Abstract
Background
Neural epidermal growth factor-like 1 protein has emerged as a significant antigen in membranous nephropathy, an important cause of nephrotic syndrome in adults. However, the prognosis of neural epidermal growth factor-like 1–associated membranous nephropathy remains limited and requires further exploration.
Methods
A Chinese retrospective cohort of 446 patients diagnosed with membranous nephropathy on renal biopsy was categorized into phospholipase A2 receptor-associated (n = 358) and phospholipase A2 receptor–negative (n = 88) membranous nephropathy groups. The phospholipase A2 receptor–negative group was subjected to neural epidermal growth factor-like 1 fluorescence staining. Multivariate regression analyses, stratified analyses, and Kaplan–Meier analyses were used to explore the correlation of neural epidermal growth factor-like 1 with the risk of end-stage renal disease.
Results
We identified 11 cases of neural epidermal growth factor-like 1-associated membranous nephropathy, accounting for 12.7% of phospholipase A2 receptor–negative membranous nephropathy cases. These patients were predominantly young women with a history of fairness cream use. In patients with phospholipase A2 receptor–negative membranous nephropathy, multivariable regression analysis showed that the neural epidermal growth factor-like 1-positive group had a higher risk of end-stage renal disease compared with the neural epidermal growth factor-like 1-negative group (hazard ratio = 3.2, p = 0.026). Stratified analysis showed that this association was particularly significant in patients with systemic lupus erythematosus (hazard ratio = 5.1, p = 0.041). Kaplan–Meier analysis also revealed a higher risk in neural epidermal growth factor-like 1–positive patients (p = 0.003).
Conclusion
Neural epidermal growth factor-like 1 expression is an independent risk factor for end-stage renal disease, especially in patients with systemic lupus erythematosus, with an incidence rate of approximately 12.7% in patients with phospholipase A2 receptor–negative membranous nephropathy.
Keywords
Background
Membranous nephropathy (MN) is a leading cause of nephrotic syndrome (NS) in adults, characterized by the thickening of the glomerular basement membrane (GBM) and changes in the renal tubules and interstitial tissue. 1 The prognosis and treatment responsiveness of patients with MN vary greatly. 2 Since the discovery of M-type phospholipase A2 receptor (PLA2R) in 2009, 3 the identification of target antigen in MN has advanced rapidly. 4 It has been established that PLA2R plays a crucial role in the diagnosis and treatment monitoring of MN. However, approximately 30% of patients with MN are PLA2R-negative, 4 and there is an urgent need to further accurately classify patients with MN and their clinical features.
In the previous decade, new target antigens of MN such as thrombospondin type-1 domain-containing 7A (THSD7A), 5 exostosin 1/exostosin 2 (EXT1/2), 6 neural epidermal growth factor-like 1 protein (NELL-1), 7 semaphorin 3B, 8 protocadherin 7, 9 and neural cell adhesion molecule 1 10 have been discovered. The classification of MN associated with different target antigens is of great significance to the diagnosis and treatment of MN and has attracted attention. 11
A recent study found that NELL-1 could be the second most common MN antigen after PLA2R. 12 Approximately 5%–10% of patients with MN have NELL-1-related MN.13,14 An association has been found between NELL-1 MN and various conditions, including the use of sulfur-containing medications such as alpha-lipoic acid, skin-whitening creams, and traditional indigenous medicines with high mercury (Hg) content. It has also been linked to underlying conditions such as malignancies, infections (e.g. human immunodeficiency virus infection), sarcoidosis, hematopoietic stem cell transplantation, graft-versus-host disease, and rheumatoid arthritis.15–17 However, there is limited information about the prognosis and clinical characteristics of NELL-1-associated MN in Chinese populations. Exploring this association is crucial for formulating targeted prevention and treatment strategies.
Materials and methods
Study design and participants
A retrospective cohort study was conducted by consecutively enrolling 532 patients with biopsy-confirmed MN treated at Shenzhen Second People’s Hospital between 1 January 2015 and 1 March 2022. Patients whose paraffin samples could not be subjected to immunofluorescence (IF) staining, whose biopsy contained fewer than 8 glomeruli (n = 3), and whose follow-up duration was <100 days were excluded (n = 83). A flowchart of the inclusion and exclusion criteria for patients is shown in Figure 1. After enrollment, we performed PLA2R IF staining on the paraffin-embedded renal tissue samples of 446 patients with MN. EXT1/2 and THSD7A immunostaining was additionally performed for all NELL1-positive samples (n = 11). For those who tested negative for PLA2R, we further conducted NELL-1 IF staining on renal tissue samples. Baseline clinicopathological data and follow-up data were collected. The clinicopathological features and prognosis of the NELL-positive/PLA2R-MN and NELL-negative/PLA2R-MN cases were analyzed in detail. Kaplan–Meier estimates and regression analysis were performed to assess the impact of NELL1 expression on the risk of end-stage renal disease (ESRD). Our study was performed in accordance with the Helsinki Declaration of 1975 as revised in 2024. 18 All patient data in this study have been rigorously de-identified. The reporting of this study conforms to the Strengthening the Reporting of Observational studies in Epidemiology (STROBE) guidelines. 19

Flow chart of the inclusion and exclusion criteria for patients with MN. MN: membranous nephropathy.
