Abstract
Acute subarachnoid hemorrhage (SAH) presentation is a very rare occurrence in spinal schwannomas. We report a case of lumbar schwannoma in a patient presenting first with acute SAH. A 40-year-old man was referred to our department 7 days after the sudden onset of headache, neck pain, and fever. SAH was suspected; however, head computed tomography (CT) findings were normal. A lumbar puncture indicated blood-stained cerebrospinal fluid (CSF). Moreover, the CSF pressure changed from 200 mmH2O to 90 mmH2O after drainage of 10 mL of CSF indicating a blockage of CSF. Subsequent magnetic resonance imaging (MRI) confirmed an intradural tumor with SAH, which also caused blockage of the CSF circulation. The patient underwent immediate surgery and fully recovered. In conclusion, the early diagnosis and total removal of the tumor and blood clot significantly improved the patient’s outcome. There is a high index of suspicion for spinal tumors resulting in SAH when there is a CSF pressure change after lumbar puncture in an SAH patient.
Keywords
Introduction
Subarachnoid hemorrhage (SAH) caused by spinal lesions is rare, accounting for approximately 0.6% to 1.5% of all cases of SAH.1,2 The more common causes of SAH from spinal lesions comprise arteriovenous malformations, saccular aneurysms of spinal arteries, lumbar puncture, coagulopathy, and trauma. On very rare occasions, spinal intradural tumors, either primary or metastatic, may cause SAH. 3 Acute SAH presentation is a very rare occurrence in spinal schwannomas, especially in the absence of compression symptoms. Here, we report a case of lumbar schwannoma in a patient presenting first with acute SAH. In conjunction with a review of the literature, we discuss the mechanism and details of the diagnostic processes for this rare and difficult diagnostic condition. The reporting of this study conforms to the CARE guidelines. 4
Case report
A 40-year-old man was referred to our department 7 days after the sudden onset of headache, neck pain, and fever. A neurological examination revealed nuchal rigidity, and the patient resisted walking or standing because of sharp headache, and neck and back pain. The muscle strength of both lower extremities was intact, and the patient’s medical history was negative for any trauma or relevant interventions. Hematological examination demonstrated hyperleukocytosis and neutrophilic granulocytosis. Considering the sudden onset of symptoms, SAH was suspected.
Head computed tomography (CT) findings were normal, and a lumbar puncture was suggested for further diagnosis. The lumbar puncture indicated that the cerebrospinal fluid (CSF) was blood-stained (Figure 1a), and a CSF analysis was performed. The total cell count was 12,600 × 106/L, and the white blood cell count was 650 × 106/L. The protein level was 3.45 g/L, and the glucose level was 2.39 mmol/L. A diagnosis of SAH was made. Meanwhile, the CSF pressure changed from 200 mmH2O to 90 mmH2O after drainage of 10 mL CSF, which indicated CSF circulation blockage, and, consequently, magnetic resonance imaging (MRI) was performed. A 2 × 1 × 1-cm mass at the L3 level was found. The lesion showed heterogeneous signal intensity and was thick, and irregular enhancement was revealed after intravenous contrast medium administration. There was also a diffuse lesion located superiorly at the adjacent level. Hyperintensity on T2- and T1-weighted images with homogeneous enhancement in postcontrast images were also shown, which was consistent with blood degradation products (Figure 1b–e).

Preoperative examination and images. Blood-stained CSF confirmed the diagnosis of SAH (a); MR images showed a 2 × 1 × 1-cm mass at the L3 level (small arrow), with a diffuse subarachnoid hemorrhage signal (big arrow) above the mass (b: T2-weighted, c: T1-weighted, d: with enhancement, e: axial image with enhancement).
The patient underwent emergent surgery for hematoma evacuation and tumor resection. Laminectomy was performed from L2 to L3 using an ultrasonic bone scalpel. The medial facet joint was exposed, and effort was taken to maintain the facet joint capsules. The spinous processes, interspinous ligaments, and ligamentum flavum of the planned laminoplasty section were kept intact. After opening the dura mater, a grayish-reddish intradural tumor was encountered at the L3 level, and a large subarachnoid blood clot was found mainly at the L2 level (Figure 2a). The blood clot was totally removed followed by a saline flush. The tumor was separated from the surrounding nerve roots and resected en bloc with careful dissection using a standard microsurgical technique (Figure 2b). After removing the tumor, the spinal dura was watertight sutured using 5-0 absorbable suture, and laminoplasty of L2–L3 was performed (Figure 2c). The surgical wound closure was then completed. Somatosensory and motor evoked potentials were monitored during the operation. Histological examination of the surgical specimen confirmed the diagnosis of schwannoma without an obvious intratumoral hemorrhagic component (Figure 2d).
