Abstract
Objective
This study was performed to assess the influence of obesity on colorectal cancer (CRC) and investigate the efficacy of Xiaotan Tongfu (XTTF) decoction to CRC treatment.
Methods
BALB/C mice were used to establish an obesity-associated CRC model by a high-fat diet and tumor implantation. The tumors were harvested from mice inoculated with CT26 cell suspension. Body weight, liver weight, hepatic metastasis, and histological changes were observed. Immunohistochemical analysis and real-time polymerase chain reaction were performed to measure the levels of insulin-like growth factor 1 (IGF-1), IGF-1 receptor (IGF-1R), and IGF binding protein 3 (IGFBP-3).
Results
Obesity influenced the secretion of IGFs and aggravated CRC, while XTTF decoction inhibited the process of hepatic metastasis in CRC by upregulating the secretion of IGF-1/IGF-1R and downregulating the secretion of IGFBP-3.
Conclusions
This study provides evidence that XTTF decoction can serve as a candidate curative treatment for CRC.
Keywords
Introduction
Colorectal cancer (CRC) is the third most frequently diagnosed cancer in men and the second in women, and it has a poor prognosis with a high mortality rate. 1 High rates of CRC in “Westernized” countries indicate that environment or lifestyle, especially the intake of high-energy foods and lack of exercise, may play a vital role in the etiology of CRC.
Mounting epidemiological evidence has consistently shown a positive association between obesity and CRC; ecological data show high rates of CRC in countries with a high prevalence of obesity. 2 Although there is convincing evidence connecting obesity with the risk of CRC, the underlying biological mechanisms have not yet been fully elucidated. 3 Most of the signaling pathways associated with the process of tumor progression focus on transcription factors that fundamentally adjust the expression of genes involved in the stimulation of inflammation, angiogenesis, and tumor metastasis. 4 Insulin resistance, adipokines, and chronic inflammation induced by obesity might play key roles in the complex metabolic pathways of colorectal carcinogenesis. 2 Supportive evidence suggests that insulin-like growth factor 1 (IGF-1) is associated with CRC, and higher levels of IGF-1 have been linked with an increased risk of CRC. 5 Surgery, chemotherapy, and radiotherapy are currently the mainstays of CRC therapy; however, the adverse effects of these therapies cannot be ignored. Novel or additional safer treatment methods are thus needed.
Traditional Chinese medicine (TCM) has been widely used and shown to be effective in CRC treatment.6,7 On the basis of studies of various types of TCM, we prepared Xiaotan Tongfu (XTTF) decoction, which has complex compositions and exerts functions mainly by Pinelliae rhizoma, Citrus reticulata Blanco, and Poria, as an effective approach to the treatment of CRC. Previous studies have suggested that Pinelliae rhizoma obviously inhibits growth of ovarian cancer SKOV3 cells. 8 Nobiletin, an active compound of Citrus reticulata Blanco, has been confirmed to suppress the proliferation and invasion of several kinds of colon cells. 9 Pachymic acid from Poria has significant anti-cancer activity in cell culture, and it can initiate apoptosis in prostate cancer cells through mitochondrial dysfunction. 10 On the basis of these findings, we speculated that XTTF decoction can be applied to the management of CRC.
In this study, we hypothesized that obesity has a positive influence on CRC while XTTF decoction can effectively relieve CRC by interfering with hepatic metastasis. We considered that its mechanism of action may involve IGFs such as IGF-1, IGF-1 receptor (IGF-1R), and IGF binding protein 3 (IGFBP-3). XTTF decoction could be valuable in the treatment of CRC.
Methods
Materials
The components of XTTF decoction were purchased from Shanghai Ley’s Pharmaceutical Co., Ltd. (Shanghai, China), and the decoction was manufactured in Changzheng Hospital. Antibodies to IGF-1, IGF-1R, and IGFBP-3 were obtained from Abcam (Cambridge, UK). Trizol reagent and transcription reagent were obtained from JRDUN Biotechnology Company (Shanghai, China).