IF for podocyte antigen staining
NELL-1 was evaluated in 3-µm formalin-fixed paraffin sections of PLA2R-negative biopsies with standard heat-induced epitope retrieval and overnight 4°C primary antibody incubation, followed by fluorescent secondary antibody and 4′,6-diamidino-2-phenylindole counterstaining. Granular GBM fluorescence visible at both low and high magnification was considered positive. EXT1/2 and THSD7A immunostaining was additionally performed for all NELL1-positive cases. The fluorescence intensity was judged by two nephrologists. IF staining showed NELL-1 granular distribution along the GBM, as seen in Figure 2(b). In this study, the paraffin-embedded kidney biopsy specimens of patients who had undergone renal biopsy at our hospital were accessed and used from the biopsy specimen bank of our hospital. We collected patients’ hospitalization data retrospectively and anonymously to protect patient privacy. The study did not involve any invasive procedures that could harm the patients, posed no risk of complications, and did not involve any form of compensation. As reported elsewhere, 19 due to the observational nature of the study, the need for informed consent was waived by the Ethics Committee of Shenzhen Second People’s Hospital. All the methods were performed in accordance with the relevant guidelines and regulations. A statement to this effect has been included in the methods section.
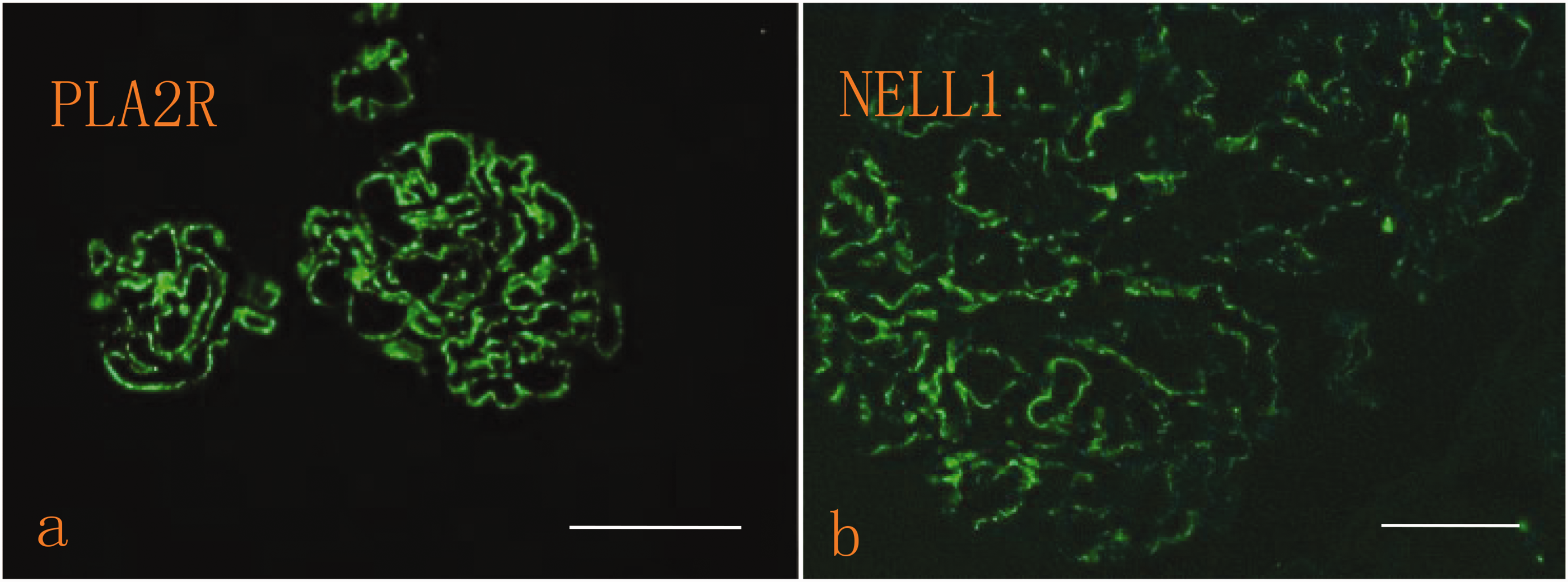
Glomerular NELL-1 and PLA2R staining with immunofluorescence. (a) Immunofluorescence analysis showing PLA2R deposits linearly along the glomerular capillary wall (150× magnification, scale bar: 100 µm) and (b) Immunofluorescence staining shows NELL-1 granular distribution along GBM (200× magnifications, scale bar: 50 µm). NELL-1: neural epidermal growth factor-like 1; GBM: glomerular basement membrane.
