Abstract
In the current era of early detection of chronic kidney disease and efficient therapeutic options for management of its complications, skeletal manifestations of renal hyperparathyroidism are increasingly rare. A 31-year-old female patient presented for evaluation of severe pain in the left forearm, right hand, right knee, right hip, and lower back following a fall sustained 3 days prior to presentation. She had a history of end-stage renal disease and received maintenance hemodialysis. Review of the medical records revealed that she had poor compliance with her diet, medications, and dialysis treatments. Laboratory values were significant for marked elevation in serum parathyroid hormone level (1735 pg/mL), as well as hyperphosphatemia and normal serum calcium levels. Interestingly, X-ray images showed generalized severe demineralization of the extremities with the pelvic computed tomography scan revealing presence of diffuse brown tumors. In addition, she had insufficiency fractures of the extremities that left her incapacitated and had to be managed conservatively due to her poor functional status and ongoing severe hyperparathyroidism. Unfortunately, the patient refused surgical removal of the parathyroid glands and was hence treated with a high-dose phosphate binder and a calcimimetic agent together with reinforcement of compliance. This case highlights the importance of metabolic assessment of patients presenting with unexpected bone complications and can be used to raise awareness of the physicians on the extreme cases of mineral bone complications secondary to renal disease that are observed rarely.
Introduction
In patients with chronic kidney disease (CKD), parathyroid hormone (PTH) levels progressively increase as an adaptive response to hyperphosphatemia. Observational studies demonstrate an association between PTH levels at extremes (less than two times and greater than nine times the normal assay limits) and mortality in these patients. 1 With early detection and treatment of CKD and its complications, it is very unusual to see skeletal manifestations of secondary hyperparathyroidism. 2 This is particularly notable in the current era of potent phosphate binders, calcimimetic agents (i.e. equivalent of medical parathyroidectomy), and use of high-flux dialyzers that provide excellent clearance of uremic toxins such as phosphate. Herein, we present a case of secondary hyperparathyroidism that presented with brittle bones and characteristic radiographic changes.
Case presentation
A 31-year-old woman presented for evaluation of pain in the left forearm, right hand, right knee, right hip, and lower back following a fall sustained 3 days prior to presentation. She had a past medical history notable for end-stage renal disease (ESRD) for which she has been receiving maintenance hemodialysis for the past 15 years, hypertension, diabetes, and congestive heart failure. She was wheelchair bound due to disabling peripheral neuropathy and chronic leg pain. She reported that she had fractured her left wrist about 3 weeks prior to presentation. She also experienced femoral fracture about a year ago, that was managed conservatively.
Physical examination revealed a blood pressure of 153/81 mm Hg and pulse rate of 101 bpm. There was moderate swelling and tenderness over the right distal femur with limited range of motion of the right knee due to pain. Left forearm splint due to previous fracture was in place. Tenderness was also noted over tip of the right index finger. There was no distal neurovascular deficit in the extremities. Medications included aspirin of 81 mg per day, lisinopril of 40 mg per day, carvedilol of 25 mg twice a day, sevelamer carbonate of 800 mg thrice a day with meals, and insulin regimen. The patient reported non-compliance with medications, diet, and hemodialysis treatment.
Initial laboratory values were significant for hemoglobin of 8.5 g/dL (12–15 g/dL), white blood cell count of 6.2 thou/mm3 (4–10 thou/mm3), platelet count of 222 thou/mm3 (150–450 thou/mm3), blood urea nitrogen of 36 mg/dL (6–20 mg/dL), and creatinine of 5.72 mg/dL (0.4–0.9 mg/dL). Serum intact PTH was found to be significantly elevated at 1735 pg/mL (15–65 pg/mL (goal 150–300 pg/mL for ESRD)) with serum calcium level of 9.5 mg/dL (8.4–10.2 mg/dL) and phosphate of 5.7 mg/dL (2.7–4.5 mg/dL). Serum 25-hydroxy vitamin D level was 24 ng/mL (>20 ng/mL).
X-ray of the right lower extremity showed diffusely decreased osseous mineralization and a comminuted distal femoral fracture (Figure 1). Similarly, X-ray of the left upper extremity revealed significant diffuse osteopenia, and X-ray of the right hand showed findings concerning for possible fracture of the distal phalanx of the index finger just proximal to the subungual tuft versus aggressive erosion at this site was reported for the right hand (Figure 2). Computed tomography (CT) scan of the abdomen and pelvis showed remarkable diffuse severe demineralization of the bones, with florid changes of renal osteodystrophy, prominent subligamentous resorption in the sacroiliac joints bilaterally, and widespread brown tumors (Figure 3).