Cell culture
Mouse colon cancer CT26 cells were purchased from Shanghai Cells Center, the Chinese Academy of Sciences (Shanghai, China). Cells were cultured in RPMI-1640 medium (GIBCO, Life Technologies, Carlsbad, CA, USA) containing 25% fetal calf serum (Hangzhou Sijiqing Biological Engineering Materials Co., Ltd., Hangzhou, China) and incubated at 37°C in an atmosphere of 5% carbon dioxide.
Animals
Male BALB/C mice aged 6 to 8 weeks and weighing 18 to 20 g were purchased from the Animal Experiment Center of the Chinese Academy of Sciences (Shanghai, China). The mice were maintained in a pathogen-free animal facility for 1 week before the experiment. All experiments were conducted in accordance with the guidelines for the care and use of laboratory animals. All experimental operations performed were approved by the Institutional Review Board of Shanghai SLAC Laboratory Animal Co., Ltd.
Establishment of high-fat linked hepatic metastasis model of CRC in mice
To optimize our findings of obesity-associated CRC in this study, we divided the animal experiments into two parts.
Part 1: The mice were randomly divided into two dietary groups (n = 20 each): those fed a regular diet and those fed a high-fat diet. Twelve weeks after the beginning of feeding, the mice were subcutaneously inoculated with a total of 1.0 × 108 CT26 cells in their right axilla to develop implantation tumors. Histologically intact tumor tissue was then harvested and implanted into the colon wall of mice to establish a model similar to human CRC. All mice were divided into four groups (n = 8 each) as follows: the regular-control group (R-C group), fed a regular diet; the fat-control group (F-C group), fed a high-fat diet; the regular-model group (R-M group), fed a regular diet and subjected to tumor implantation; and the fat-model group (F-M group), fed a high-fat diet and subjected to tumor implantation.
Part 2: The same preliminary treatment as in Part 1 was performed to establish an obesity-associated CRC model, and all mice were divided into three groups (n = 8 each): the control group, untreated and fed a regular diet; the model group, fed a high-fat diet and subjected to tumor implantation; and the XTTF group, fed a high-fat diet, subjected to tumor implantation, and then treated with XTTF decoction (3.536 mg/(g.d), once daily).
All mice were continuously fed the same regular diet. After 4 weeks, all mice were killed by spinal dislocation.
Histological analysis and immunohistochemistry (IHC) analysis
The liver tissues were fixed in 10% phosphate-buffered formalin, embedded in paraffin, and sectioned at 5 μm. The sections were then stained with hematoxylin and eosin. The slides were observed and photographed with a microscope (Olympus BH2; Olympus, Tokyo, Japan).
The 5-μm-thick sections obtained from the formalin-fixed and paraffin-embedded tissue specimens were routinely dewaxed and rehydrated followed by endogenous peroxidase blocking. The sections were blocked with 20% goat serum albumin (Dako, Glostrup, Denmark) followed by incubation with antibodies to IGF-1, IGF-1R, and IGFBP-3 for 2 hours. The sections were then incubated with HRP-R/M (Dako) for 30 minutes according to the manufacturer’s instructions and counterstained with hematoxylin. Positive results were stained brown and counted at ×400 magnification.
RNA extraction and real-time polymerase chain reaction
Total RNA was isolated from the liver using Trizol reagent according to the manufacturer’s instructions. The RNA was reversed-transcribed into cDNA, and the reverse transcriptase product was amplified by gene-specific primer pairs; glyceraldehyde 3-phosphate dehydrogenase was used as a normalizing control. The primers (Applied Biosystems, Foster City, CA, USA) used for detection are shown in Table 1. The amplification conditions were as follows: 40 cycles of 2 minutes of pre-denaturation at 94°C and 25 s of denaturation at 94°C followed by 30 s of annealing. Reactions were performed in duplicate, and data were analyzed using the 2−△△Ct method. 11
PCR primers for detection.

