Abstract
Follicular dendritic cell sarcoma (FDCS) of the nasopharynx is a rare malignant tumor that has been described in only a few case reports, and its differential diagnoses include diverse clinicopathologic entities. FDCS is often initially misdiagnosed, especially when examining small biopsy specimens. We herein report a case of FDCS arising in the nasopharynx that was initially misdiagnosed as a nerve sheath tumor. A 44-year-old woman presented with persistent obstruction of the left nasal cavity and underwent an excisional biopsy. The specimen demonstrated morphologic and immunohistochemical features of FDCS. In situ hybridization for Epstein–Barr virus-encoded RNA was negative. The patient was treated with chemotherapy and radiotherapy. The sarcoma recurred near the original site more than 3 years after the initial treatment and was completely resected. At the time of this writing, the patient had remained disease-free for 1 year after resection. This case is being reported to improve the clinical recognition of FDCS.
Introduction
Follicular dendritic cell sarcoma (FDCS) was first reported by Monda et al. 1 in 1986. It is derived from mesenchymal stem cells and is grouped with histiocytic and dendritic cell neoplasms in the World Health Organization’s classification of tumors. 2 FDCS presents with lymph node disease in 31% of patients, extra-nodal disease in 58%, and both nodal and extra-nodal disease in 10%. 3 The extra-nodal distribution is very wide-ranging, and the occurrence of extra-nodal FDCS has mainly been reported in the tonsils, gastrointestinal tract, soft tissues, and mediastinum. 4 The nasopharynx is an extremely rare location for FDCS, and most such cases occur sporadically. 5 These cases are quite challenging to diagnose because sarcomatoid carcinoma is much more common. We herein report a case of FDCS in the nasopharynx and provide a literature review on this rare disorder, exploring the clinicopathological characteristics, differential diagnosis, and prognosis of the tumor to improve the diagnostic accuracy of the tumor while providing a theoretical basis for its treatment. The reporting of this study conforms to the CARE guidelines. 6
Case presentation
Clinical findings
A 44-year-old woman presented with a 4-month history of persistent obstruction of the left nasal cavity accompanied by occasional dizziness without runny nose or bleeding. She had previously been in good health and had no history of family disease. Computed tomography showed an irregular soft tissue shadow on the left lateral wall of the nasopharynx extending into the left nasal cavity and involving the oropharynx (Figure 1(a)). Contrast-enhanced computed tomography showed uneven obvious enhancement (Figure 1(b)). Subsequent nasal endoscopy examination showed a pale red, grayish-whitish neoplasm involving the left nasopharynx. The surface was not smooth; it was blood-stained and protruding into the nasal cavity. The patient then underwent an excisional biopsy. A pathological diagnosis of classic FDCS was ultimately made based on the histologic morphology and the immunohistochemical features of the tumor. The patient was treated with chemotherapy (CHOPx7; cyclophosphamide, doxorubicin, vincristine, and prednisone) followed by 30 radiotherapy sessions. At that time, the patient did not undergo surgical resection because the tumor site was not surgically accessible. The sarcoma recurred near the original site more than 3 years after the initial treatment and was completely resected. At the time of this writing, the patient had remained disease-free for 1 year after resection.

Follicular dendritic cell sarcoma of the nasopharynx. (a) On non-contrast-enhanced computed tomography, an irregular soft tissue shadow on the left lateral wall of the nasopharynx extended into the left nasal cavity and involved the oropharynx. (b) On contrast-enhanced computed tomography, the lesion was significantly enhanced.
