Abstract
Background
Abemaciclib, an oral kinase inhibitor, is used to treat hormone receptor–positive and HER2-negative breast cancer patients. However, there has been a decrease in studies reporting adverse reactions to abemaciclib-related kidney injuries. Thus, this study was aimed at assessing its safety profile using a large-scale pharmacovigilance database.
Methods
Abemaciclib-related adverse drug reaction reports from the Food and Drug Administration Adverse Event Reporting System were obtained and scrutinized, and adverse drug reactions were selected using reporting odds ratio, the proportional reporting ratio methods, empirical Bayes geometric mean and UK Medicines and Healthcare products Regulatory Agency methods.
Results
We selected 10,757 matched reports associated with abemaciclib, among which we found eight adverse reactions about kidney injuries correlated with abeamciclib, such as increased blood creatinine, renal disorder, decreased glomerular filtration rate, increased blood urea, hydronephrosis, abnormal renal function test, increased creatinine renal clearance and increased cystatin C. A demographic analysis of reported cases of abemaciclib-associated renal injury revealed that the majority were female, aged ≥46 years and had taken the drug ≥30 days.
Conclusion
This study highlights the characteristics of adverse reactions with abemaciclib and those associated with renal damage, which are crucial for safety studies on the clinical use of this drug.
Plain language summary
Objective
Abemaciclib is an oral kinase inhibitor commonly used to treat hormone receptor–positive and HER2-negative breast cancer. Although it has shown efficacy in treating breast cancer, there have been concerns about its potential to cause kidney injuries. Despite this, studies addressing the adverse effects of abemaciclib on kidney function have been limited. This study aimed to assess the safety profile of abemaciclib, focusing on its association with kidney-related adverse events.
Methods
We conducted a retrospective analysis of adverse drug reactions (ADRs) related to abemaciclib using the Food and Drug Administration Adverse Event Reporting System (FAERS). The study focused on ADRs associated with kidney injuries, and we employed several disproportionality analysis methods to identify potential signals of kidney-related adverse reactions. These methods included the reporting odds ratio (ROR), proportional reporting ratio (PRR), empirical Bayes geometric mean (EBGM), and UK Medicines and Healthcare products Regulatory Agency (MHRA) methods.
Results
Our analysis identified a total of 10,757 reports associated with abemaciclib. Among these, we found eight distinct kidney-related adverse reactions, including increased blood creatinine, renal disorders, decreased glomerular filtration rate, increased blood urea, and abnormal renal function tests. A demographic analysis revealed that the majority of the affected patients were female, over 46 years of age, and had been taking abemaciclib for more than 30 days.
Conclusion
This study provides valuable insights into the kidney-related adverse effects of abemaciclib. The findings suggest that kidney injuries may be an underreported side effect of this medication, particularly among women and those on prolonged treatment regimens. Further research is needed to better understand the renal safety of abemaciclib and to develop guidelines for managing potential kidney-related risks in patients undergoing treatment.
Keywords
Objective
Abemaciclib, approved by the Food and Drug Administration (FDA) in 2017 in the United States, is an oral kinase inhibitor (kinase inhibitor indicated) currently used as a single agent or in combination with other agents for the treatment of hormone receptor (HR)–positive, human epidermal growth factor receptor 2 (HER2)–negative breast cancer patients. 1
Breast cancer is a common type of cancer in women, accounting for 32% of new cancers diagnosed in women in the United States in 2024 alone, and is a significant cause of cancer-related deaths in women; according to a 2021 statistic, for women, breast cancer causes the second highest number of cancer-related deaths after lung cancer. As for men, although the incidence of breast cancer is not as high as for women (approximately 1% of all breast cancer cases), it is still a cancer that should not be ignored as the incidence of breast cancer in men is increasing with the aging of the population. 2 Meanwhile, in breast cancer, aberrant cell cycle regulation has been identified as an important mechanism for breast cancer progression and has been used as a potential therapeutic target. 3
Abemaciclib, a new generation cyclin-dependent kinases (CDK) inhibitor, selectively inhibits CDK4/6 in targeting breast cancer, targeting the cyclin-CDK-retinoblastoma (Rb) pathway, and inducing cell cycle arrest to achieve therapeutic effect. 4
The use of selective CDK inhibitors such as abemaciclib is an effective strategy for the treatment of breast cancer, and in a randomised, double-blind, phase III study comparing abemaciclib plus NSAI (anastrozole or letrozole) versus placebo plus NSAI in postmenopausal women with HR+, HER2- ABC without prior systemic therapy in the advanced setting. 5 The final results showed numerical improvement in chemotherapy-free survival with the addition of abemaciclib.