PCR, polymerase chain reaction; IGF-1, insulin-like growth factor 1; IGF-1R, insulin-like growth factor 1 receptor; IGFBP-3, insulin-like growth factor binding protein 3; GAPDH, glyceraldehyde 3-phosphate dehydrogenase.
Statistical analysis
Analysis of variance was performed using SPSS Version 16.0 (SPSS Inc., Chicago, IL, USA). Values are presented as mean ± standard deviation. Statistical significance between groups was determined by Student’s t-test or one-way analysis of variance. A p value of ≤0.05 was considered statistically significant.
Ethics
All animal experiments were conducted according to the ethical guidelines of Shanghai SLAC Laboratory Animal Co., Ltd.; Shanghai Laboratory Animal Center of the Chinese Academy of Sciences; and the “3R” principle. All efforts were made to minimize animal suffering. The research project was approved by the Ethics Committee of Shanghai SLAC Laboratory Animal Co., Ltd. and Shanghai Laboratory Animal Center of the Chinese Academy of Sciences.
Results
Obesity increases the growth of solid tumors and hepatic metastases in CRC
As stated in the Introduction section, obesity has a great influence on the evolution of CRC. To further explore and verify this relationship, we implanted tumor tissue into mice to establish a hepatic metastatic CRC model that paralleled that in humans. Figure 1 shows that the high-fat diet caused an increase in body weight, while obesity conversely resulted in great weight loss in mice with CRC. In a comparison of the F-M group with the other groups, we found that tumors were significantly larger and heavier, the liver structures were more severely damaged, and hepatic metastases were more widespread and severe (p < 0.05 for all). These findings indicate that obesity might accelerate the severity of CRC.

Obesity increases the growth of solid tumors and hepatic metastases in CRC. (a) A high-fat diet induced obesity in BALB/C mice. ***p < 0.001 vs. R-C group. Obesity-associated CRC increased (b) tumor weight (*p < 0.05 vs. R-M group) and (c) hepatic metastases in CRC (***p < 0.001 vs. R-M group). (d) Histological analysis of tumor sections with hematoxylin and eosin staining. The mean ± standard deviation was used to obtain the data in this figure using SPSS software. CRC, colorectal cancer; R-C group, regular-control group (fed a regular diet); F-C group, fat-control group (fed a high-fat diet); R-M group, regular-model group (fed a regular diet and subjected to tumor implantation); F-M group, fat-model group (fed a high-fat diet and subjected to tumor implantation).
Obesity upregulates protein expression and mRNA levels of IGF-1 and IGF-1R and downregulates level of IGFBP-3 in CRC
The levels of IGFs are closely associated with an increased risk of CRC. Figure 2(a) shows that the protein expression of IGF-1 was higher in the R-M and F-C groups than in the R-C group and was highest in the F-M group. The mRNA level of IGF-1 was significantly better in the F-M than R-C group (p < 0.05), while there was no obvious difference between the other groups (Figure 2(d)). The same findings were obtained for IGF-1R (Figure 2(b), (e)). The IGFBP-3 levels were lower in the R-M and F-C groups than in the R-C group and were lowest in the F-M group (Figure 2(c), (f)). These findings suggest that obesity and CRC have an effect on IGF levels to a certain degree and that obesity can promote upregulation of the protein expression and mRNA level of IGF-1 and IGF-1R while downregulating these levels of IGFBP-3, thus having a positive influence on CRC.

Effect of obesity on protein expression and mRNA levels of IGF-1, IGF-1R, and IGFBP-3 of BALB/C mice. Protein expression of (a) IGF-1, (b) IGF-1R, and (c) IGFBP-3 was quantified following immunohistochemical analysis. The mRNA levels of (d) IGF-1, (e) IGF-1R, and (f) IGFBP-3 were detected by real-time polymerase chain reaction. The mean ± standard deviation was used to obtain the data in this figure using SPSS software. *p < 0.05, **p <0.01, and ***p < 0.001 vs. R-C group. IGF-1, insulin-like growth factor 1; IGF-1R, insulin-like growth factor 1 receptor; IGFBP-3, insulin-like growth factor binding protein 3; R-C group, regular-control group (fed a regular diet); F-C group, fat-control group (fed a high-fat diet); R-M group, regular-model group (fed a regular diet and subjected to tumor implantation); F-M group, fat-model group (fed a high-fat diet and subjected to tumor implantation).
