Abstract
The prognosis of renal pelvis cancer is poor and therapeutic options are limited, especially for patients with advanced disease. In this report, we present a case of advanced renal pelvis carcinoma in a male patient in his 60s, characterized by an activating mutation in ERBB2. Clinical evaluation resulted in a pathological diagnosis of renal pelvis carcinoma with liver metastasis. Immunohistochemistry staining results suggested that CK, P63, and PAX8 were positively expressed, while Sy, CK7, CK20, S100, PAX8, and HEP1 were negatively expressed. Furthermore, next-generation sequencing results showed an activating mutation in the ERBB2 gene. The patient initially received a trastuzumab-based combination therapy, which led to a significant reduction in ERBB2 mutation frequency and a stable condition after three treatment cycles. However, following continuous treatment for 4 months, the patient developed drug resistance that resulted in disease relapse. Subsequently, the patient received apatinib treatment, but the therapeutic response was not satisfactory. The patient’s condition underwent rapid deterioration and he ultimately succumbed to the disease. This case underscores the potential benefit of trastuzumab for treating ERBB2-mutated advanced renal pelvis cancer, but further highlights that overcoming drug resistance remains a crucial challenge for long-term efficacy.
Introduction
Urothelial carcinoma (UC) is the predominant cancer type within the genitourinary system, with the bladder being its primary occurrence site.1,2 Involvement of both the renal pelvis and bladder is observed in approximately 5% of cases.3,4 Renal pelvis cancer prognosis is generally poor, with limited therapeutic options, particularly for patients in advanced stages. Therefore, there is a need to explore new effective treatment options.
The ERBB2 gene encodes an important cell surface protein receptor, recognized as human epidermal growth factor receptor 2 (HER2).5,6 The presence of activating mutations or amplifications within ERBB2 contributes to an escalated count of HER2 receptors on the cellular exterior and intensified receptor signaling functionality. This, in turn, fosters unregulated cellular proliferation and growth, a characteristic hallmark of malignant conditions.7,8 Anomalies in the ERBB2 gene can potentially induce excessive HER2 activation, establishing a clear association with diverse types of malignancies, predominantly breast and gastric cancers.9,10 However, evaluating ERBB2 expression is not currently a standard facet of routine diagnostic procedures for UC. Precisely estimating the subset of UC patients that exhibit ERBB2 positivity, which comprises those who could potentially benefit from anti-HER2 targeted therapeutics, remains uncertain. 11
Studies have predicted that ERBB2-activating mutations may exhibit sensitivity to anti-HER2 monoclonal antibodies, such as trastuzumab and pertuzumab, which have Food and Drug Administration approval for treating breast and gastric cancers.12–14 Peyromaure et al. documented their preliminary work with trastuzumab in metastatic transitional cell carcinoma of the urinary tract, with their findings suggesting that using trastuzumab potentially offers a safe and efficacious therapeutic avenue for managing this cancer type. 15 ERBB2 mutations have also been found in some patients with refractory renal pelvis carcinoma.16,17 Notably, bladder cancer patients with an ERBB2 mutation showed a favorable response to trastuzumab.18,19 However, there are limited reports on the use of trastuzumab in renal pelvis carcinoma.
In this report, we present the case of an advanced renal pelvis carcinoma patient with an ERBB2 insertion mutation who experienced benefits from trastuzumab therapy. Our case contributes to this field by providing supplementary information for the treatment of clinical patients.
Case report
In October 2016, a male patient in his early 60s was pathologically diagnosed with renal pelvis carcinoma with liver metastasis from positron emission tomography-computed tomography (PET-CT) and liver biopsy findings (Figure 1). The imaging diagnostic findings revealed noteworthy characteristics, including a thickened wall of the calices within the left renal pelvis, which exhibited enhancement and heightened metabolic activity. Left renal pelvis cancer was initially suspected, coupled with an observed delay in the excretory function of the left kidney. Notably, heightened metabolic activity was evident in multiple smaller lymph nodes adjacent to the abdominal aorta at the level of the left renal hilus, prompting consideration for possible metastasis. The presence of numerous hypermetabolic nodules within both the lungs and liver further suggested metastatic tumor involvement. Enlargement of the right hilar lymph node with augmented metabolic activity provided additional evidence. Immunohistochemistry staining results showed that choline kinase (CK), protein 63 (P63), and paired-box gene 8 (PAX8) were positively expressed, while synemin (SYNM), cytokeratin 7 (CK7), CK20, S100, PAX8, and human ezrin peptide 1 (HEP1) were negatively expressed. These results, combined with the patient’s clinical presentation of a left renal tumor, demonstrated a high likelihood of renal cancer metastasis.
