Abstract
Single-organ cutaneous small-vessel vasculitis is a specific type of vasculitis. Its recurrence rate is approximately 10%, and it is a serious health issue that can lead to increased healthcare costs. There have been no reports of successful treatment of recurrent necrotizing single-organ cutaneous small-vessel vasculitis in children. We report the case of an 11-year-old Chinese girl who presented with recurrently elevated anti-streptococcal O titers and worsening cutaneous necrosis of the lower extremities. She was diagnosed with streptococcal infection-related recurrent severe single-organ cutaneous small-vessel vasculitis and was successfully treated using a “cocktail” therapy, including antibiotics, plasma exchange, and immunosuppressants. She was followed up for 4 years with no recurrence. In conclusion, recurrent necrotizing single-organ cutaneous small-vessel vasculitis in children can be successfully treated with a “cocktail” therapy containing plasma exchange.
Keywords
Introduction
Vasculitis encompasses a wide spectrum of disorders caused by tissue inflammation damage and ischemia of the vessel walls or perivascular tissues. 1 It can occur in one or more organs, including the skin and kidneys. According to the size and structure of the related vessels, vasculitis can be divided into large-, medium-, small-, and variable-vessel vasculitis. Moreover, it can be divided into single-organ vasculitis and vasculitis associated with systemic disease; this condition is associated with different etiologies, depending on the location of the involved organ or the etiology of the related vessels. 2
Different types of vasculitis have different pathophysiologies, clinical characteristics, and prognosis. Cutaneous vasculitis (CV) consists of a series of disorders characterized by predominantly cutaneous involvement with varying degrees of systemic symptoms. CV is caused by inflammation involving small and medium blood vessels in the skin and is classified as primary CV or secondary CV. Examples of secondary CV include CV secondary to systemic vasculitis, infections, drug use, and malignancies. 3 CV may manifest in various forms, such as erythema, papules, purpura, ecchymosis, ulceration, necrosis, gangrene, livedo reticularis, and sunken scar, depending on the size and depth of the affected vessels. 4
Although certain types of vasculitis can occur in both children and adults, there are some differences in clinical manifestations, etiology, treatment, and prognosis between childhood vasculitis and adult vasculitis. CV is rare in children and mainly involves allergic vasculitis and polyarteritis nodosa. 5 Single-organ cutaneous small-vessel vasculitis (SoCSVV), also termed leukocytoclastic vasculitis, is a specific type of vasculitis defined by the 2012 International Chapel Hill Consensus Conference (2012 CHCC) nomenclature system. It is associated with ulcerative colitis and Crohn disease. 6 In particular, recurrent cutaneous small-vessel vasculitis (CSVV) associated with infections has rarely been reported in children, which poses a considerable challenge for clinical diagnosis and treatment. Here, the clinical manifestations of a pediatric case of streptococcal infection-related recurrent SoCSVV were reported; moreover, the clinical manifestations and treatment of SoCSVV were reviewed.
Case presentation
Chief complaints
A Chinese girl, aged approximately 10 years, who presented with recurrently elevated anti-streptococcal O (ASO) titers and worsening cutaneous necrosis of the lower extremities was admitted to the West China Second University Hospital in May 2023. She was diagnosed with streptococcal infection-related recurrent severe SoCSVV. Approval for publication of this case report was obtained from the West China Second University Hospital. We also obtained written informed consent from the patient for the publication of this report. The reporting of this study conforms to Case Report (CARE) guidelines. 7
History of present illness
The patient had swelling and pain in her hands and feet. After 10 days, she developed a fever and cutaneous rash. The clinical manifestations progressively worsened with the development of palpable purpura and ecchymosis, and she was subsequently admitted to Lu Xian Hospital (a local county hospital) and treated with antibiotics (flucloxacillin) and dexamethasone for 5 days. Laboratory examination revealed an elevated ASO titer at 843 (normal value: <166) IU/mL, indicating a streptococcal infection. The white blood cell (WBC) count was 36.49 × 109/L (normal value: 4–10 × 109/L), and the C-reactive protein (CRP) level was 154.13 (normal value: <8) mg/L.
