Abstract
Oral vancomycin is mainly used to treat and prevent active Clostridium difficile infection. Because it is widely believed that there is a very low absorption rate via the gastrointestinal tract, reports of adverse reactions following oral vancomycin administration are rare. This case report describes for the first time a case of antibiotic-associated diarrhoea in a 2-month-old infant treated with oral vancomycin. After oral vancomycin treatment, the number of eosinophils increased significantly and the levels gradually recovered after drug withdrawal. A review and analysis of the previously reported adverse reactions caused by oral vancomycin and eosinophilia caused by vancomycin confirm the need for physicians to pay close attention to vancomycin-related adverse reactions, to monitor the required concentration and to measure eosinophil counts in patients with rash-related adverse reactions. Patients with concomitant diseases and children should be monitored for adverse events as it is possible that they have increased gastrointestinal absorption of vancomycin following oral administration. When vancomycin causes eosinophilia, fever and rash, physicians should be alert to the possibility of organ damage.
Introduction
Vancomycin is a glycopeptide antibiotic that is widely used to treat infections caused by severe gram positive and other antibiotic-resistant bacteria, especially methicillin-resistant Staphylococcus aureus infection. The common adverse reactions of vancomycin include nephrotoxicity, ototoxicity, fever, vancomycin flushing reaction and other symptoms, 1 but there are a growing number of reports of eosinophilia associated with vancomycin use.2–8 Oral vancomycin is mainly used to treat and prevent active Clostridium difficile infection.9,10 Because its absorption rate in the intestinal tract is generally considered to be extremely low, adverse drug events associated with intestinal administration are infrequently reported.11–13 Eosinophilia caused by oral vancomycin in infants with C. difficile infection has not been reported previously. This current case report is the first to describe this adverse event in an infant that received oral vancomycin. In addition, this article summarized the clinical characteristics of the adverse reactions of oral vancomycin and the related cases of vancomycin-induced eosinophil elevation. The aim of the current report was to highlight the possible clinical presentations of this condition in order to facilitate early diagnosis and treatment.
Case report
In March 2019, a 2-month-old female was admitted to the Department of Gastroenterology, Wuhan Maternal and Child Healthcare Hospital, Tongji Medical College, Huazhong University of Science & Technology, Wuhan, Hubei Province, China presenting with diarrhoea for 15 days and bloody stools for 1 week. In the later stage, the child had yellow watery stools, approximately 20 times/day, accompanied by low fever, with the highest temperature of 37.7°C. The stools became bloody 1 week ago. Montmorillonite powder, probiotics and oral rehydration salts were ineffective. When she was admitted to hospital, her condition was slightly poor, her diet was approximately 500 ml per day, her sleep was not stable and her urine output was 25 ml/kg per day.
There was no family history. In January 2019, she was hospitalized in the Department of Paediatric Surgery of Tongji Hospital, Wuhan, Hubei Province China and underwent intestinal resection and anastomosis due to small intestinal atresia in the neonatal period. After the operation, her surgical incision was infected and she was successively administered the following drugs: 80 mg cefoperazone sodium and 20 mg tazobactam sodium intravenous (i.v.) every 12 h for 10 days; 55 mg meropenem i.v. every 8 h for 7 days; 28 mg teicoplanin i.v. once a day for 7 days; 85 mg amoxicillin clavulanate potassium i.v. every 8 h for 6 days; and 20 mg metronidazole i.v. every 8 h for 21 days. She was discharged from hospital in good condition. Diarrhoea occurred on the 4th day after stopping antibiotic treatment.
A physical examination recorded the following: temperature, 37 °C; pulse rate, 132 beats/min; respiration rate, 32 breaths/min; blood pressure, 85/54 mmHg; conscious mind; decreased skin elasticity; no rash was found; abdominal examination showed no muscle tension; active bowel sounds; no anal fissure; and all other physical examinations were negative. Routine blood tests recorded the following: white blood cells (WBC), 17.00 × 109/l; neutrophil %, 60.2%; eosinophil %, 1.2%; haemoglobin (Hb), 107 g/l; C-reactive protein (CRP), 17 mg/l; alanine aminotransferase (ALT), 32 U/l; aspartate aminotransferase (AST), 28 U/l. Analysis of renal function, myocardial enzymes and electrolytes showed the following: no obvious abnormalities were found. Ultrasonography showed gas accumulation in the intestine, but there was no evidence of necrotizing enterocolitis and intussusception. Routine stool tests showed the following: occult blood +, but other indices were negative; no abnormalities were found on stool culture.
