Abstract
Pneumatosis of the portal vein is considered a rare imaging sign rather than a disease. It usually occurs in patients with digestive tract diseases such as intestinal obstructive diseases, mesenteric vascular diseases, closed abdominal trauma, and liver transplantation. Because of its high mortality rate, it is also termed the “sign of death.” Hawthorn contains tannic acid, and seafood is rich in calcium, iron, carbon, iodine, and other minerals and proteins. Thus, consuming both hawthorn and seafood together can result in the formation of an indigestible complex in the body, acting as the main pathogenic factor in patients with intestinal obstruction. We herein describe a patient with duodenal obstruction caused by hawthorn who developed the hepatic portal venous gas sign and was cured by nonsurgical treatment.
Keywords
Introduction
Portal venous pneumatosis is a rare imaging sign caused primarily by abnormal accumulation of gas in the portal vein and its intrahepatic branches, and it may develop from several different causes. In 1955, Wolfe and Evans 1 first reported hepatic portal venous gas (HPVG) in newborns, and in 1960, Susman and Senturia 2 first reported HPVG in an adult. The mortality rate in the early stage can be as high as 75%; hence, HPVG is also termed the “sign of death.” It is not an independent disease but is instead often considered a symptom of various digestive system diseases. Intestinal obstructive disease, mesenteric vascular disease, closed abdominal trauma, liver transplantation, and other diseases can effectively lead to this imaging sign. We herein describe a patient with duodenal obstruction who developed the HPVG sign and was cured by nonsurgical treatment. Nonsurgical treatment in such cases reduces the high risk associated with surgical treatment, improves long-term outcomes after treatment, reduces the cost of treatment for this disease, and increases patient compliance. The reporting of this study conforms to the CARE guidelines. 3
Case presentation
A man in his early 80s had a history of gastroesophageal reflux, gastritis, old myocardial infarction, diabetes, and prostate cancer. He presented with a 3-day history of intermittent abdominal distension, abdominal pain, nausea, and vomiting. These symptoms had not been relieved by administration of oral epprazole, Armillaria oral liquid, or pancreatin enteric-coated capsules (which had not been prescribed by local doctors, and the usage and dosage of these drugs were unknown). Therefore, the patient was admitted to our hospital for diagnosis and treatment.
Initially, the patient and his family requested an endoscopic examination. Because of the patient’s previous heart attack, however, performing endoscopy under anesthesia was high-risk. Based on the patient’s current symptoms and signs as well as his history, we considered the possibility of ileus, but we did not exclude other diseases. Because he was not suitable for gastroenteroscopy, we prioritized abdominal computed tomography (CT) to not only confirm the disease and exclude most other digestive diseases but also provide guidance for further treatment. The results of abdominal CT (without intravenous or oral contrast) indicated gas accumulation in the portal vein, splenic vein, and superior mesenteric vein (Figure 1). Obstruction was also observed at the flexor ligament of the duodenum (Figure 2). Therefore, the patient was admitted to our hospital.