Her clinical condition did not improve; therefore, she was admitted to the Children’s Hospital Affiliated to Chongqing Medical University with a fever, pain and swelling in the elbows and knees, and rashes on her limbs and face. Cutaneous histopathological examination revealed subcutaneous arteriole vasculitis and perivasculitis. After approximately 2 months, routine examinations showed an obviously increased ASO titer of 4953 IU/mL and a mildly increased erythrocyte sedimentation rate (ESR) of 62–81 (normal value: 0–20) mm/1 h. Pharyngeal swab testing revealed gram-positive cocci. Anti-nuclear antibody (ANA) spectrum and anti-neutrophil cytoplasmic antibodies (ANCA) were within normal limits. Blood culture and bone marrow culture results were normal. She was discharged after 20 days of antibiotic and glucose therapy, which resulted in a return to normal body temperature, pain relief, and alleviation of swelling and rash symptoms.
After half a month, due to the recurrent cutaneous ecchymosis and necrosis, accompanied with mouth ulcer, she was admitted to a local hospital again and treated with glucosteriod drugs and aspirin. She also underwent skin debridement for the cutaneous necrosis in the lower limbs. After 1 month of therapy, she asked for discharge from the hospital and then voluntarily stopped therapy. One month later, after the last discharge, she was admitted to our hospital due to recurrence of severe SoCSVV.
History of past illness
The patient was healthy and had unremarkable medical history of upper respiratory tract infection, drug use, or vaccination.
Personal and family history
Personal and family history did not reveal any notable findings.
Physical examination
One month after the last discharge, physical examination upon admission revealed cutaneous rash (Figure 1), swelling and pain in her limbs, and skin necrosis over a large area of her lower limbs (Figure 2). The patient had mild diarrhea, with mostly 1–2 bowel movements per day, which were pasty.

Recurrent single-organ cutaneous small-vessel vasculitis with streptococcal infection in a child. (a) An 11-year-old Chinese girl with single-organ cutaneous small-vessel vasculitis was included in this study and (b) skin lesion in the lower limbs of the patient.
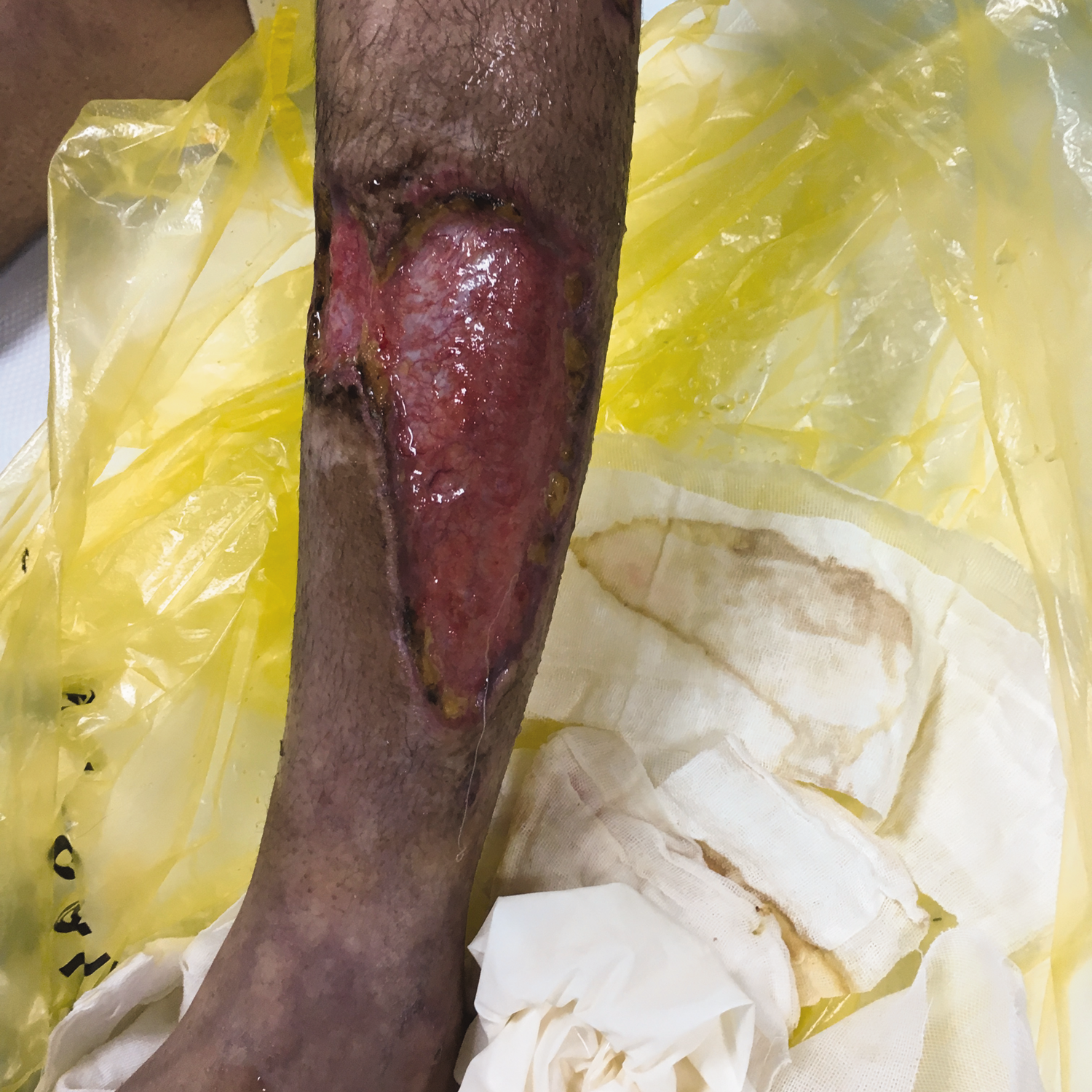
An enlarged area of skin necrosis in the lower limbs of the patient.
Laboratory examinations