Diagnostic criteria
The diagnosis of MN is based on pathological parameters assessed using light microscopy, immunofluorescence, and electron microscopy that show diffuse thickening of the basement membrane along the GBM epithelial cells. 20 The diagnosis of lupus MN (LMN) and scoring of the Austin activity (total score 24) were performed according to relevant indicators by a professional renal pathologist, 21 and the diagnosis of hepatitis B virus–associated MN (HBV-MN) was confirmed by the presence of HBV antigen in renal tissue sections.21,22 The pathological diagnosis was affirmed by two pathologists.
Molecular docking of inorganic Hg with NELL1 protein
We retrieved the 2D mol2 file of inorganic Hg from the Public Chemical (PubChem) database and employed AutoDockTools, version 1.5.6, to scrutinize the spatial conformation, assign charges and atomic types, and designate flexible bonds as rotatable, subsequently saving the file in pdbqt format to serve as the ligand. We conducted a search within the Universal Protein Resource (UniProt) database for the target’s three-dimensional conformation and procured the three-dimensional structure of protein NELL1 from the Protein Data Bank, storing it in pdb format. We used AutoDockTools, version 1.5.6 to eliminate water molecules, introduce hydrogen atoms, assign charges, and perform optimization. We established a default grid box and saved the refined protein structure in pdb format to act as the receptor. We employed AutoDock Vina, version 1.1.2 for the molecular docking of the protein and the small molecule and employed PyMOL for visualizing the docking outcomes. Binding energy ranging from −5 to 0 kcal/mol signified the presence of binding affinity, that from −5 to −7 kcal/mol indicated a favorable binding interaction, and that less than −7.5 kcal/mol denoted an exceptionally strong binding capacity.
Outcomes
The primary clinical outcome of this study was the renal composite endpoint event of ESRD, defined as an estimated glomerular filtration rate (eGFR) <15 mL/min/1.73 m2 or 50% lower than that baseline or the initiation of renal replacement therapy. 23 The secondary clinical outcome was the achievement of proteinuria remission. Partial remission is defined as stable renal function with a ≥50% reduction in proteinuria to a level between 0.3 and 3.5 g/24 h. Complete remission (CR) is characterized by proteinuria <0.3 g/24 h. Patients who do not meet either criterion are classified having non-remission. 23
Statistical analyses
All the statistical analyses were performed using the Empower Stats and R software, version 3.4.1. This research used mixed-effects model analysis to interpolate missing data. Data following normal distribution are presented as mean ± SD, while non-normally distributed data are expressed as median (P25, P75) values. The Mann–Whitney U test was used for non-normally distributed two-group comparisons, and the Kruskal–Wallis test was employed for multiple-group comparisons. The association between NELL1 expression and the risk of progression to ESRD was assessed using Kaplan–Meier estimation and regression analysis. For all tests, p value of <0.05 was considered statistically significant.
Results
The baseline characteristics of the study population are shown in Table 1. We analyzed 532 renal biopsies diagnosed with MN between 2015 and 2022 at the Shenzhen Second People’s Hospital to investigate the prevalence and clinical characteristics of NELL-1–associated MN among Chinese patients with PLA2R-negative MN. All biopsies were analyzed at the Guangzhou Jinyu Medical Laboratory. As shown in Figure 1, 446 patients who met the inclusion criteria and were diagnosed with MN on renal biopsy were included in the study. Of these, 358 patients (80.3%) had PLA2R-positive MN, while 88 (19.5%) had PLA2R-negative MN (Figure 1). Within the PLA2R-negative MN group, 11 patients (12.7%) tested positive for NELL-1. As demonstrated in Figure 2, IF staining was used to visualize the distribution patterns of PLA2R and NELL-1 proteins within the glomerular structure. The results indicated that PLA2R exhibited a distinct linear distribution along the glomerular capillary walls, consistent with its known role in MN pathogenesis. In contrast, NELL-1 was observed to have a granular distribution pattern within the basement membrane. This differential localization may suggest distinct pathophysiological roles for these proteins in glomerular diseases, PLA2R being associated with immune-mediated injury and NELL-1 potentially involved in basement membrane remodeling or other localized processes. Additionally, in the analysis of the secondary etiology of MN, we found that in the 77 cases of the NELL-negative/PLA2R-negative group, 49 (7/11, 63.6%) were those of LMN, 9.09% were those of HBV-MN, and the remaining 27.27% were those of MN of unknown etiology. Similarly, in the NELL-positive/PLA2R-negative group, 63.6% patients had LMN, 1 (9%) had tumor-related MN, and 9 (81.8%) had a history of long-term use of skin fairness cream (Figure 1). As shown in Table 1, patients with NELL-1–positive/PLA2R-negative MN exhibited a higher prevalence of NS, and a significantly greater proportion of them progressed to ESRD compared with patients with NELL-1–negative/PLA2R-negative.
