Abstract
Objective
Renal impairment is a significant complication of systemic lupus erythematosus (SLE). Additionally, infection in patients with end-stage renal disease (ESRD) attributable to SLE is common, and it increases the risk of mortality. This study explored the infection profile and risk factors for mortality in patients with ESRD attributable to SLE.
Methods
In this retrospective, observational study of 125 hospitalized patients, demographic, clinical, laboratory, treatment, and prognosis data were retrieved and analyzed.
Results
The 125 cases included 98 pulmonary infections (78.4%), 14 urinary infections (11.2%), and 13 intestinal infections (10.4%). Twenty-six patients died within 1 month after enrollment. Univariate Cox regression and Kaplan–Meier analyses revealed several possible indicators potentially influencing patient survival. Furthermore, multivariate Cox regression analysis identified a higher SLE Disease Activity Index-2000 score, recent higher-dose glucocorticoid use, hypertension, and catheter indwelling as risk factors for higher mortality.
Conclusions
Infections were common in patients with advanced SLE and ESRD, and several risk factors might increase the risk of mortality. Once infection is identified, empiric antibiotics should be initiated immediately, and subsequent antibiotics should be applied per the results of drug sensitivity testing to clear the infection.
Keywords
Introduction
Systemic lupus erythematosus (SLE) is a chronic multisystem autoimmune disease. Renal involvement such as lupus nephritis (LN) is a significant complication of SLE.1,2 It is estimated that up to 40% to 70% of patients with SLE might develop renal disease during the course of their illness, with manifestations varying from mild proteinuria to acute or chronic declining renal function.3–6 The definition and classification of chronic kidney disease (CKD) were proposed by the National Kidney Foundation Kidney Disease Outcomes Quality Initiative. According to this definition, end-stage renal disease (ESRD) refers to a chronic renal disease with estimated glomerular filtration rate (eGFR) <15 mL/min/1.73 m2, the need for maintenance hemodialysis/peritoneal dialysis, or renal transplantation.7,8
ESRD can increase the risk of hospitalization and mortality in patients with SLE. Moreover, patients with SLE and CKD are approximately 2-fold more likely to experience cardiovascular and cerebrovascular events, septic shock, or death. 9 Once infection occurs in patients with SLE and ESRD, the treatment is complicated, and the prognosis may be much worse, which can bring significant challenges to clinicians.10,11 In recent years, increasing attention has been paid to the infectious complications of patients with SLE. At present, the infection burden is high in patients with SLE or ESRD, but there is little information on the infection burden in patients with coincident ESRD and SLE. This study analyzed the pathogenic bacteria and drug sensitivity of patients with coincident SLE, ESRD, and infection. We also explored the clinical characteristics and risk factors affecting their prognosis to provide a reasonable referral for clinical prevention and anti-infection treatment strategies in this scenario.
Materials and methods
Study design and setting
This was a two-center retrospective study of patients with biopsy-proven LN diagnosed between January 2001 and December 2020. This study was conducted according to STROBE guidelines. 12 Hospitalized patients with ESRD from two neighboring medical centers sharing the same patient sources, namely Jiao Tong University Affiliated Sixth People’s Hospital (Shanghai, China) and Shanghai Punan Hospital of Pudong New District (Shanghai, China), were enrolled. All patients received kidney biopsy before enrollment and fulfilled the 1997 ACR classification criteria for SLE.1,3 The patients were divided into two subgroups per their final clinical infection outcomes (survivors/non-survivors) within 1 month after enrollment. LN was described histologically using World Health Organization criteria before 2006 and subsequently per International Society of Nephrology and Renal Pathology Society criteria. 13 SLE disease activity was assessed after hospitalization, calculated using the SLE Disease Activity Index-2000 (SLEDAI-2K) as described elsewhere. 14 eGFR was calculated using the Chronic Kidney Disease Epidemiology Collaboration formula.7,8 Because of the study’s retrospective nature, signed informed consent for the review of electronic medical records was required in the inpatient registration stage before enrollment. The Ethics Committees of both centers approved the study (Nos. 2020-37 & SPNH-2020.6; approval date: June 26, 2020). The exclusion criteria were as follows: malignancy requiring urgent or long-term therapy; receipt of any organ transplant (including kidney transplantation); and incomplete co-morbidity, renal function, or pharmacotherapy data. We declared that all procedures performed in the presents study followed the ethical standards of the institutional and/or national research committee and complied with the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards.
