Abstract
Objective
To assess the ability of markers of inflammation to identify the solid or micropapillary components of stage IA lung adenocarcinoma and their effects on prognosis.
Methods
We performed a retrospective study of clinicopathologic data from 654 patients with stage IA lung adenocarcinoma collected between 2013 and 2019. Logistic regression analysis was used to identify independent predictors of these components, and we also evaluated the relationship between markers of inflammation and recurrence.
Results
Micropapillary-positive participants had high preoperative neutrophil-to-lymphocyte ratios. There were no significant differences in the levels of markers of systemic inflammation between the participants with or without a solid component. Multivariate analysis showed that preoperative neutrophil-to-lymphocyte ratio (odds ratio [OR] = 2.094; 95% confidence interval [CI], 1.668–2.628), tumor size (OR = 1.386; 95% CI, 1.044–1.842), and carcinoembryonic antigen concentration (OR = 1.067; 95% CI, 1.017–1.119) were independent predictors of a micropapillary component. There were no significant correlations between markers of systemic inflammation and the recurrence of stage IA lung adenocarcinoma.
Conclusions
Preoperative neutrophil-to-lymphocyte ratio independently predicts a micropapillary component of stage IA lung adenocarcinoma. Therefore, the potential use of preoperative neutrophil-to-lymphocyte ratio in the optimization of surgical strategies for the treatment of stage IA lung adenocarcinoma should be further studied.
Keywords
Background
Lung cancer is a highly prevalent malignant tumor that is associated with a high mortality rate in China and worldwide.1,2 In 2022, lung cancer ranked first among all new cases of malignant tumors in China, accounting for 18.06% of the total. The number of deaths caused by lung cancer also ranked first, accounting for 23.9% of the total number of deaths caused by malignant tumors. 2 In the United States, lung cancer ranks second among new tumors and is the leading cause of mortality among patients with malignancies. 1 The great popularity of the use of computed tomography (CT) scanning in clinical practice has increased the probability of detecting early-stage lung cancer, which mostly manifests as lung adenocarcinoma. 1 Based on their predominant histopathologic components, invasive lung adenocarcinomas can be further subdivided into lepidic, acinar, papillary, micropapillary, or solid-predominant adenocarcinomas. 3 Patients with the solid- or micropapillary-predominant subtypes are at a higher risk of tumor recurrence or metastasis than those with the other subtypes, even if they are at the same stage or have undergone the same radical surgical treatment.4–6 Moreover, even a small solid or micropapillary component (>1% or >5% of the entire tumor, respectively) may be associated with an unfavorable outcome.7,8 Furthermore, the most appropriate surgical intervention for use in patients with stage IA lung adenocarcinoma may differ according to its histologic components. Some previous studies have shown that the outcomes of patients undergoing sublobar resection are similar to those of patients undergoing lobectomy for the treatment of stage IA lung adenocarcinoma. 9 However, others have shown that sublobar resection is associated with a worse prognosis than lobectomy when an adenocarcinoma has solid or micropapillary components.10,11 Such contradictory findings emphasize the necessity of characterizing the tumor histologically, either preoperatively or intraoperatively. However, despite frequent previous efforts to identify predictors of the histologic component of lung adenocarcinomas,12–14 no promising markers have been identified that could be used pre- or intraoperatively.
Systemic inflammation is deeply involved in the development and progression of tumors. 15 Markers of systemic inflammation, including the neutrophil-to-lymphocyte ratio (NLR), lymphocyte-to-monocyte ratio (LMR), and platelet-to-lymphocyte ratio (PLR), have been shown to be useful prognostic indicators in patients with various tumors, including lung adenocarcinoma.16–18 In addition, some studies have shown that the NLR and PLR are valuable indicators of the pathologic grade of bladder cancers 19 and gliomas. 20 However, it is unknown whether these markers may be predictors of the presence of certain components, and especially the solid or micropapillary components, of stage IA lung adenocarcinomas.