Histopathological findings
Gross examination revealed a grayish-whitish solid mass that measured 2.5 ×2.5 × 1.0 cm. Microscopically, the tumor was composed of uniform, spindled tumor cells forming fascicular, storiform whorls. The neoplastic cells exhibited mild cellular atypia, unclear borders, a moderate amount of eosinophilic cytoplasm, nuclei with a vesicular chromatin pattern, visible small nucleoli, and a clear nuclear membrane. Binuclear or multinucleated giant cells were seen in the tumor. Occasional mitoses were present, but no area of coagulative necrosis was identified. Some lymphocytic infiltrates were present between tumor cells (Figure 2(a)–(c)). The tumor was immunoreactive for CD23 (Figure 2(d)), CD21 (Figure 2(e)), and CD68 (Figure 2(f)) and false-positive for S100 (Figure 2(g)-1). We initially believed that the tumor was positive for S100, but on closer inspection, we noticed that the background of the tissue sections was stained too dark and that large numbers of vacuoles were present. When we tested the immunohistochemical markers again, only a few cells in a small area showed perinuclear positivity (Figure 2(g)-2). However, only nuclear or nuclear and cytoplasmic staining could be judged as positive. Staining was negative for PCK, EMA, CK19, CD20, TDT, CD1a, CD99, HMB45, melan-A, SMA, desmin, and CD34. In situ hybridization for Epstein–Barr virus (EBV)-encoded RNA was performed. The tumor was negative for EBV. Figures are not presented because the data were negative.

Cytomorphology and immunohistochemistry of the follicular dendritic cell sarcoma. (a) The tumor was composed of uniform spindled tumor cells forming fascicles, storiform arrays, and whorls (hematoxylin–eosin; magnification: 200×). (b) The neoplastic cells showed mild cellular atypia and had unclear borders, a moderate amount of eosinophilic cytoplasm, nuclei with a vesicular chromatin pattern, visible small nucleoli, and a clear nuclear membrane (hematoxylin–eosin; magnification: 400×). (c) Occasional mitoses were present (hematoxylin–eosin; magnification: 400×). The tumor was positive for (d) CD23, (e) CD21, and (f) CD68 (magnification: 200×). Because of technical factors, staining for S100 presented a difference (g1, 2) (magnification: 200×).
Ethics
The study was approved by the ethics committee of the Xiantao First People’s Hospital Affiliated to Yangtze University. Written informed consent from the patient was obtained before the presentation of the study.
Discussion
Extra-nodal FDCS is a rare neoplasm, especially in the head and neck. A Medline search of FDCS revealed 9 cases (see Table 1), for a total of 10 cases with the addition of ours. Among these cases, FDCS occurred in adults (age range, 23–70 years; median age, 42 years), and there was no sex predilection (male:female ratio, 6:4). The tumor size was usually ≤3 cm in its greatest dimension. Two cases occurred on a background of Castleman's disease.7,8 Nevertheless, the pathogenesis of FDCS remains unclear. It has been proposed that the pathogenesis of FDCS may be akin to that of colorectal cancer in the sequence of cell hyperplasia-dysplasia-neoplasia, whereas others have suggested that the occurrence of FDCS may be associated with EBV infection and autoimmune disorders and may originate from a common mesenchymal stem cell. 3 However, among the 10 cases of nasopharyngeal FDCS, 5 tumors (cases 2, 4, 7, 8, and ours) were negative for EBV-encoded RNA by in situ hybridization. In one case (case 1), EBV genomes were detected by polymerase chain reaction. 9 In other cases, data were unavailable. Therefore, the association between EBV and FDCS of the nasopharyngeal region warrants further investigation. Clinical manifestations are nonspecific and include nasal congestion, blood-stained nasal discharge, a sensation of blockage in the ears, paresthesia of the roof of the mouth, and sinus headaches.
Summaries of nine cases of extra-nodal FDCS of the nasopharyngeal region from the English-language literature.