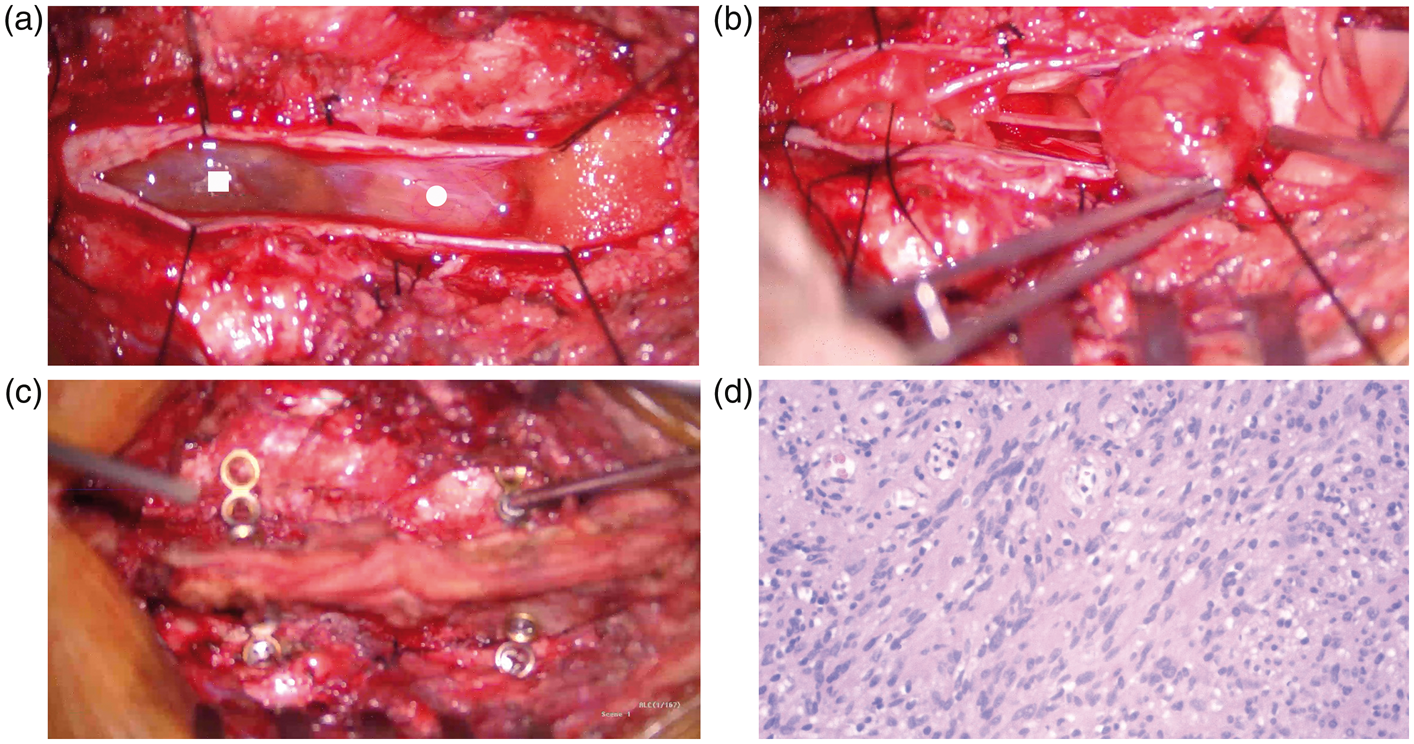
Intraoperative and pathological images. A subarachnoid blood clot (rectangle) and tumor (circle) were encountered after opening the spinal dura (a). The blood clot was totally removed and the tumor was separated from the surrounding nerve roots (b). Laminoplasty of L2–L3 was subsequently performed (c). Histological examination confirmed the diagnosis of schwannoma with no intratumoral hemorrhagic component (d) (hematoxylin and eosin staining, ×200).
The patient had an uneventful postoperative course, and no inflammatory reaction or CSF leakage was encountered. There was remarkable relief in his neck and back pain, and 5 days after the operation, he was able to walk for exercise. He was discharged on the 7th postoperative day and made a full recovery and returned to his daily activities. At the 1-year follow-up, there was no tumor or hemorrhage signal in MR images (Figure 3a–b). The patient was satisfied with the treatment.

Follow-up MR images. The patient made a full recovery with no tumor or hemorrhage signal at the 1-year follow up (a: T2-weighted, b: T1-weighted).