Baseline characteristics, treatments, and outcomes.

Results are expressed as median and interquartile range value or as or mean ± SD values.
ACEI/ARB; angiotensin-converting enzyme inhibitor/angiotensin II receptor blocker; ALB: albumin; ANA: antinuclear antibody; C1q: complement component 1q; C3: complement component 3; CR: complete remission; eGFR: estimated glomerular filtration rate; ESRD: end-stage renal disease; FSGS: focal segmental glomerulosclerosis; FU: follow-up; Hb: hemoglobin; HBV-MN: hepatitis B virus–associated membranous nephropathy; IATt: immunosuppressive antiproteinuric treatment; IF: immunofluorescence; IFTS: interstitial fibrosis and tubular atrophy score; IgA: immunoglobulin A; IgG: immunoglobulin G; IgM: immunoglobulin M; LMN: lupus membranous nephropathy; MN: membranous nephropathy; NELL-1: neural epidermal growth factor-like 1; PR: partial remission; TC: total cholesterol; UA: uric acid; UPCR: urinary protein/creatinine ratio.
NELL1 as an independent risk factor for ESRD in patients with PLA2R-negative MN
To comprehensively evaluate the prognostic factors for patients with PLA2R-negative MN, we constructed a multivariate regression model (Table 2). Our analysis indicated a significant link between ESRD and NELL1 expression, indicating that NELL1 is a key prognostic indicator for patients with MN (hazard ratio (HR) = 6.1, 95% confidence interval (CI): 2.2–12.4, p = 0.007). After adjusting for age, gender, systemic lupus erythematosus (SLE) status, presence of NS, immunosuppressive therapy, and eGFR, NELL1 expression remained an independent risk factor for ESRD in patients with PLA2R-negative MN (HR = 3.2, 95% CI: 2.3–11.6, p = 0.026) (Table 2). Furthermore, stratified analysis was used to explore the link between NELL1 expression and ESRD based on the history of SLE. In the subgroup without SLE history (n = 32), the HR for ESRD in patients with NELL1-positive MN was 2.1 times higher compared with that in patients with NELL1-negative. However, this result showed a non-significant p value of 0.996 (95% CI: 0.5–13.89), indicating no statistically significant association between NELL1 expression and the risk of ESRD in individuals without SLE history. Conversely, in the subgroup with SLE history (n = 56), patients with NELL1-positive MN exhibited a 5.1-fold increased risk of developing ESRD compared with NELL1-negative patients (95% CI: 1.02–29.93, p = 0.041), representing a statistically significant difference (Table 3). This suggests that NELL1 expression might serve as a more relevant biomarker for ESRD risk in patients with SLE history, potentially due to the interplay between SLE-related immune mechanisms and the pathophysiological processes involving NELL1 in the kidneys. Figure 3 presents a Kaplan–Meier survival analysis of ESRD-free survival in patients with PLA2R-negative MN, stratified by NELL1 antigen staining status. The analysis revealed a significant difference in ESRD-free survival between patients with NELL1-positive and NELL1-negative, as indicated by the p value of 0.003. Patients with NELL1-positive showed a lower ESRD-free survival rate over time compared with patients with NELL1-negative, suggesting that NELL1 positivity was associated with an increased risk of progression to ESRD in this cohort.
Cox regression analyses for the time to ESRD.

Adj HR: adjusted hazard ratio; ALB: albumin; ANA: antinuclear antibody; CI: confidence interval; eGFR: estimated glomerular filtration rate; ESRD: end-stage renal disease; FSGS: focal segmental glomerulosclerosis; HR: hazard ratio; NELL-1: neural epidermal growth factor-like 1; UPCR: urinary protein/creatinine ratio.
Stratified analyses based on SLE history to evaluate the association between NELL1 expression and ESRD risk.

CI: confidence interval; ESRD: end-stage renal disease; HR: hazard ratio; NELL-1: neural epidermal growth factor-like 1; SLE: systemic lupus erythematosus.