Data collection
Demographic, clinical, laboratory, and biopsy data and treatment strategies (including SLE-related treatment, ESRD-related treatment, and antibiotic therapy) were retrieved from the patients’ electronic medical records. The clinical course of SLE, recent therapy within 3 months before enrollment (including glucocorticoids, combined cyclophosphamide, cyclosporine, hydroxychloroquine, other immunosuppressants, dosage, and treatment regimens) and other clinical data of the enrolled patients were recorded. The cumulative dose of all glucocorticoids in different dosage forms and usages were calculated after they were mathematically converted to prednisone 3 months before enrollment, and the final unit used was milligrams. Meanwhile, creatinine, hypersensitive C-reactive protein (CRP), procalcitonin, hemoglobin, serum albumin, and other biochemical indices were recorded.
Diagnosis of infection
The combination of clinical symptoms, physical signs, and laboratory and microbiological data of the patients was used to make a comprehensive judgment on whether patients had infection. Mycobacterium tuberculosis infection was diagnosed or dismissed per the purified protein derivative skin test, T-SPOT test, and response to anti-tuberculosis treatment. In patients suspicious for infection who had fever, cough, frequent or urgent urination, painful urination, diarrhea, and other symptoms, if the fungus was found in blood culture or living tissue, deep fungal infection was diagnosed after contamination or colonization was appropriately excluded. All patients with interstitial pneumonia or eccentric cavity indicated by chest X-ray or computed tomography were highly suspicious for pulmonary fungal infection and monitored closely. Positive culture of specimens from aseptic body fluids such as blood, cerebrospinal fluid, ascites, pleural effusion, joint cavity effusion, or biopsy specimens with a positive culture and tissue invasion evidence was used as the gold standard for the diagnosis of invasive candidiasis. For non-sterile specimens, positive culture results multiple times for the same site several times or isolation of the same species of Candida simultaneously necessitated a diagnosis of possible invasive candidiasis. All patients suspicious for invasive candidiasis were advised to undergo blood fungal culture.
Pathogenic bacterium culture and drug sensitivity testing
Samples collecting, drug sensitivity testing, and treatment protocols were performed as per the standard of care in the hospitals. The dose of anti-infection medicine was adjusted per the drugs’ metabolic characteristics and patients’ renal function. Hemodialysis, peritoneal dialysis, or continuous renal replacement therapy was performed per the patients’ condition. For patients on hemodialysis, anti-infection drugs were used after dialysis. The duration, location, prognosis, and outcome of infection were extensively analyzed.
Infection-specific mortality
The causes of death were subjected to an in-depth discussion among all research team members. The outcome and timing of 1-month infection-specific mortality after enrollment in this study were analyzed after dismissing other possible causes of death such as acute massive cerebral infarction, acute cerebral hemorrhage, acute myocardial infarction, and malignant arrhythmia.
Statistical analysis
Unless otherwise stated, the continuous variables in our study were reported as the mean ± standard deviation for normal distribution, and the categorical data were reported as percentages. Student’s t-test and the Mann–Whitney U test were applied for normally distributed and non-normally distributed variables, respectively. The chi-squared test was applied to analyze categorical variables. Survival curves were plotted according to the Kaplan–Meier method and compared using the log-rank test. The univariate Cox proportional hazards model and multivariate Cox proportional hazards regression model (Forward: LR method) were used to identify significant prognostic factors. Statistical significance was set at P < 0.05 (two-sided). All data were analyzed using IBM SPSS statistical software for Windows, version 26.0 (IBM, Armonk, NY, USA), and Prism software, version 9.0 (GraphPad, San Diego, CA, USA).
