Abstract
Hepatic portal venous gas is often referred to as the “sign of death” because it signifies a very poor prognosis if appropriate treatments are not promptly administered. The etiologies of hepatic portal venous gas are diverse and include severe complex abdominal infections, mesenteric ischemia, diving, and complications of endoscopic surgery, and the clinical manifestations are inconsistent among individual patients. Thus, whether emergency surgery should be performed remains controversial. In this report, we present three cases of hepatic portal venous gas. The patients initially exhibited symptoms consistent with severe shock of unknown etiology and were treated in the intensive care unit upon admission. We rapidly identified the cause of each individual patient’s condition and selected problem-directed intervention measures based on active organ support, antishock support, and anti-infection treatments. Two patients recovered and were discharged without sequelae, whereas one patient died of refractory infection and multiple organ failure. We hope that this report will serve as a valuable reference for decision-making when critical care physicians encounter similar patients.
Keywords
Introduction
Hepatic portal venous gas (HPVG) is an imaging feature associated with various diseases and was first reported by Wolfe and Evans in 1955. 1 Early reviews of HPVG reports indicated that HPVG was associated with a mortality rate of 75%. 2 However, with the popularization and advancement of radiological technology, the detection rate of HPVG has increased, even in patients with some nonemergency conditions.3–5 In a summary of reported clinical cases, Kinoshita et al. 6 stated that most cases involved ischemia or necrosis of the intestinal tract. Consequently, emergency surgery for HPVG has gradually attracted more attention, and a 2001 survey revealed a reduced overall mortality rate of 39%. 7 Nevertheless, the choice between conservative and surgical treatment of HPVG remains controversial8–10 because patients frequently present with severe infection and severe shock alongside abdominal discomfort, further complicating decision-making.
In our previous clinical work, we treated three patients in critical condition presenting with severe shock of unknown origin and signs of HPVG. The etiologies of their conditions were diving-induced decompression sickness (DCS), bowel necrosis secondary to chronic appendicitis, and sepsis after orthotopic liver transplantation, and their treatment results were satisfactory. In this report, we share our diagnostic and treatment experiences in the treatment of similar patients with the aim of providing a beneficial reference for clinicians. The reporting of this study conforms to the CARE guidelines. 11
Case report
Patient 1