Repeated laboratory examinations revealed an increased WBC count of 15.9–18.3 × 109/L, CRP level of 55–68.6 mg/L, and ESR level of 25–34 mm/h after 10 days of admission. The ASO titers were 423–684 IU/mL, indicating persistent streptococcal infection. The serum levels of C3 and C4 were normal. The urine sediment examination results were normal. The serum levels of ANA, anti-cardiolipin antibody (ACA), and lupus anticoagulant antibodies were all normal. ANA spectrum and ANCA were also within normal limits. The results of blood and bone marrow cultures were normal. There was no other major organ system involvement. Moreover, test results for hepatitis B virus, human immunodeficiency virus, and hepatitis C virus were negative.
Biopsy and immunofluorescence staining
Microscopic examination showed the following results: 1. Muscle: skeletal muscle fibers showed cross-striations, with no significant atrophy observed; 2. Skin: excessive keratinization of the epidermis was observed. A few lymphocytes infiltrated around the superficial dermis. The endothelium of small arteries in the subcutaneous tissue was markedly swollen, with lumen obstruction observed and neutrophils, eosinophils, and nuclear debris visible within the lumen. The vessel walls were thickened, with infiltration of neutrophils and eosinophils observed within the wall. One vessel wall showed fibrinoid necrosis. Epithelioid cells clustered around the vessels. Neutrophils and eosinophils infiltrated around the adipose tissue and skin appendages, with visible nuclear fragmentation. Immunohistochemistry showed the following results: CD31 (+), leukocyte common antigen (LCA) leukocytes (+), and smooth muscle actin (SMA) (+). Special staining results included Masson collagen (+).
Final diagnosis
Based on the medical history, clinical manifestations, and laboratory examinations, a diagnosis of streptococcal infection-related recurrent severe SoCSVV was established.
Treatment
A “cocktail” therapy was administered for 30 days, consisting of antibiotics (intravenous infusion of regular doses of penicillin and cefoperazone tazobactam for 7 days), plasma exchange (three times; fresh frozen plasma, 2000 mL, once every other day), and prednisone (60 mg, once a day), followed by an intravenous infusion of high-dose methylprednisolone (500 mg, once a day for 3 days) with oral mycophenolate mofetil (MMF; 1 g each time, 2 times a day). Patient consent was obtained before initiating treatment.