However, the use of abemaciclib is accompanied by a variety of adverse effects, and the known adverse effects are diarrhoea, neutropenia, interstitial lung disease or pneumonitis, hepatotoxicity, venous thromboembolism, etc. 6 Regarding the effect on the kidney, the specification only mentions that it may lead to a transient increase in blood creatinine, but in clinical use, we have found that patients treated with abemaciclib may develop renal damage, manifested as increased blood creatinine, decreased glomerular filtration rate, increased blood urea, and increased cystatin C. 1
Therefore, we extracted and screened the reports related to abemaciclib in the FDA Adverse Event Reporting System (FAERS) from 2017q3 to 2024q2 and analysed the reports of adverse reactions that may be related to abemaciclib, aiming to investigate the relationship between abemaciclib and adverse reactions of renal damage.
Method
Data for this study were obtained from the FAERS database. FAERS supports post-market safety monitoring of marketed drugs and therapeutic biologics and contains reports of adverse reactions received by the FDA from manufacturers, as well as from consumers and professionals involved in the healthcare business. Since our data are derived from a public database (FAERS), any identifiable patient information has already been removed. Additionally, we will recheck the data to ensure that no patient-identifiable information is present, safeguarding patient privacy. In this study, data from 2017q3 to 2024q2 were obtained from the FAERS database and were screened for data on adverse event reports related to abemaciclib in the kidney. Our screening method was to de-emphasise the data according to the FDA-recommended method and to identify the role played by the drug in the adverse events as primary suspect drug (PS). We determined the PS by four calculation methods (reporting odds ratio [ROR], proportional reporting ratio [PRR], Medicines and Healthcare Products Regulatory Agency [MHRA] and Empirical Bayes Geometric Mean [EBGM]) to calculate the signal intensity of each preferred term (PT, level medical terminology describing the event, using the Medical Dictionary for Regulatory Activities [MedDRA]). The study was conducted in accordance with the Helsinki Declaration of 1975, as revised in 2013, and the reporting of this study conformed to Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 7
Data extraction and identification
The data for this study were obtained from the ASCII data packages in the FAERS database (updated quarterly), and we chose the data for a total of 28 quarters from 2017q3 to 2024q2. The data package in each quarter includes seven types of data files: DEMO contains patient demographic and administrative information, a single record for each event report; DRUG contains drug/biologic information for as many medications as were reported for the event (1 or more per event); REAC contains all terms coded for the adverse event (1 or more). For more information on MedDRA, please contact the MSSO Help Desk at

Flow chart.
Statistical analysis
In view of the constraints inherent in the FAERS database, which encompasses spontaneous reports and is deficient in comprehensive real-world adverse drug reaction data, it is not feasible to directly calculate the incidence of adverse drug reaction events. Nevertheless, disproportionality analysis is an effective method for identifying signals of adverse drug reaction events in retrospective pharmacological studies. In order to overcome the limitations of single algorithms and to enhance the reliability and accuracy of the resulting data mining, multiple algorithms were employed for the analysis of the data. 8 Consequently, the identification of adverse event signals was performed using disproportionality analysis (DPA) with the ROR, 9 the PRR methods, EBGM based on disproportionality analysis and UK MHRA methods.
The ROR method employs a two-sided test with a 95% confidence interval (CI), wherein a lower limit exceeding one signifies a signal, contingent on the number of reports (N) being equal to or greater than three. In order for the PRR method to generate a signal, the following criteria must be met: a minimum number of reports (N) of three, a PRR value of two or higher, and a variance (χ2) of four or higher. The EBGM method, although less sensitive, permits stratified analysis of population factors. In comparison to the ROR and PRR methods, the Bayesian method exhibits several advantages, including high specificity, a stable signal, and a low probability of misclassification. 10 Consequently, it can be considered a more prudent method. In regard to the MHRA method, adverse events with a lower limit of the 95% CI greater than one and reported in at least three cases using the ROR method, and with PRR > 2, C2 > 4, and reported in at least three cases using the MHRA method, were defined as adverse drug event signals. 11 The FAERS data employed in this study and analysis process were collected and conducted using the RStudio software, version 4.3.1 (Table 1).