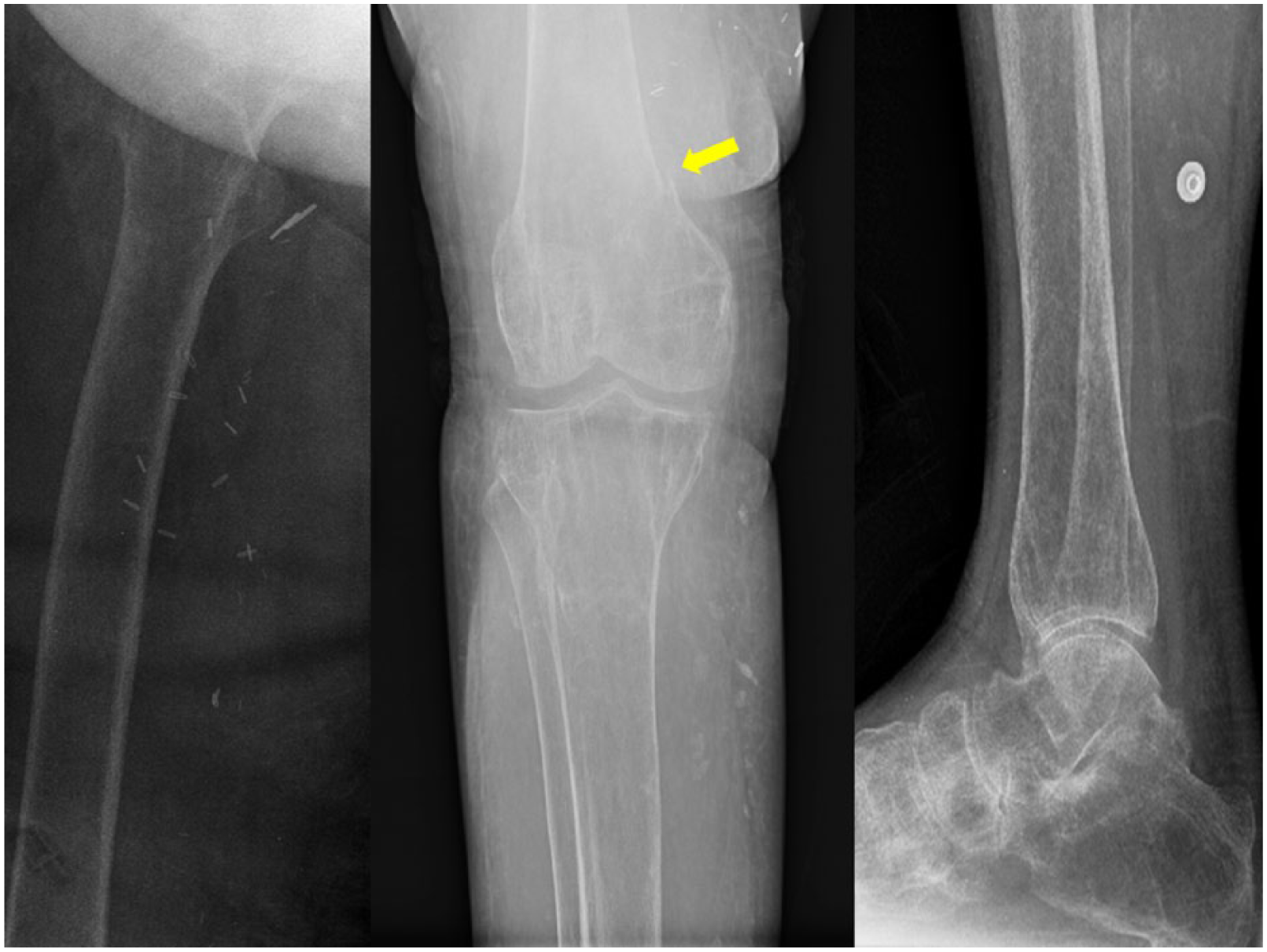
X-ray of the right lower extremity. Diffusely decreased osseous mineralization. There is a comminuted distal femoral fracture (arrow).