In the present study, we aimed to determine whether markers of inflammation could be measured preoperatively as predictors of the presence of solid or micropapillary components of stage IA lung adenocarcinomas and to evaluate their association with the prognosis of the patient.
Methods
Patient selection
Data were retrospectively retrieved for consecutive patients who underwent curative surgical treatment and were histologically confirmed to have stage IA adenocarcinoma in our center between January 2013 and December 2019. Patients were excluded if (1) the histologic type of their tumor was re-diagnosed as adenocarcinoma in situ, the tumor was minimally invasive, it was an invasive mucinous adenocarcinoma, or it was an adenocarcinoma of stage >IA, according to the 2021 World Health Organization (WHO) guidelines; 21 (2) if they had a history of tumor or multiple primary lung cancers; (3) had a concomitant disease that could have affected the peripheral blood data, including autoimmune disorders, hematologic disorders, and active infections; (4) peripheral blood testing was performed ≥30 days preoperatively; and (5) if insufficient medical information was available. Propensity score matching was conducted to reduce patient selection bias. All the patient details were de-identified. The study was approved by the Ethics Committee of Zhoushan Hospital (approval number: 2021-167), and the requirement for informed consent was waived. The study and manuscript comply with the applicable EQUATOR guidelines, and the reporting of the study conforms to the STROBE guidelines. 22
Data collection
Data were obtained from the medical records of the participants and included their sex, age, smoking status, blood cell counts (neutrophils, lymphocytes, monocytes, and platelets), preoperative carcinoembryonic antigen (CEA) concentrations, tumor size, surgical procedures undergone, and outcome data. The NLR, LMR, and PLR were calculated as the ratios of the neutrophil count to the lymphocyte count, the lymphocyte count to the monocyte count, and the platelet count to the lymphocyte count, respectively. Tumor size referred to the largest dimension of the entire lesion measured in the lung window.
Histologic evaluation
Histologic slides were re-examined by two pathologists, as per the WHO 2021 classification recommendations. 21 Each component was graded in 5% increments. Specifically, for solid or micropapillary components, a proportion of ≥1% was regarded as positive. 7 The participants were subsequently grouped as follows: solid-negative and micropapillary-negative (S−M−), micropapillary-negative and solid-positive (S+M−), solid-negative and micropapillary-positive (S−M+), and positive for both (S+M+).
Recurrence and follow-up
Follow-up examinations were usually conducted every 3 months during the first year following tumor resection, and every 6 months from the second year onwards. Tumor recurrence was initially diagnosed on the basis of appropriate symptoms, the results of a physical examination, and laboratory or imaging findings, and was subsequently confirmed through tissue biopsy, or through close follow-up of the participants if it was difficult to obtain pathologic samples. The recurrence time refers to the time interval between surgery and the first diagnosis of a recurrence. The acquisition of follow-up data ended in December 2023.
Statistical analysis
Data are summarized as numbers (categorical variables) or mean ± standard deviation (continuous variables). Inter-group univariate comparisons were performed using the chi-square test, Fisher’s exact test, independent-samples Student’s t-test, or one-way analysis of variance, followed by the least significant difference test, as appropriate. The relationships between variables were assessed using bivariate correlation analysis. Predictors of the presence of a micropapillary component were identified using logistic regression analysis. The diagnostic accuracy and optimal cut-off values for the predictors were determined using receiver operating characteristic (ROC) curve analysis. Many different causes of death were recorded that were not related to tumor recurrence. Therefore, we used a competitive risk model to calculate the cumulative incidence of recurrence (CIR), 23 which considers deaths unrelated to tumor recurrence as competing events. Statistical analyses were conducted using IBM SPSS version 23.0 (IBM Corp., Armonk, NY, USA) and R version 4.0.0 (www.r-project.org). P < 0.05 was deemed to indicate statistical significance.
