Abstract
Objective
To systematically identify drug-induced esophageal ulcer and address existing knowledge gaps.
Methods
We conducted a real-world study using the Food and Drug Administration Adverse Event Reporting System database from Q1 2004 to Q4 2024. Disproportionality analyses were performed to evaluate drug-induced esophageal ulcer risk. Data for each drug were merged and screened using the RxNax platform to recalculate drug exposure, proportional reporting ratio, reporting odds ratio, and chi-squared values. High-risk drugs classified by anatomical therapeutic chemical classification system were cross-checked with Side Effect Resource, product labels, and published literature.
Results
A total of 12,763 drug-induced esophageal ulcer cases were identified. Forty-nine high-risk drugs, spanning nine anatomical therapeutic chemical classification system classes, were detected, including antineoplastics and immunomodulators (Class L), systemic anti-infectives (Class J), and drugs affecting the musculoskeletal system (Class M). Aspirin, alendronic acid, and doxycycline were most frequently reported; doxycycline, clindamycin, and alendronic acid generated the strongest safety signals. Antineoplastics/immunomodulators (anatomical therapeutic chemical classification system L) had the highest cumulative proportional reporting ratio, primarily driven by tacrolimus, mycophenolic acid, and sunitinib. Notably, tacrolimus, sunitinib, meclofenamic acid, clopidogrel, and erlotinib were associated with esophageal ulcers but remain unlabeled for this risk.
Conclusions
This study provides a comprehensive drug-induced esophageal ulcer risk profile, highlighting high-risk drugs beyond commonly recognized agents. These findings can inform safer prescribing practices, patient counseling, and targeted pharmacovigilance.
Keywords
Introduction
The esophagus, a critical passageway for food from the mouth to the stomach, is normally protected by its intact mucosa. However, drug-induced esophageal ulcer (DEU)—defined as damage and ulceration of the esophageal mucosa caused by medications—poses a serious health threat. Typical clinical manifestations include sudden onset of retrosternal pain, dysphagia, and hematemesis, while endoscopic examination often reveals mucosal erythema, erosions, and microscopically distributed esophageal ulcers.1,2 The pathomechanism of DEU involves direct chemical irritation of the esophageal mucosa by drugs, localized physical retention, and indirect effects mediated by systemic pharmacological actions. 3 DEU is frequently misdiagnosed or diagnosed late because its symptoms closely resemble those of gastroesophageal reflux disease. 4 Misdiagnosis may lead to unnecessary anti-reflux therapy, increasing the burden on the healthcare system. In addition, DEU can result in severe complications, including transmural perforation, stricture formation, and occult hemorrhage.5,6 Case reports of DEU caused by drugs such as tetracycline, clindamycin, and potassium chloride date back to 1979. 7 In recent years, attention has also turned to the occurrence of DEU in the adolescent population. 8
Despite this, awareness of DEU risk remains limited. Clinicians often overlook DEU as a potential cause of chest pain or odynophagia, and its diagnosis is frequently underestimated because most patients report self-limiting symptoms. 9 Existing studies on DEU have several limitations. Case reports are neither sufficiently representative nor do they provide quantitative risk indicators.10–14 Small clinical studies are usually single-center, limiting control over confounding factors.8,15 In vitro or animal experiments do not accurately reflect human esophageal dynamics and often involve high drug concentrations, which restricts their clinical relevance. Collectively, these limitations make the current evidence insufficient to guide practice effectively.
DEU is a serious adverse drug reaction (ADR) that, although easily overlooked, should not be underestimated. Nevertheless, studies investigating associated drug–risk signals remain insufficiently systematic. The present study is a real-world pharmacovigilance analysis of DEU based on the Food and Drug Administration (FDA) Adverse Event Reporting System (FAERS) database. It aimed to expand the spectrum of risk-associated drugs and identify potential high-risk medications that have been underemphasized in previous literature.
