Abstract
Smooth muscle tumors are benign tumors originating from smooth muscle tissues, mostly occurring in the uterus and digestive tract; however, smooth muscle tumors occurring in the perianal sphincter are very rare. A 48-year-old woman presented with perianal swelling and pain and was misdiagnosed with a perianal abscess after examination at a foreign hospital. Upon further examination, the patient was diagnosed with a perianal smooth muscle tumor, with a size of approximately 6.5 × 5.5 × 4 cm. We succeeded in surgically removing the tumor. The patient was followed up for 2 years after the operation, and her anus functioned well without recurrence. Perianal abscess and perianal smooth muscle tumors are difficult to differentiate in the clinic and need to be identified via imaging. In clinical practice, smooth muscle tumors should be differentiated from perianal abscess and diseases such as rectal mesenchymal tumors and smooth muscle sarcomas.
Introduction
Smooth muscle tumors (SMT) are benign growths originating from smooth muscle tissues, accounting for 3.8% of all benign soft tissue tumors, 1 and are prevalent in the uterus and digestive tract. 2 Overall, 7% of all SMT are located in the gastrointestinal tract, with the stomach and small intestine being the main organs involved; these tumors rarely occur in the rectum and even more rarely in the perianal area, accounting for approximately 3% of all SMT of the gastrointestinal tract. 3 Ezinger classified SMT into the following categories: superficial SMT, vascular SMT, and deep SMT. The morphology of deep soft tissue SMT is characterized by well-defined and fibrous pseudo-coated membranes with mucous vitreous degeneration and degenerative changes, 4 which is consistent with the features of the patient in this case. Due to the lack of specific manifestations, it is often easy to miss or misdiagnose this tumor. 2 SMT in the perianal area need to be differentiated from perianal abscess (PA) and diseases such as rectal mesenchymal tumors and smooth muscle sarcomas (SMS).
Case report
A woman in her late 40s presented with initial symptoms of perianal distension and pain and had no fever, blood in the stool, or other symptoms following perianal ultrasound at a foreign hospital, which suggested PA formation. The laboratory parameters were as follows: leukocyte count, 14.31 × 109/L; neutrophil count, 10.35 × 109/L; and C-reactive protein level, 44.57 mg/L. PA was diagnosed in the hospital, and surgical treatment was performed. No pus was observed during intraoperative probing, but a perianal tumor was identified, making the diagnosis unclear. Therefore, the surgery was terminated. The patient was admitted to Weihaiwei People’s Hospital in late 2020. Magnetic resonance imaging (MRI) and enhanced computed tomography (CT) showed a tumor in the sciatic rectal fossa, moderate heterogeneous enhancement with regular margins, and extensive necrosis in the center. Moreover, the tumor was poorly disintegrated from the bowel wall at the deep level of the external sphincter, extending and pushing away from the external sphincter into the sciorectal fossa. The possibility of a rectal mesenchymal tumor or neurogenic tumor was not excluded based on the images (Figure 1). To clarify the nature of the tumor and provide guidance for subsequent diagnosis and treatment, a puncture was performed under ultrasound guidance on the right side of the anal canal using a puncture needle, and a portion of the tumor tissue was removed for biopsy (Figure 2). Based on pathology, the combination of immunohistochemistry showed an origin in the smooth muscle (Figure 3).

(a) Coronal T2W1 sequence showing poor disintegration of the bowel wall at the level of the deeper part of the tumor with the external sphincter, extending and pushing away the external sphincter to the sciorectal fossa and (b) Sag T2W1 sequence showing strong tumor–perianal sphincter association.

Ultrasound-guided puncture biopsy.

Pathology results. The combination of immunohistochemistry showed an origin in the smooth muscle.
