Abstract
Background
The prevalence of hyperuricemia is increasing, posing a significant health concern, particularly in China, where it affects 13.3% of the population. Traditional nutritional perspectives do not fully explain hyperuricemia, and the relationship between immune status and hyperuricemia remains unclear.
Objective
This study aimed to examine the association between the immunonutritional index—hemoglobin, albumin, lymphocytes, and platelets (HALP) score—and hyperuricemia.
Methods
In this retrospective cross-sectional study, clinical data from 12,598 adults at Guangdong Provincial Hospital of Chinese Medicine were analyzed using SPSS and R.
Results
Among the total participants, 3422 had hyperuricemia. Significant differences in immune-related indices were observed, with the HALP score showing a higher area under the curve (0.675, 95% confidence interval: 0.665–0.686) than traditional markers. HALP was significantly and nonlinearly associated with hyperuricemia risk, with an inflection point of 53.85. Below this threshold, each unit increase in HALP was associated with a 1.9% increase in risk (p < 0.001).
Conclusion
This cross-sectional study identified a nonlinear association between the HALP score and the prevalence of hyperuricemia. These findings suggest that HALP serves as a novel and easily accessible indicator of hyperuricemia, potentially offering insights into its immunometabolic mechanisms for future research. However, further large-scale prospective studies are necessary to confirm this association.
Keywords
Introduction
Hyperuricemia (HUA) 1 refers to a group of metabolic disorders characterized by abnormally elevated levels of uric acid (UA) in the body resulting from purine metabolism dysfunction. When UA concentrations exceed their saturation threshold in the blood or interstitial fluids, monosodium urate crystals may precipitate and deposit in the joints, internal organs, and other tissues. These crystals can trigger inflammatory responses, cause tissue damage, and potentially lead to acute organ dysfunction, such as acute kidney injury.
Epidemiological investigations 2 have indicated that the prevalence of HUA is steadily increasing, and HUA has become the second most common endocrine metabolic disorder after type 2 diabetes. In China, its prevalence has reached as high as 13.3%. 1 Multiple large-scale retrospective data analyses have shown that HUA is closely associated with various systemic conditions, including long-term mortality in hypertensive patients, 3 all-cause mortality in patients with osteoarthritis, 4 atherosclerotic cardiovascular disease, 5 and diabetic nephropathy. 6 However, current therapeutic options for HUA remain limited, highlighting the urgent need for novel strategies in the prevention and management of this condition.
UA 7 is primarily synthesized in the liver, intestines, and vascular endothelium, with approximately 30%–40% 1 excreted through the intestines. Recent studies have shown that impaired intestinal excretion 8 is a key contributor to the development of HUA and emphasized the crucial role of gut microbiota 9 in regulating UA metabolism. The gut microbiome not only influences various endocrine and metabolic disorders10,11 but is also closely associated with several inflammatory conditions. 12 Both dietary factors 13 and host characteristics affect the composition of gut microbiota, while alterations in the gut microbial profile14,15 can impact the host’s absorption and utilization of dietary components. Studies have indicated that 9 Lactobacillus, a key member of the gut microbiota, can inhibit the activity of xanthine oxidase (XO) and purine nucleoside phosphorylase while enhancing nucleoside hydrolase RihA-C activity, thereby reducing urate synthesis. Additionally, it upregulates the expression of the ABCG2 transporter protein, facilitating UA excretion. Recent research in gut microecology has revealed 16 the influence of microbial communities on the pathogenesis and progression of gout and HUA, underscoring their potential as therapeutic targets. These findings suggest that HUA is not merely a disorder of nutrient metabolism but a complex pathological process involving three key dimensions: immune regulation, nutrient absorption, and substance metabolism, characterized by distinct immunometabolic features.
Currently, the management of HUA and gout remains challenging, primarily due to the lack of reliable tools for assessing long-term serum UA levels, the susceptibility of UA concentrations to short-term dietary fluctuations, and the absence of clear guidelines regarding monitoring frequency. These limitations complicate the development of effective treatment and management strategies. However, traditional inflammatory markers—such as the neutrophil-to-lymphocyte ratio (NLR), monocyte-to-lymphocyte ratio (MLR), systemic immune-inflammation index (SII), platelet-to-lymphocyte ratio (PLR), and pan-immune-inflammation value (PIV)—have proven inadequate in capturing this immuno-nutritional perspective.