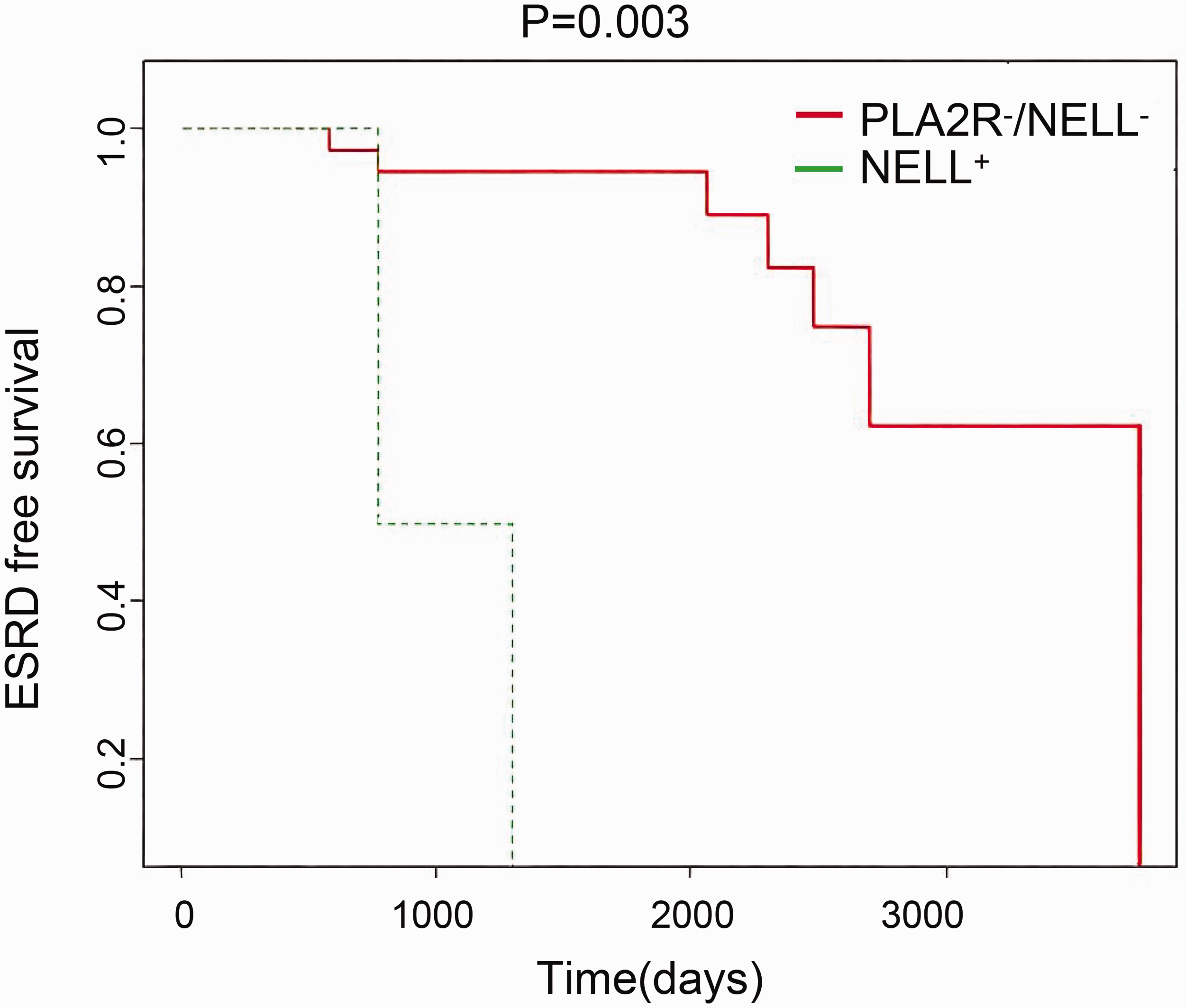
Kaplan–Meier analysis of ESRD-free survival in patients with PLA2R-negative MN. ESRD: end-stage renal disease; MN: membranous nephropathy.