Results
Demographics and disease characteristics
In total, 125 patients were consecutively enrolled in this study. Two subgroups of patients from two centers were subjected to homogeneity testing for basic demographics and disease characteristics. The patients with homogeneity were divided into two distinct groups per their final clinical infection outcomes within 1 month after enrollment, namely survivors (n = 99) and non-survivors (n = 26). The demographic data, clinical characteristics, and immunomodulatory medications among the enrolled patients were summarized (Table 1). Regarding the clinical data, the age and sex distribution; rates of pneumococcal and seasonal influenza vaccination; duration of ESRD; CRP level; receipt of methotrexate, leflunomide, mycophenolate mofetil, cyclosporine, and azathioprine; and rate of co-morbid diabetes did not differ between the groups. Hemoglobin, albumin, globulin, and serum creatinine levels; SLEDAI-2K scores; the proportions of patients with glycosylated hemoglobin >7%, procalcitonin >0.5 µg/L, and co-morbid hypertension; and the doses of medication within 3 months after enrollment significantly differed between the groups (all P < 0.05).
Demographic data, clinical characteristics, and immunomodulatory medications among the enrolled patients.

ESRD, end-stage renal disease; Class III, focal lupus nephritis; Class IV, diffuse lupus nephritis; Class V, membranous lupus nephritis; Class VI, advanced sclerotic lupus nephritis; CRP, C-reactive protein; SLEDAI-2K, Systemic Lupus Erythematosus Disease Activity Index 2000.
Data are presented as the number of cases and percentage or mean ± standard deviation.
Composition and proportion of pathogenic microorganisms in patients with ESRD and infection
Among 125 patients, 98, 14, and 13 had pulmonary infection (78.4%), urinary infection (11.2%), and intestinal infection (10.4%), respectively. Of these patients, 119 cultures were performed, and 58 patients (48.7%) were positive for pathogenic microorganisms. Among them, 29 patients were infected by gram-positive bacteria, 18 patients were infected by gram-negative bacteria, and 11 patients had fungal infections (five, four, one, and one case of Candida albicans, Aspergillus, Candida tropicalis, and Candida parapsilosis infection, respectively; Table 2).
Results of detected pathogenic microorganisms in infected patients with end-stage renal disease.
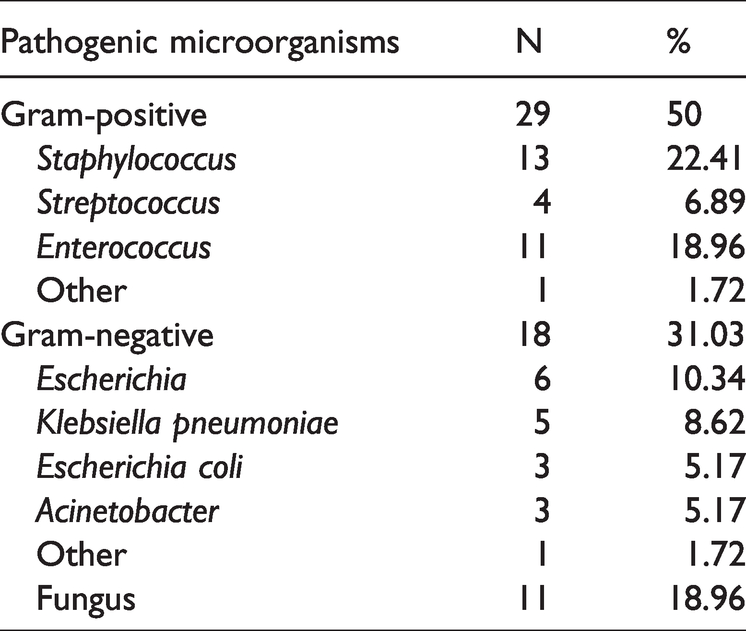
Drug sensitivity analysis for gram-stained bacteria
All gram-negative bacteria in our cultured samples were sensitive to meropenem and imipenem. The bacteria also displayed varying degrees of sensitivity to other drugs including piperacillin/sulbactam, cefoperazone/sulbactam, ampicillin/sulbactam, levofloxacin, cefepime, gentamicin, aztreonam, amikacin, minocycline, and ceftazidime. However, many of the isolated gram-negative bacteria were resistant to ceftazidime, cefotaxime, ciprofloxacin, cefuroxime, and sulfamethoxazole. These results were essential to the empirical anti-infection treatment strategy in a similar clinical scenario. In our study, most gram-positive bacteria, such as Staphylococcus, Streptococcus, and Enterococcus, were most sensitive to linezolid, vancomycin, and teicoplanin, followed by rifampicin, ampicillin/sulbactam, cefazolin, levofloxacin, and gentamicin. The rates of sensitivity to clindamycin, oxacillin, and penicillin were 20.69%, 17.24%, and 13.79%, respectively (Supplementary Table 1).