In recent years, Alshuweishi et al. 17 proposed the HALP score, a novel immunonutritional biomarker that integrates hemoglobin level (Hb), albumin level (ALB), lymphocyte count (LYM), and platelet count (PLT). The HALP score has been associated with cardiovascular and cerebrovascular diseases,18,19 various malignancies, 20 and cancer prognosis. 21 In a retrospective analysis of clinical data from 7192 participants, Alshuweishi et al. 17 reported an association between the HALP score and dyslipidemia. However, no studies to date have investigated the relationship between the HALP score and HUA. Therefore, this study aimed to explore the potential association between the HALP score and HUA. By clarifying this relationship, we aim to evaluate whether the HALP score could serve as a simple, accessible, and cost-effective biomarker for identifying individuals at increased risk of HUA, thereby informing patient management and public health interventions.
Materials and methods
Data collection
This cross-sectional study retrospectively analyzed data from adults who underwent routine health examinations at Guangdong Provincial Hospital of Chinese Medicine between January 2023 and December 2023. As a major tertiary care institution and one of the largest traditional Chinese medicine centers in Southern China, this hospital serves a diverse patient population, providing a representative sample for investigation. This study was conducted in accordance with the Declaration of Helsinki (1975). To ensure patient privacy, all collected data were anonymized, and any personally identifiable information was removed. Only individuals with complete clinical and laboratory records were included. The availability of key variables—such as age, sex, blood cell analysis (including white blood cell count (WBC), neutrophil count (NEUT), LYM, monocyte count (MONO), red blood cell count (RBC), Hb, and PLT), serum ALB, and serum UA (SUA)—was assessed. Participants with missing baseline demographic information and missing data on Hb, ALB, LYM, PLT, and SUA levels as well as those younger than 18 years were excluded. A total of 12,598 participants were enrolled, including 6214 males and 6384 females.
According to the Chinese Guidelines for the Diagnosis and Treatment of Hyperuricemia and Gout (2019), 22 HUA was defined as SUA levels of >420 μmol/L. Participants with SUA levels >420 μmol/L were classified into the HUA group, while those with SUA levels ≤420 μmol/L were classified into the non-HUA group.
Statistical analysis
The Kolmogorov–Smirnov test (p < 0.001) indicated that the continuous variables were not normally distributed. Continuous variables in both the HUA and non-HUA groups were expressed as medians and interquartile ranges (P25, P75) and compared using the Mann–Whitney U test. Binary logistic regression was used to examine the associations between immune-inflammatory indices and HUA in adults. Receiver operating characteristic (ROC) curves were plotted, and the area under the curve (AUC) was calculated. The optimal cutoff points were determined using the Youden index.
The following formulas were used to calculate PLR, NLR, MLR, SII, PIV, and HALP: (a) PLR = PLT/LYM; (b) NLR =NEUT/LYM; (c) MLR = MONO/LYM; (d) SII = (NEUT × PLT)/LYM; and (e) HALP = Hb × ALB × LYM/PLT.
A restricted cubic spline (RCS) regression model was used to analyze the relationship between the HALP score and HUA. Based on the minimum Akaike Information Criterion (AIC), a three-knot model was selected. If a nonlinear relationship was detected, threshold effect analysis was used to identify inflection points between the HALP score and HUA. The threshold was estimated by testing all possible values, and the value with the highest likelihood was selected as the inflection point. A two-piecewise logistic regression model was used on either side of the threshold to examine the association between the HALP score and HUA risk. A likelihood ratio test was used to compare the segmented model with the original model. All statistical analyses were conducted using SPSS 18.0 for Windows (SPSS Inc., Chicago, IL, USA), R (version 4.2.1), and Zstats. A significance level of α = 0.05 was used, with p < 0.05 considered statistically significant. The reporting of this study conforms to the Strengthening the Reporting of Observational studies in Epidemiology (STROBE) guidelines. 23
Results
A total of 12,598 adults were included in this study, with 3422 in the HUA group and 9176 in the non-HUA group. The median age of the cohort was 41.0 (interquartile range: 33.0–51.0) years, with females constituting 50.7% of the population. Baseline characteristics differed between the two groups in terms of sex, WBC, NEUT, LYM, MONO, RBC, Hb, and ALB (Table 1). Common immune-related indices, including PLR, NLR, MLR, SII, PIV, and HALP, were calculated, all of which showed statistically significant differences between the two groups (Table 2). A binary logistic regression model was constructed using age, male sex, PLR, NLR, MLR, SII, PIV, and HALP as variables (Table 3). In multivariable regression analysis, all composite indices were entered into the model and adjusted for age and sex. The MLR was not significant in the multivariable analysis (p > 0.05), suggesting that the association between MLR and HUA risk was attenuated after adjustment. PLR, SII, PIV, and HALP remained statistically significant in both univariate and multivariate analyses.