XTTF decoction significantly increases mice body weight and inhibits growth of solid tumors
We administered XTTF decoction at 3.536 mg/(g.d) to assess the curative effect of this decoction on CRC. After the mice had been killed, we observed that the tumors had grown continuously and that the final tumors were visibly larger in the model group than in the other groups. As shown in Figure 3(a), the mice in the XTTF group showed a significantly higher body weight than those in the model group (p < 0.05), whereas there was no obvious difference in body weight between the control group and model group. The tumor weight was obviously lower in the XTTF group than in the model group (Figure 3(c)). These data suggest that XTTF decoction can ameliorate the body weight loss and decrease the tumor growth of CRC in mice.
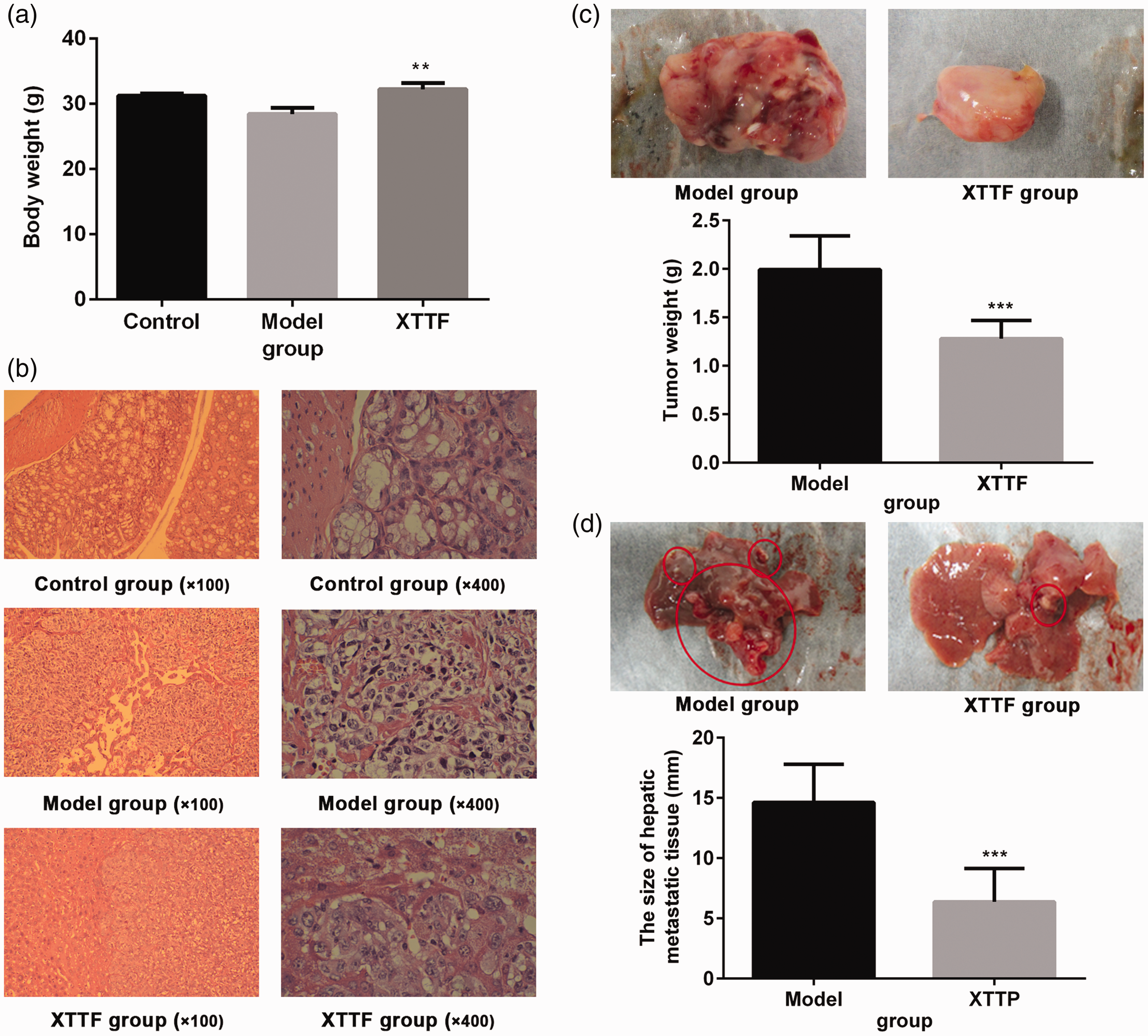
XTTF decoction inhibits the progress of tumor growth and hepatic metastases in obesity-associated colorectal cancer. Changes in (a) body weight and (b) tumor section of mice in the model group after administration of XTTF decoction. (c) XTTF decoction significantly decreased the tumor weight and the size of the hepatic metastatic tissue. The mean ± standard deviation was used to obtain the data in this figure using SPSS software. **p < 0.01 and ***p < 0.001 vs. model group.
XTTF decoction suppresses hepatic metastases of CRC in mice