The child had a history of intestinal surgery and had been treated with many antibiotics for a long period of time. At the same time, she had the following clinical manifestations: severe diarrhoea; low fever; WBC count >15 × 109/l; and no other pathogenic bacterial infection was found. Although there was no evidence of C. difficile, antibiotic-related diarrhoea was the most likely diagnosis. In addition, it should be noted that the patient was fed with deeply hydrolyzed milk powder and amino acid milk powder after surgery, so it is unlikely that the eosinophil elevation was caused by cow's milk protein allergy. On the same day (in March 2019), she was given 40 mg/kg per day vancomycin orally (actual 34 mg) four times a day (Vianex S.A., Athens, Greece; specification: 500 mg/bottle; batch number: H20140174) and 25 mg metronidazole i.v. twice a day. The diarrhoea improved.
On day 4 of hospitalization, routine blood tests recorded the following: WBC, 23.27 × 109/l, neutrophil %, 29.4%; eosinophil %, 35%; eosinophil count, 8.14 × 109/l; Hb, 101 g/l. On day 7 of hospitalization, routine blood tests recorded the following: WBC, 16.59 × 109/l; neutrophil %, 30%; eosinophil %, 23.9%; eosinophil count, 3.97 × 109/l; Hb, 101 g/l; ALT, 37 U/l; AST, 30 U/l. Renal function indices were normal. After the clinical symptoms were obviously improved, she was discharged from hospital. She continued to be administered 40 mg/kg per day vancomycin and 25 mg metronidazole twice a day orally for 1 week. These drugs were then discontinued. An outpatient examination at 2 weeks after drug withdrawal showed the following: WBC, 9.6 × 109/l; neutrophil %, 42%; eosinophil %, 2.1%; eosinophil count, 0.2 × 109/l; Hb, 97 g/l.
This study was approved by the institutional review board of Wuhan Children's Hospital, Tongji Medical College, Huazhong University of Science & Technology (no. 2022R053-E02). The reporting of this study conforms to CARE guidelines. 14 The parents of the infant who participated in this case report provided written informed consent for publication.
Discussion
The current case was assessed according to the Naranjo scale as follows: 15 (i) there was a conclusive report before the conclusion of eosinophilia caused by vancomycin (1 point); (ii) the adverse drug reaction (ADR) occurred after oral vancomycin (2 points); (iii) the ADR was relieved after stopping oral vancomycin (1 point); (iv) the child had used metronidazole many times in the past and there was no history of eosinophilia. Presenting after oral vancomycin and considering an ADR caused by oral vancomycin is not another cause that can cause this ADR alone (2 points); (v) the number of eosinophils in routine blood tests before and after medication is the objective evidence to confirm the adverse reaction (1 point). In conclusion, according to the causal evaluation of the ADR, the final score was 6 (i.e. probably related), which suggested that the current case’s eosinophilia was probably an adverse reaction caused by oral vancomycin.
The clinical application of oral vancomycin is gradually expanding, but this route of administration has always been considered to be associated with poor intestinal absorption, high intestinal drug concentration and no need to monitor blood drug concentration.16,17 A previous study confirmed that oral vancomycin is rarely absorbed from the gastrointestinal system even in severe diseases or renal failure. 18 However, some studies have confirmed that vancomycin can be detected in the blood of individuals infected by C. difficile, which may be related to the increased absorption of vancomycin by areas of the gastrointestinal tract that have inflammation.19,20 In published studies, the frequency of detection of serum vancomycin levels in patients receiving oral therapy ranged from 2% to 68%.18,21,22
There are few reported cases of adverse reactions related to oral vancomycin worldwide; and the reported cases are mainly skin adverse reactions, such as vancomycin flushing reaction, 23 maculopapule, 24 urticaria 19 and linear immunoglobulin (Ig)A bullous dermatosis. 25 The pathogenesis of different rash types is different. For example, vancomycin flushing reaction is caused by non-IgE-mediated mast cell degranulation and excessive histamine release; 26 and linear IgA bullous dermatosis is type IV delayed type hypersensitivity, with pathological changes characterized by the linear deposition of IgA along the basement membrane zone. 26 The main clinical manifestations of IgE-mediated hypersensitivity induced by oral vancomycin are local or systemic maculopapule and urticaria. 26 Table 1 and Table 2 present a summary and analysis of the case reports (including this current case) of the adverse reactions caused by oral vancomycin,11,16,19,20,23–25,27–34 which can occur at ages ranging from 2 months to 82 years (median age, 59 years). Skin changes were the most common (13 of 16 patients; 81.3%). In addition, there were two cases with blood dyscrasia, one case with ototoxicity and one case with laryngeal obstruction. This current case is the first report of eosinophilia caused by oral vancomycin in an infant. A previous study reported that the risk factors of the systemic absorption of oral vancomycin included renal insufficiency, severe C. difficile infection, high vancomycin dose (>500 mg/day), long-term treatment (>10 days), intensive care unit admission, use of vancomycin retention enema and gastrointestinal inflammation. 32 All 16 patients described in this summary of the published literature had underlying diseases, which was basically consistent with the aforementioned research. Fortunately, except for one patient with serious underlying diseases and a poor prognosis after taking vancomycin orally for a long period of time, 25 all of the other 15 patients achieved good outcomes after stopping vancomycin and/or receiving other treatments.11,16,19,20,23,24,27–34
Ig, immunoglobulin; CDI, Clostridium difficile infection.