Results
Characteristics of the participants
In total, 218 patients with either solid or micropapillary components to their tumors were identified, and 436 patients with neither solid nor micropapillary components were matched at a 1:2 ratio with their nearest equivalent. Of the total number, 334 were men and 320 were women. The mean age of the participants was 62.74 ± 9.07 years. A total of 188 participants had a history of smoking, and 165 and 489 underwent sublobar resection and lobectomy, respectively. The mean tumor size was 1.99 ± 0.72 cm and the mean CEA concentration was 3.36 ± 4.24 ng/mL. Histopathologic examination led to 436, 90, 107, and 21 participants being placed in the S−M−, S+M−, S−M+, and S+M+ groups, respectively. The mean preoperative NLR, LMR, and PLR were 2.12 ± 0.99, 4.88 ± 1.64, and 128.78 ± 51.15, respectively. By December 2023, tumor recurrence had been reported for 72 participants, and 20 had died for other reasons. The median duration of follow-up for the survivors with no signs of tumor recurrence was 43.0 (range, 10–113.5) months.
Relationship between markers of systemic inflammation and clinicopathologic factors
Initially, we compared the NLR, PLR, and LMR of the four groups. ANOVA revealed significant differences in the preoperative NLR (F = 23.772, P < 0.001), and LSD testing showed that the preoperative NLR was significantly higher in the S−M+ group than in the S−M− group (P < 0.001), but there was no difference between the S+M− and S−M− groups. There were no significant differences in the preoperative LMR or PLR among the four groups (F = 0.547 and F = 1.181, respectively). The participants were then allocated to two further groups—micropapillary-positive (M+ group, n = 104) and micropapillary-negative (M− group, n = 458) groups—to investigate the relationship of each ratio with the micropapillary component alone. The preoperative NLR of the M+ group was significantly higher than that of the M− group (P < 0.001), but there were no significant differences in the preoperative LMR or PLR.
With respect to the other clinicopathologic characteristics evaluated, the preoperative NLR significantly correlated with the age of the participants (P = 0.013), but there was no correlation of preoperative NLR with sex, smoking status, CEA concentration, or tumor size. The preoperative PLR closely correlated with sex (P = 0.01) and smoking status (P = 0.008), but there was no correlation with age, CEA concentration, or tumor size. The preoperative LMR closely correlated with age (P < 0.001), sex (P < 0.001), and smoking status (P < 0.001), but there was no correlation with CEA concentration or tumor size (Table 1).
Relationships of the clinicopathologic characteristics of the participants with the preoperative NLR, PLR, and LMR.

Bivariate correlation analysis. bIndependent sample t-test. Significant P-values (<0.05) are shown in bold.
NLR, neutrophil-to-lymphocyte ratio; PLR, platelet-to-lymphocyte ratio; LMR, lymphocyte-to-monocyte ratio; CEA, carcinoembryonic antigen.
Clinicopathologic predictors of the presence of a micropapillary component
The predictive values of clinicopathologic factors for the presence of a micropapillary component were determined using logistic regression analysis (Table 2).
Results of the logistic regression analyses to identify predictors of stage IA lung adenocarcinoma with a micropapillary component.

Significant P-values (<0.05) are shown in bold.
HR, hazard Ratio; CI, confidence Interval; CEA, carcinoembryonic antigen; NLR, neutrophil-to-lymphocyte ratio; LMR, lymphocyte-to-monocyte ratio; PLR, platelet-to-lymphocyte ratio.
Univariate analysis revealed that sex (P = 0.026), smoking status (P = 0.011), CEA concentration (P = 0.001), tumor size (P = 0.001), and preoperative NLR (P < 0.001) were significantly associated with the existence of a micropapillary component. Conversely, there were no significant associations of preoperative LMR, preoperative PLR, or age with such a component. Multivariate analysis confirmed that preoperative NLR (odds ratio [OR] = 2.094; 95% CI, 1.668 to 2.628; P < 0.001), tumor size (OR = 1.386; 95% CI, 1.044 to 1.842; P = 0.024), and CEA concentration (OR = 1.067; 95% CI, 1.017 to 1.119; P = 0.008) were independent predictors of a micropapillary component in stage IA lung adenocarcinomas. The predictive efficiency of each variable was assessed using a ROC curve. The optimal cut-off value of the preoperative NLR was 2.55 (area under the curve [AUC], 0.723; sensitivity, 50.8%; specificity, 84.2%), that for CEA concentration was 6.73 ng/mL (AUC, 0.519; sensitivity, 16.4%; specificity, 99.5%), that for tumor size was 2.15 cm (AUC, 0.587; sensitivity, 47.7%; specificity, 66.2%); and that for the combined score was 0.226 (AUC, 0.737; sensitivity, 56.3%; specificity, 81.4%). The combined score was associated with the largest AUC for the prediction of stage IA lung adenocarcinoma with a micropapillary component (Figure 1). However, these data imply that the predictive ability of the preoperative NLR and the combined score are unsatisfactory.