Data and methods
Data sources
This retrospective study was conducted using the FAERS database. FAERS is designed to support post-market surveillance of drugs and therapeutic biologics and contains information on ADRs and medication errors collected by the FDA.16–19 Data are voluntarily submitted by healthcare professionals and consumers and include demographic characteristics, drug information, clinical outcomes, and other relevant details. We queried the FAERS database using the OpenVigil 2.1 pharmacovigilance analytics platform. OpenVigil 2.1 is a publicly available tool that imports raw FAERS data and enables data extraction, cleaning, mining, and analysis.20,21
Definition of ADRs and drugs
ADRs in the FAERS database were coded according to the Preferred Term (PT) in the Medical Dictionary for Regulatory Activities (MedDRA, Version 26.1). The PT hierarchy at level 4, which is most commonly used, was applied in this study. We searched for “Oesophageal ulcer” (MedDRA code: 10030201) in the PT column to identify drugs associated with esophageal ulceration. All corresponding reports were then downloaded. For statistical analysis, the generic drug name was used as the unique identifier. Many FAERS reports use brand names; these were converted to generic names using the DrugBank database. If a drug name could not be retrieved in DrugBank (https://go.drugbank.com/drugs), the report was considered incorrect and removed manually.
Determine the relevant anatomical therapeutic chemical classification system (ATC) code for drugs
Drugs that met the screening criteria were checked against the ATC code on the World Health Organization (WHO) ATC/DDD index website (https://www.whocc.no/atc_ddd_index/). The ATC system, the official classification system of the WHO for medicines, consists of five hierarchical levels. The first level is a single letter representing the anatomical classification. The second level is a two-digit code for therapeutic classification. The third level is a single letter indicating pharmacological classification. The fourth level is a single letter for chemical classification, and the fifth level is a two-digit number specifying the compound. Individual drugs may have multiple ATC codes; for this study, the ATC code corresponding to the first indication in the drug specification was selected. In this study, drugs causing DEU were classified anatomically using the first level of the ATC code and pharmacologically using the third level. A sunrise diagram was then created based on the proportional reporting ratio (PRR) value of each drug.
Excavation signal evaluation
To comprehensively evaluate the validity of risk signals for adverse events, we searched for “esophageal ulcer” on the Side Effect Resource (SIDER) website (http://sideeffects.embl.de/) and reviewed records of drugs reported to cause DEU. Drug inserts were individually examined to determine whether DEU-related ADRs were documented. Additionally, PubMed and China Knowledge Network were searched for studies or reports of related adverse events. The results were analyzed to evaluate the mining signals for adverse events, and a summary table was created to present the findings. This study was conducted in accordance with the Declaration of Helsinki (1975, revised 2024). No AI-generated content or AI-assisted data analysis tools were used in the conception, design, execution, or interpretation of this research. The reporting of this study conforms to Standards for QUality Improvement Reporting Excellence (SQUIRE) guidelines.16,19,22
Ethics statement
This study analyzed data from a publicly available and anonymized resource. The FAERS database is designed to support FDA-approved drug safety surveillance programs and is accessible for research purposes. Therefore, this study did not require Institutional Review Board approval, nor did it require obtaining informed consent from patients.
Statistical analysis
Disproportionality analysis was used to generate hypotheses regarding possible associations between drugs and esophageal ulcer. This method calculates the ratio of observed to expected drug–ADR reports, with significant imbalance indicated when a prespecified threshold is exceeded. ROR and PRR are the two most commonly used measures in disproportionality analysis, valued for their simplicity and consistent results. The calculation formulas and criteria are listed in Table 1. Higher ROR or PRR values indicate a stronger statistical association between the suspected drug and the ADR. ROR and PRR values were initially calculated using OpenVigil 2.1, and subsequent data processing and analysis were performed using Microsoft Excel 2019.
The statistics used for detecting drugs with high-risk DEU.

Equation: a, number of reports containing both the target drug and the target ADR; b, number of reports containing other ADRs of the target drug; c, number of reports containing the target ADR for other drugs; d, number of reports containing other drugs and other ADRs.