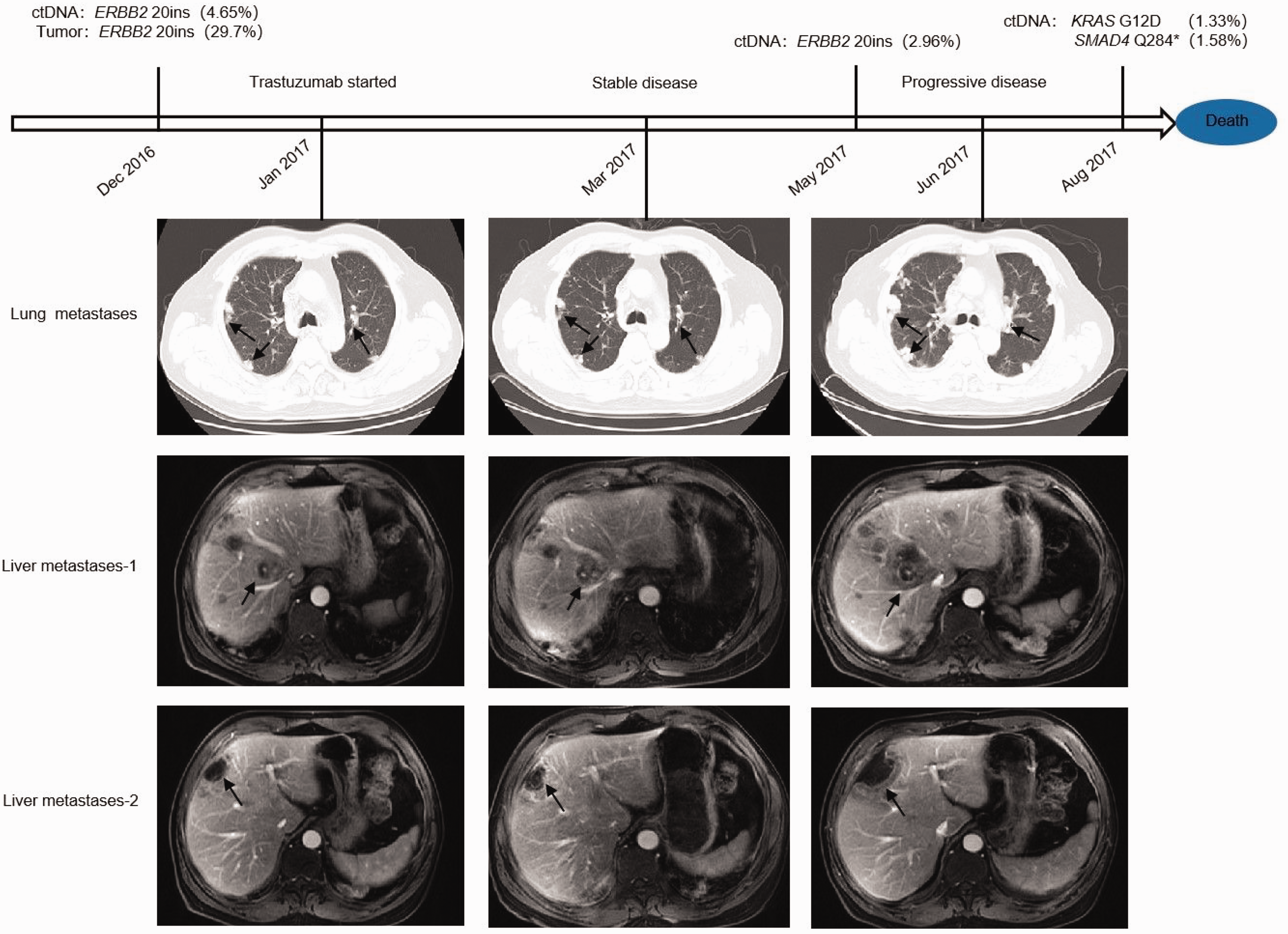
Treatment timeline and computed tomography results.