Case details of patients with NELL-1-associated MN in the cohort
As shown in Table 4, our study cohort included 11 patients diagnosed with NELL-1-associated MN. Their median age was 34 years, with a broad age range spanning from 22 to 64 years. The median urinary protein excretion was 5.6 g/24 h (Q1–Q3: 3.5–6.9 g/day), indicative of substantial protein loss in this cohort. None of the EXT1/2/THSD7A stains were positive in any NELL1-positive case. The median eGFR was 92.3 (Q1–Q3: 42–119) mL/min/1.73 m2. Serum albumin levels were also measured and showed a median value of 23.4 (Q1–Q3: 23.1–25.9) g/L. Six patients were treated with immunotherapy, and 2 patients progressed to ESRD during the follow-up period. Antinuclear antibody positivity was observed in 90.9% of the patients with NELL1-positive MN. Notably, all patients exhibited granular positivity for NELL-1 in the glomerular capillary loop deposits (Figure 2). All cases of NELL-1-associated MN demonstrated segmental or incomplete bright granular staining for IgG (2–3+/3), and IgG1 staining was also positive in the capillary loops. This suggests a distinct immunological pattern in the pathogenesis of NELL-1-associated MN. Patient 6 presented with significant proteinuria and was concurrently diagnosed with thyroid nodule classified as Thyroid Imaging Reporting and Data System category 4b and NELL-1-associated MN. Her blood sample was positive for anti-NELL1 antibodies. After 345 days of immunotherapy, her renal function remained stable; however, marked proteinuria persisted. Patient 1 was a middle-aged man diagnosed with SLE and NELL1-associated MN, presenting with massive proteinuria and stage 4 chronic kidney disease. Thrombotic microangiopathy was identified on kidney biopsy. Despite treatment with corticosteroids and intravenous immunoglobulin, the patient developed severe pneumonia. His proteinuria remained unresolved, culminating in ESRD, and hemodialysis treatment was initiated after 225 days. Patients 2 and 4 were diagnosed with SLE and exhibited substantial proteinuria; they were managed with angiotensin-converting enzyme inhibitor/angiotensin II receptor blocker therapy alone. This conservative approach was chosen due to their compromised baseline renal function and lack of acute pathological features in renal biopsy specimens. Despite adherence to this regimen, both patients continued to experience persistent proteinuria throughout the 2-year follow-up period. Notably, patient 4 advanced to ESRD within 357 days of initiating treatment. Additionally, patient 11, after 308 days of immunosuppressive therapy, maintained stable renal function but did not achieve proteinuria remission. Furthermore, patients 3, 5, 8, and 10 exhibited stable renal function and achieved CR of urinary protein during the follow-up period.
Characteristics and outcomes of patients with NELL1-positive MN.

ANA: antinuclear antibody; C1q: complement component 1q; C3: complement component 3; eGFR: estimated glomerular filtration rate; IC: immune complex C; IF: immunofluorescence; IgA: immunoglobulin A; EXT1/2: exostosin 1/exostosin 2; IgG: immunoglobulin G; IgG1: immunoglobulin G subclass 1; IgG2: immunoglobulin G subclass 2; IgG3: immunoglobulin G subclass 3; IgG4: immunoglobulin G subclass 4; IgM: immunoglobulin M; LMN: lupus membranous nephropathy; MN: membranous nephropathy; NELL-1: neural epidermal growth factor-like 1; SLE: systemic lupus erythematosus; THSD7A: thrombospondin type-1 domain-containing 7A.
In addition, 81.8% of the identified patients with NELL-1–associated MN reported having used skin fairness creams. This correlation underscores a potential link between these cosmetic products and the development of MN, which warrants attention. This study employed molecular docking techniques to explore the interaction between Hg, a potentially harmful substance commonly found in skin fairness creams, and NELL1 protein. The analysis demonstrated that Hg established hydrophobic interactions with key amino acid residues LYS96, LYS80, and THR94, with interaction distances of 2.3 pm, 2.4 pm, and 2.2 pm, respectively (Supplementary Figure 1). These short interaction distances substantiate a strong and intimate binding between Hg and NELL1 protein.
Discussion
NELL-1-associated MN has become the second most common MN target antigen after PLA2R-associated MN. This study investigated the clinicopathological features and prognostic characteristics of patients with PLA2R-negative MN based on NELL1 expression. Cases with positive glomerular NELL-1 staining accounted for 12.7% of PLA2R-negative MN cases. Multivariable regression models and stratified analyses demonstrated that NELL1 expression is a significant prognostic factor for ESRD in patients with PLA2R-negative MN, particularly in those with a history of SLE. Kaplan–Meier analysis further supported these findings, indicating that NELL1-positive patients are at a higher risk of ESRD.
In our real-world cohort, NELL1 positivity was observed in 2% of all MN cases, a figure that aligns closely with recent real-world multicenter data from China (1.3%–2.5%); however, it is substantially lower than the percentages reported in earlier, tertiary-care or referral-based series from Western and Japanese populations (5%–10%).22,24 This discrepancy could primarily be attributable to regional and ethnic differences in antigen exposure and referral patterns. NS occurred in only 30% of NELL-1-negative patients, while it occurred in 70%–80% of patients with MN, reflecting ethnic variations and the result of our early-biopsy policy that captures sub-nephrotic disease. Previous studies have indicated that compared with patients with PLA2R-positive MN, those with PLA2R-negative MN are more likely to achieve spontaneous remission. Specifically, within one study, the proportion of patients with PLA2R-negative MN who experienced spontaneous remission within the initial 6 months of follow-up was 18.2%, significantly higher than that observed in the PLA2R-positive cohort (3.4%). A meta-analysis revealed that the likelihood of remission in patients with PLA2R-negative MN is 1.31 times higher compared with that in patients with PLA2R-positive. 25 However, despite the relatively better prognosis for PLA2R-negative patients, 35%–40% of patients treated conservatively may still progress to renal failure within 10 years. 26 In clinical practice, doctors usually first recommend conservative treatment for these patients, focusing on symptomatic relief and observation rather than immediate aggressive immunosuppressive therapy.