95% CI: 95% confidence interval; ADR: adverse drug reaction; DEU: drug-induced esophageal ulcer; N: number of reports; PRR: proportional reporting ratio; ROR: reporting odds ratio; χ²: chi-squared.
Results
Drugs with DEU-related adverse event signals and their ATC codes
Using OpenVigil 2.1 for data mining, a total of 12,763 cases were reported in the FAERS database for drugs identified as primary causes of DEU. After recalculating and screening by merging similar drugs via the RxNorm database to meet the inclusion criteria of this study, 49 high-risk drugs were selected for analysis. ATC codes for these 49 drugs were sequentially queried, and the results, sorted by frequency from highest to lowest, are presented in Table 2.
The positive signaling drugs that cause adverse reactions of esophageal ulcers.
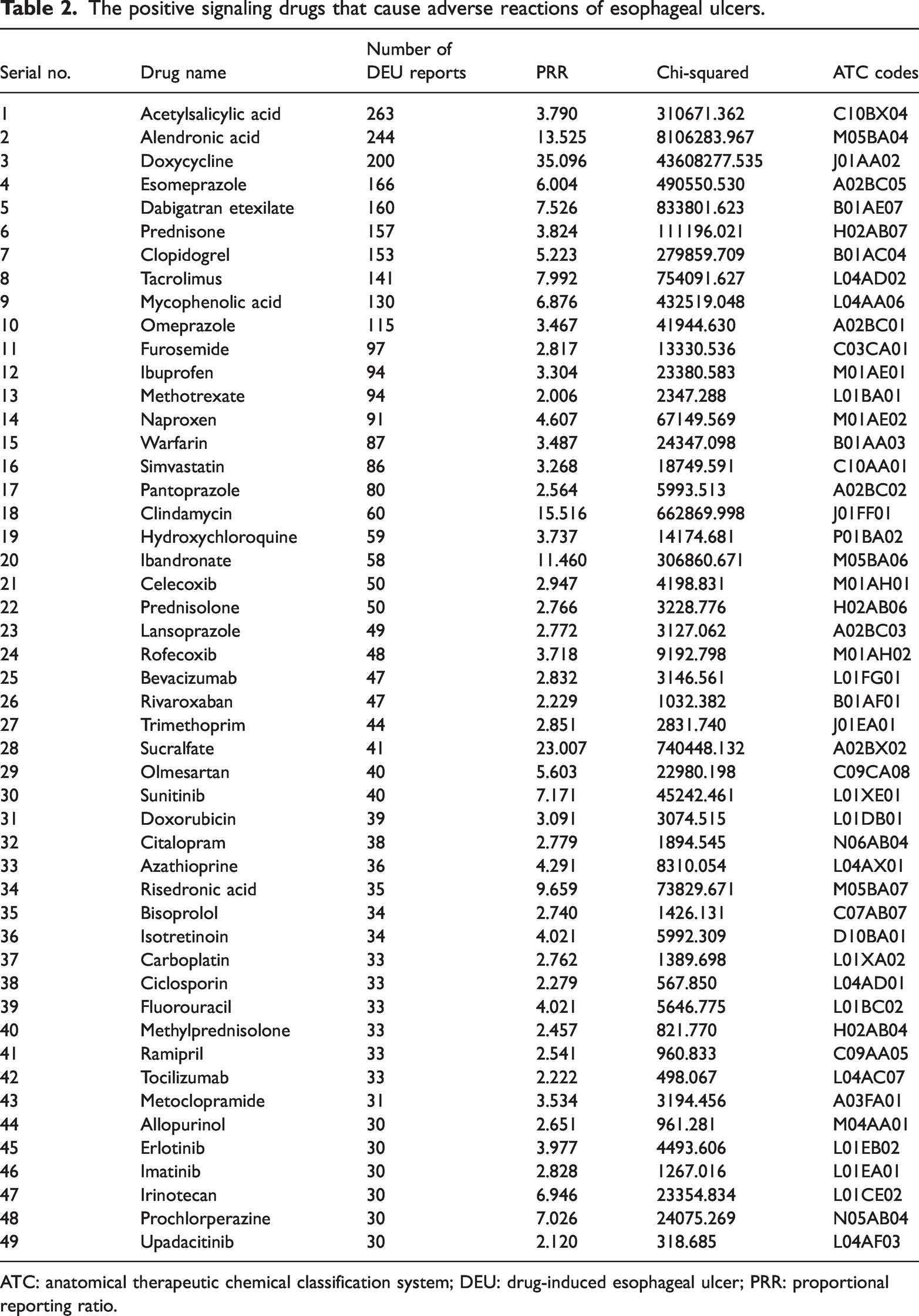
ATC: anatomical therapeutic chemical classification system; DEU: drug-induced esophageal ulcer; PRR: proportional reporting ratio.