Comparison of baseline characteristics in the adult population.

ALB: albumin level; Hb: hemoglobin level; HUA: hyperuricemia; LYM: lymphocyte count; MONO: monocyte count; NEUT: neutrophil count; PLT: platelet count; UA: uric acid.
Comparison of novel immune-related indicators between the two groups.

HALP: hemoglobin, albumin, lymphocytes, and platelets; HUA: hyperuricemia; MLR: monocyte-to-lymphocyte ratio; NLR: neutrophil-to-lymphocyte ratio; PIV: pan-immune-inflammation value; PLR: platelet-to-lymphocyte ratio; SII: systemic immune-inflammation.
Independent predictors of HUA in the adult population: a binary logistic regression analysis.

CI: confidence interval; HALP: hemoglobin, albumin, lymphocytes, and platelets; HUA: hyperuricemia; MLR: monocyte-to-lymphocyte ratio; NLR: neutrophil-to-lymphocyte ratio; OR: odds ratio; PIV: pan-immune-inflammation value; PLR: platelet-to-lymphocyte ratio; SII: systemic immune-inflammation.
These indicators were included in ROC curve analysis, and AUCs were calculated (Figure 1). The AUCs for PLR, SII, and PIV were 0.394 (95% confidence interval (CI): 0.383–0.405), 0.473 (95% CI: 0.462–0.484), and 0.553 (95% CI: 0.542–0.564), respectively. Based on the Youden index, the optimal cutoff points for predicting HUA were as follows: PLR = 40.45 (sensitivity = 0.999, specificity = 0.001), SII =111.68 (sensitivity = 0.998, specificity = 0.004), and PIV = 125.76 (sensitivity =0.696, specificity = 0.396). The HALP score demonstrated a higher AUC of 0.675 (95% CI: 0.665–0.686) and an optimal cutoff point of 57.87 (sensitivity = 0.692, specificity =0.572), indicating a higher predictive value for HUA than traditional immune-inflammatory markers. The diagonal line in Figure 1 represents the reference line (AUC = 0.50).
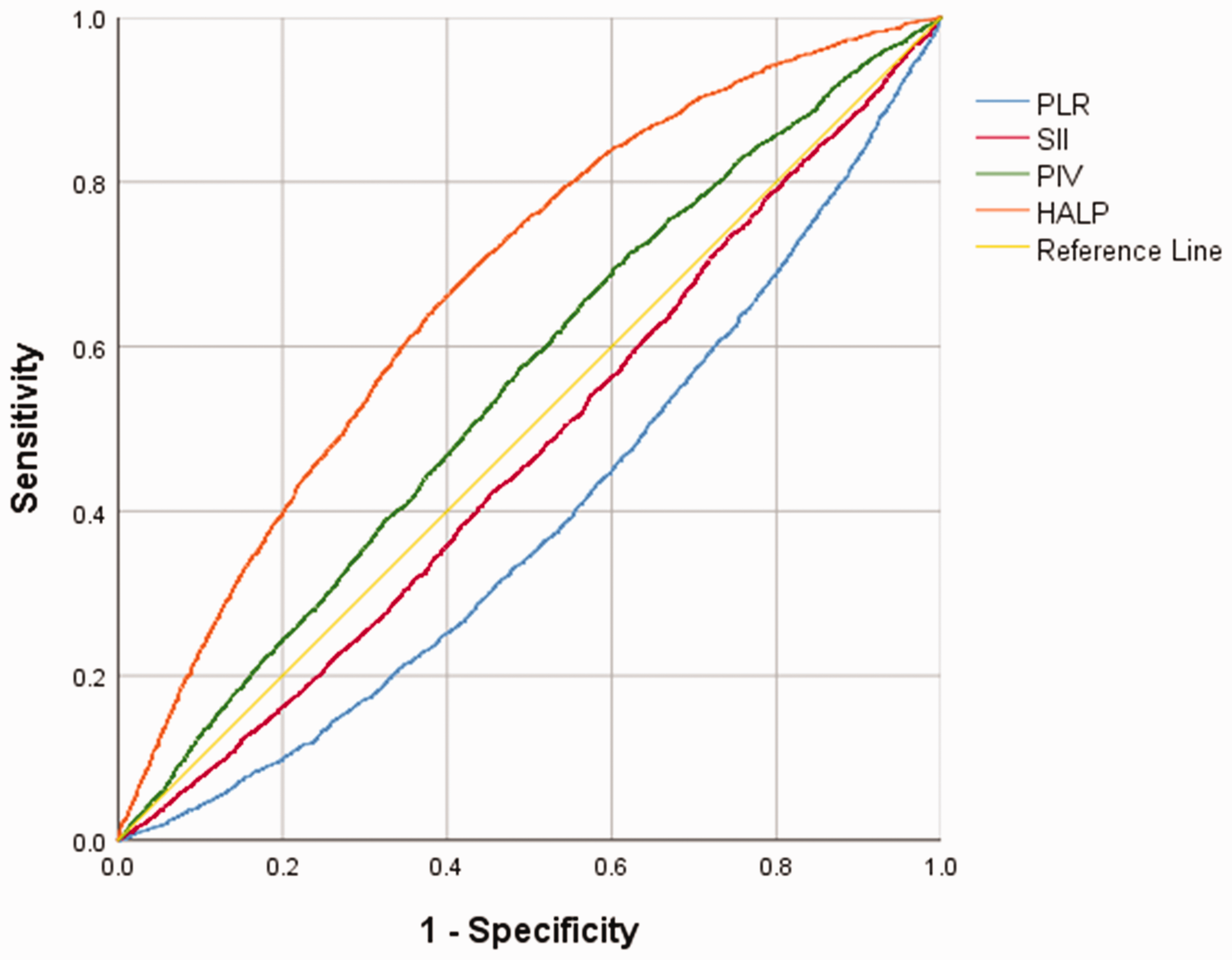