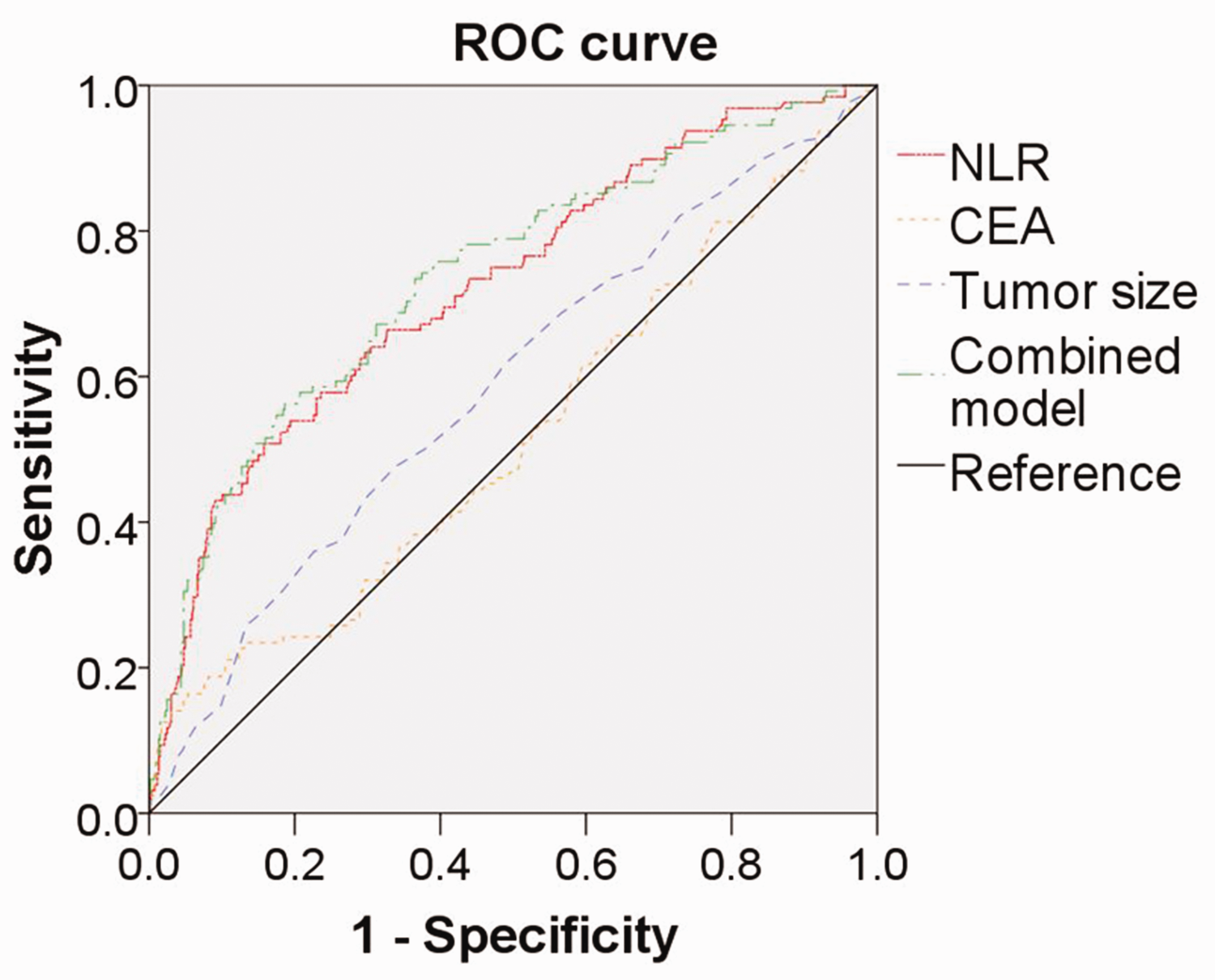
ROC curves for preoperative NLR, CEA concentration, tumor size, and a combined score for the prediction of the presence of a micropapillary component in stage IA lung adenocarcinomas. NLR, neutrophil-to-lymphocyte ratio; CEA, serum carcinoembryonic antigen; ROC, receiver operating characteristic.