Cox regression analysis for possible risk factors associated with lethal infections
In 125 hospitalized patients with SLE and infection, the antibiotic strategies were adjusted per the results of drug sensitivity testing. After meticulous care, 99 patients experienced clinical improvement, and they were discharged within 1 month. Conversely, 26 patients died within 1 month of severe infection. Among the 26 patients who succumbed to infection, 8, 7, and 11 were infected by fungi, gram-positive bacteria, and gram-negative bacteria, respectively. The differences in clinical characteristics between the surviving and non-surviving patients were analyzed. Kaplan–Meier curves for overall survival according to specific indicators and univariate Cox regression analysis were used first for variable selection to determine the risk factors associated with death. The Kaplan–Meier curves illustrated that higher pre-dialysis serum creatinine levels (>530 µmol/L), anemia (hemoglobin <90 g/L), hypoproteinemia (albumin <30 g/L), catheter indwelling state, higher glucocorticoid dosage in the last 3 months (average dose >15 mg/day), higher SLEDAI scores (SLEDAI-2K ≥15), hypertension (>140/90 mmHg), and diabetes were associated with higher mortality (P < 0.1, Figure 1). Furthermore, multivariate Cox regression adjusted for age, sex, and other confounders was conducted to detect the independent risk factors associated with lethal infection in patients with ESRD attributable to SLE. The result illustrated that only a higher SLEDAI score (SLEDAI-2K ≥15), a higher glucocorticoid dosage in the last 3 months (equivalent prednisone dose >15 mg/day), hypertension (>140/90 mmHg), and a catheter indwelling state were independent risk factors associated with mortality attributable to infection in this cohort (Table 3).

Kaplan–Meier curves for overall survival according to specific indicators.
Univariate and multivariate Cox proportional hazards regression model analysis of overall survival.
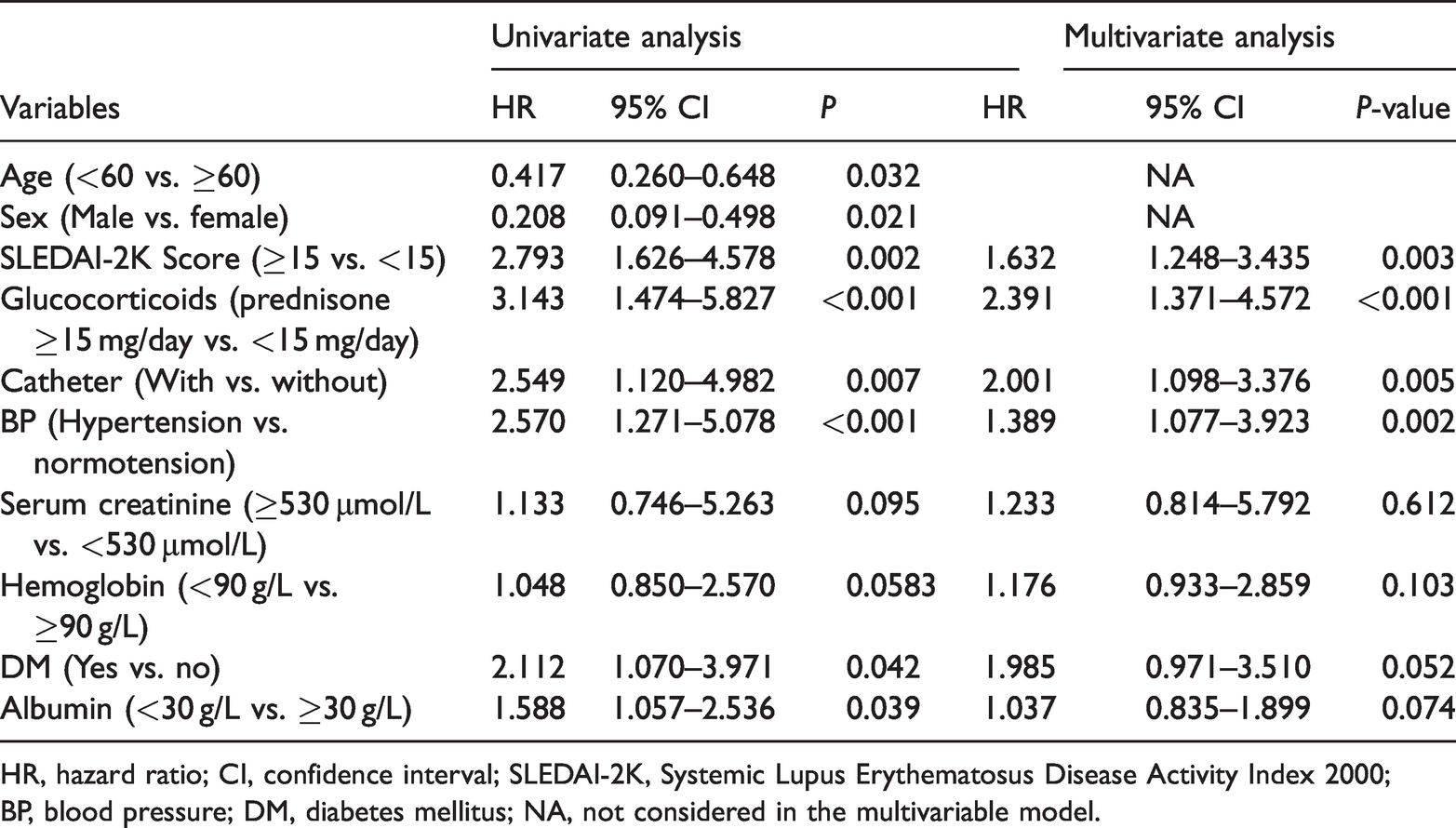
HR, hazard ratio; CI, confidence interval; SLEDAI-2K, Systemic Lupus Erythematosus Disease Activity Index 2000; BP, blood pressure; DM, diabetes mellitus; NA, not considered in the multivariable model.
Discussion