ROC curves for PLR, SII, PIV, and HALP scores in diagnosing HUA. HALP: hemoglobin, albumin, lymphocytes, and platelets; PIV: pan-immune-inflammation value; PLR: platelet-to-lymphocyte ratio; ROC: receiver operating characteristic; SII: systemic immune-inflammation.
RCS regression identified a nonlinear relationship between the HALP score and HUA risk (Figure 2). Logistic regression combined with a two-piecewise logistic regression model confirmed the nonlinear relationship between the HALP score and the risk of HUA (log-likelihood ratio test p < 0.05) (Table 4).

Restricted cubic spline regression analysis of the relationship between HALP as a continuous variable and HUA. The blue solid line represents the estimated adjusted OR, while the shaded area indicates the 95% CI. The dashed line signifies an OR or risk ratio of 1.0. The spline curve is adjusted for age and sex.
Threshold effect analysis of HALP score on HUA.

OR: odds ratio; CI: confidence interval; HALP: hemoglobin, albumin, lymphocytes, and platelets; HUA: hyperuricemia.
The inflection point between HALP and HUA was 53.85. When the HALP score was <53.85, each one unit increase in HALP was associated with a 1.9% increase in the adjusted odds of HUA (adjusted OR: 1.019, 95% CI: 1.012–1.026).When the HALP score was ≥53.85, each one unit increase in HALP was associated with a 0.3% increase in the adjusted odds of HUA (adjusted OR: 1.003, 95% CI: 1.000–1.005).
Discussion
To the best of our knowledge, this is the first large-sample cross-sectional study that investigated the association between the HALP score and HUA. We found a nonlinear relationship between the HALP score and HUA risk, indicating that within a certain range, HALP scores are significantly associated with the risk of HUA. These findings may provide new insights into the pathogenesis and clinical management of HUA.