After preoperative preparation, we made a paranal curved incision and found that the tumor was located in the sciatic-rectal fossa, the upper edge of the tumor was adherent to the puborectalis muscle, the lower edge was adherent to the internal anal sphincter, and the root of the tumor was in close proximity to the deeper part of the external sphincter muscle and the rectal wall, with a rich blood supply (Figure 4). Following blunt dissection, the tumor was completely resected, the pelvic floor was reconstructed, and the puborectalis muscle and internal and external sphincters were repaired. The size of the tumor was approximately 6.5 × 5.5 × 4 cm (Figure 5), and postoperative pathology suggested the presence of an SMT with mucinous degeneration. Immunohistochemical analysis revealed SMA (+), Desmin (+), CD-34 (+), CD117 (−), Dog-1 (−), S-100 (−), and Ki67 < 2%+ (Figure 6). The patient recovered well postoperatively, had good anal function, and showed no signs of recurrence at the 2-year postoperative follow-up. The reporting of this study conforms to the Case Report (CARE) guidelines. 5 This case has been authorized for publication by the patient.

Photograph of the tumor during surgery.
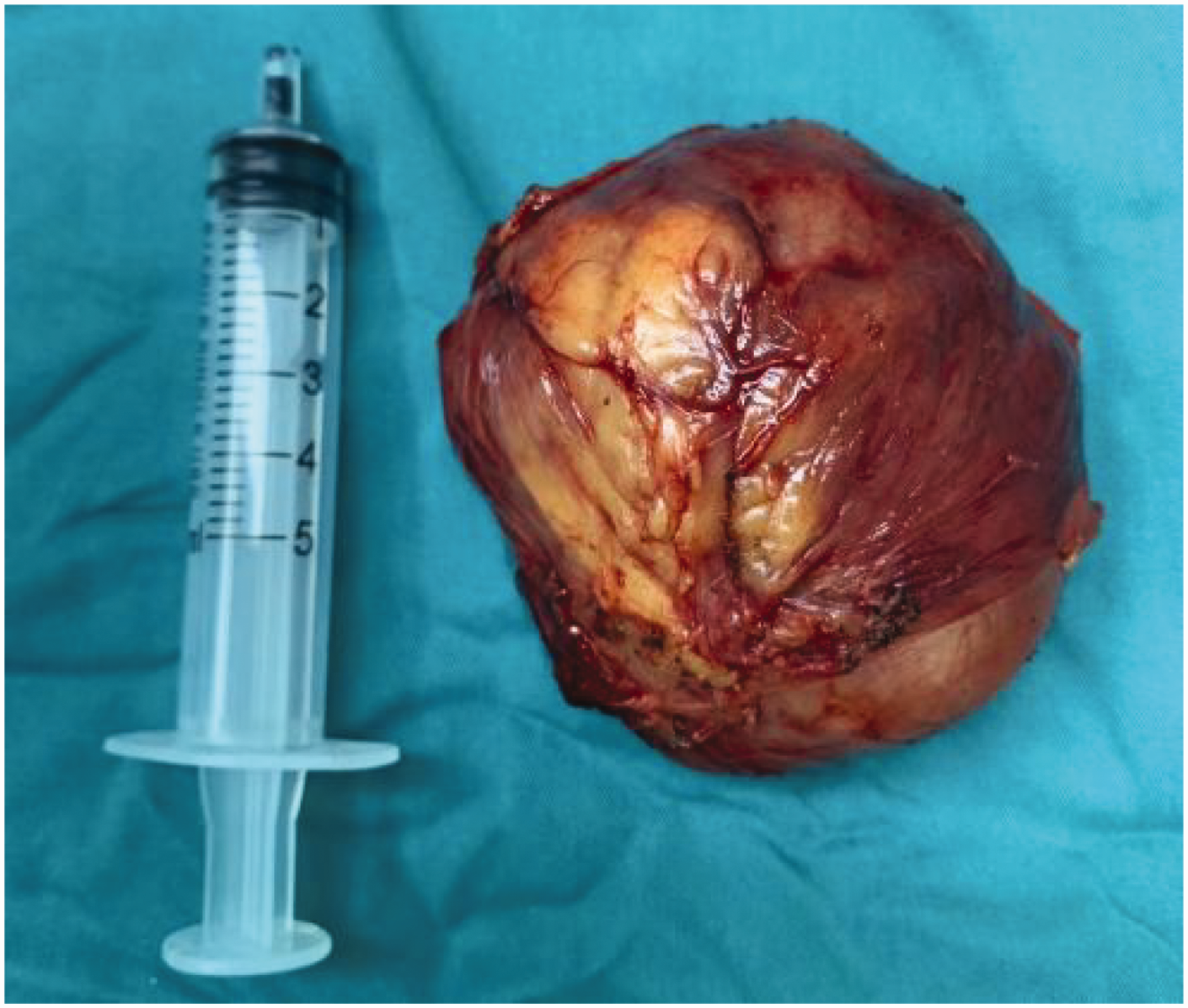
Tumor removal.