A middle-aged male occupational diver developed lower limb weakness and bilateral tingling after a dive. Medical assistance was not sought until his symptoms worsened. On the second day after the onset of symptoms, the patient became confused and was transported to the emergency department of our hospital by ambulance. A whole-body computed tomography (CT) scan revealed no abnormal signs except for the presence of venous gas emboli in the liver (Figure 1). His blood pressure began to decrease, progressing to severe shock and necessitating a moderate dose of noradrenaline after active fluid infusion. The patient was subsequently transferred to the intensive care unit (ICU). He had a 10-year history of diabetes but had not been receiving regular treatment. We obtained the patient’s consent for treatment.
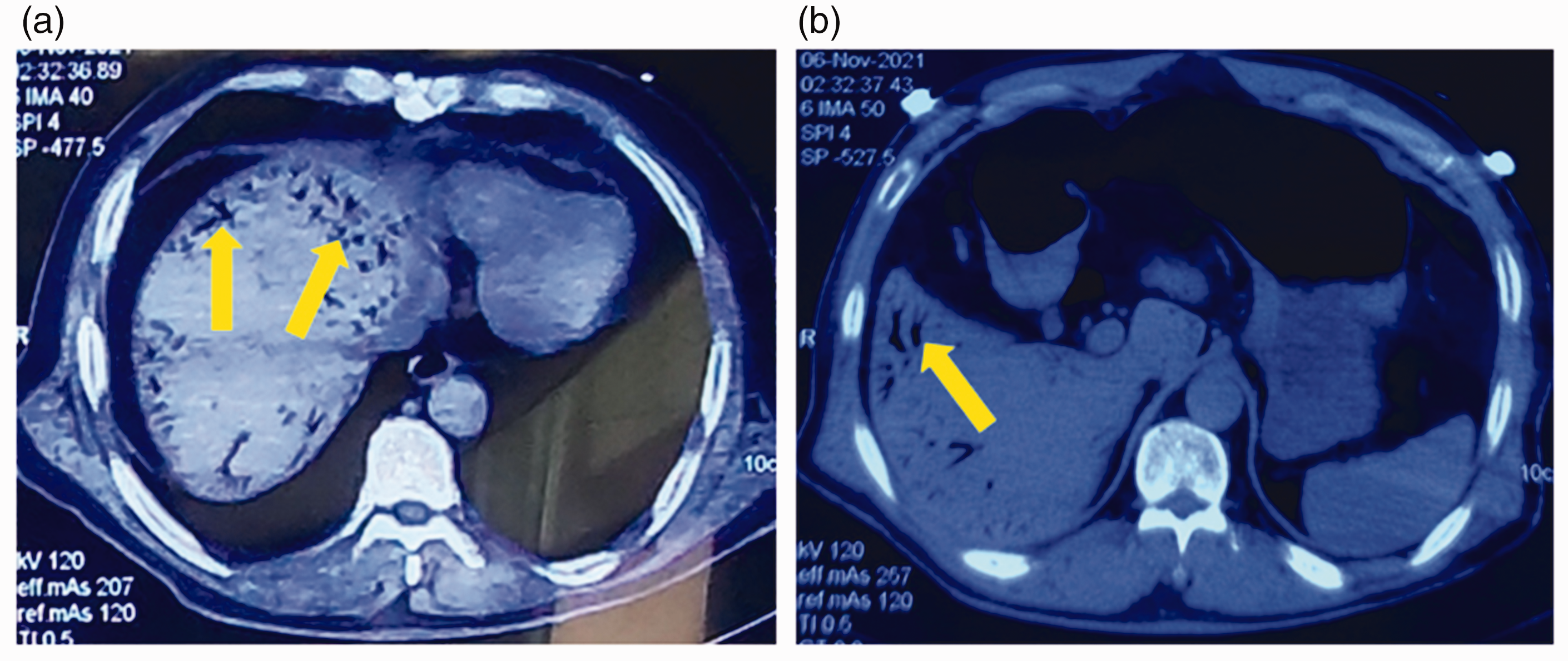
Computed tomography imaging manifestations of Patient 1 on admission. (a) Venous gas emboli in the liver (yellow arrow) and (b) gastric retention and gas in the bowel.
On physical examination, the patient was in a state of mild coma (Glasgow coma scale score of 8) and his body temperature was 39.5°C, respiratory rate was 24 breaths/minute, pulse rate was 100 beats/minute, and blood pressure was 107/79 mmHg while undergoing noradrenaline support at 0.5 μg/kg/minute. He was receiving oxygen support at 3 L/minute through a protective endotracheal tube, and his oxygen saturation (pulse oximetry) was 88%. Laboratory examination revealed the following: white blood cell count, 30.01 × 109/L; lymphocyte count, 2.95 × 109/L; hemoglobin level, 207 g/L; pH, 7.292; arterial oxygen saturation, 87.8%; standard bicarbonate, 18.9; lactic acid level, 4.6 mmol/L; procalcitonin (PCT) level, 15.02 ng/mL; C-reactive protein level, 114 mg/L; creatinine level, 119 µmol/L; creatine kinase level, 2138 U/L; activated partial thromboplastin time, 31.7 s; and D-dimer level, 4.1 g/L. Considering his history of diving, symptoms, and imaging findings, we strongly suspected DCS. In addition, he was concurrently diagnosed with septic shock, diabetic ketoacidosis, and rhabdomyolysis. However, we were unable to identify the source of infection or any specific evidence of pathogenic bacteria. Notably, we found no evidence of acute abdominal disease. According to the treatment principles for DCS, it was necessary to initiate hyperbaric oxygen (HBO2) therapy. However, his respiratory and circulatory failure worsened, requiring us to prioritize active life-support treatments such as ventilator support, empirical anti-infection treatment using imipenem, antishock support, precise fluid resuscitation, glycemic control using insulin, and diuresis over HBO2 therapy to stabilize his condition. On day 5, his blood pressure stabilized and his consciousness improved. Although he could not be completely weaned from the ventilator, we commenced HBO2 therapy following the specific scheme established by the U.S. Navy 12 because we determined that the potential benefits outweighed the associated risks. By day 10, his condition had significantly improved, and he no longer required organ support treatment. He was subsequently transferred to the HBO2 department for further treatment. On day 18, the patient was discharged without sequelae.