Discussion
Spinal pathology is a rare event in SAH. Vascular malformations and trauma are the most common causes of SAH; 5 however, tumors and hemorrhagic diatheses may also result in SAH. Among the primary spinal tumors, conus ependymoma is the most common cause associated with SAH. 6 Spinal intradural schwannomas may also result in SAH; however, these are a very rare cause, especially without spinal compression symptoms. In this summary, we identified 8 cases of SAH from a spinal schwannoma reported in the last 20 years7–14 (Table 1).
Summary of the reported cases of spinal schwannomas with SAH.
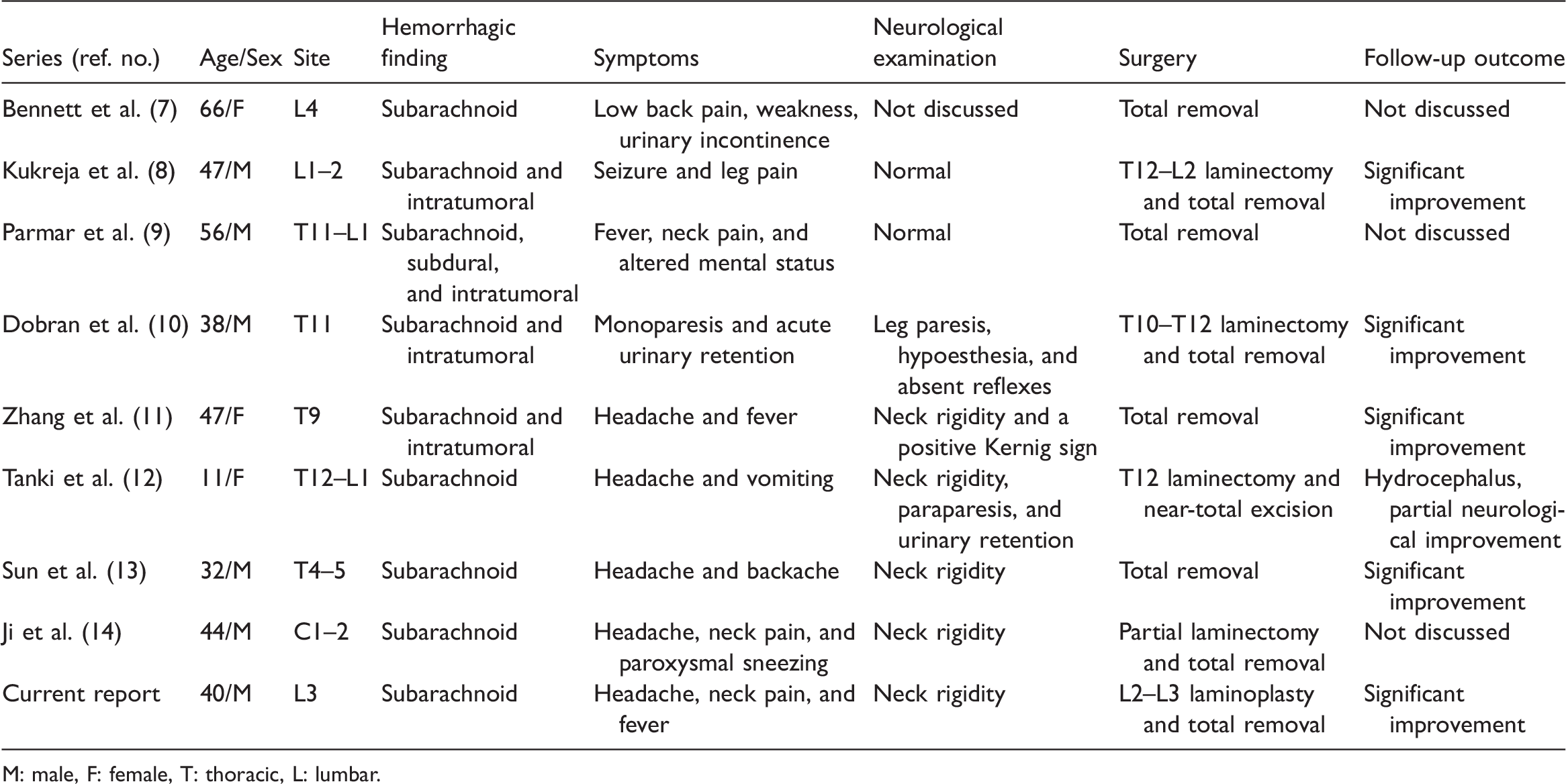
M: male, F: female, T: thoracic, L: lumbar.