The patient’s weight remained relatively stable and he did not manifest any notable symptoms, such as lumbar or abdominal discomfort, except for mild hematuria. It should also be highlighted that the patient had a smoking record spanning approximately three decades, as smoking is a risk factor for UC.20,21
The patient’s initial chemotherapy regimen consisted of gemcitabine (1.7 g) on days 1 to 8 and cisplatin (40 mg) on days 1 to 3. Unfortunately, after four cycles of chemotherapy, the disease remained uncontrolled and rapidly progressed, with the development of multiple new metastases in the lungs and liver. To explore further therapeutic options, we collected the patient’s circulating tumor DNA (ctDNA) and tumor tissue for next-generation sequencing (NGS) analysis. The results revealed an activating non-frameshift insertion mutation (c.2310_2311insGCATACGTGATG/p.E770delinsEAYVM, allele frequency 4.65%) in exon 20 of the ERBB2/HER2 gene, along with mutations in several other genes (Table 1).
Allele frequency (af, 4.65%) in exon 20 of ERBB2/HER2 together with mutations in several other genes.

NGS, next-generation sequencing; RUNX1, runt-related transcription factor 1; KMT2C, lysine methyltransferase 2C; FGFR2, fibroblast growth factor receptor 2; SMARCB1, SWI/SNF related, matrix associated, actin dependent regulator of chromatin, subfamily b, member 1; SMAD4, SMAD family member 4; N/A, not available (the mutation was not observed); ctDNA, circulating tumor DNA.
The ERBB2-activating mutation was predicted to be sensitive to anti-HER2 monoclonal antibodies, such as trastuzumab and pertuzumab, which have been approved by the Food and Drug Administration for treating breast cancer and gastric cancer.22,23 The patient started a trastuzumab-based therapy in January 2017. The treatment regimen included trastuzumab and cisplatin (40 mg) on days 1 to 3 and xeloda (1 g) twice daily on days 1 to 14. After three cycles of treatment, the patient achieved a stable disease status and continued with this regimen. The mutation frequencies of these mutations and levels of carcinoembryonic antigen (CEA) were dynamically monitored in December 2016 and May and August 2017 (Table 1; Figure 2; Supplementary Table 1).
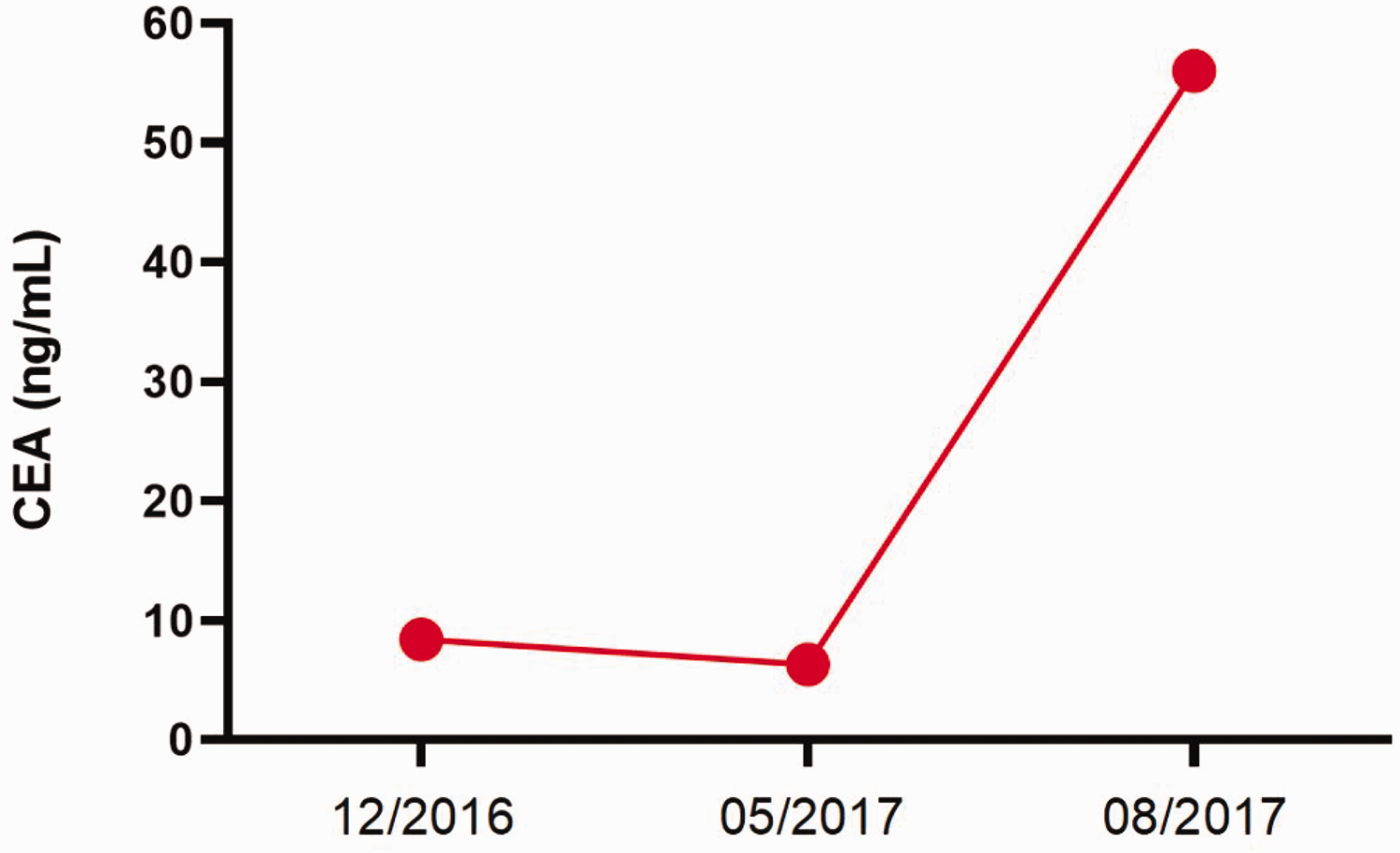