The patient’s first abdominal CT examination (without intravenous/oral contrast) revealed gas accumulation in the portal vein, splenic vein, and superior mesenteric vein.
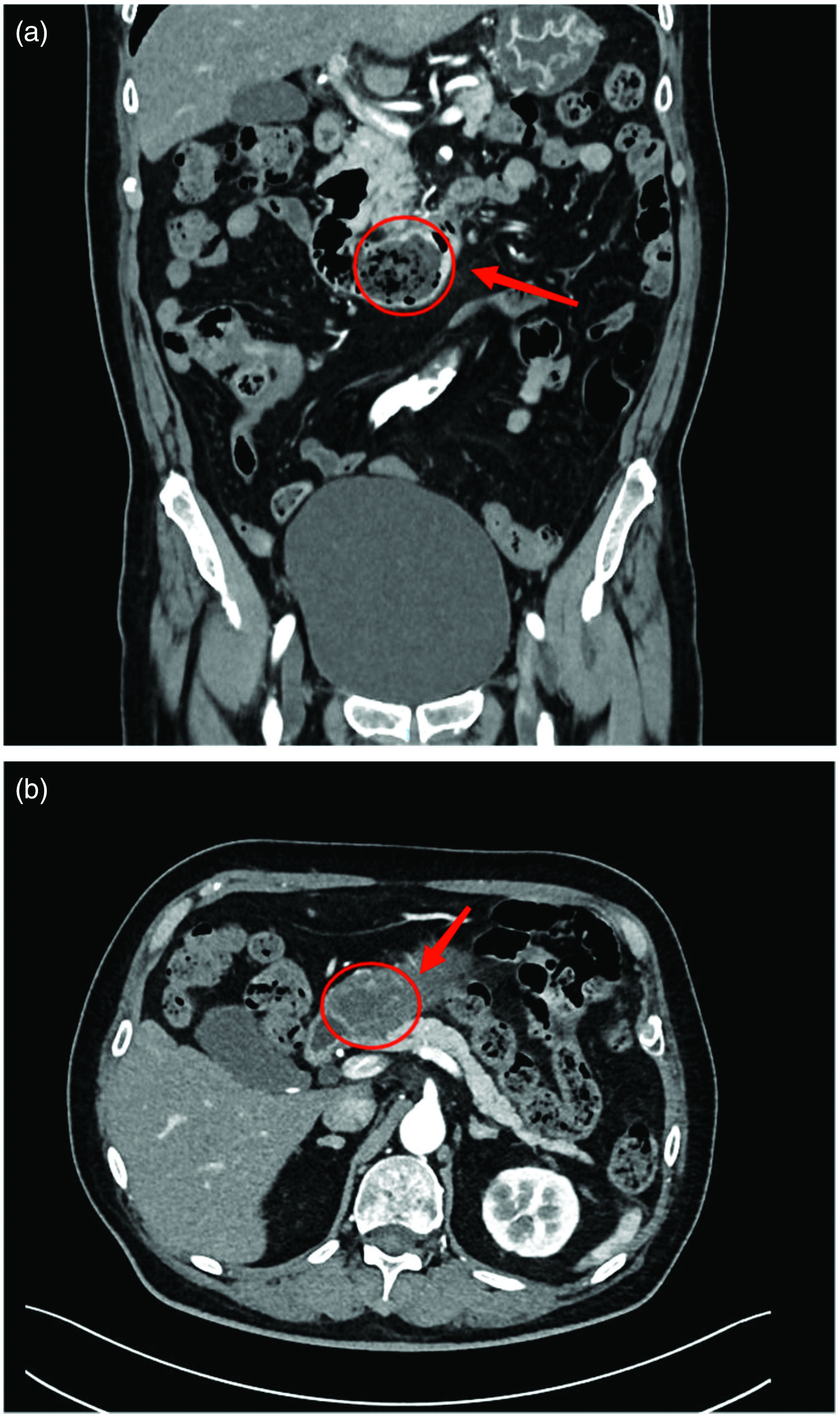
(a) Sagittal view of abdominal CT (without intravenous/oral contrast): obstruction can be observed at the flexor ligament of the duodenum (arrow) and (b) Cross section of abdominal CT (without intravenous/oral contrast): obstruction with associated edema can be seen in the duodenum (arrow).
On the first day, physical examination showed no signs of previous abdominal surgery or scars; a full, slightly raised abdomen with no abnormalities in the shape of the gastric or intestinal regions and no peristaltic wave; and no abdominal wall varices. No tenderness or rebound pain was noted throughout the whole abdomen. Percussion of the whole abdomen produced a drum sound, and no shifting dullness was noted. In addition, no obvious abnormality was found in the rest of the physical examination. At the time of admission, the patient’s temperature was 36.2°C (no fever), blood pressure was 121/73 mmHg, and heart rate was 74 beats/minute. The patient’s laboratory and physical examination results are shown in Table 1. Blood cultures with both anaerobic and aerobic identification were negative, and the troponin, amino-terminal B-type brain natriuretic peptide, and neutrophil apolipoprotein concentrations were not significantly abnormal. Conservative treatment was prescribed, and the patient was immediately treated with meropenem to prevent infection. The patient was also treated by fasting (water), anti-inflammatory therapy, acid suppression, rehydration, and nutritional support according to his symptoms.
Physical and laboratory examination results.

Reference range of each laboratory finding is given in parentheses.
The next day, the patient underwent an urgent high-throughput test for the various infectious pathogens, and the test results were negative. Gastroscopy (microscopic examination) was then carried out, which revealed that the esophagus exhibited flaky erosion and that yellow-green turbid gastric juice was present in the stomach. The mucosa of the gastric fundus and body was smooth. The mucosa of the gastric horn and antrum showed scattered patchy hyperemia, local edema, and attached viscous secretion. The pylorus was smooth and unobstructed. No abnormalities were found at the bulb and posterior part of the duodenum. To substantially reduce the pressure in the intestinal cavity, we implanted an endoscopic intestinal obstruction catheter (the decompression effect of which is superior to that of conventional short tubes). The intestinal obstruction catheter was inserted through the nose, and the distal end of the descending duodenum was placed inside with the assistance of endoscopy. Abdominal CT (with intravenous/oral contrast) was performed, which revealed that the intrahepatic and intravenous gas accumulation had disappeared and that no abnormal enhancement shadow was present (Figure 3).