SLE is essentially a disease that can affect multiple systems and present various symptoms simultaneously. Lupus nephritis can manifest as hematuria, albuminuria, or acute or chronic kidney injury.15–18 In recent years, the prognosis of LN has been substantially improved, and the risk of mortality has been greatly reduced by induction remission and maintenance therapy. 19 At present, the application of glucocorticoids and immunosuppressants is generally accepted in the current guidelines globally. Conversely, the subsequent complications, especially infection, have gradually attracted attention. The incidence of infection in patients with LN, especially in those with coincident ESRD, is exceptionally high, estimated at 36% to 45%.20–22 Moreover, infection is one of the most important causes of death in patients with LN after treatment with different immunosuppressants, including glucocorticoids, cyclophosphamide, hydroxychloroquine, rituximab, or other biological agents. 23 Many cases of SLE are complicated by various infections attributable to the primary disease or compromised immune function caused by drug treatment.24,25 Differentiating infections from high disease activity is crucial in patients with autoimmune diseases such as SLE.26,27
Our study demonstrated that pulmonary infection was most common, followed by urinary and intestinal infection, which was consistent with the results of other research. 21 Multiple causes of pulmonary infection can be postulated. First, patients had exudative inflammatory lesions caused by small pulmonary vessels. As the natural lung barrier was damaged, it served as a good medium for pathogenic bacteria to thrive. Second, patients with SLE sometimes develop proteinuria and malnutrition, leading to rapid water and sodium retention, heart failure, and rapid pulmonary edema. The application of glucocorticoids and immunosuppressants reduced patients’ immunity and increased the incidence of infection. Ye et al. demonstrated that the primary pathogens of patients with SLE were Streptococcus pneumonia, Haemophilus influenzae, Pseudomonas aeruginosa, and Staphylococcus aureus. 28 Lin et al. claimed that the most common infection pathogens in their study were bacteria and fungi. The main infecting bacteria were Staphylococcus aureus, Pseudomonas aeruginosa, and Escherichia coli. 22 In this study, we found that the most common pathogens in infected patients with SLE and ESRD were gram-positive bacteria. This was not consistent with previous findings, possibly because our enrolled patients had lower rates of immunosuppressant use and higher rates of dialysis and central venous catheter indwelling. As the rate of fungal infection was relatively high and the outcomes were unfavorable, we emphasized that fungal infection should not be ignored in patients with advanced LN who are suspicious for combined infection. We should be vigilant for those covert infections.
