antibiotics have some efficacy against radiation-induced mucositis.
Previous studies found that cefuroxime sodium may cause immune hemolysis, 16 thereby reducing HGB and hematocrit levels. The patients in the present study showed varying levels of RBC and HGB after antibiotic treatment. The insidious onset of the disease means that most patients are at a middle-late stage when they are diagnosed and therefore require chemotherapy, which can cause myelosuppression. The administration of antibiotics during or after chemotherapy may further exacerbate myelosuppression. However, some studies showed that the use of prophylactic antibiotics during anti-tumor treatment effectively decreased the probabilities of fever and infection in patients with solid tumors. 17 Prophylactic antibiotics are usually given for myelosuppression of ≥grade 4 or above, and antibiotics are recommended for patients with accompanying fever. However, the use of prophylactic antibiotics in patients with ≤grade 3 myelosuppression remains controversial. In addition, whether antibiotics should be given for severe mucositis with negative bacterial cultures is also debatable. In the current study, the efficacy rate of antibiotics in patients who tested negative for bacterial cultures was 71.4% compared with 90.9% in patients who tested positive for bacterial cultures. We suggest that bacterial cultures may appear negative because only the dominant bacteria are cultured under the laboratory culture conditions.
In this study, the 3-year OS rate of the 42 patients with severe mucositis who used antibiotics was 78.17%, which was lower than that in patients without antibiotics (87.85%). Multivariate analysis also identified antibiotics as an independent risk factor affecting survival in NPC patients. Few studies are currently investigating the role of antibiotics in promoting tumor recurrence and metastasis, and the effects on prognosis. However, a recent clinical study
8
examined the effects of antibiotics against Gram-positive bacteria on outcomes in patients with tumors of the hemolymphatic system and found that the overall response rate among 800 patients with chronic lymphocytic leukemia who received antibiotics against Gram-positive bacteria was less than that of patients who did not receive antibiotics (74.3% vs. 90.2%, respectively; n = 45). In addition, patients who were given antibiotics showed faster disease progression. Multivariate analysis showed that antibiotics were associated with reduced progression-free survival (HR 2.090,
This study was limited by the fact that it was a retrospective analysis and may thus have included bias. In addition, our examination of the effects of antibiotics on prognosis were limited to blood cells, and further more detailed research is required. Finally, the follow-up duration of this study was short and more recent follow-up data were unavailable.
The present results confirmed that T stage, N stage, and type of treatment are primary factors affecting the prognosis in patients with NPC undergoing radiotherapy, and that close monitoring of RBC counts and HGB levels are needed in patients who develop severe radiation-induced mucositis and are treated with antibiotics. These patients may require nutrient supplementation, anemia correction, and close follow-up.
Footnotes
Acknowledgments
We are thankful for the participation of all the patients enrolled in this study. We would also like to thank Editage for their writing support.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This work was supported by the National Natural Science Foundation of China (Nos. 81602390 and 81960186), Guangxi Natural Science Foundation (Nos. 2016GXNSFCB 380003 and 2017GXNSFAA198013), Open Research Project of Key Laboratory of High-Incidence-Tumor Prevention & Treatment (Guangxi Medical University), Guangxi Key Research & Development Program (GuiKeAB1850010), Guangxi Medical and Health Appropriate Technology Development and Promotion Application Project (S2018039), Ministry of Education, and Scientific Research Project of Guangxi Health and Family Planning Commission (No. Z2014629).