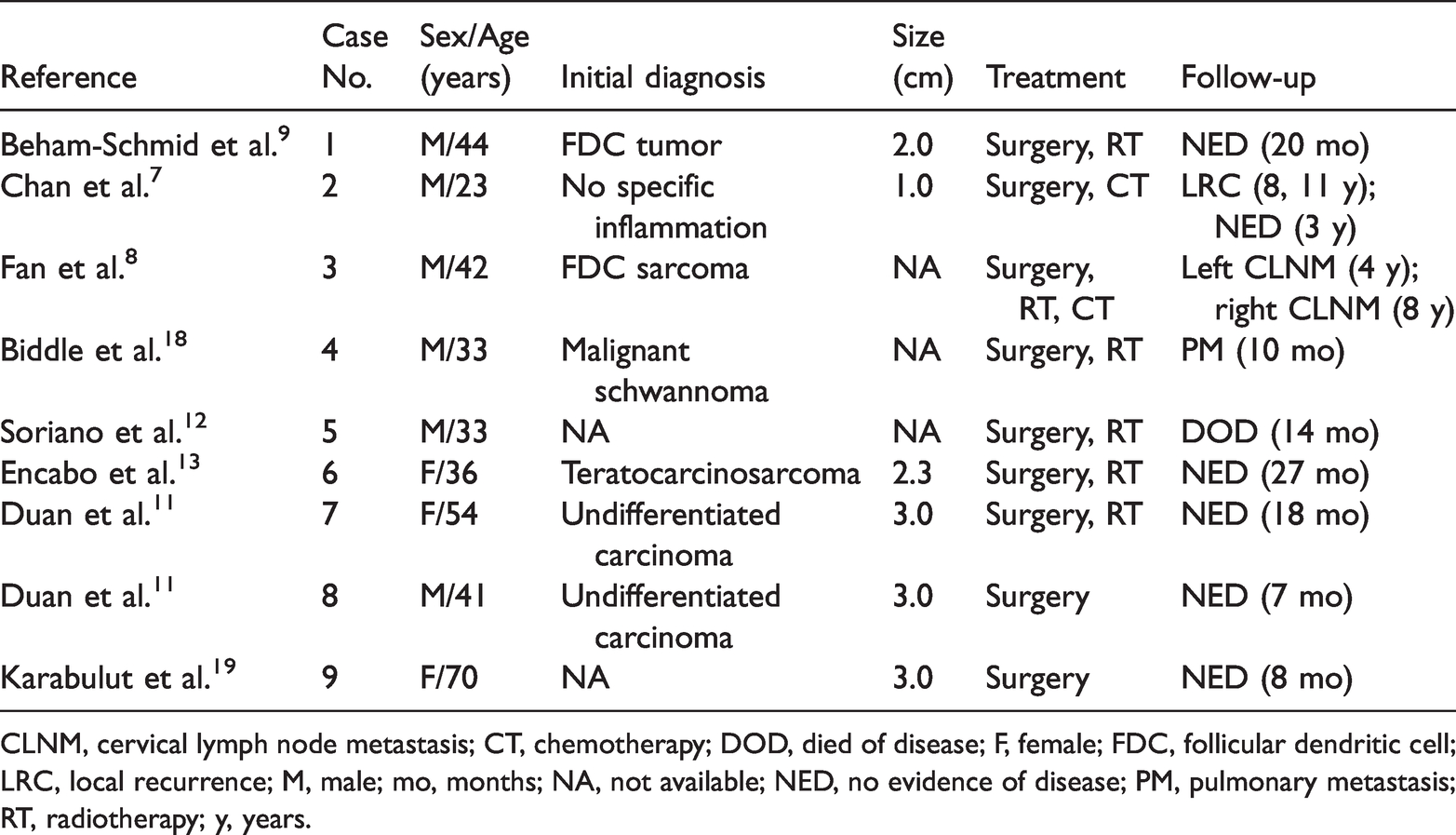
CLNM, cervical lymph node metastasis; CT, chemotherapy; DOD, died of disease; F, female; FDC, follicular dendritic cell; LRC, local recurrence; M, male; mo, months; NA, not available; NED, no evidence of disease; PM, pulmonary metastasis; RT, radiotherapy; y, years.