Formulas and threshold values of ROR, PRR, MHRA and EBGM.

ADE: adverse drug event; CI: confidence interval; EBGM: Empirical Bayes Geometric Mean; MGPS: multi-item gamma poisson shrinker; MHRA: Medicines and Healthcare products Regulatory Agency; PRR: proportional reporting ratio; PT: preferred term; ROR: reporting odds ratio.
Meanwhile, we further selected five adverse reactions of abemaciclib-associated renal injury that were not mentioned in the specification, on top of the eight previously identified adverse reactions related to renal function (decreased glomerular filtration rate, increased cystatin C, renal disorder, abnormal renal function test, increased blood urea), the data in their DEMOyyQq and THERyyQq were extracted and organised into a table, where the columns ‘time_of_onset_group’ were calculated to categorise the results into groups after calculation (‘event_dt’ - ‘start_dt’) (event_dt:date the adverse event occurred or began, start_dt:date the therapy was started), in which, the data that does not satisfy the format of YYYY/MM/DD will be discarded, and ‘start_dt’ if there is more than one, then the earliest reported time will be selected (Table 2).
Features of abemaciclib-associated kidney injuries adverse reactions cases not mentioned by drug instructions.

AT: Austria; BR: Brazil; CH: Switzerland; CN: China or consumer; CZ: Czechia; DE: Germany; EG: Egypt; ES: Spain; FR: France; GB: United Kingdom; HK: Hong Kong; HP: PH, pharmacist; IN: India; JP: Japan; KR: Korea, South; MD: physician; OT: other health-professional; PH: pharmacist; PL: Poland; RO: Romania; US: United States.
Results
We screened 10,757 matched reports (25,617 by PTs) from FAERS. Overall, the top 30 adverse reactions for abemaciclib included diarrhoea, fatigue, nausea, vomiting, malignant neoplasm progression, decreased appetite, white blood cell count decreased, dehydration, anaemia, neutropenia, and so on. Among these, the most frequent PT was diarrhoea (a = 3136, 12.24%, ROR = 12.82 [95% CI, 12.34–13.31], EBGM = 10.79), which accounted for 12.24% of all reported PTs, followed by fatigue (a = 962, 3.76%, ROR = 2.86 [95% CI, 2.68–3.05], EBGM = 2.77), and then nausea (a = 932, 3.64%, ROR = 3.07 [95% CI, 2.87–3.28], EBGM = 2.96; Table 3).
Top 30 clinical adverse reactions of abemaciclib ranked by frequency (N) at the PT’s level in FAERS database calculated by disproportionality analysis.