Full abdominal CT re-examination revealed that both the intrahepatic and intravenous gas accumulation had disappeared, and no abnormal enhancement shadow was found on the enhanced scan.
On the fourth day, the intestinal obstruction catheter was rechecked. A standing abdominal plain film showed that a liquid–gas plane was present at the head of the left middle and upper abdominal intestinal obstruction catheter, the local intestinal cavity was filled with gas, the intestinal cavity was slightly expanded, and the mucosa was widened. Injection of contrast agent through the intestinal obstruction catheter revealed anti-peristaltic flow of most of the contrast agent along the intestinal duct where the intestinal obstruction catheter was located; additionally, the mucosa was annular, the intestinal cavity was slightly expanded, and the mucosa was widened. The head of the ileus catheter was located at the beginning of the jejunum and was peristaltic. Films were taken at 60 and 240 minutes. In the left abdomen, two to three groups of small intestine and a local small intestinal intraluminal oval-shaped filling defect were observed. The distal contrast agent passed easily through the barrier with no leakage from the intestine into the abdominal cavity, and the distal intestinal cavity was clearly depicted by the contrast agent (Figure 4). Based on the gastrointestinal contrast findings, we considered the presence of duodenal obstruction descending into the jejunum in this patient. On the seventh day of admission, the symptoms of pain, vomiting, distension, and inability to defecate and flatulate disappeared, and the intestinal obstruction was relieved.

Digestive tract radiography showed an oval-shaped filling defect in the left abdomen along with two to three groups of small intestine. Obstruction of the passage of contrast agent occurred at the distal end with no leakage from the intestine into the abdominal cavity, and the distal intestinal cavity was clearly depicted by the contrast agent. The duodenal obstruction was considered to have possibly descended to the jejunum.
Discussion
HPVG is an extremely rare imaging sign. The gas can enter the portal vein system for various reasons, and the duration of this sign is relatively short. It is not an independent disease but is usually accompanied by digestive tract diseases such as intestinal obstructive disease, mesenteric vascular disease, 4 gastric ulcers5,6 gastric cancer, 7 peptic ulcers, 4 endoscopic and radiation surgery.8–13 Previous reports have indicated that HPVG is often associated with severe intestinal diseases and has a mortality rate of up to 75%. 4 It is also often considered an indication for emergency abdominal surgery and has been termed the “sign of death.” As a result of the enormous progress and development in the field of medicine, Kinoshita et al. 4 reviewed the previous case literature in 2001. Their research and analyses showed that the total mortality rate of HPVG was 39%, but when accompanied by severe basic diseases such as intestinal necrosis, the mortality of HPVG could reach 75%. 4 The mechanisms underlying the development of HPVG remain unclear, and the treatment of the disease needs to be further explored.
Although the pathogenesis of HPVG is still unclear, two main theories have been proposed: the bacterial theory and the mechanical theory. The bacterial theory states that various factors can lead to the breeding of large numbers of intestinal bacteria, which produce a large amount of gas and damage the intestinal mucosal barrier. The gas produced by the bacteria can effectively enter the vascular system to form HPVG. 14 The mechanical theory states that the destruction of the endothelial barrier between the mesenteric vein and the intraluminal gas that accumulates by several factors can cause extensive damage to the mucosa and increase the intestinal pressure. This leads to destruction of the intestinal mucosa and entrance of the intestinal gas into the vascular system to form HPVG. 6 We also found that 86% of patients with HPVG had a positive blood culture. Therefore, blood culture examination of patients with HPVG is of great importance. 15 Interestingly, some prior studies have also revealed that because carbon dioxide accounts for a relatively high proportion of gas components in the portal vein system, this soluble gas can continue to dissolve and disappear in the vascular system even after the different factors leading to the gas production have been reduced or resolved.6,16,17
The clinical characteristics of patients with HPVG are usually determined by the underlying cause of this imaging sign, and no significant difference has been found between symptoms in male and female patients. Patients with mild symptoms may develop nausea, vomiting, abdominal pain, diarrhea, abdominal distension, and decreased bowel sounds. By contrast, patients with severe symptoms may develop intestinal septicemia, bloody stool, acidosis, shock, and other associated symptoms. However, some patients have no obvious clinical symptoms. 15