aTreatment data only available for 14 patients.
Ig, immunoglobulin.
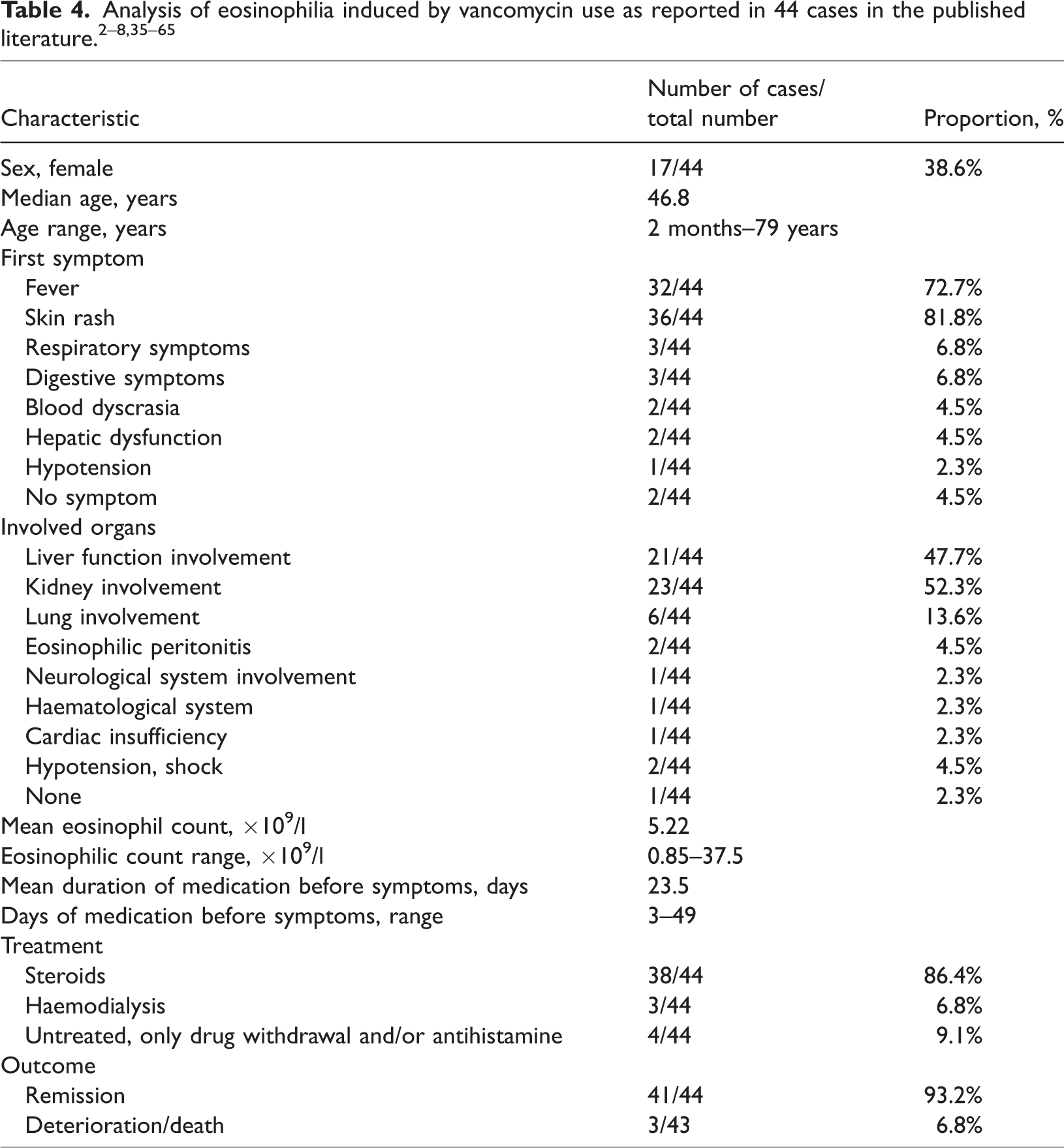
The effects of vancomycin on the haematological system include anaemia, leukopenia, thrombocytopenia and eosinophilia. However, there are few cases of eosinophilia caused by vancomycin in both the clinic and the published literature. With the increasing use of vancomycin in clinical practice, it is particularly important to summarize this type of literature. Table 3 and Table 4 present a summary and analysis of 44 cases of eosinophilia caused by vancomycin.2–8,35–65 Of these 44 cases, drug reaction with eosinophilia and systemic symptoms (DRESS), also known as drug-induced hypersensitivity syndrome, was found in 35 cases.2–8,35–57 In other cases, some patients had increased eosinophil counts and involvement of other organs such as kidney, lung and heart function due to intravenous use of vancomycin.58,62,63 Although the reported cases were been clearly diagnosed as DRESS, they were considered to have this syndrome based on the case data. A comprehensive review all of the 44 published cases with eosinophilia caused by vancomycin demonstrated the following (Table 4):2–8,35–65 male patients were more often affected (27 of 44 patients; 61.4%); the median age was 46.8 years (range, 2 months to 59 years); the median time to vancomycin-related symptoms was 23.5 days (range, 3–49 days); and the median eosinophil count was 5.22 × 109/l (range, 0.85–37.5 × 109/l). Fever (32 of 44 patients; 72.7%) and/or rash (36 of 44 patients; 81.8%) were the first symptoms of eosinophilia in patients with vancomycin treatment; and another two cases had hypotension or were asymptomatic after intraperitoneal injection of vancomycin.60,61 Organs and other systems were involved in the 44 cases; including the liver (21 of 44 patients; 47.7%), kidney dysfunction or failure (23 of 44 patients; 52.3%), lung involvement (six of 44 patients; 13.6%), peritonitis (two of 44 patients; 4.5%), neurological system (meningitis; one of 44 patients; 2.3%) and cardiac insufficiency (one of 44 patients; 2.3%). Therefore, when vancomycin causes fever, rash and eosinophilia in the clinic, physicians should be alert to the possibility of organ damage and closely monitor for this complication.
ALT, alanine aminotransferase.
When a patient presents with suspected vancomycin-related adverse reactions, the first treatment is to stop the vancomycin and then give steroids, antihistamines and other treatments according to the condition. Due to the prolonged half-life of vancomycin, the current summary of 44 cases showed that severe refractory cases need renal replacement therapy (three of 44 patients; 6.8%)47,54,62 or even liver transplantation (one of 44 patients; 2.3%). 40 A total of 41 of 44 patients (93.2%) achieved a good outcome and were considered to have gone into remission. This proportion was higher than previously reported. 66 The three patients that experienced deterioration or death were over 60 years old.