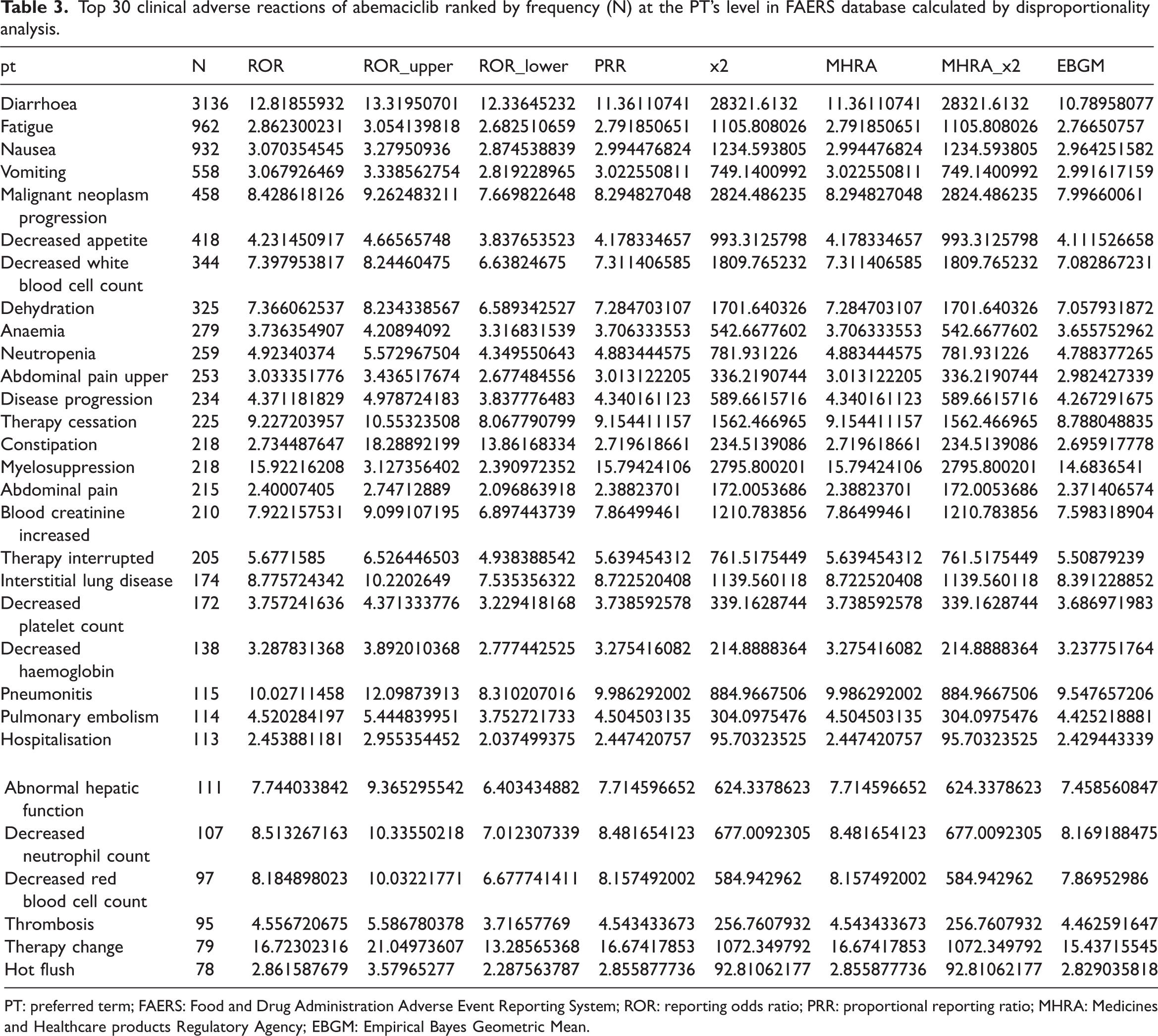
PT: preferred term; FAERS: Food and Drug Administration Adverse Event Reporting System; ROR: reporting odds ratio; PRR: proportional reporting ratio; MHRA: Medicines and Healthcare products Regulatory Agency; EBGM: Empirical Bayes Geometric Mean.
Based on disproportionate signalling analysis, the top 30 adverse reactions associated with abemaciclib included pseudocirrhosis, cystatin C increased, tumour marker abnormal, creatinine renal clearance increased, cell marker increased, dairy intolerance, asymptomatic coronavirus disease 2019, radiation pneumonitis, embolism venous, lymphangiosis carcinomatosa, and so on. Of these, the signal intensities of the top four adverse reactions showed a substantial lead in signal intensity compared with the others (ROR > 40, EBGM > 30; Table 4).
Top 30 clinical adverse reactions of abemaciclib ranked by ROR at the PT’s level in FAERS database calculated by disproportionality analysis.

COVID-19: coronavirus disease 2019; PT: preferred term; FAERS: Food and Drug Administration Adverse Event Reporting System; ROR: reporting odds ratio; PRR: proportional reporting ratio; MHRA: Medicines and Healthcare products Regulatory Agency; EBGM: Empirical Bayes Geometric Mean.