Patient 2
A man in his 20s was referred to our hospital because of recurring fever and persistent pain in his lower right abdomen. He had undergone appendicectomy for acute appendicitis at a local hospital 1 year previously. One week before his admission to our hospital, a CT examination of the lower abdomen was conducted when he visited another healthcare facility, revealing wall thickening and edema in the terminal ileum, cecum, and ascending colon. Additionally, it showed small areas with gas density around the cecum and a small amount of fluid collection in the pelvic cavity. Therefore, the patient was initially diagnosed with typhlitis and received antimicrobial treatment and other symptomatic interventions in the gastrointestinal surgery department. However, his symptoms were not alleviated, and his inflammatory responses, including excessive perspiration and polypnea, became increasingly severe. The day before his admission to our hospital, his body temperature spiked to 41°C and he developed bright red bloody stools, with an estimated total blood loss of approximately 800 mL. For more effective treatment, the patient was transferred to the ICU of our hospital. We obtained the patient’s consent for treatment.
Upon admission, the patient was conscious but in low spirits. He was able to cooperate with the physical examination. His vital signs were unstable, characterized mainly by a high heart rate of approximately 120 beats/minute and low blood pressure (mean arterial pressure of <65 mmHg) despite mild to moderate doses of vasoactive agents and active fluid infusion to maintain his blood pressure. Laboratory tests revealed the following: white blood cell count, 0.63 × 109/L; leukocyte count, 0.41 × 109/L; lymphocyte count, 0.20 × 109/L; platelet count 58 × 109/L; PCT level, 132.29 ng/mL; C-reactive protein level, 203.57 mg/L; alanine aminotransferase (ALT) level, 110 U/L; total bilirubin (TBIL) level, 65.2 μmol/L; albumin level, 25.8 g/L; gamma-glutamyl transpeptidase level, 134 U/L; D-dimer level, >20 µg/mL; and activated partial thromboplastin time, 53.7 s. An enhanced CT scan of the entire abdomen was performed to further clarify the diagnosis. The results showed multiple pneumatosis in the portal vein and its branches, wall necrosis in the small intestine and cecum, liver injury and cholecystitis, circumscribed peritonitis, and a small amount of fluid in the abdominal cavity (Figure 2). We immediately organized multidisciplinary consultations with experts in intensive care, gastrointestinal disease, hepatology, gastroenterology, and radiology. Following these consultations, gastrointestinal surgeons performed an emergency exploratory laparotomy at midnight after the patient’s admission. They identified wall torsion and angulation at the terminal ileum, located approximately 50 cm from the ileocecal junction, which had already undergone ischemia and necrosis (Figure 2). They performed an enterectomy of the affected intestinal segment. The following day, next-generation sequencing (NGS) revealed Klebsiella michiganensis in a blood specimen. The patient’s treatment regimen included meropenem (2 g every 8 hours) in combination with ornidazole (0.5 g every 12 hours) as part of the antimicrobial strategy. Other supportive treatments included anti-inflammatory interventions, immune regulation, liver protection, acid suppression, fluid resuscitation, circulation support, maintenance of homeostasis, and nutritional support. The patient’s blood pressure gradually stabilized, and he was weaned off vasoactive drug support. Infection-associated indices, including the white blood cell count, PCT level, and C-reactive protein level, began to return to normal levels, indicating control over septic shock. However, the patient continued to exhibit an intermittent fever, and his TBIL and ALT levels steadily increased, suggesting worsening acute liver injury. Therefore, based on the available treatments, we enhanced the liver protection measures, relieved the bile duct spasms, and managed the jaundice. On day 10, we conducted an abdominal CT re-examination and found that the pneumatosis in the portal vein and its branches had disappeared and that the liver injury had improved compared with the previous enhanced CT results. The TBIL level also began to decline. On day 12, with the infection indices fully under control, we initiated de-escalation therapy. Finally, on day 20, the patient had completely recovered and was discharged from the hospital.

Computed tomography imaging manifestations of Patient 2 on admission. (a) Small number of venous gas emboli in the liver (yellow arrow). (b) Wall necrosis affecting part of the small intestine and cecum, ascitic fluid, and gas in the bowel and (c) postoperative wall torsion and angulation at the terminal ileum located far from the ileocecal junction, with an approximately 50-cm portion affected by already-existing ischemia and necrosis.