Although drug sensitivity analysis results are important for treating patients with infection, culturing pathogenic bacteria often takes a long time, and the positive rate is not high for many reasons. Thus, most patients need to be given regular and effective empirical antibiotic treatment in the early stage of infection. The antibiotics considered in anti-infection treatment should cover gram-positive bacteria, especially Staphylococcus aureus, and gram-negative bacteria, including Klebsiella and Enterobacter. Therefore, when selecting empiric antibiotics, once the infecting pathogen is identified, it is recommended to combine potentially effective antibiotics to strengthen the anti-infection effect for early control. This strategy should have a role in prognosis improvement. Patients with severe SLE often have fungal infection, and the most frequently observed pathogen was invasive Aspergillus pneumoniae, especially in patients with recent high-dose glucocorticoid and immunosuppressant therapy.
SLE can increase the risk of infection for many reasons. 29 In our study, many patients did not receive a high dose of glucocorticoid or immunosuppressive therapy because of their reduced renal function or low SLEDAI, and thus, their prognosis was slightly better than that in patients who received high-dose glucocorticoids. Therefore, balancing the application course and dosage of glucocorticoids and immunosuppressants in patients with SLE remains a major challenge to solve. Our study also detected several risk factors for higher mortality in this cohort. Notably, we found that the control of hypertension in patients on dialysis was associated with improved survival.
As a two-center retrospective study, our study had limitations including possible selection bias. It would be more persuasive if a larger-sample, multicenter clinical study was conducted. Meanwhile, as a retrospective, observational study, it can be challenging to judge whether each patient’s infection was community- or hospital-acquired. The strength of our study lies in that most of the enrolled patients had chronic renal failure stage or required dialysis, differentiating them from their counterparts with normal renal function. Such patients are more likely transferred to nephrology or intensive care units. Their characteristics and distinctive medical surroundings differ from what the rheumatologists might encounter.20,30–32 At present, there are no adequate studies on the profile of pathogenic bacteria and drug sensitivity of infected patients with ESRD or dialysis dependence because of LN.33–35
Our study summarized and analyzed the pathogenic microbiology, clinical characteristics, and prognostic factors of infected patients with severe SLE and ESRD, thereby providing a reasonable referral for the prevention and treatment of such patients. We conclude that patients with SLE and ESRD are prone to infection, most commonly pulmonary infection, and most of the pathogens are bacteria. However, fungal and opportunistic infection can occur. Thus, it is necessary to be cautious of various pathogenic microbial infections, which have higher mortality and which can lead to poor prognoses. When necessary, prophylactic therapy should be initiated.
Conclusions
LN is one of the critical manifestations of SLE. Some cases of LN progress to ESRD, and infection is common in such patients. This study retrospectively analyzed the clinical data, pathogenic bacteria, and drug sensitivity of 125 infected patients with SLE and ESRD. Meanwhile, the risk factors for mortality in these patients were analyzed. We recommend that once infection is identified, empiric antibiotics should be immediately initiated, and subsequent antibiotics should be selected according to the results of drug sensitivity analysis. Meanwhile, clinical workers need to reevaluate or adjust the patients’ ongoing immunoregulatory strategy to ensure that the infection is cleared.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605221118702 - Supplemental material for Infection profile and risk factors for mortality in patients with end-stage renal disease attributable to systemic lupus erythematosus: a two-center integrated study
Supplemental material, sj-pdf-1-imr-10.1177_03000605221118702 for Infection profile and risk factors for mortality in patients with end-stage renal disease attributable to systemic lupus erythematosus: a two-center integrated study by Yuqiang Chen, Lisha Zhang, Qin Xue and Niansong Wang in Journal of International Medical Research
Footnotes
Author contributions
CYQ and WNS conceived and designed the study. ZLS and XQ contributed to data collection. CYQ, XQ, and WNS analyzed the data. CYQ and ZLS contributed materials and analysis tools. CYQ wrote the manuscript. All authors have read and approved the final version of the manuscript and agreed with the order of the authors.
Data availability
Supplemental material (Supplementary Table 1) for this article is available online.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was partially funded by the National Nature Science Foundation of China (NSFC81300602).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