Among these reports, there were 285 reports of renal adverse reactions, with a total of 334 PTs reported, ranging from our selected eight renal function-related adverse reactions (increased blood creatinine, renal disorder, decreased glomerular filtration rate, increased blood urea, hydronephrosis, abnormal renal function test, increased creatinine renal clearance and increased cystatin C), the number of reported adverse reactions for blood creatinine increased were faultily reported relative to other abemaciclib-related renal function-related adverse reactions (a = 210, 62.87%, ROR = 7.92 [95% CI, 6.90–9.10], EBGM = 7.60), accounting for 62.87% of all renal function-related adverse reactions, while second was renal disorder (a = 47, 14.07%, ROR = 2.89 [95% CI, 2.16–3.85], EBGM = 2.86). According to the results of signal intensity analysis, cystatin C increased (a = 3, 0.90%, ROR = 41.71 [95% CI, 11.98–145.14], EBGM = 34.52), creatinine renal clearance increased (a = 6, 1.80%, ROR = 40.27 [95% CI, 16.72–97.01], EBGM = 33.53) were in TOP1, TOP2. The third was blood creatinine increased (a = 210, 62.87%, ROR = 7.92 [95% CI, 6.90–9.10], EBGM = 7.60). Kidney injuries associated clinical adverse reactions of abemaciclib ranked by ROR, the first and the second PTs signal intensities were higher (ROR > 40, EBGM > 30) (Table 5, Table 6).
Kidney injuries associated with clinical adverse reactions of abemaciclib ranked by frequency (N) at the PT’s level in FAERS database calculated by disproportionality analysis.

PT: preferred term; FAERS: Food and Drug Administration Adverse Event Reporting System; ROR: reporting odds ratio; PRR: proportional reporting ratio; MHRA: Medicines and Healthcare products Regulatory Agency; EBGM: Empirical Bayes Geometric Mean.
Kidney injuries associated with clinical adverse reactions of abemaciclib ranked by ROR at the PT’s level in FAERS database calculated by disproportionality analysis.

PT: preferred term; FAERS: Food and Drug Administration Adverse Event Reporting System; ROR: reporting odds ratio; PRR: proportional reporting ratio; MHRA: Medicines and Healthcare products Regulatory Agency; EBGM: Empirical Bayes Geometric Mean.
For the five specifications we selected (including increased blood urea, increased cystatin C, decreased glomerular filtration rate, renal disorder and abnormal renal function test), there was no mention of abemaciclib. We analysed the demographic data for the five adverse reactions of abemaciclib associated with renal injury that were not mentioned in the specification (including increased blood urea, increased cystatin C, decreased glomerular filtration rate, renal disorder and abnormal renal function test) by extracting their corresponding DEMO files. For these adverse reactions, we found that the reported PTs were reported by patients concentrated in the 65+ years age group, with all reported reports being 46 years and older. The gender of the reported patients was predominantly female (83.18%), with all of the patients with a PT of abnormal renal function test being female. The most reported patients were consumers (occp_cod = CN), and the country of the reported patients was concentrated in the United States (reporter_country = US).
The column ‘time_of_onset_group’ is calculated and grouped from the data in DEMO and THER, and represents the grouping of the time of the patient’s adverse reaction minus the time of the patient’s initiation of the medication, i.e., it can be interpreted as the amount of time after which the patient would have experienced an adverse reaction after the administration of abemaciclib. Overall, the five adverse reactions were more frequently categorised in the groups of 39–180 days (a = 32, 44.4%) and >180 days (a = 33, 45.2%), with only one occurrence of an adverse reaction, renal disorder, being categorised in the 0–7 days group.
Discussion
Diarrhoea was the most frequent adverse reaction in the current study (a = 3136), a phase 2 MONARCH 1 study evaluating the use of abemaciclib monotherapy in the treatment of women with refractory hormone receptor–positive (HR+), HER2- and metastatic breast cancer. Metastatic breast cancer, diarrhoea was the most common adverse reaction (all grades 90.2%, grade 3 19.7%, grade 2 28.8%, grade 1 41.7%) in the phase 2 MONARCH 1 study. 12 This is consistent with the results we obtained. However, it is worth noting that when ranked in terms of signal intensity, the intensity of diarrhoea was in the 19th place, and the intensity value was not high compared with other adverse reactions (a = 3136, ROR =12.82 [95% CI,12.34–13.31], EBGM =10.79), probably because diarrhoea is not very specific as an adverse drug reaction, and most of the combined medications can cause diarrhoea.