Patient 3
A man in his 60s struggling with alcohol addiction presented to our hospital. An incidental physical examination conducted 8 months prior had revealed a hypoechoic nodule in his liver. Two days before his admission, he had begun experiencing upper abdominal pain. At a local hospital, a CT scan was performed to identify possible signs of a large malignant tumor coupled with fracture and hemorrhage in the liver. In addition, diagnostic paracentesis revealed that the bloody fluid collected from the abdomen did not clot. After approval by the organ transplantation ethics committee, the patient was transferred to our hospital while awaiting a suitable liver donor. We obtained patient consent to treatment. The day after his admission, total hepatectomy and orthotopic liver transplantation were successfully performed. However, because of the complex nature of the operation, prolonged anhepatic phase, substantial bleeding, low body temperature, metabolic acidosis, and severe coagulation dysfunction, the patient was transferred to the ICU immediately postoperatively. After extensive organ support, clotting substrate supplementation, blood transfusions, and hemostasis using recombinant factor VIIa and antifibrinolysis, his hemoglobin level gradually increased, and his clotting function improved by day 3. However, his blood pressure became unstable again after a short recovery time. Invasive hemodynamic monitoring revealed a low cardiac index and afterload, and his infection-associated indices began fluctuating, raising concerns about the possibility of septic shock. Therefore, we initiated a combined anti-infection therapeutic strategy that included the use of imipenem, polymyxin, daptomycin, and caspofungin. By day 7, the patient’s vital signs had improved without any circulatory or respiratory support. His consciousness gradually returned, and dynamic laboratory tests indicated that his TBIL and ALT levels were trending downward, suggesting that the donor liver was functioning as expected. Although no evidence of a specific etiology was obtained using traditional methods or NGS for pathogen detection, his PCT level remained high. On day 12, his condition was stable and the infection-associated indices appeared to have improved to some extent. We adjusted the anti-infection strategy by discontinuing the polymyxin and daptomycin, after which the endotracheal tube was extubated. However, on day 15, the patient’s white blood cell count markedly increased to 18.69 × 109/L, PCT to 5.53 ng/mL, TBIL to 77.5 μmol/L, ALT to 86 U/L, and cholinesterase to 2643 U/L and his blood ammonia level decreased to 9 µmol/L. Circulatory deterioration began, and we strongly suspected worsening of a secondary infection. Consequently, we escalated the use of empiric anti-infection drugs, reintroducing polymyxin after collecting blood and drainage fluid samples for NGS.
On day 16, the patient’s oxygen saturation (pulse oximetry) and mean arterial pressure decreased, necessitating tracheal reintubation. Laboratory tests revealed a continuous increase in the infection-associated indices and worsening of donor liver function, particularly excretion and detoxification. Bedside sonography revealed nonuniform echogenicity of the donor liver. On day 17, the patient developed multiple organ dysfunction involving the central nervous system, respiratory system, circulatory system, liver, kidneys, and coagulation system. In addition, his abdominal distension became more prominent, and a total abdominal CT scan showed pneumatosis at the portal vein along with multiple hypodense lesions distributed throughout the donor liver (Figure 3). Bacterial translocation from the intestinal flora was suspected as the primary cause of sepsis. Considering the patient’s unstable vital signs and risk of impending disseminated intravascular coagulation, the surgical experts deemed him ineligible for further surgery. Although the anti-infection strategy covered the known pathogenic bacteria, NGS revealed carbapenem-resistant K. pneumoniae. Despite these efforts, the patient’s condition continued to deteriorate, and his circulatory failure and refractory metabolic acidosis remained unresponsive to therapy. The patient died on day 18.