Carcinoembryonic antigen status of the patient’s blood test results was consistent with the variations of mutation allele frequencies in the next-generation sequencing results.
The results demonstrated that the trastuzumab-based treatment was initially beneficial to the patient, as evidenced by a significant reduction in CEA levels and decreased frequency of ERBB2 mutations from January to May 2017. However, after 4 months of trastuzumab treatment, the patient developed drug resistance that led to a rapid deterioration of his condition. CEA levels markedly increased and the ERBB2 mutation frequency began to rise. In response to this, the patient commenced apatinib treatment in June 2017. Unfortunately, despite this intervention, the patient’s condition could not be controlled and he succumbed to the disease.
The reporting of this study conforms to CARE guidelines. 24 This case report involving human participants was reviewed and approved by the Shanghai Eastern Hepatobiliary Surgery Hospital Ethics Committee. Furthermore, verbal informed consent was meticulously acquired from both the patient and his family for the proposed treatment regimen. This article diligently safeguards patient privacy by employing de-identification measures to ensure the confidentiality of personal information. The patient’s family provided approval for the publication of this case report and the associated images.
Discussion
In this report, we presented a case of an advanced renal pelvis cancer patient with a novel ERBB2 insertion mutation (E770delinsEAYVM) who initially responded to trastuzumab treatment and achieved a stable disease status for 4 months. This novel ERBB2 mutation is potentially a trastuzumab-sensitive mutation in renal pelvis cancer. ERBB2 insertion mutations have been observed in various types of tumors, including breast cancer, gastric cancer, and lung cancer.25–27 These mutations can result in a constitutively active form of the HER2 protein, leading to increased cell proliferation, survival, and metastatic potential. Yang et al. demonstrated that ERBB2 insertion mutations possess the potential to heighten drug resistance in certain breast cancer cases. 28 However, our understanding of ERBB2 insertion mutations in renal pelvic carcinoma remains limited, with an inadequate number of pertinent case reports on the subject.