For all abemaciclib-related adverse reactions, after calculating the signal intensity by four calculations, there were four with higher signal intensity and they were pseudocirrhosis (a = 5, ROR = 44.24 [95% CI, 16.75–116.82], EBGM = 36.22), cystatin C increased (a = 3, ROR = 41.71 [95% CI,11.98–145.13], EBGM = 34.52), tumour marker abnormal (a = 11, ROR = 40.41 [95% CI, 21.10–77.36], EBGM = 33.62), creatinine renal clearance increased (a = 6, ROR = 40.27 [95% CI, 12.72–97.01], EBGM = 33.53). For the adverse reaction pseudocirrhosis, the number of reported PTs was 5, but the signal intensity was high, 12 pseudocirrhosis often occurs after systemic chemotherapy for liver metastases of breast cancer, with a prevalence of 38%–81% in patients with liver metastases of breast cancer, 13 therefore, the pseudocirrhosis, although high in signal, may not be associated with abemaciclib. Given that abemaciclib is a drug primarily used for the treatment of breast cancer, it is possible that it causes Tumour marker abnormalities as a result of normal pharmacological effects or disease factors, which is consistent to the result of a research from Dr Klein, ME. 14
Cystatin C is a protein that is filtered relatively freely by glomeruli, it is not affected by non-renal factors and is a surrogate marker of renal function. 15 In the present study, the number of reported cases of adverse reaction cystatin C increased was 3, but it had a high signal (ROR = 41.71, EBGM = 34.52), which suggests that the incidence of this adverse reaction may be low, but its occurrence may be highly correlated with abemaciclib and this PTs is not mentioned in the inserts, so it is recommended for the Healthcare professionals, in the case of cystatin C increased in patients on abemaciclib, can target their suspicion to abemaciclib.
In general, in previous research, increased creatinine renal clearance is a reflection of damage in early diabetic nephropathy. 16 In our study, we found that among the patients using abemaciclib, six cases also reported increased creatinine renal clearance with higher signals, so we suspect that abemaciclib may cause kidney damage similar to diabetic nephropathy. However, due to the small number of reported cases, we do not exclude the possibility that the patients may have comorbid diabetic nephropathy themselves.
In a multicenter study evaluating the safety of abemaciclib, kidney-related adverse reactions were primarily characterized by increased creatinine levels, which is consistent with our findings. 17 For the eight adverse reactions on the kidney selected in this study, they accounted for approximately 2.65% of all reported reports (in terms of number of reports) and 1.30% of all PTs. It is noteworthy that among these adverse reactions concerning the kidney, only blood creatinine increase was explicitly mentioned in the specification as an adverse reaction after the use of abemaciclib, while the others were not explicitly stated in the specification. Abemaciclib and its major metabolically active agents are known to cause adverse reactions in the kidney through the inhibition of the transporters OCT2 (SLC22A2), MATE1 (SLC47A1), and MATE 2-K (SLC47A2), 18 and inhibit renal tubular secretion of creatinine, which results in the elevation of blood creatinine (SCr) that can be observed with clinical use of abemaciclib (approximately 15%–40%), but which can be reversed with cessation of abemaciclib. 19 Therefore, glomerular filtration rate (GFR) may be high if calculated using SCr in patients on abemaciclib, and this may not be clinically significant for the patient. It may be more appropriate to use other markers (such as blood urea nitrogen [BUN], cystatin C) to assess renal function in patients on abemaciclib. 20 In addition, biomarkers such as kidney injury molecule 1 (KIM-1) may also be appropriate for assessing renal function in patients treated with abemaciclib.21,22
For our five selected adverse reactions to abemaciclib-related renal injury that were not mentioned in the specification, we found that the reported patients were over 45 years of age, which we hypothesize is related to abemaciclib’s primary indication of breast cancer, which is predominantly clustered in the age range of 60–69 years (ductal carcinoma in situ cases 31%, invasive cases 29%; United States, 2024), with a median age of 62 years. 23 Similarly, reported patients were predominantly female, which may also be related to the gender distribution of breast cancer incidence (men account for approximately 1% of breast cancer cases). 24
The fact that adverse reactions were most frequently reported by consumers suggests that abemaciclib-associated adverse reactions to renal injury may generally occur in non-healthcare settings, and therefore, physicians and pharmacists should educate patients to be concerned about whether they are experiencing any abnormalities in renal function, concern about the amount of urine they are urinating, the presence of foamy urine, regular checkups of renal function when abemaciclib is given to a patient for use as a treatment. Patients should undergo regular examination of renal function, and if there is any abnormality, they should come to the hospital for timely consultation. It can also be surmised that the higher frequency of discovery by patients is likely to be detected during non-short-term cycles of drug use, so we turn our discussion to the length of time between drug use and the occurrence of an adverse reaction.