It has been established that imaging diagnosis of HPVG is usually accomplished by X-ray photography, ultrasound, or CT. However, the centripetal flow of bile prevents the gas from migrating outward; thus, the gas can exist in the center of the liver, more than 2 cm from the liver envelope, and it is usually considered to be biliary gas. By contrast, the gas in the portal vein within the liver can be transported to small venous branches through centrifugal flow. Therefore, on abdominal CT and X-ray examinations, the presence of a branched transparent substance within 2 cm of the liver capsule can be used to diagnose HPVG.18,19 In 1978, HPVG was first detected on a routine plain abdominal film. 20 However, this diagnostic method has certain major drawbacks. HPVG can only be diagnosed when a large amount of gas has accumulated in the vascular system. Therefore, after the diagnosis, the patient’s condition can be profoundly serious, with a high risk of mortality and a poor prognosis. Schulze et al. 21 conducted a prospective study to compare the potential sensitivity of conventional radiography, CT, color Doppler flow imaging, and ultrasound in detecting gas in the hepatic venous system. The results showed that both CT and color Doppler flow imaging had significantly higher sensitivity than the other diagnostic methods. Overall, CT is currently considered the best diagnostic method for diagnosing HPVG, and it is also regarded as having the highest sensitivity for HPVG.21,22
In the early years after its discovery, HPVG was generally regarded as an indication for surgery. However, Yoo et al. 23 studied the treatment of 18 patients with HPVG and showed that surgical treatment was not the treatment of choice. The best treatment method and the prognosis of HPVG are primarily determined by the clinical manifestations, imaging, laboratory evidence, and potential pathologic causes.6,24,25 Gastric emphysema, 26 chronic intestinal pseudo-obstruction, 27 intestinal obstruction, gastric ulceration, and complications of endoscopic surgery can all be conservatively treated to achieve a good prognosis. Therefore, for patients without clear surgical indications and with extremely high surgical risk, conservative treatment is undoubtedly the best choice. It can improve both patient acceptance and the long-term prognosis.
In this case, we diagnosed the patient’s underlying condition by collecting a detailed medical history and performing relevant imaging examinations. The symptoms of intestinal obstruction associated with HPVG may include abdominal pain, abdominal distension, nausea, and vomiting. Through further analysis, we considered that the cause of the HPVG in this patient could be attributed to the mechanical theory. The patient’s medical history was again recorded after admission. We found that the patient had eaten crab and hawthorn 10 days before the onset of the disease. Research and clinical experience have established that hawthorn contains tannic acid and that seafood is rich in calcium, iron, carbon, iodine, and other minerals as well as proteins. Additionally, tannic acid contains a large number of hydroxyl and carboxyl groups, allowing it to react with substances such as proteins to produce poorly soluble compounds. 28 Therefore, when hawthorn and crab are consumed together, they can form an indigestible complex comprising tannin protein, calcium tannate, and other substances, resulting in the development of intestinal obstruction. This obstruction can then increase the pressure in the intestinal cavity and destroy the intestinal barrier. Gas can enter the vascular system through the damaged mucosa, thus forming the HPVG sign. In the present case, we administered meropenem to prevent infection and provided conservative treatment such as fasting (water), anti-inflammatory therapy, acid suppression, rehydration, and nutritional support according to the patient’s symptoms. The HPVG sign finally disappeared and the intestinal obstruction was relieved along with all symptoms and signs. The long-term prognosis was thus improved.
Conclusion
HPVG usually occurs in combination with various digestive tract diseases, such as gastric ulcers, intestinal obstruction, and gastric cancer, among others. It is not always fatal, and surgical treatment is not the only the treatment option available. Its prognosis and treatment mainly depend on the diagnosis of the primary disease. Compared with X-ray photography and ultrasound, abdominal CT has emerged as the best imaging method for diagnosing HPVG. It plays an irreplaceable role in achieving a clear diagnosis, reducing mortality, and significantly improving the prognosis.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605231180540 - Supplemental material for Bezoar as a cause of portal vein pneumatosis: a case report
Supplemental material, sj-pdf-1-imr-10.1177_03000605231180540 for Bezoar as a cause of portal vein pneumatosis: a case report by Qing Zhang, Heyu Meng, Yanqiu Chen and Fanbo Meng: on behalf of the Hellenic Headache Society in Journal of International Medical Research
Supplemental Material
sj-pdf-2-imr-10.1177_03000605231180540 - Supplemental material for Bezoar as a cause of portal vein pneumatosis: a case report
Supplemental material, sj-pdf-2-imr-10.1177_03000605231180540 for Bezoar as a cause of portal vein pneumatosis: a case report by Qing Zhang, Heyu Meng, Yanqiu Chen and Fanbo Meng: on behalf of the Hellenic Headache Society in Journal of International Medical Research
Footnotes
Data availability statement
All data supporting the conclusions of this article are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Ethics statement
Written informed consent was provided by the patient’s family. All details have been de-identified such that the identity of the patient may not be ascertained in any way. The requirement for review board approval was waived because of the nature of this study (case report).
Funding
This research received no external funding.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