As shown in Figure 3(d), we observed several white nodules on the liver surface in the model group, while few such nodules were found in the XTTF group. Under the microscope (Figure 3(b)), the hepatic lobule structure was markedly damaged, and no normal liver tissue was seen. Most tissues were necrotic, and infiltration of a large number of metastatic cancer cells was observed. The cytoplasm was small in volume and clumped, and this finding was closely associated with the macroscopic formation of cancer nodules. In the XTTF group, the hepatic lobule structure was relatively integrated with slight infiltration of only a few cancer cells. The histological analysis suggested that XTTF decoction can effectively suppress hepatic metastases of CRC in BALB/C mice.
XTTF decoction downregulates protein expression and mRNA level of IGF-1 and IGF-1R and upregulates IGFBP-3 in hepatic metastases of CRC in mice
In an effort to determine whether XTTF decoction has a positive influence on hepatic metastases through the IGF system, IHC analysis and real-time polymerase chain reaction were used to detect protein expression and mRNA levels of IGF-1, IGF-1R, and IGFBP-3 in hepatic metastases.
As shown in Figure 4(a)–(c), the protein expression of IGF-1 and IGF-1R was dramatically lower in the XTTF group than model group, and the protein expression of IGFBP-3 was obviously increased in the XTTF group. IHC analysis revealed that XTTF decoction could significantly downregulate the protein expression of IGF-1 and IGF-1R and upregulate the protein expression of IGFBP-3 in hepatic metastases of CRC in BALB/C mice (p < 0.05).