Schwannomas are common, benign neoplastic lesions that tend to occur in patients between the fourth and seventh decades of life. The incidence accounts for approximately 30% of all spinal tumors. Hemorrhage from spinal schwannomas into the subarachnoid space is unusual. According to previous studies, two main theories have been proposed to explain the mechanism: the vascular theory and the mechanical theory. 15 According to the vascular theory, the ectatic hyalinized vessels of the tumor may undergo spontaneous thrombosis, which is followed by distal tumor necrosis complicated with hemorrhage. The second theory postulates that abnormal traction on the vascular attachments between the tumor and normal neural tissue injures the superficial vascular tissue, which results in hemorrhage. Kukreja et al. also demonstrated that degenerative changes in the spine may contribute to mechanical stress on the tumor. 8 According to previous studies, in approximately 50% of cases, the appearance of symptoms was related to exertion, which can be explained by the mechanical theory. 15 However, in accordance with our literature case summary, only three patients (33%) had a clear history of spine exertion. In our case, the presence of SAH developed spontaneously without previous tumor compression symptoms, and the patient reported no history of exertion. Therefore, our case cannot be explained by the mechanical theory alone. In addition, there was no sign of tumor necrosis or intratumoral hemorrhage according to histological examination. We hypothesized that neovascularization along with mechanical stress at the interface between the subdural tumor and spinal dura caused damage to the fragile ectatic vessels, followed by dissection of the blood into the subarachnoid space. This mechanism may also cause rupture of the spinal arachnoid and subsequent spinal subdural hematoma, which has been occasionally reported in ependymoma. 9 More studies are needed to provide a better understanding of the mechanism.
The clinical presentation of SAH can vary greatly. Patients may present with headache, seizures, vomiting, fever, mental status changes, and even sudden death. 8 Spinal schwannoma can be a “silent” tumor that presents with no spinal cord or nerve root compression symptoms. These characteristics result in a diagnostic challenge. Typically, the onset symptoms of spinal SAH are sudden. When there is an acute onset of symptoms, arteriovenous malformation or malignant disease is usually suspected. However, some circumstances can alert the surgeon to consider the possibility of spinal SAH, such as radicular back pain followed by headache without loss of consciousness. In addition, the sudden worsening of spinal neurological function suggests a spinal lesion. The level of vertebral pain can refer to the site of bleeding. Previously, Parmar et al. illustrated that 28% of spinal nerve sheath tumors that caused SAH presented with intracranial symptoms. 9 However, our literature review revealed that seven patients (77%) exhibited intracranial clinical symptoms.8–14 As arterial aneurysm and vascular malformation are the more common causes of SAH, an angiographic study may be the preferential examination to confirm the diagnosis. In our patient, spinal infection or SAH from vascular malformation was considered first. After a lumbar puncture, bloody CSF confirmed the diagnosis of SAH. Importantly, the CSF pressure change after drainage of 10 mL of CSF indicated that there was an intradural tumor blockage at a relatively lower spinal level, rather than vascular malformation. Consequently, spine MRI was suggested to demonstrate the underlying pathology instead of angiography, and a lumbar intradural mass was found. The subsequent surgical decompression and total removal of the mass and blood clot improved the patient’s outcome. Our case demonstrated that early diagnosis is very important. The details of the patient’s history and symptoms, especially the CSF pressure changes, provided great help to guide the clinicians in appropriate management.
Conclusion
This report emphasizes the importance of a high index of suspicion for an obstruction of the spinal subarachnoid space when there is a CSF pressure change after lumbar puncture in an SAH patient, which may result from spinal tumors. The early diagnosis and total removal of the tumor and blood clot significantly improved the patient’s outcome.
Footnotes
Acknowledgments
The authors thank the patient and his family who generously agreed to be interviewed for this research. We also thank the pathologists and radiologists for their contributions to the diagnosis of the disease.
Authors' contributions
Dongao Zhang designed the study and performed the manuscript writing and the literature review. Tao Fan supervised and revised the manuscript. Wayne Fan and Yinqian Wang helped in reviewing the manuscript. All authors have read and approved the final manuscript.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Ethics statement
Our ethics committee does not require approval for case reports. Written informed consent was obtained from the patient for publication of this case report and accompanying images.
Availability of data and materials
The datasets used or analyzed during the present study are available from the corresponding author on reasonable request.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Beijing Municipal Science and Technology Commission (No. Z191100006619040) and The Capital Health Research and Development of Special (No. 2020-2-8011).