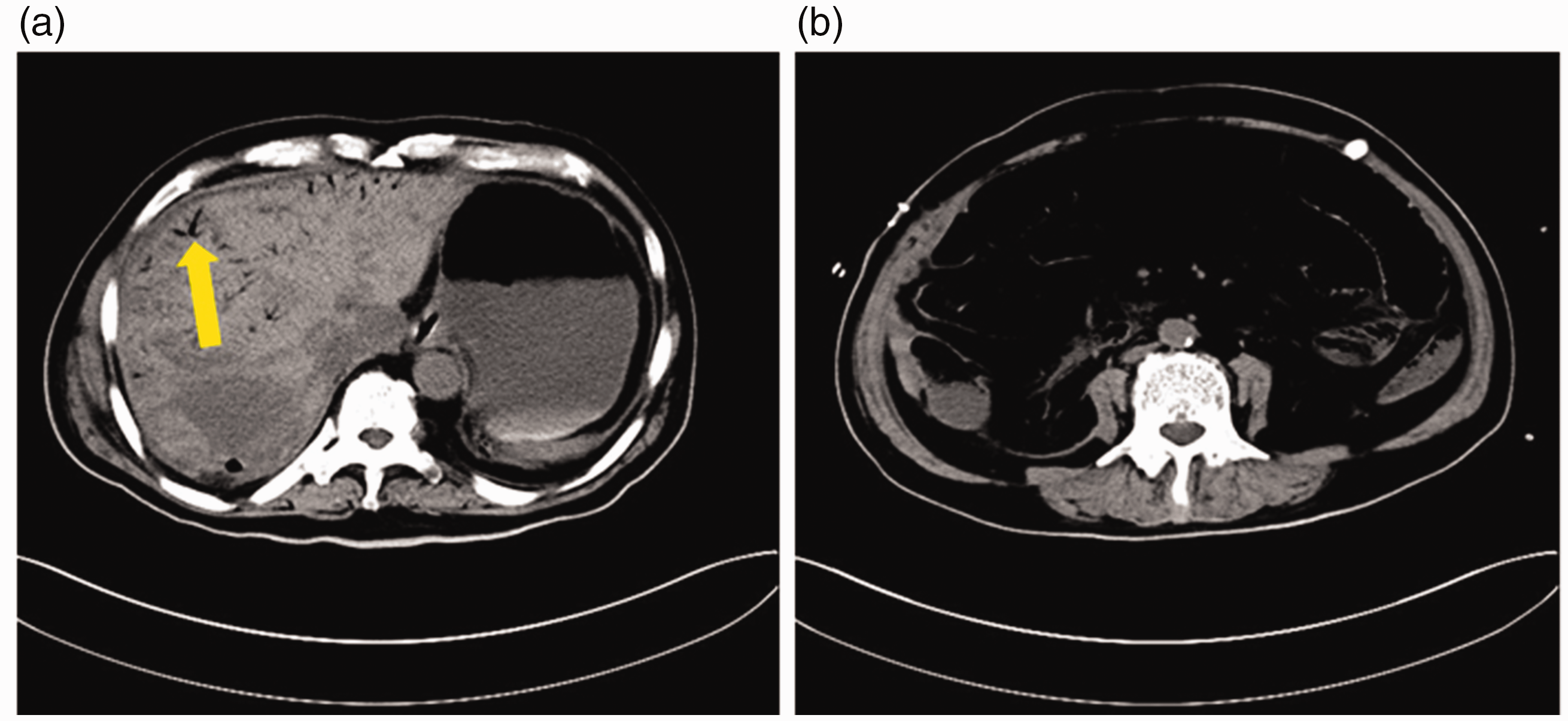
Computed tomography imaging manifestations of Patient 3 upon the change in his condition after liver transplantation. (a) Venous gas emboli (yellow arrow) and multiple low densities in the liver and (b) extensive gas accumulation in the bowel.
Discussion
HPVG is a rare imaging sign characterized by an abnormal accumulation of gas within the portal vein and its intrahepatic branches. 2 It can manifest secondary to various conditions, including digestive tract disease, intestinal obstructive disease, mesenteric vascular disease, anastomotic leakage, closed abdominal trauma, and liver transplantation.13,14 Because of its association with a poor prognosis, HPVG is commonly called “the sign of death.” 15 Emergency surgery can improve the prognosis, particularly in cases caused by intestinal ischemia, reducing the mortality rate to approximately 16.5%. 7 However, HPVG itself is not an absolute surgical indication; the decision to pursue surgery mainly depends on the specific etiology.16,17 Indeed, decision-making regarding HPVG is not easy if complicated by other unstable factors. In our previous study, we managed three patients in critical condition who had severe shock of unknown origin in combination with HPVG, and different etiologies were confirmed by subsequent examinations. No similar reports covering a series of critical cases of HPVG and severe shock resulting from three uncommon etiologies have been published to date. According to current studies, the pathogenesis of HPVG can be summarized as infection by aerogenic bacteria and the accumulation of additional gases from the bloodstream.8,18
HPVG often occurs when the barrier function in the gastrointestinal tract is destroyed, leading to bacterial translocation.19,20 This forms a reliable basis for understanding the pathophysiology of HPVG complicated by septic shock. In Patient 1, inert gas diffusion was the main cause of HPVG, which developed secondary to inappropriate decompression during diving. The patient also developed severe shock at an early stage, although we did not find the exact foci of infection, and there were no abnormal signs in the gastrointestinal tract on abdominal CT examination. Translocation of the intestinal flora was highly suspected on the premise of stimulation of DCS and underlying type 2 diabetes. After administering active organ support, anti-infection agents, and antishock therapies, delayed HBO2 therapy was administered and the prognosis was satisfactory. Patient 2 presented a classic case of HPVG that was mainly associated with chronic inflammation of the cecum originating from a residual abscess from a previous appendectomy. We adopted individualized therapy including intestinal resection, anti-infection treatment, and fluid resuscitation. Severe shock recurred shortly after the emergency surgery. Patient 3 had a massive liver tumor and underwent liver transplantation. However, his disease course and therapy were not smooth. After receiving active organ support and anti-infection therapy in the ICU, his condition temporarily improved, and the donated liver began to function. Owing to the influence of multiple factors, including immunosuppression, hypoperfusion, bacterial translocation, and a prolonged ICU stay, the infection was not fundamentally controlled and gradually progressed to refractory septic shock. The patient developed abdominal distension in the final stage, and features of HPVG were discovered on abdominal CT examination. Unfortunately, the patient was unable to undergo surgery. In these three patients, HPVG was accompanied by severe shock arising from three different causes, different problem-oriented treatment strategies based on the specific etiology were adopted, and positive outcomes were obtained in two of the patients.