UA 7 is primarily synthesized in the liver, intestines, and vascular endothelium. It is not only the end product of exogenous purine metabolism from dietary intake but is also endogenously produced through the metabolism of purines from damaged, dead, or apoptotic cells. The development of HUA is influenced by multiple factors, including diet, genetics, metabolic status, and medication history. With advances in research,1,24 an increasing number of scholars have emphasized that HUA has significant immune-related components. The HALP score, composed of Hb, ALB, LYM, and PLT, is considered valuable in evaluating the immunonutritional status.
Hemoglobin is an important carrier of oxygen in the human body 25 and also serves as a major transporter and buffer of nitric oxide compounds. 26 Therefore, it is considered one of the key biochemical indicators reflecting the body’s nutritional status. Current evidence suggests that Hb is negatively associated with endothelial function. 26 Elevated Hb may cause endothelial dysfunction and is identified as an independent risk factor for metabolic syndrome 25 and gestational diabetes mellitus. 27 Fang et al. 28 analyzed clinical data from 33,183 adults to explore the association between Hb and SUA under different glycemic statuses. They found that the SUA levels tended to increase with rising Hb in adults younger than 60 years with normal glucose levels or prediabetes. Similar findings were reported by He et al, 29 who also observed a positive correlation between UA and Hb. Individuals with Hb ≥170 g/L should therefore monitor SUA levels closely to reduce cardiovascular disease risk. Our results showed that Hb was significantly higher in the HUA group than in the non-HUA group (p < 0.001).
Scholars have suggested that Hb can indirectly reflect blood viscosity.25,30 Elevated Hb may lead to increased blood viscosity, which can cause circulatory stagnation, reduce renal blood flow, and subsequently impair UA excretion. In addition, pathological increases in Hb 25 may indicate the presence of hypoxia in the body, and higher Hb levels often imply a greater inflammatory burden. UA, one of the major antioxidants in the human body, scavenges up to 55% of extracellular free radicals 31 and also possesses pro-oxidative properties. As the final product of purine metabolism, UA synthesis depends on the catalytic activity of xanthine dehydrogenase (XDH) and XO. 32 Under hypoxic conditions, XDH is converted into XO. 31 Excessive XO production can disrupt the balance between antioxidants and pro-oxidants, thereby promoting the development of inflammatory states.
ALB is considered a negative acute-phase protein. 33 During conditions of malnutrition (such as cancer-related cachexia) and acute inflammatory responses, ALB tends to decrease to varying degrees. However, in our study, ALB was significantly higher in the HUA group than in the non-HUA group (p < 0.001).
We speculate that several possible mechanisms may explain this finding. ALB, the most abundant protein in the human body, reflects both nutritional status and hepatic synthetic function. 34 As the liver is a major site of UA production, enhanced hepatic synthesis may contribute to HUA development. In addition to endogenous UA production, HUA is closely associated with dietary factors. An unbalanced diet may lead to elevated ALB and UA levels. Furthermore, albumin can bind to various ligands and has free radical-scavenging capabilities, which may confer specific antioxidant properties. 35 It also plays important roles in maintaining plasma oncotic pressure and exerting anti-inflammatory effects. 36 Elevated ALB may represent a pathological compensatory response to a high inflammatory burden within the body. However, this hypothesis requires further experimental validation.
Research has revealed that UA 37 can increase the expression of immune cell markers CD40 and CD86, thereby initiating immune responses by enhancing antigen presentation to T cells and providing costimulatory signals. Yu et al. 37 reported that high levels of soluble UA enhance Th2-dominated immune responses, suggesting that UA may act as an immunoadjuvant in the treatment of certain diseases. Our study data indicate that LYM was higher in the HUA group than in the non-HUA group (p < 0.001), consistent with previous findings.
In addition to Hb, ALB, and LYM, PLT is part of the HALP score. HUA 38 is associated with coagulation disorders; UA can induce inflammatory states and vascular damage through oxidative stress reactions, potentially leading to thrombus formation. PLT plays a crucial role in this process. Several retrospective studies 38 have shown that HUA is often accompanied by abnormal PLT, with a positive correlation observed. 39 However, in our study, PLT did not differ significantly between the two groups (p = 0.116). This may be attributed to the broad age range of the sample and the complexity of comorbidities, which could introduce variance in the study.