The pathogenesis of eosinophilia induced by vancomycin remains unclear, but it is speculated to be related to increased inflammatory cytokines, especially interleukin-5, which reach a peak a few days before the peak of eosinophilia. 67 It is thought that inflammatory cytokines might be involved in organ damage and the subsequent induction of eosinophilia. 67 This current case report described the induction of eosinophilia by oral vancomycin in a 2-month-old infant without it causing organ damage. The lack of organ damage in the current case might be related to the infant's immune system not being fully developed so it did not initiate a strong response. With the wide application of vancomycin in the clinic, understanding its association with adverse reactions can help avoid organ damage in a timely manner.
It should be noted that repeated use of metronidazole did not cause eosinophil elevation in the current case, so it was inferred that eosinophil elevation in this infant was related to the newly added oral vancomycin treatment. However, there was no evidence to prove that the eosinophilia observed in this current patient was caused by the combined action of oral vancomycin and metronidazole, although this remains a small possibility.
In conclusion, a case of eosinophilia induced by oral vancomycin in an infant was reported for the first time in this current case report. A summary and analysis of the previously reported adverse reactions caused by oral vancomycin and eosinophilia caused by vancomycin confirm the need for physicians to pay close attention to vancomycin-related adverse reactions, to monitor the required concentration and to measure eosinophil counts in patients with rash-related adverse reactions.
Footnotes
Author contributions
Study concepts: Yali Wu, Fang Wang; literature research: Yali Wu, Shan Guo, Fang Wang; clinical information collection: Yali Wu, Fang Wang; data analysis/interpretation: Yali Wu, Shan Guo; manuscript preparation: Fang Wang, Wei Yin; manuscript final version approval: Fang Wang.
Declaration of conflicting interests
The authors declare that there are no conflicts of interest.
Funding
The authors disclose receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a grant from the Chen Xiao-ping Foundation for the Development of Science and Technology of Hubei Province (no. CXPJJH121002-202130).