Signal mining for DEU-related adverse events
The 49 drugs that met the screening criteria were visualized in a sunburst chart (Figure 1). The inner circle represents the first ATC level (anatomical major group), the middle circle shows pharmacological subgroups (level 3), and the outer circle displays individual drugs. The width of each slice corresponds to the PRR value of the drug, with larger slices indicating stronger disproportionate signals. This visualization reflects the relative likelihood that each drug type may trigger DEUs.
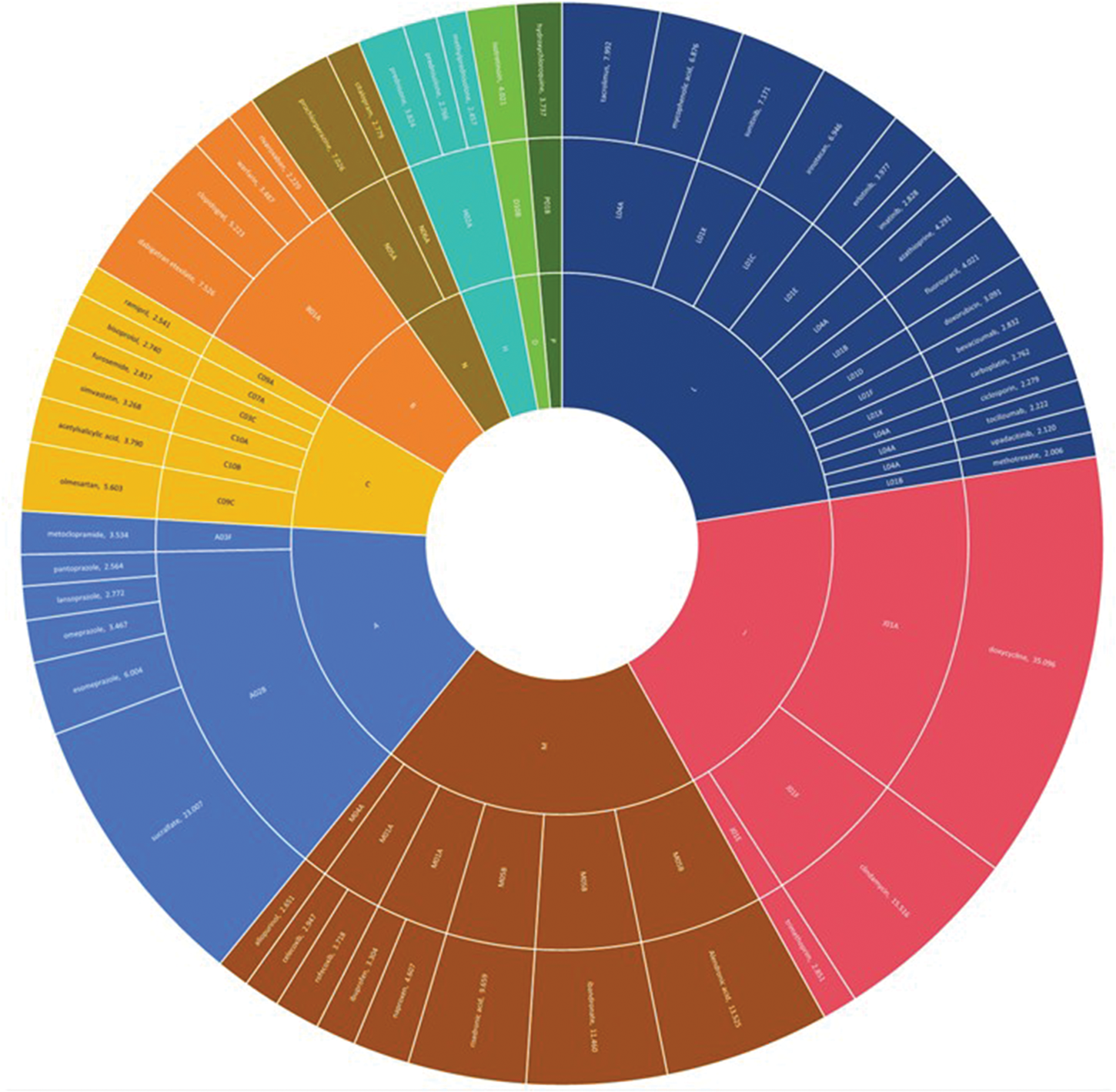
Sunburst chart of suspected drugs causing DEU, classified and grouped by ATC. A: alimentary tract and metabolism; B: blood and blood-forming organs; C: cardiovascular system; D: dermatologicals; H: systemic hormonal preparations, excluding sex hormones and insulins; J: anti-infectives for systemic use; L: antineoplastic and immunomodulating agents; M: musculoskeletal system; N: nervous system; P: antiparasitic products, insecticides, and repellents. ATC: anatomical therapeutic chemical classification system; DEU: drug-induced esophageal ulcer.
Drugs were classified according to the first ATC level, and cumulative PRR values for each class are shown in Figure 2. DEU signal intensity was highest for antineoplastic and immunomodulating agents (L), followed by anti-infectives for systemic use (J), drugs affecting the musculoskeletal system (M), digestive tract and metabolism (A), cardiovascular system (C), blood and hematopoietic organs (B), nervous system (N), endocrine system (H), dermatological drugs (D), and antiparasitic agents (P).

Funnel plot of cumulative PRR values for the first ATC-level classification of each drug. ATC: anatomical therapeutic chemical classification system; PRR: proportional reporting ratio.