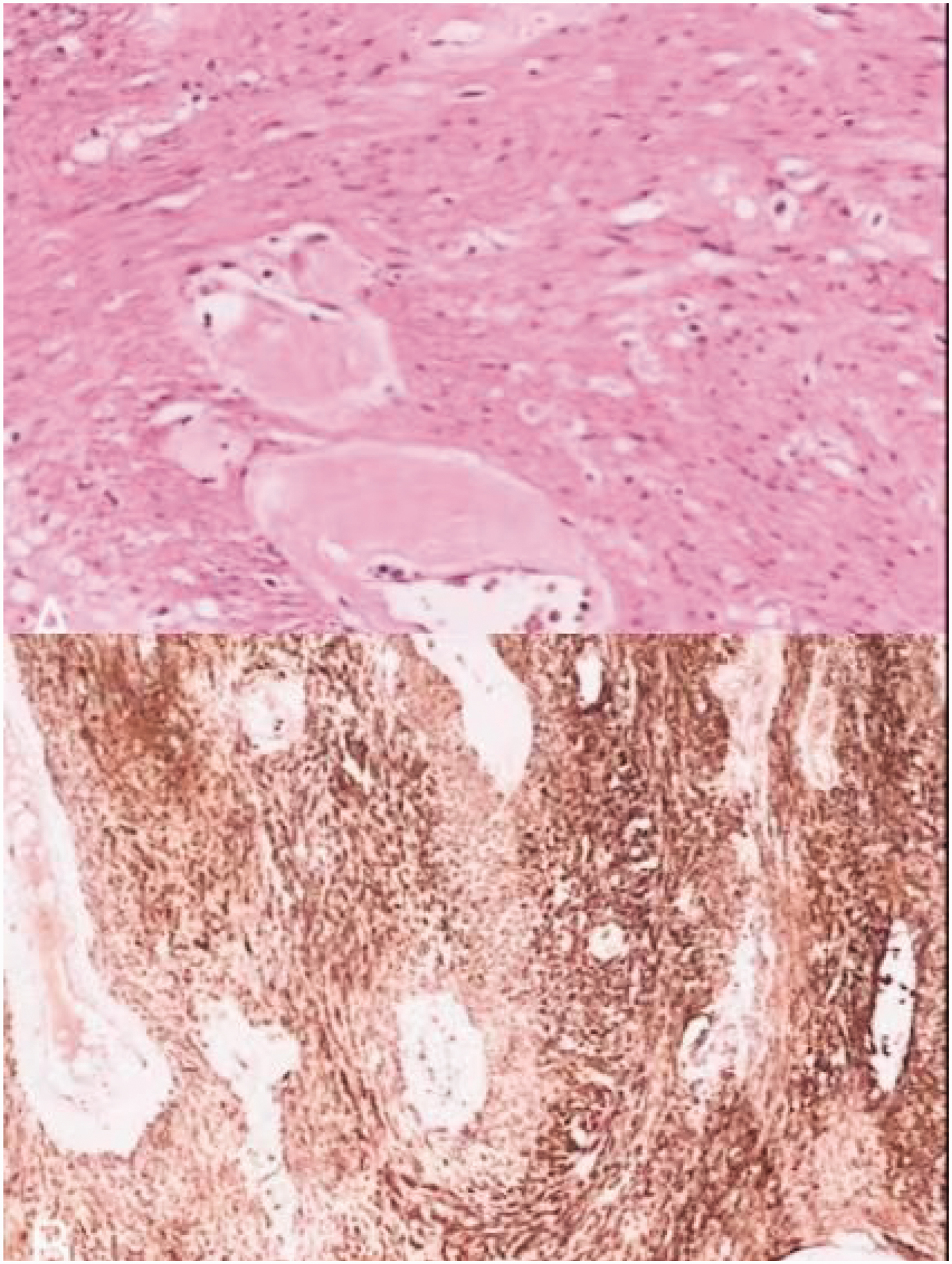
Postoperative pathology.