Following trastuzumab administration, the patient exhibited a positive clinical response, with the disease remaining stable. Subsequent ctDNA sequencing analysis revealed a decline in the maximum allele frequency (MAF) of the ERBB2 E770delinsEAYVM mutation throughout the treatment course. This observation is consistent with previous reports that have linked a decreased MAF of sensitive mutations or target genes to a favorable response in colon cancer and non-small cell lung cancer.29,30 Similar trends were initially observed in our case. However, despite the disease stability and minimal changes in tumor volume during treatment, the MAF of the ERBB2 insertion mutation and those of several other mutations continued to decline. These observations were difficult to understand, until four months later when the patient developed drug resistance and experienced tumor progression.
A third peripheral ctDNA analysis was conducted, which revealed two novel mutations: KRAS G12D (allele frequency 1.33%) and SMAD4 Q284* (allele frequency 1.58%) (Table 1). Because KRAS is downstream of HER2 in the signaling pathway, activating mutations in KRAS may explain the development of trastuzumab resistance in this case. Additionally, research has shown that loss of SMAD4 function can contribute to reduced sensitivity to EGFR/ERBB2-targeted drugs.31–33 Unfortunately, because of the limited availability of effective therapeutics against KRAS and SMAD4 mutations, we were unable to control the disease progression.
Our observations in this case align with the concept that the clonal architecture of a tumor undergoes dynamic changes during treatment. We believe that the novel ERBB2 insertion mutation was sensitive to trastuzumab and the subclone harboring this mutation was eliminated during treatment. However, a new subclone carrying the SMAD4 and KRAS mutations, which exhibited resistance to trastuzumab, gained a growth advantage under the selective pressure of trastuzumab. Consequently, the elimination of the trastuzumab-sensitive subclone and emergence of the trastuzumab-resistant subclone resulted in stable tumor volume. These findings underscore the importance of dynamically monitoring the mutation status during treatment, as it can provide a better understanding of the tumor's status and aid in optimizing treatment regimens in a timely manner.
Conclusion
The advent of cancer genome sequencing has led to significant advancements in various NGS platforms. In our study, we performed NGS testing on cell-free ctDNA and tumor tissue samples from a patient with advanced renal pelvis cancer, which supported the identification of an ERBB2 activating mutation. The patient subsequently received trastuzumab treatment, which resulted in a period of stable disease for 4 months. This case highlights the potential efficacy and clinical application of trastuzumab in renal pelvis cancer, a disease with limited treatment options. Moreover, trastuzumab-based therapy appears to be a safe and effective choice for metastatic renal pelvis cancer. Prospective trials are warranted to further investigate and validate this therapeutic option.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605231204502 - Supplemental material for Advanced renal pelvis carcinoma patient with an ERBB2 insertion mutation: a case report
Supplemental material, sj-pdf-1-imr-10.1177_03000605231204502 for Advanced renal pelvis carcinoma patient with an ERBB2 insertion mutation: a case report by Peipei Shang, Cheng Lou, Lei Yin, Chenjie Tao, Yulong Dong, Guang Yang and Zhengang Yuan in Journal of International Medical Research
Supplemental Material
sj-pdf-2-imr-10.1177_03000605231204502 - Supplemental material for Advanced renal pelvis carcinoma patient with an ERBB2 insertion mutation: a case report
Supplemental material, sj-pdf-2-imr-10.1177_03000605231204502 for Advanced renal pelvis carcinoma patient with an ERBB2 insertion mutation: a case report by Peipei Shang, Cheng Lou, Lei Yin, Chenjie Tao, Yulong Dong, Guang Yang and Zhengang Yuan in Journal of International Medical Research
Footnotes
Acknowledgements
We extend our heartfelt appreciation to the Shanghai Eastern Hepatobiliary Surgery Hospital for their invaluable support. We would also like to extend our gratitude to the patient and his family for their dedication and efforts. Most importantly, we wish to convey our profound thanks to the patients for their unwavering support in advancing the realm of medicine.
Author contributions
PS proposed writing this case report. All authors participated in the preparation and review of the paper.
Data availability statement
The datasets used and analyzed during the current study are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