Signal evaluation results
A search of SIDER 4.1 for “esophageal ulcer” identified 50 drugs associated with DEU. By combining the signal mining results from OpenVigil 2.1, drug insert information, and the SIDER 4.1 search results, the top 20 suspected drugs with the strongest DEU signals are presented in Table 3.
The top 20 positive signaling drugs associated with DEU ranked by PRR.

ATC: anatomical therapeutic chemical classification system; DEU: drug-induced esophageal ulcer; PRR: proportional reporting ratio; SIDER: Side Effect Resource.
Discussion
OpenVigil 2.1, as a publicly available pharmacovigilance tool, overcomes technical limitations by integrating standardized MedDRA term mapping, a multi-algorithm signal detection framework, and a built-in time-series analysis module. It allows direct import and analysis of raw ADR data from the FAERS database.20,21 This systematic study not only confirms DEU risk associated with well-established medications but also identifies significant safety signals for drugs not commonly recognized for this adverse effect.
A total of 12,763 DEU-related adverse event reports were identified, and 49 suspected drugs were analyzed. These drugs were broadly distributed across nine major ATC classifications, including antitumor and immunomodulating agents (Class L), systemic anti-infectives (Class J), and musculoskeletal system drugs (Class M). The three most frequently reported drugs were aspirin (frequency = 263), alendronate (frequency = 244), and doxycycline (frequency = 200). The three drugs with the highest signal intensities were doxycycline (PRR = 35.096), aluminum thioglycollate (PRR = 23.007), and clindamycin (PRR = 15.516). These results are consistent with prior knowledge that certain drugs carry a high risk of DEU.