This study identified NELL1 staining positivity in kidney tissue samples as a vital risk factor for ESRD progression in patients with PLA2R-negative MN (HR = 3.2, 95% CI: 2.3–11.6, p = 0.026). The early aggressive intervention is necessary in NELL1-positive MN and warrants the attention of nephrologists. Serum NELL-1 antibody levels have been reported to decrease over time in patients with MN treated with rituximab, which indicated that NELL-1 may be similar to PLA2R and can be used as a serological marker. 27 Future studies will need to test the optimal strategy and the potential role of anti-NELL-1 antibody in the diagnosis and prediction of kidney disease progression in patients with MN. The identification of target antigens in MN is of great significance for patient prognosis and is helpful in the formulation of effective and individualized treatment plans. In sum, these findings emphasize the significance of early detection and precise targeting of the underlying etiology in the management of NELL-1 MN. Notably, none of the NELL1-positive biopsies exhibited concomitant EXT1/2 and THSD7A positivity (0/11). Given the limited sample size of this study, larger multicenter cohorts are warranted to establish the true prevalence and prognostic significance of EXT1/2 and NELL1 co-expression.
We observed that NELL-1-associated MN was associated with SLE (63.6%), findings that align with previous research. 24 Of 11 cases of NELL1-associated MN, 7 had concurrent LMN. A Mayo Clinical cohort study of 374 patients with LMN found that approximately 32.6% of patients with LMN had EXT1/2-positive LMN and showed better prognosis than patients with EXT1/2-negative LMN.28,29 Identification of target antigens in LMN is of great significance for patient prognosis and helps in formulating effective treatment plans. Our study performed stratified analysis based on the presence of SLE. In the SLE = 1 subgroup (n = 56), patients with NELL1-positive MN had a higher ESRD risk compared with NELL1-negative patients (HR = 5.1, p = 0.041, Table 3), suggesting that NELL1 is a more relevant ESRD biomarker in patients with SLE history, likely due to interactions between SLE-related immunity and NELL1 renal pathophysiology. Compared with previous reports, NELL1-associated MN in patients without autoimmune comorbidities or LMN demonstrates favorable renal outcomes, underscoring the necessity of stratifying prognosis according to the underlying etiology.
In addition, compared with the NELL-1 MN study cohort from Beijing Anzhen Hospital, 22 both Chinese cohorts showed a higher prevalence among young women. In our series, an association with the use of fairness creams was found in 9 of 11 patients with NELL1-associated MN. The cohort that used fairness creams comprised younger participants, with a female predominance and had clinical manifestations of NS, which may be related to the interaction of Hg with NELL-1 protein. Chronic Hg poisoning, resulting from the long-term use of fairness creams, is well-documented in the literature as a cause of kidney disease. 29 It often presents with the clinical features of NS. MN and minimal change disease are the two most common pathological types associated with Hg-induced nephropathy. Recent studies have indicated a possible connection between the use of skin fairness creams and NELL1-associated MN. 30 In an Indian study of patients with NELL1 MN, 13 of 15 patients reported using such creams before their symptoms began. 30 In addition, molecular docking indicated a favorable binding affinity between Hg and NELL-1 protein. This suggests that Hg binds to the NELL1 antigen on the GBM, leading to immune complex deposition and the induction of an autoimmune response. Inorganic Hg compounds have been used in skin-lightening products since ancient times. 31 No patient exhibited all of the following three factors: SLE, long-term use of skin fairness cream, and thyroid carcinoma; 6 patients exhibited both SLE and cream use, 1 had SLE alone, and 1 reported the use of cream and had thyroid carcinoma. Given the limited sample size, causal inferences cannot be drawn, and larger multicenter studies are required to clarify the relative contributions of autoimmune, environmental, and neoplastic factors.
Hg that is added to whitening cosmetics can inhibit the activity of enzymes in the epidermis, preventing the formation of melanin, thereby providing a whitening effect. Some unscrupulous manufacturers, to highlight the whitening effect in the short term, add excessive Hg to facial creams or masks. 32 This mercury is absorbed through the skin and enters the bloodstream. The kidney is the main organ responsible for the excretion and accumulation of Hg, and long-term or short-term intake of large amounts of Hg can lead to nephrotoxicity and kidney damage. In a study that analyzed data from the National Health and Nutrition Examination Survey, 33 it was found that compared with other ethnicities, Asians have a geometric mean of total Hg (B-tHg) in their blood that is approximately 3–4 times higher. Additionally, non–United States (US)-born Asians have about 1.5 times more Hg accumulation in their kidneys compared with US-born Asians. Interestingly, the B-tHg levels in US-born Asians decrease with age, while those in non–US-born Asians show an increasing trend. These differences may be attributed to varying dietary habits, living environments, or genetic backgrounds. The docking simulation predicted a high-affinity binding site for Hg2+ on NELL1; however, direct tissue Hg quantification was not performed. Consequently, we cannot confirm whether this interaction is present in all 11 NELL1-positive cases.