Discussion
SMT, which were first described by Virchow in 1854 as benign tumors, 6 originate from smooth muscle fibers and are found in the uterus and digestive tract; the main organs involved in the digestive tract are the stomach and small intestine. However, the rectum may be involved in rare cases and the perianal area in even fewer cases, accounting for approximately 1/2000 of benign and malignant tumors of the rectum. 7 These tumors have been previously classified as gastrointestinal mesenchymal neoplasms and are currently classified separately based on their own specific immunohistochemical pattern. From a clinical and pathologic viewpoint, Ezinger classified SMT into the following categories: superficial SMT, vascular SMT, and deep SMT. The morphology of deep soft tissue SMT is characterized by well-defined and fibrous pseudo-coated membranes with mucous vitreous degeneration and degenerative changes, 4 which is also consistent with the features of this case.
SMT in the rectal and perianal regions have different growth patterns, namely, intraluminal, intramural, and extraluminal. Most SMT in the rectum grow intraluminally, and those in the perianal region tend to grow extraluminally due to their origins in the mucosal muscularis mucosae of the rectum, intrinsic muscularis mucosae of the intestinal wall, or internal sphincter of the rectal muscle, which is a continuation of the rectal muscle. 1 Thus, these tumors often present with perianal pain and discomfort and a falling sensation and may be characterized by the presence of a palpable, homogeneous, solid mass. The patient in this case also had a solid perianal mass. In this case, the patient experienced perianal distension as the first symptom, which was misdiagnosed as a PA at the first visit and was diagnosed as an SMT only after gradually improving auxiliary examinations.
In clinical work, the diagnosis of SMT should be differentiated from diseases such as gastrointestinal mesenchymal tumors and SMS, including PA and many mesenchymal tumors. PA is a common morbidity characterized by acute onset and severe pain that is often accompanied with chills and fever. 2 When the local tension of the abscess reaches a certain degree, the skin may rupture and pus may overflow. There is a difference between SMT and PA in terms of response to treatment. Perianal SMT have a short postoperative recovery period, exhibit low recurrence rate, and require a radical surgical procedure with few complications. 8 For PA, attention should be paid to acute-phase drainage and infection control, with rapid symptomatic relief after treatment. However, there is a high risk of recurrence and formation of anal fistulas, indicating the need for long-term follow-ups. 9 Although the patient in this case did not have a fever or other discomfort in the early stage, the white blood cell count, neutrophil percentage, and C-reactive protein levels were elevated; thus, the patient was suspected of having a PA. In particular, for deep abscesses located in the deep posterior anal canal space or the isciorectal fossa, the clinical manifestations are similar to those of perianal SMT, such as the appearance of the skin without obvious abnormalities. However, on physical examination, buttock/perineum distension and pain, pressure pain on palpation, or no obvious symptoms may be noted. 10 In this case, the patient with SMT in the perianal region experienced initial symptoms of perianal distension and pain, and there were no other systemic symptoms. There was no abnormality upon visual diagnosis, and finger palpation reached the right side of the intra-anal stromal ligamentous tissue. The first symptom of perianal SMT was swelling and pain in the perianal area without other systemic symptoms. There was no abnormality upon visual examination, and the right side of the tough tissue in the anus could be detected upon palpation, accompanied with tenderness and pain. Interestingly, perianal SMT can also be associated with perianal infectious diseases. Dagmura et al. reported the case of a patient who presented with recurrent perianal purulent overflow; upon further examination, a fistula was observed passing through the collateral sphincter as an SMT. 11 PA and perianal SMT are difficult to differentiate in the clinic and need to be identified via imaging. On MR or CT, perianal SMT have clear, regular borders and show uniform enhancement on weighted images. 12 Since the tissue surrounding PA is often accompanied with edema, MRI shows blurred borders and uneven enhancement density. If clinicians can overcome the challenges associated with the diagnosis and treatment of the disease and can carefully analyze the clinical manifestations and examination results, the misdiagnosis rate can be greatly reduced.