Relationships between clinicopathologic characteristics of the participants and the CIR for tumors
Univariate analysis identified the histologic grouping (P < 0.001), sex (P = 0.016), age (P = 0.01), and the CEA concentration (P < 0.001) as significant predictors of the CIR. However, no strong associations were found of CIR with smoking status, tumor size, the surgical procedure undergone, the preoperative NLR, the preoperative LMR, or the preoperative PLR. Multivariate competing risk proportional hazards model analysis confirmed that the independent risk factors for CIR were CEA concentration (hazard ratio [HR] = 1.069; 95% CI, 1.03 to 1.107; P < 0.001), age (HR = 1.036; 95% CI, 1 to 1.068; P = 0.026), sex (HR = 0.527; 95% CI, 0.32 to 0.868; P = 0.012) and the existence of a solid or micropapillary component (S−M− as the reference group; S−M+: HR = 2.723; 95% CI, 1.26 to 5.875; P = 0.011; S+M−: HR = 7.278; 95% CI, 4.05 to 13.077; P < 0.001; S+M+: HR = 9.626; 95% CI, 4.31 to 21.492; P < 0.001) (Table 3).
Relationships of the clinicopathologic characteristics of the participants with stage IA lung adenocarcinoma with their CIR.

Significant P-values (<0.05) are shown in bold.
CI, confidence interval; HR, hazard ratio; CEA, carcinoembryonic antigen; S−M−, solid and micropapillary negative; S+M−, solid positive, micropapillary negative; S−M+, solid negative, micropapillary positive; S+M+, solid and micropapillary positive; NLR, neutrophil-to-lymphocyte ratio; LMR, lymphocyte-to-monocyte ratio; PLR, platelet-to-lymphocyte ratio; CIR, cumulative incidence of recurrence.
Discussion
In the present study, we have evaluated the preoperative use of several markers of systemic inflammation for the prediction of specific cellular components and their associations with the outcomes of stage IA lung adenocarcinoma using a relatively large sample. Only the preoperative NLR was found to provide information regarding the micropapillary component, and none of the markers assessed were found to be associated with the CIR in patients with stage IA lung adenocarcinoma.
Because limited tumor resection has become an acceptable approach to the treatment of small peripheral lung nodules, the accurate pre- or intraoperative identification of solid and micropapillary components, which may not be suitable for limited resection,10,11 is now extremely important for optimal surgical planning. However, the accuracy with which solid or micropapillary components can currently be identified on the basis of preoperative needle biopsy samples or intraoperative frozen sections is unsatisfactory.24,25 Previous research has demonstrated that some CT scan image features are useful for the prediction of micropapillary or solid components.12,13 However, some of these characteristics are subjective and qualitative, and therefore poor reproducibility significantly limits their widescale clinical use. Cha et al. 26 reported that a solid appearance, a tumor diameter of ≥2.5 cm, a maximum standardized uptake value of 7, and a tumor of stage >I are independent indicators of the presence of micropapillary and solid components. However, limited resection is not appropriate for patients with these characteristics. Moreover, owing to its high cost, positron emission tomography is not yet a routine examination for patients with early-stage lung cancer in China. A previous study explored the relationship between certain genes and a microcapillary component to tumors, and showed that high expression of chemokine ligand 14 is present in patients with a micropapillary component, meaning that this could serve as a biomarker of the presence of a micropapillary component of lung adenocarcinoma. 14 However, such an assessment would be expensive and difficult to perform.