This finding carries substantial weight for clinical practice. Although induction regimens for class V lupus nephritis (LN-V) differ from those for idiopathic MN, our data demonstrate that NELL1 positivity identifies a high-risk subset within PLA2R-negative MN irrespective of the SLE status. Prospective trials comparing rituximab and standard LN-V protocols specifically in NELL1-positive, proteinuric patients are warranted to translate these findings into evidence-based practice. In addition, it helps mitigate the effect of environmental risk factor. The suspected connection between Hg in skin-whitening creams and NELL-1–associated MN necessitates better patient education to alert them about harmful substances and encourage their avoidance. Nonetheless, given the small cohort of patients with NELL1-associated MN, additional research is imperative to validate our observations and expand upon these preliminary insights.
Limitations
Owing to the retrospective design, our study has certain limitations. First, the serum samples were not systematically archived; hence, we were unable to quantify circulating anti-NELL1 antibodies and could not correlate antibody titers with clinical activity or outcomes. Second, paired blood and 24-hour urinary Hg measurements were unavailable, precluding direct assessment of Hg exposure intensity. Third, although experimental data indicate that Hg drives a Th2-biased autoimmune response via up-regulation of interleukin-4 (IL-4), 34 we lacked IL-4 measurements; consequently, the mechanistic link between Hg exposure and NELL1-associated MN could not be formally tested. Moreover, causal relationships between specific regimens and ESRD cannot be inferred. Prospective cohorts with protocolized biospecimen collection are warranted to address these gaps and delineate the exposure–immune–pathology axis more precisely. Larger multicenter and longer prospective studies are needed to clarify the link between NELL-1 and LMN and confirm the long-term prognosis of NELL-1-associated MN.
Conclusions
In sum, our real-world data indicate that approximately 12.7% of patients with PLA2R-negative MN have NELL-1-associated MN. These patients are predominantly young women with clinical manifestations of NS and a history of skin fairness cream use. High NELL1 expression appears to be independently associated with an increased risk of ESRD, particularly in those with concomitant SLE, and may aid early risk stratification. Owing to the retrospective design, these findings need prospective validation.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605251379806 - Supplemental material for Neural epidermal growth factor-like 1 expression predicts end-stage renal disease in phospholipase A2 receptor–negative membranous nephropathy: A retrospective cohort stratified by systemic lupus erythematosus
Supplemental material, sj-pdf-1-imr-10.1177_03000605251379806 for Neural epidermal growth factor-like 1 expression predicts end-stage renal disease in phospholipase A2 receptor–negative membranous nephropathy: A retrospective cohort stratified by systemic lupus erythematosus by Qingqing Rao, Yuan Cheng, Shuling Yue, Haiying Song, Wangyang Li, Dongli Qi and Qijun Wan in Journal of International Medical Research
Footnotes
Acknowledgments
The authors are grateful to the Guangzhou KingMed Diagnostic Laboratory Group that assisted in staining of renal tissue target antigens.
Author contributions
Qingqing Rao, Yuan Cheng, and Shuling Yue conceived and planned the research, performed the experiments, analyzed the data, wrote the initial draft, and revised the manuscript. Haiying Song planned the study. Wangyang Li and Dongli Qi collected the data. Qijun Wan planned the study and reviewed the manuscript. All authors approved the final version of the manuscript.
Data availability statement
The datasets presented in this article are not readily available because the data collected for this study were derived from clinical records, which are not available for sharing. Requests to access the datasets should be directed to Qijun Wan at
Declaration of conflicting interest
The authors declare that there are no conflicts of interest.
Ethics statement
The retrospective nature of this study did not require Institutional Review Board approval. Our study was approved by the Ethics Committee of Shenzhen Second People’s Hospital (ethics approval number 20210620213357018-FS01).
Funding
The authors disclose receipt of the following financial support for the research, authorship, and publication of this article. This work was supported by the Sanming Project of Medicine in Shenzhen (Project No.: SZSM202211013), the Shenzhen High-level Hospital Construction Fund, the Shenzhen Key Medical Discipline Construction Fund (Project No.: SZXK009), the National Natural Science Foundation of China (Project No.: 81900639), the Research Program of the Shenzhen Science and Technology (Project No.: JCYJ20190806162807125), the Cohort Study of Podocyte Antigen Expression and Renal Function Progression in Chinese Patients with Membranous Nephropathy (Project No.: 20213357018), and the Shenzhen Second People’s Hospital Clinical Research Fund of Guangdong Province High-level Hospital Construction Project (Grant No.: 20213357012).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