Histologically, FDCS is composed of neoplastic follicular dendritic cells and close-combined lymphoid cells. FDCS can be classified into two types: classic FDCS and inflammatory pseudotumor-like FDCS. 10 Classic FDCS shows two main morphological features. In one pattern, the tumor consists of spindled to ovoid cells arranged in fascicles, storiform arrays, whorls, or nodules. The neoplastic cells show indistinct cell borders and a moderate amount of eosinophilic cytoplasm. The nuclei are oval or elongated and contain vesicular or granular finely dispersed chromatin, a delicate nuclear membrane, and visible nucleoli. Binuclear or multinucleated giant cells can be seen in the tumor. Lymphocytes infiltrate throughout the tumor. In the other pattern, the tumor cells present an epithelioid morphology, show diffuse sheer growth or are separated by prominent fibrovascular septa, are syncytial-appearing with ovoid nuclei, and have distinct small nucleoli. Lymphocytic cells are scattered within the tumor. Although the lymphocytes may be sparse, lymphocyte infiltration has been regarded as a characteristic of FDCS.11 High-grade features can be found in some cases, with obvious nuclear atypia, atypical mitoses, and coagulative necrosis. 12 In our review of the 10 herein-described cases, all tumors presented as classic FDCS. Three tumors (cases 6, 7, and 8) showed epithelioid morphology features and were initially diagnosed as undifferentiated carcinoma 11 or teratocarcinosarcoma. 13 One tumor (case 5) showed predominantly high-grade histologic features. 12 Four tumors (cases 2, 3, 4, and our own) were composed of spindled cells forming fascicles, storiform arrays, or whorls, and two tumors (case 4 and the our case) were initially diagnosed as malignant schwannoma because they were immunoreactive for S100 (in our own case, S100 was false-positive). The data for cases 1 and 9 were not available. Therefore, the morphology is consistent with the literature.
Immunohistochemical examination is essential to reach a definitive diagnosis of FDCS. FDCS is often positive for one or more FDC markers, such as CD21, CD23, or CD35. Li et al. found that the positivity rates for CD21, CD35, and CD23 were 85%, 55%, and 31.7%, respectively. 14 Therefore, if there is a high index of suspicion for FDCS, a group of immunohistochemical indicators is recommended. CXCL13 and D2-40 are commonly positive but are not specific. Staining for vimentin, desmoplakin, clusterin, and fascin is also often positive. CD68, EMA, and S100 are variably positive.
Extra-nodal FDCS is still an under-recognized entity, especially in the head and neck. It poses a potential diagnostic pitfall for pathologists. FDCS composed of spindle cells may be confused with nerve sheath tumors, gastrointestinal stromal tumors, inflammatory myofibroblastic tumors, meningioma, and type A thymoma. FDCS shows epithelioid morphology features that may be confused with poorly differentiated or undifferentiated carcinoma, malignant melanoma, lymphoma, or other tumors derived from dendritic cells. The presence of lymphocytes in the tumor cells is an important clue to considering the use of immunohistochemical markers of FDCS in the differential diagnosis. It is also important to distinguish FDCS from inflammatory pseudotumor-like FDCS. However, this tumor is often found in the abdominal organs, particularly in the liver and spleen, and is consistently associated with the EBV. 15
Although FDCS has an indolent course, local recurrence is quite common, with a recurrence rate of 40% to 50%. 16 In our case, the sarcoma recurred near the original site more than 3 years after the initial treatment. Nevertheless, treatment recommendations for FDCS are not well-defined because of the low incidence of the tumor. Most cases are treated by surgical resection with or without adjuvant chemotherapy and/or radiation therapy. Gounder et al. 5 suggested that adjuvant or neoadjuvant treatment of FDCS did not affect overall survival. In contrast, Soriano et al. 12 reported that a combination of chemotherapy, surgery, and radiation may be more effective. For patients who cannot undergo surgical resection, lymphoma-type regimens may be used; this is what was initially used in our case. 16 However, the tumor recurred near the original site more than 3 years after treatment. Evaluation of more cases would help to establish standard treatment protocols. In addition, several studies have suggested that the following aspects contribute to a worse prognosis of FDCS: tumors of ≥6 cm, coagulative necrosis, nuclear mitosis of ≥5/10 HPF, and obvious cellular atypia.3,12,17 However, whether these are adverse prognostic factors for nasopharyngeal FDCS remains unclear; more cases need to be studied.