Although previous studies often focused on specific drug classes, our analysis systematically screened 20 years of data (2004–2024) across all drug classes. Beyond confirming established associations, a key contribution of this study is the identification of significant DEU signals for drugs not currently listed in standard adverse effect databases (e.g. SIDER) or in their prescribing information. Notably, strong disproportionate signals were observed for tacrolimus, mycophenolic acid, sunitinib, clopidogrel, and erlotinib. These findings highlight potential “blind spots” in official safety profiles, particularly for high-risk populations such as immunocompromised or oncology patients. Clinicians should exercise heightened vigilance and counsel patients on esophageal symptoms when prescribing these agents, even in the absence of prior warnings. For regulators and the pharmacovigilance community, these signals warrant further investigation and consideration for label updates.
Retrospective studies have shown that antibiotics and nonsteroidal anti-inflammatory drugs (NSAIDs) are major causes of drug-induced esophagitis, consistent with our findings. 23 Literature also documents DEU associated with bisphosphonates; one study reported that 10.7% of patients taking oral bisphosphonates developed gastric or esophageal symptoms. 24 Furthermore, the combination of bisphosphonates with antithrombotic drugs or NSAIDs increases mucosal injury and symptom severity.25,26 Disproportionality analysis of the FAERS database similarly identified esophageal ulceration as a major adverse event of bisphosphonates. 26 Therefore, antibiotics, NSAIDs, and bisphosphonates should be considered high-risk medications for DEU. Particular caution is warranted when bisphosphonates are co-administered with antithrombotic drugs/NSAIDs; the clinical necessity of such combinations should be critically evaluated, and patient guidance on proper medication use should be strengthened.
Doxycycline, a high-frequency, high-risk drug, exhibited a signal intensity far exceeding that of the traditionally recognized high-risk drug alendronate (PRR =13.525), suggesting that previous literature may have underestimated the risk of some drugs in this class. Carlborg et al. 27 conducted a clinical and experimental study on tetracycline-induced DEU as early as 1983, in which 40 of 100 patients taking oral tetracycline developed sudden onset of intense retrosternal pain and dysphagia. All patients had significant peri-esophageal ulceration. History, barium meal, esophagoscopy, biopsy, and esophageal manometry revealed no other obvious etiology, and the localized corrosive effect of tetracycline was implicated. Experimental testing in the cat esophagus further confirmed the severe localized corrosive effect of tetracycline. Additional case reports have also documented DEU caused by doxycycline.13,28,29 Early clinical studies and animal experiments have established the mechanism of localized corrosion for tetracyclines, and as a derivative, doxycycline warrants heightened vigilance.
In this study, antitumor and immunomodulating agents (Class L) ranked first in cumulative PRR values in the ATC Class I classification, involving 12 drugs (24.5%, 12/49). The top three drugs most likely to cause DEUs in this category were tacrolimus, mycophenolic acid, and sunitinib. Tacrolimus ranked first in both frequency and signal strength. This drug was not listed in the SIDER database, and its prescribing information mentions gastrointestinal ulceration and perforation, oral mucositis, and ulceration, but does not specify esophageal ulceration. The first case report of tacrolimus-induced acute esophageal necrosis in 2020 highlights the potential for immunosuppressants to cause this rare adverse reaction. 30 Therefore, tacrolimus-induced DEU should be considered a significant concern requiring clinical vigilance. Other high-signal immunomodulators, including fluorouracil and mesalazine, have also been reported in the literature.31,32 Among antitumor drugs, sunitinib exhibited the highest signal intensity, and Jeanniard-Malet et al. 33 reported three cases of severe ulcerative esophagitis induced by sunitinib. A study comparing erlotinib plus gemcitabine versus gemcitabine alone in advanced pancreatic cancer found a higher frequency of ulcers in the combination therapy group. 34 Literature searches have also identified novel antitumor agents such as bevacizumab and cetuximab as associated with esophageal ulcers.35,36 Real-world data from this study, combined with increasing literature reports, indicate that the DEU risk associated with antitumor and immunomodulatory agents has been previously under-recognized and should be a focus of clinical concern. High-risk antitumor agents in patients with peptic ulcer disease require careful assessment, and monitoring for DEUs should be intensified. These agents are particularly relevant in chronic, high-stakes conditions (e.g. organ transplantation, cardiovascular disease, oncology), where early recognition of DEU can prevent serious complications. Clinicians are encouraged to enhance monitoring and patient education to identify DEU risk promptly.