Outcome and follow-up
The patient’s CRP (5 mg/L) level and ESR (16 mm/h) decreased to normal levels after administering “cocktail” therapy for 2 months. The swelling and pain in her limbs and cutaneous rashes were resolved and fresh granulation tissue was observed in the area of previous skin necrosis after 20 hospital days.
After discharge, the treatment with oral prednisone and MMF was maintained for 12 months with tapered doses. Two months after discharge, her clinical symptoms completely disappeared. Five months after discharge, the previous large area of skin lesion was healed (Figure 3). She completely recovered from CV in the following year. She was followed up for 4 years without any recurrence of the disease.

Pathological changes in the skin lesion at 5 months after discharge.
Discussion
The clinical classification of vasculitis is mainly based on the 2012 CHCC nomenclature system. 2 For childhood vasculitis, a modified classification system based on the earlier 1994 CHCC system was proposed by the European League against Rheumatism and Pediatric Rheumatology European Society in 2005. 8 It has long been recognized that CV may be a single-organ disease limited to the skin or a symptom of many different clinical disorders. The types of CV are summarized in Supplementary Table 1. One of the major changes to the 2012 CHCC nomenclature is the introduction of a new major category—single-organ vasculitis—vasculitis that affects the blood vessels of a single organ, with no indication of the involvement of other organs. 9 For diseases confined to the skin, the term SoCSVV has been coined. 10 Here, we report, for the first time, the successful treatment of recurrent severe SoCSVV associated with streptococcal infection in a pediatric patient using a cocktail therapy.
Currently, only three studies have reported on the characterization of SoCSVV in a large sample of adults with CV.8–10 Previously, the term hypersensitivity vasculitis (HV), defined by the American College of Rheumatology in 1990, 11 was used to denote vasculitis of small blood vessels that is primarily, but not exclusively, cutaneous. In the 1994 CHCC system, the term “cutaneous leucocytoclastic angiitis” was coined to denote small-vessel vasculitis confined to the skin and not associated with any other primary vasculitis such as Henoch–Schoenlein purpura, granulomatosis with polyangiitis, or cryoglobulinemia. 12 However, it has been recognized that this term is also problematic because histological features are not always consistent with this clinical phenotype. 13 A more comprehensive term “leukocytoclastic vasculitis” (LCV) is also used in clinical practice and medical articles. Compared with HV or LCV, SoCSVV is a more specific definition. Ozen et al. 8 conducted a study among 250 adult HV patients at a Spanish university referral center between 1976 and 2011 and showed that only 60 (24%) patients met the diagnostic criteria for SoCSVV. Bouiller et al. 9 found that in 2000, only 29 of the 112 (25.9%) adult LCV patients in a French university hospital met the SoCSVV criteria. Pastuszczak et al. 10 , who conducted a study at a Poland university hospital from 2010 to 2015, reported that 24 of the 30 (80%) adult LCV patients met the SoCSVV criteria. These results consistently show that SoCSVV is a more restricted entity than HV or LCV because SoCSVV is confined to the skin without involvement of any other organs and always requires histological confirmation.
The disease-inducing or -triggering factors remain unknown for nearly a half of the SoCSVV patients. Some cases might be post-infectious, drug-induced, or vaccination-related. 14 It is generally accepted that most vasculitic lesions are mediated by immunopathologic mechanisms that can be categorized as hypersensitivity reactions or immune complex (IC) deposition/type III hypersensitivity reactions.3,15 IC deposition activates complement and, in turn, induces neutrophil chemotaxis and mast cell degranulation. 3 Immigrated neutrophils release proteolytic enzymes and free oxygen radicals, leading to vessel wall damage. In addition, increased adhesion between endothelial cells and inflammatory cells due to increased expression of adhesion molecules plays a role in CV pathogenesis. 13 Moreover, the exact mechanism of SoCSVV remains unknown. Although the patient in this case had no clear medical history of upper respiratory tract infection, drug use, or vaccination, her ASO titer was significantly increased on day 11 after disease onset, with a value of 843 IU/mL. Additionally, the ASO titer level fluctuated between 423 and 4953 IU/mL during the course of the disease, indicating streptococcal infection.