For column ‘time_of_onset_group’, we note that almost all of these adverse reactions do not occur within a week but mostly in the time period of one month to six months, which suggests that these five adverse reactions may be related to the accumulation of the drug dose, with a certain latency period, and do not appear immediately after the use of the drug, but occur after a period of time of use of the drug or the accumulation of the dose to a certain amount. Therefore, for patients using abemaciclib, more attention should be paid to regular follow-up examinations, as well as the patient’s own attention to the condition of their kidneys at home. 25 Specifically, the adverse reactions of increased blood urea and abnormal renal function tests were more concentrated in the group of 31–180 days compared with the other adverse reactions.
Blood urea is an indicator associated with a high-protein diet, so a patient’s diet may also affect this indicator, and a high-protein diet may also affect a patient’s renal function, 24 so we recommend that patients on abemaciclib control their protein intake at each meal. The renal function test is a comprehensive test that includes urea, blood urea nitrogen (BUN), creatinine and other indicators, which can reflect the basic condition of patients’ renal function. The abnormal renal function test adverse reactions are located in the 31–180 group of time_onset_group, which indicates that for patients using abemaciclib, they may have adverse reactions in the 31–180 group. In patients using abemaciclib, renal function impairment may occur more frequently after one month of dosing. 26
The limitations of this study are that there may be a situation of bias in data analysis because we used study data from the FAERS database, whose adverse event reports were submitted by reporters with different backgrounds; the data content was not fully harmonised, and there is this parts of missing data. Moreover, these reports are past reporting experiences, and we could not extrapolate the current reported patient’s. We also observed that in this study, the incidence of adverse reactions in females was higher than that in males. However, due to the relatively small number of reports collected from males, we cannot conclude that abemaciclib-associated adverse reactions are more prevalent in females. Our results are intended as a complementary study to explore post-marketing adverse reactions to this drug.
Conclusion
By analysing the adverse event reports related to abemaciclib in FAERS, the adverse reactions and our selected renal-related adverse reactions and five renal damage-related (including increased blood urea, increased cystatin C, decreased GFR, renal disorder and abnormal renal function test), we can make the following summary of abemaciclib users who experienced renal damage-related adverse reactions: the gender of the reported cases was predominantly female, and all of the reported cases were over 46 years of age. The majority of the reported individuals were from the United States and were of consumer status.
For renal-related adverse reactions, the adverse reaction of increased blood creatinine may be related to the pharmacological effects of abemaciclib itself. We reasoned that increased cystatin C may be an unspecified, potential adverse reaction of abemaciclib. In addition, we found that adverse reactions associated with renal impairment usually reappeared after a period of time on the drug (>31 days), and therefore we urge healthcare professionals to require long-term follow-up observation of patients using a course of abemaciclib for more than 30 days, with follow-up visits to monitor their renal function, especially cystatin C and blood creatinine.
Footnotes
Acknowledgements
We highly appreciate every author who contributed to the manuscript.
Author contributions
Data availability statement
Declaration of conflicting interests
The authors declare no conflicts of interest.
Funding
The study is funded by Medical Scientific Research Foundation of Guangdong Province of China (B2023022), Foundation of Guangdong Provincial People’s Hospital (8220130280, 8237120025) and Fundamental and Applied Basic Research Program of Guangdong Province (2021A1515220031).
Informed consent statement
Not applicable.
Institutional review board statement
This study used publicly available safety ICSR data that were provided in an anonymous form and were already compliant with ethical standards. Therefore, no further ethical evaluation was necessary.