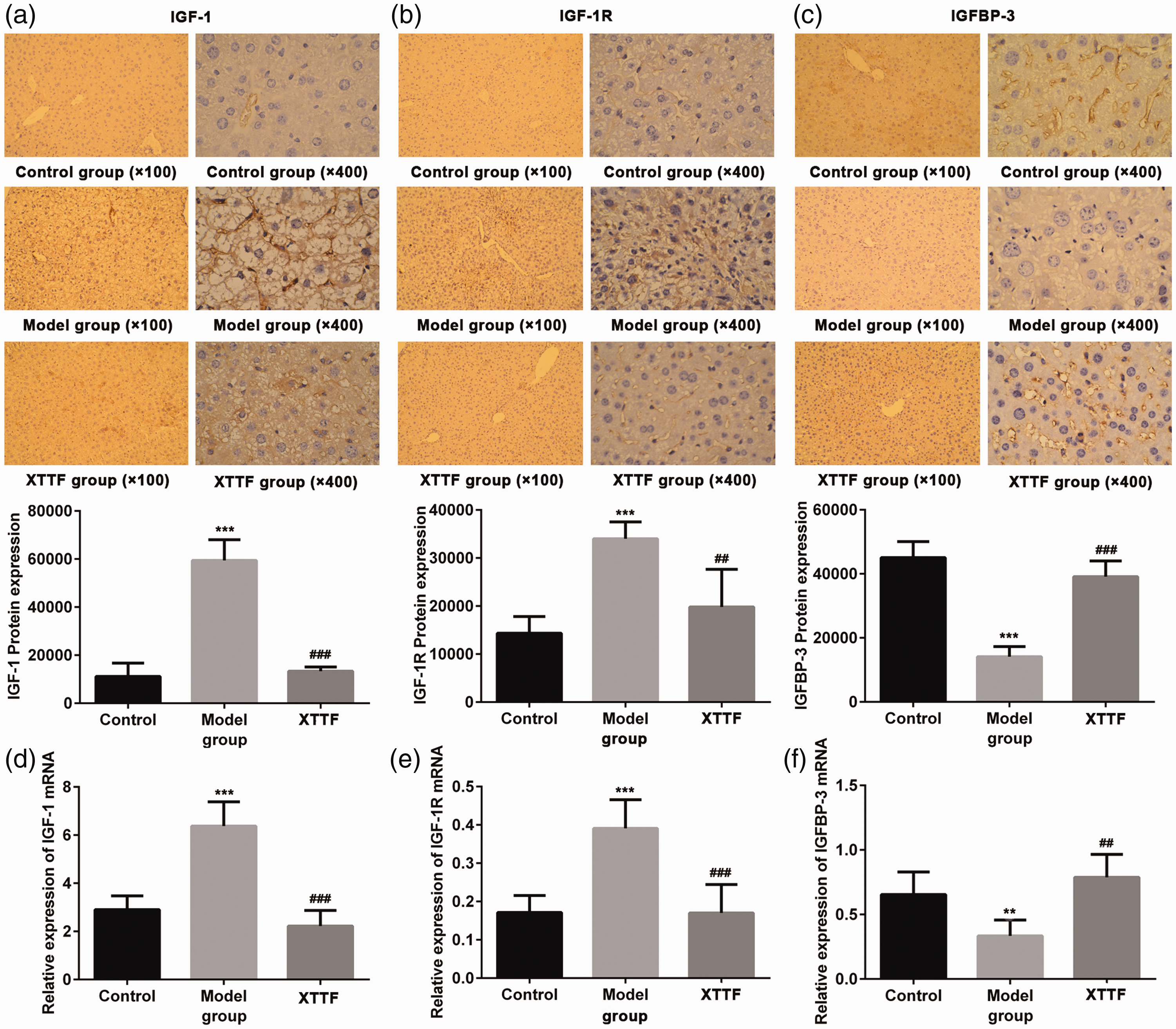
Effect of XTTF decoction on protein expression and mRNA level of IGF-1, IGF-1R, and IGFBP-3 in obesity-associated colorectal cancer. Protein expression of (a) IGF-1, (b) IGF-1R, and (c) IGFBP-3 was quantified following immunohistochemical analysis. The mRNA levels of (d) IGF-1, (e) IGF-1R, and (f) IGFBP-3 were detected by real-time polymerase chain reaction. The mean ± standard deviation was used to obtain the data in this figure using SPSS software. **p < 0.01 and ***p < 0.001 vs. control group; ##p < 0.01 and ###p < 0.001 vs. model group. IGF-1, insulin-like growth factor 1; IGF-1R, insulin-like growth factor 1 receptor; IGFBP-3, insulin-like growth factor binding protein 3.