The clinical manifestations of CV vary greatly. Clinical manifestations in children with vasculitis (common or specific) are summarized in Supplementary Table 2. Many patients who are finally diagnosed with a specific type of vasculitis often have nonspecific systemic symptoms such as fever and skin lesions at the beginning. 15 The characteristics of CV lesions involving the superficial vessels of the derma include erythema, papules, and purpura. The manifestations of ecchymosis purpura, bullosa, and blood blister usually involve the reticular vessels of the dermis. 16 In SoCSVV cases, the most frequently observed skin-limited clinical manifestations are palpable purpura (ranging from 76% to 100%), as reported in the three retrospective re-analyses of adult SoCSVV.8–10 Other skin lesions including red macules, urticaria vasculitis, ulcers, and nodules were also observed, but at much lower frequencies. In addition to skin lesions, fever was observed in 18% and 31% of SoCSVV cases in studies reported by Ozen et al. 8 and Bouiller et al., 9 respectively. Notably, skin necrosis or pustules are rarely observed in cases of CSVV.10,17 In the current case, the child had recurrent fever and skin necrosis following the development of palpable purpura and ecchymosis, without the involvement of other organs. The clinical manifestations and laboratory examinations fulfilled the definition of typical SoCSVV related to streptococcal infection. The reason she was taken to multiple hospitals and hospitalized four times for disease recurrence may be related to treatment interruption.
SoCSVV is usually a benign disease with a good prognosis. Bouiller et al. 9 did not observe any case of relapse among the 29 SoCSVV patients in their study. However, Ozen et al. 8 and Pastuszczak et al. 10 reported recurrence rates of 8.3% (5/60) and 25% (6/24) in SoCSVV patients, respectively.8,10 Combining these three studies, the overall recurrence rate of SoCSVV was 9.7%. Recurrence is a serious problem that leads to increased healthcare costs. The patient in the current case relapsed repeatedly before seeking treatment at our hospital.
Generally, CV treatment depends on the type and severity of the disease and the risk of permanent organ damage. The primary goal in CV management is the identification of the triggering causes of vasculitis (e.g. infection and drug use). For infected patients, treatment for the underlying infection is necessary. 18 For cases that might be related to drug use, drug withdrawal is needed. For relieving the symptom of pruritus or burning, nonsteroidal anti-inflammatory drugs or antihistamines are used.19,20 For CV limited to the skin, anticoagulants, glucocorticoids, immunosuppressants, antihistamines, and nonsteroidal anti- inflammatory drugs are mainly used.21,22 In some severe cases, the use of intravenous immunoglobulin infusion, infliximab, and hydroxychloroquine has been reported and recommended.23,24 Etiology, treatment, and prognosis in children with CV are summarized in Supplementary Table 3.
Refractory CV patients have been treated with some steroid-sparing agents, including dapsone, colchicine, cyclophosphamide, methotrexate, MMF, intravenous immunoglobulin (IVIG), and anti-tumor necrosis factor agents. 25 CV patients with cutaneous necrosis may need prompt and aggressive vasodilation therapy with calcium channel blockers, nifedipine, prostaglandins, or pentoxifylline.26–28 Surgical debridement may be necessary in some severe cases. 28 Plasma exchange is used as an effective therapy for severe vasculitis with multiple organ involvement. 28 In our case, the patient was treated with antibiotics, glucocorticoids, and aspirin, with cutaneous debridement before hospital admission. However, the vessel vasculitis relapsed multiple times and aggravated with an enlarged area of skin necrosis, serious exudation, and a significantly increased inflammatory index. After admission to our hospital, we considered that this was a rare and intractable case of recurrent SoCSVV. If the disease was not treated promptly and aggressively, it might have resulted in the involvement of other organs.