SMS is a malignant tumor with abnormal cell morphology, no clear envelope, and the possibility of distant metastasis. Benign SMS usually present as well-defined, slow-growing, painless masses, whereas malignant SMS are fast-growing, may be associated with pain or ulceration, and have invasive and metastatic potential. Differentiation between the two usually relies on histopathologic examination, including features such as cellular anisotropy, nuclear division index, and necrosis.13,14 In this case, from the early perianal ultrasound examination to the late MRI and enhanced CT, the disease could be identified from a morphological viewpoint, and the possibility of SMS could be excluded. Although SMT are benign, data reported by Vorobyov et al. demonstrated that recurrent SMT are capable of transforming to SMS after multiple surgeries. 15 Most studies have shown that the risk of malignant transformation in typical SMT is <1%, but the risk increases in recurrent cases (approximately 2%–5%). 16 Recurrence and malignant regression of tumors appear to be associated with incomplete resection or tumors with a size >5 cm. 12 Adequate and complete resection of the tumor is the key to reducing the rate of recurrence of deep soft tissue SMT. 11
In our patient, after initial misdiagnosis, the diagnosis of a perianal SMT was finally confirmed. Based on preoperative CT, MRI, and intraoperative exploration, it was confirmed that the root of the tumor was located deep within the external sphincter at the level of the rectal musculature, growing outward into the lumen and encroaching upon the external sphincter complex up to the sciatico-rectal fossa. In this case, the SMT was large. Owing to the unique location of the perianal SMT, it was necessary to ensure careful dissection and complete resection to ensure the integrity of the peritoneum and prevent recurrence and damage to the anal sphincter complex. If the sphincter is damaged during resection due to unavoidable factors, such as tumor invasion, the sphincter should be repaired in a timely manner to prevent the occurrence of anal dysfunction.
Perianal SMT are often misdiagnosed as PA due to atypical symptoms and low morbidity. Undiagnosed SMT may gradually increase in size and press on the rectum or anal canal, resulting in difficulty in defecation, sensation of anal swelling, or even intestinal obstruction. Early diagnosis via imaging (e.g. high-resolution ultrasound or MRI) can reduce the rate of misdiagnosis, and early surgical resection is more likely to preserve anal sphincter function and reduce the risk of postoperative incontinence. The rarity of perianal SMT makes them a “diagnostic pitfall” in clinical practice, and this case report can improve clinicians’ ability to identify rare perianal diseases, reduce misdiagnosis, and further optimize the diagnostic and treatment process.
Footnotes
Acknowledgements
The authors would like to thank the patient and her family for providing consent to use her photograph/video in this article.
Authors’ contributions
YG, YS, and XL were the patient’s attending physicians, reviewed the literature, and contributed to manuscript drafting; XL, TW, MY, CZ, and HL were responsible for the revision of the manuscript for important intellectual content; all authors issued final approval for the version to be submitted.
Data availability statement
The authors confirm that the data supporting the findings of this study are available within the article.
Declaration of conflicting interests
All authors have completed the ICMJE uniform disclosure form. The authors have no conflicts of interest to declare.
Funding
This study was supported by Weihai Traditional Chinese Medicine Science and Technology Program, No.2024 II-8.