Notably, thioglycollate, prochlorperazine, olmesartan ester, and azathioprine are not documented as causing DEU in drug inserts, the SIDER database, or relevant literature. The strong signals identified in this study suggest these potential risks warrant further validation, though they may represent false-positive signals. Conversely, tacrolimus, sunitinib, mycophenolic acid, clopidogrel, and erlotinib do not list esophageal ulceration in their prescribing information, yet real-world data and literature evidence indicate a clear risk. These findings help compensate for delays in drug insert updates, alert clinicians to previously underappreciated DEU risks, and enrich the catalog of major suspected drugs associated with DEUs in clinical practice.
The study also found that proton pump inhibitors (PPIs), such as esomeprazole (PRR = 6.004) and omeprazole (PRR =3.467), ranked as high-risk medications, which contradicts the previous notion that PPIs reduce the risk of DEU. Possible explanations include: (a) PPI users often concurrently take other high-risk medications (e.g. NSAIDs or antiplatelet agents), and confounding factors may not be fully controlled; (b) long-term acid suppression may lead to bacterial overgrowth in the stomach, which can secondarily damage the esophagus; and (c) PPIs themselves may interfere with the esophageal mucosal barrier via unknown mechanisms. This finding highlights the need for clinicians to re-evaluate the dual role of PPIs in DEU and avoid blind co-administration.
The mechanisms underlying DEUs are diverse and vary across drug classes. Bisphosphonates (e.g. alendronate, ibandronate) can cause direct chemical irritation of the mucosa, primarily due to their high acidity and the tendency of tablets to remain in the esophagus. Endoscopic findings typically reveal erosions or ulcerations, exudative inflammation, and esophageal wall thickening. 37 Patients who do not follow proper administration instructions—taking the medication upright and with sufficient water—may experience prolonged drug adhesion to the esophageal wall, leading to localized pH reduction and chemical burns. Novel drug delivery systems, such as nanostructured floating rafts, may offer a future approach to prevent bisphosphonate-induced DEU. 38 Antibiotics (e.g. doxycycline, clindamycin) form an acidic solution when in contact with water and can cause localized ulceration without stricture formation, which typically resolves upon drug discontinuation. 39 Antineoplastic agents (e.g. fluorouracil, sunitinib) impair esophageal mucosal repair by inhibiting cell proliferation (e.g. fluorouracil interferes with DNA synthesis) and may also induce systemic immunosuppression, increasing the risk of opportunistic infections.31 Sunitinib, a multi-targeted tyrosine kinase inhibitor, may exacerbate mucosal ischemia by inhibiting angiogenesis. 33 Immunosuppressants (e.g. tacrolimus, meclofenamic acid) reduce mucosal immune defenses by inhibiting T-cell function or purinergic synthesis, promoting opportunistic infections (e.g. cytomegalovirus) and leading to ulcer formation.11,30,32 NSAIDs contribute to ulcer formation by reducing prostaglandin-mediated mucosal protection and causing direct cellular damage. 40 Anticoagulants (e.g. dabigatran etexilate, warfarin) may synergistically increase bleeding risk when combined with NSAIDs or glucocorticoids. 41
Patient-related factors predominantly involve those that prolong the transit time of drugs in the esophagus. 3 Age-related reductions in salivation, anatomical changes in the esophagus, taking medications at bedtime, and motor impairments may all increase esophageal transit time. 42 The most significant factor, however, is taking medications with insufficient water or while lying down. 43 To prevent drug-induced esophagitis, patients should follow medication instructions carefully: remain upright for at least 30 min after ingestion, take medications with at least 180 ml of water, and consider liquid formulations for bedridden patients or those with dysphagia. 44