The results shown in Figure 4(d) and (e) indicate that the mRNA levels of IGF-1 and IGF-1R were obviously lower in the XTTF group than in the model group, whereas the mRNA level of IGFBP-3 was significantly increased in the XTTF group (p < 0.05). These results indicate that XTTF decoction can downregulate the mRNA levels of IGF-1 and IGF-1R and upregulate that of IGFBP-3 in hepatic metastases of CRC in BALB/C mice.
Discussion
CRC is one of the most common cancers encountered in the clinical setting and has a poor prognosis and high mortality rate. Most cases of cancer mortality are due to metastasis rather than the tumor itself; thus, hepatic metastasis is an important factor of mortality in patients with CRC.
In an effort to identify effective therapies for CRC, we must establish a stable, feasible, and repeatable animal model to simulate human CRC. In the present study, we implanted intact tumor tissue harvested from mice inoculated with a CT26 cell suspension into the colon wall of mice, similar to human CRC.12–14 After 2 weeks, obvious nodules had formed and continued to become larger throughout the study period. This result indicated that we successfully established a simple and reproducible CRC model with a high rate of hepatic metastasis.
Increasingly more research is showing that IGFs are a key potential target in the therapy of cancer. IGF-1, an important mitogenic factor, is activated by insulin resistance. It can promote cell proliferation and inhibit cell apoptosis. 15 IGF-1 combines with IGF-1R and exerts both autocrine and paracrine functions. IGFBP-3, a growth factor that promotes cell apoptosis, competitively binds with IGF-1R and functions through accommodating IGF-1 or even functions independent of IGFs. 16 IGFBPs can form complexes with IGF-1, accompanied by a reduction in the amount of free IGF-1 available and a decrease in IGF-1 mediated DNA synthesis. 17 Obesity, especially abdominal fat, is a primary cause of insulin resistance. 18 Obesity-induced hyperinsulinemia can increase hepatic synthesis of IGF-1 and reduce the secretion of IGFBP, resulting in improved levels of circulating IGF-1. 12 In the present study, the levels of IGF-1 and IGF-1R were improved and the levels of IGFBP-3 were reduced in the model group, demonstrating the presence of a close association between obesity and CRC and the possibility that obesity promotes hepatic metastasis in the process of CRC.
Furthermore, in one study, the levels of IGF-1 mRNA in human colon cancer specimens were reportedly 3- to 5-fold higher than those in normal colonic epithelium. 19 Mouse experiments have also demonstrated that decreases in the amount of IGF-1 and activity of IGF-1R in the tissue or blood circulation can inhibit the growth of CRC cells.20,21 In the present study, we used XTTF decoction to treat obesity-associated CRC in mice. The mice in the treatment group exhibited much better physical conditions and liver indexes. XTTF decoction significantly decreased the activity of IGF-1 and IGF-1R and obviously improved the IGFBP-3 activity. On the basis of these findings, we consider that XTTF decoction, which provides abundant nutriment and is an effective constituent to substances such as polysaccharides, probably inhibits hepatic metastasis and inhibits the process of CRC by downregulating IGF-1 and IGF-1R and upregulating IGFBP-3.
In summary, the results of this study demonstrate that obesity can stimulate tumor growth and hepatic metastasis, increase protein expression and mRNA levels of IGF-1 and IGF-1R, and decrease IGFBP-3 levels, all of which can be reversed by XTTF decoction. Thus, XTTF decoction is beneficial to bodily health and can effectively inhibit hepatic metastasis in CRC cancer, suggesting that XTTF decoction can serve as a potential treatment of CRC.
Declaration of conflicting interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Footnotes
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Nature Science Foundation of China “The role and mechanism of Xiaotan Sanjie Decoction on Cell migration and Invasion in Gastric Cancer based on CAFs proliferation and related factor expression in tumor microenvironment” (81603524) and the Traditional Chinese Medicine Scientific Research Funds of Shanghai Municipal Health Bureau “The Clinical Effects of ‘Dispersing Phlegm and Eliminating Stagnation’ Decoction used by Professor Pinkang Wei on patients with moderate and advanced gastric cancer” (2012L092B).