The choice between emergency surgery and conservative treatment for patients with HPVG remains controversial. 21 The decision to perform surgery is largely influenced by a deeper understanding of the etiology of HPVG. Most cases are initially associated with intestinal ischemia secondary to various diseases.2,13 However, with the popularization of CT examination, the detection rate of HPVG has increased. Some nonischemic conditions, such as diving, accidental inhalation of inert gases, and complications from endoscopic surgery, can also trigger HPVG. Hence, some physicians have advocated nonsurgical treatment.22,23 With the accumulation of clinical cases and experience, increasingly more studies are being performed, including case–control studies and reports based on large-sample databases. 24 For example, Liu et al. 25 analyzed the characteristics of 131 patients with HPVG using the PubMed and Medline databases in 2020 and found significant differences between patients treated surgically and those managed conservatively, highlighting the necessity of surgery. Capolupo et al. 26 proposed that surgical treatment is the preferred option for clinically unstable patients or those with evidence of peritonitis or bowel perforation. For stable patients, nonoperative management should be considered. Notably, the definition of “stable disease” is not clear, which is a distinct limitation. Identifying the etiology is vital for decision-making. When a patient’s condition is critical and the cause is unknown or there is clear evidence of intestinal ischemia, active exploratory laparotomy should be prioritized. In this study, all patients presented with severe shock in the early stages of HPVG, and we adopted an active and effective antishock strategy to identify the specific etiology. Decision-making regarding surgery was guided by multidisciplinary consultations, with critical care physicians playing a predominant role.
Conclusions
We have herein described three critical cases of HPVG that initially manifested as severe shock in the early stages. To maintain these patients’ conditions, we implemented active anti-infection, antishock, and organ support treatments. We considered various possible etiologies, including diving, chronic appendicitis, and sepsis, which eventually led to liver transplantation in one of the patients. Two patients fully recovered, and one died because of an infection that could not be corrected. We trust that this report will serve as a valuable reference for decision-making in the management of patients with HPVG concurrent with severe septic shock.
Footnotes
Author contributions
Anwei Liu and Jiao Shen acquired the data, managed the patient, and drafted the manuscript.
Liansheng Long managed the patient and performed the surgical treatment.
Xuezhi Shi managed the patient.
Qiang Wen provided technical guidance.
Zhiguo Pan conceived and designed the study.
All authors critically revised the manuscript, approved the final version to be published, and agree to be accountable for all aspects of the work.
Data availability statement
The data that support the findings of this study are available from the corresponding author, Zhiguo Pan, upon reasonable request.
Declaration of conflicting interests
The authors declare that there are no conflicts of interest.
Ethics statement
Because this was a retrospective study involving anonymized data, the requirement for ethics approval was waived.
Informed consent for publication
All patients provided verbal consent for publication of the report.
Funding
This work was supported by the Medical Scientific Research Foundation of Guangdong Province (No. A2023255), the Science and Technology Program of Guangzhou City (No. 2024A04J4557), and the National Natural Science Foundation of China (No. 81873116).