X-ray of the (a) left shoulder, (b) elbow, and (c) right hand. Marked diffuse demineralization with findings concerning for possible fracture of the distal phalanx of the index finger just proximal to the subungual tuft versus aggressive erosion at this site (arrow).

CT scan of the pelvis showing diffuse severe demineralization of the bones with florid changes of secondary hyperparathyroidism from renal osteodystrophy. Prominent subligamentous resorption in the sacroiliac joints bilaterally and widened joint space (arrow) and widespread brown tumors (chevrons).
With normal serum calcium, high serum phosphate together with severely high PTH level, and history of non-compliance with medications and the dialysis treatment, the diagnosis of secondary (renal) hyperparathyroidism was made. Chart review revealed a history of long-standing severe hyperphosphatemia in the past explaining the presence of severe hyperparathyroidism despite current mild hyperphosphatemia (possibly now evolving toward more autonomous PTH secretion).
Typical imaging stigmata of hyperparathyroidism including diffuse demineralization, insufficiency fractures, and brown tumors supported the diagnosis in this young woman.
Her fractures were managed with non-operative treatment given her poor baseline functional status and severe hyperparathyroidism. The patient refused surgical parathyroidectomy. A high-dose phosphate binder (i.e. sevelamer carbonate) and also an oral calcimimetic (i.e. cinacalcet) were started for management of hyperphosphatemia and hyperparathyroidism. We reinforced compliance with low phosphate diet, medications, and hemodialysis treatment.
Discussion
In advanced kidney disease, the phosphorus clearance decreases significantly as the glomerular filtration rate (GFR) drops progressively. Even in patients with an estimated GFR of greater than 30 mL/min, subclinical and postprandial hyperphosphatemia has been observed, which is thought to be the major cause of secondary hyperparathyroidism. PTH stimulates the osteoclasts and leads to bone resorption, which, in turn, increases the serum concentration of calcium and phosphorus. Moreover, PTH indirectly increases intestinal calcium and phosphorus absorption by stimulating 1,25-dihydroxy vitamin D production. 3 Calcium has a negative feedback effect on the parathyroid glands through the calcium-sensing receptor; 4 activation of this receptor is the primary mechanism of action of cinacalcet.
The first step in the management of secondary hyperparathyroidism due to kidney disease is to optimize the levels of serum phosphate and calcium. Hyperphosphatemia can be addressed by dietary restriction and addition of phosphate binders if needed. Calcitriol supplementation is effective in suppressing high PTH levels, but caution needs to be exercised as it can increase the absorption of both calcium and phosphate from the gut and may increase calcium phosphate product. Vitamin D analogs such as paricalcitol are other alternatives. Current guidelines for management of CKD complications recommend checking serum 25-hydroxyvitamin D and replacing if deficient. However, replacement of nutritional vitamin D in the ESRD population with no residual renal function is controversial, although the data show that 25-hydroxyvitamin D levels below 15 ng/mL are associated with a greater severity of secondary hyperparathyroidism even in ESRD patients receiving dialysis. 5 In patients with persistently elevated PTH levels in spite of optimizing serum phosphate and calcium levels, addition of calcimimetic drug cinacalcet should be considered. It should not be started if serum calcium is lower than 8.4 mg/dL as it can cause severe hypocalcemia. In our patient with serum PTH levels of >1000 pg/mL, the likelihood of improvement is low with hyperphosphatemia control alone, and so we chose to add cinacalcet. In patients who have markedly elevated PTH levels refractory to medical management especially with related signs and symptoms (such as bone pain, weakness, and pruritis), surgical parathyroidectomy should be considered if they are acceptable surgical candidates. 6 Parathyroidectomy has been suggested to reverse the bone resorption and result in complete regression of brown tumors. 7 In this context, it is of note that cinacalcet has failed to improve all-cause or cardiovascular mortality, but could reduce the need for parathyroidectomy in patients with ESRD. 8
Conclusion
Patients with CKD, especially those on dialysis, are at high risk for secondary hyperparathyroidism and consequent bone fractures. The accumulation of phosphate, reduction in active vitamin D production, and the tendency to hypocalcemia act as stimuli for the increased secretion of PTH. These laboratory parameters need to be periodically monitored and optimized early in the course of CKD. Appropriate patient education and adherence to therapy play a vital role in the management of this bone-eating disease, which can otherwise contribute to significant morbidity.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Our institution does not require ethical approval for reporting individual cases or case series.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