The HALP score is a comprehensive assessment index based on inflammatory status (LYM, PLT) and overall nutritional status (Hb, ALB). The results of this study suggest that compared with simple inflammatory markers, the HALP score can better assess the overall nutritional status and immune-inflammatory state of individuals with HUA. Notably, we found a nonlinear relationship between the HALP score and HUA risk, with an inflection point of 53.85. When HALP is below 53.85, each one unit increase in HALP corresponds to a 1.9% increase in the adjusted OR (adjusted OR: 1.019; 95% CI: 1.012–1.026) for HUA. This finding suggests that HALP not only holds potential value for early warning of HUA but also serves as an effective tool for clinical screening of HUA, providing a basis for early intervention in high-risk populations. In addition, the nonlinear relationship of the HALP score suggests that HUA is not only a manifestation of metabolic disorder but also a result of the interplay between immune and nutritional status, offering important clues for further understanding the multifaceted mechanisms of this disease.
The clinical management of HUA remains challenging due to limited treatment options and high disease complexity. The frequency of SUA retesting in HUA patients and the clinical value of single measurements remain controversial. Some patients may show normal SUA levels on a single test and discontinue further monitoring while still experiencing a high inflammatory burden. This can complicate HUA disease management and increase the risk of acute and chronic complications. Therefore, relying solely on SUA levels for disease assessment may not comprehensively capture the complexity of the disease, particularly in early or chronic stages.
Our study revealed a close association between HALP scores and HUA in an immune-inflammatory context, suggesting its potential as a novel biomarker for further investigation. These findings suggest that by reflecting both inflammatory and nutritional status, the HALP score could assist in identifying patients with normal single SUA measurements who might harbor a higher risk, thus warranting greater attention in future clinical research aimed at optimizing management strategies. The nonlinear association observed in our study provides a rationale to explore whether the HALP score could aid in the comprehensive evaluation of patients with HUA or gout and inform individualized monitoring strategies in future prospective studies. For instance, the inflection point identified (53.85) suggests that patients with lower HALP scores may represent a distinct subgroup with a different level of association with HUA, possibly reflecting a higher metabolic-inflammatory burden. This hypothesis-generating finding warrants further investigation to determine whether it could inform monitoring frequency. This finding does not imply that patients with higher HALP scores require no further monitoring; continued attention to SUA dynamics and metabolic health remains universally important. As with all cross-sectional findings, potential clinical implications regarding monitoring strategies require validation in future prospective studies before they can be considered for clinical application.
However, this study has certain limitations. First, it is a single-center retrospective cross-sectional study, which may be subject to selection and recall biases. Second, participants’ medical history, medication history, smoking, and drinking habits were not comprehensively collected, which may have influenced the results and introduced bias to the conclusions. The data were derived from a specific region’s health examination population, primarily focusing on middle-aged individuals, which may limit the applicability of the findings to other age groups. Additionally, the study design precludes causal inference and can only reveal associations between HALP scores and HUA. Therefore, future multicenter prospective cohort studies with larger sample sizes are needed to further validate these findings and enhance their applicability and generalizability in clinical practice.
Footnotes
Acknowledgments
None.
Author contributions
Yan M: Writing–original draft, data curation, writing–review and editing. Shi W: Writing–original draft, data curation. Wang H: Writing–original draft. Li X: Writing–review and editing.
Author claim
All the authors indicate that this article has not been published in any journal and is not considered for publication in other journals.
Data availability
Data will be made available on request.
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Ethical statement
This study involves human participants and has been reviewed and approved by the Ethics Committee of Guangdong Provincial Hospital of Traditional Chinese Medicine (No.ZE2025-196). In accordance with relevant regulations and with the approval of the ethics committee, a waiver of informed consent was granted for this study.
Funding
This study was funded by the Qihuang Scholars Studio (No.: National Administration of Traditional Chinese Medicine Human Resources [2022] No. 6) and the Lin Dingkun Guangdong Province Famous Traditional Chinese Medicine Practitioner Inheritance Studio (No. 0103030912).