Previous studies have shown that plasma exchange in children with severe systemic vasculitis led to immediate reduction in inflammatory markers, effective symptom relief, and disease remission.29,30 Considering the child’s severe clinical condition and the medical history of recurrence, we developed a “cocktail” regime. The patient received intravenous infusion of antibiotics, plasma exchange, and oral glucocorticoids followed by high-dose methylprednisolone pulse therapy. The ASO titers, CRP levels, and ESR significantly decreased after the treatment and remained at low levels following maintenance therapy with oral glucocorticoids together with MMF. Her clinical symptoms completely disappeared 2 months after discharge, and the previous large area of skin affected by lesion healed 5 months after discharge. The oral drug administration was discontinued after 12 months. The patient presented no side effects related to the therapy used. Of note, there was no relapse of the disease at the 4-year follow-up.
Based on the literature review, this case of SoCSVV shares similarities with several reported purpura cases while exhibiting unique features. Similar to the case reported by Miray Kisla Ekinci et al., 31 our patient presented with palpable purpura and systemic involvement, although our case demonstrated more severe cutaneous necrosis and required more aggressive treatment, including plasma exchange. The histopathological findings of leukocytoclastic vasculitis with fibrinoid necrosis align with those of complex purpura cases described by Gehlhausen et al., 32 particularly regarding vascular occlusion and inflammatory infiltrate patterns. However, unlike thrombotic thrombocytopenic purpura cases caused by influenza 33 or hepatitis E virus, 34 our patient showed no evidence of ADAMTS13 deficiency or thrombotic microangiopathy. The recurrent nature of symptoms resembles the chronic recurrent palpable purpura reported by Youakim et al. 35 Our patient uniquely demonstrated progression to severe cutaneous necrosis requiring surgical debridement. The therapeutic response to combined immunosuppressants (MMF and steroids) parallels successful treatments reported by Miray Kisla Ekinci et al. 31 and Zhang et al., 36 although our regimen included additional plasma exchange that was not described in those cases. This case expands the spectrum of infectious-associated vasculitis by documenting a severe recurrent form linked to persistent streptococcal infection (evidenced by sustained ASO elevation), which has not been previously reported with this clinical progression pattern.37,38
There are certain limitations in this study. First, this report is based on a single case; more patients need to be studied to confirm these findings. Second, the recurrence in our patient persisted for a long time. Studies on the involvement of possible causative factors and standard and prompt therapy are needed for managing streptococcal infections in SoCSVV patients. Third, early molecular diagnosis may contribute to streptococcal infection in SoCSVV patients.
Conclusion
To the best of our knowledge, there are no previous reports of successful treatment of recurrent severe SoCSVV in children. Our experience with this case showed that a “cocktail” therapy, involving antibiotics treatment combined with blood purification and immunosuppressive treatment, is effective in preventing SoCSVV recurrence in pediatric patients.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605251368334 - Supplemental material for Recurrent single-organ cutaneous small-vessel vasculitis with streptococcal infection in a pediatric patient: A case report
Supplemental material, sj-pdf-1-imr-10.1177_03000605251368334 for Recurrent single-organ cutaneous small-vessel vasculitis with streptococcal infection in a pediatric patient: A case report by Min Yang, Xiuying Chen, Zhihua Zhang, Lijuan Hu, Yannan Guo and Songhui Zhai in Journal of International Medical Research
Footnotes
Acknowledgements
Not applicable.
Author contributions
Zhai SH was the guarantor of integrity of the entire study; Yang M, Chen XY, and Zhai SH were involved in study concepts and design; Hu LJ and Guo YN provided intellectual content and performed literature research; Yang M, Chen XY, Zhang ZH, and Zhai SH were responsible for clinical studies; Chen XY, Hu LJ, and Guo YN performed experimental studies; Chen XY, Hu LJ, and Zhang ZH contributed to data acquisition; Chen XY and Yang M were involved in data analysis; Yang M, Chen XY, and Zhai SH performed manuscript preparation; Yang M and Zhai SH were responsible for manuscript editing; and Zhai SH undertook manuscript review. All authors have read and approved the final manuscript.
Data availability statement
Data may be available upon reasonable request by contacting the corresponding author.
Declaration of conflicting interests
The authors declare that they have no conflicts of interest.
Funding
This study was funded by Science and Technology Bureau of Sichuan Province (No. 2019YFS0240).
Informed consent statement
The patient’s mother provided written informed consent. We also obtained written consent for the publication of the case report from the patient’s mother.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