However, several limitations of this study should be acknowledged. First, the spontaneous reporting nature of FAERS data may result in underreporting or overreporting.16–18 FAERS accepts reports from patients and their families, which may affect the clinical accuracy of the information but also reflects patients’ real-world experiences. The FDA encourages patient reporting to capture a broader range of potential signals. Second, behavioral factors such as medication adherence and body position cannot be obtained from the database. Third, the high frequency of missing or inconsistent data in the “time-to-onset” field prevented meaningful analysis of DEU latency. This limitation underscores the hypothesis-generating nature of pharmacovigilance signals from spontaneous reports, which are better suited for identifying potential associations than for defining precise temporal patterns. Fourth, concomitant medications may act as confounding factors for DEU, and further research is needed to clarify these interactions. Serious, unusual, or highly publicized adverse events are more likely to be reported than mild or common ones, which may lead to overestimation of risk for certain drugs (e.g. newly marketed agents) and underestimation for others. Fifth, FAERS reports are heterogeneous and often lack critical clinical details, such as complete medication history, patient comorbidities, or endoscopic confirmation of ulcers, limiting the ability to control for confounding factors. A low report count does not necessarily indicate absence of risk, as many events—particularly mild ones or those attributed to other causes—may go unreported. This is a fundamental limitation of all passive surveillance systems. There may also be significant delays between the onset of an adverse event and its submission to FAERS, which can affect the timeliness of signal detection for newly marketed drugs. Disproportionality analyses identify statistical “signals” of drug-event associations reported more frequently than expected, but they cannot establish definitive causal relationships. PRR and ROR methods cannot fully eliminate confounding bias, which may introduce some degree of distortion into the results. Therefore, clinical decisions should not be made solely on these findings, though enhanced monitoring is advised. It is important to emphasize that this study is hypothesis-generating in nature and should be interpreted alongside clinical judgment and other supporting evidence. Pharmacists, with their expertise in medication management and risk mitigation, are integral members of patient safety teams and should participate in all adverse event management processes.19,45–47 Future research should focus on validating signal strength through prospective cohort studies, investigating underlying mechanisms using esophageal mucosal biopsies and drug concentration monitoring, and developing multi-dimensional risk prediction models, potentially leveraging machine learning. Additionally, optimization of dosage forms for high-risk drugs represents a promising strategy to reduce DEU incidence.
Conclusion
This large-scale pharmacovigilance study suggests that a broader spectrum of drugs may be associated with DEU than previously recognized, particularly among immunomodulating and antineoplastic agents. The findings may inform more targeted patient counseling regarding proper medication administration and highlight the need for focused monitoring in high-risk populations. These results require validation in prospective clinical studies to further clarify causality and quantify risk.
Footnotes
Acknowledgment
None.
Author contributions
Study concept and design: LYJ and QJ; Investigation: WZ and LYJ; Acquisition of data: LYJ and JYW; Analysis and interpretation of data: LYJ and JH; Drafting of the manuscript: LYJ and KLM; Review of the manuscript: WZ, JH, JYW, KLM, LYJ, and QJ. All authors contributed to the data collection, had full access to the data room having all the data in the study, and accept responsibility to submit for publication.
Consent to participate
Not applicable.
Data availability statement
Data for the FAERS database were obtained and extracted from 2004 to 2024. The open-source database used to conduct the study is available on the website.
Declaration of conflicting interests
All the authors have declared no conflicts of interest.
Ethics approval
Not applicable.
Funding
This study was supported by the 2025 Shining Across China–Medicinal Research Capacity Building Fund Project (No. Z04J2025E170), the Hospital Pharmacy Research Project of Zhejiang Pharmaceutical Association (No. 2025ZYY74), the Zhejiang Provincial Natural Science Foundation (No. LYQ20H300001), the Clinical Research Project of Wujieping Medical Foundation (No. 320.6750.2025-6-15), and the Quzhou Technology Projects of China (No. 2023K141).