Markers of systemic inflammation can be easily evaluated following a blood count. We found that patients with a solid tumor component had no significant differences in their preoperative NLR, PLR, or LMR from those without, whereas patients with a micropapillary tumor component had a higher preoperative NLR than those without. Moreover, multivariate analysis confirmed that preoperative NLR is an independent indicator of the presence of a micropapillary component. To the best of our knowledge, this is the first study to clearly show an association between preoperative NLR and a micropapillary component of stage IA lung adenocarcinoma. However, it must be emphasized that, because the AUC value calculated was relatively low, caution should still be exercised when using the preoperative NLR to identify the existence of a micropapillary component in patients who are considered to have stage IA lung adenocarcinoma. A previous study demonstrated that deep learning models with radiomic signatures perform well at identifying a micropapillary component within a lung adenocarcinoma. 27 Furthermore, the systemic immune inflammation index (SII) 28 and the preoperative C-reactive protein/albumin (CRP/Alb) ratio, 29 which can be easily calculated using data obtained from peripheral blood tests, also have prognostic value in patients with cancer. Thus, the use of a combination of preoperative NLR and other parameters, such as radiologic features, SII, and CRP/Alb ratio, may improve the effectiveness of such predictive models, and we will investigate this possibility in the future. In addition, we have shown that although the CEA concentration and tumor size are independent indicators of the presence of a micropapillary component, their AUCs are low. This may be because of the inclusion of patients in the micropapillary-negative group with a solid component to their tumor, given that there is abundant evidence that the presence of a solid tumor component is often associated with a high CEA concentration 30 and a large tumor. 26 Therefore, in future research, we intend to create more appropriate groups to more accurately evaluate the use of the CEA concentration and tumor size as potential indicators.
The specific mechanism underpinning the relationship between preoperative NLR and a micropapillary tumor component remains unclear. Previous studies have shown that smokers have a higher NLR than non-smokers, 31 but this trend was not identified in the present study. The results show that a micropapillary component was more common in non-smokers, which is consistent with the results of another previous study. 32 Taking these findings together, a high preoperative NLR, which was found to be associated with stage IA adenocarcinomas with a micropapillary component in the present study, may develop independently of smoking status. Tumors secrete various proinflammatory molecules that could stimulate the proliferation of peripheral blood neutrophils. 33 Furthermore, large numbers of neutrophils could contribute to the progression of cancer through various mechanisms.34,35 In contrast, lymphocytes are a key mediator of immune surveillance targeting tumor cells. 36 The presence of a small number of lymphocytes is associated with a poorer immune response to tumors, and this is often a characteristic of patients with advanced tumors. 37 Therefore, NLR is commonly recognized to be an indicator of tumor progression.15,16 The micropapillary component of lung adenocarcinoma has been reported to be generated from the papillary component, which implies that the appearance of a micropapillary component is a manifestation of the progression of lung adenocarcinoma, 38 which may at least in part explain why the preoperative NLR is high in patients with stage IA lung adenocarcinoma containing a micropapillary component. However, patients who have a tumor that contains another component that has highly aggressive behavior, such as a solid component, were found not to have high preoperative NLRs in the present study. The mucin 1 (MUC1) gene is principally expressed in the micropapillary component of tumors. 39 Its product is a powerful chemoattractant that is involved in the recruitment of neutrophils, and therefore this may be responsible for the high preoperative NLR in patients with stage IA lung adenocarcinomas containing a micropapillary component.39,40
Markers of systemic inflammation are believed to have prognostic potential for advanced tumors.16–18 Hiren et al. analyzed data for 279 patients with stage IV non-small cell lung cancer (NSCLC) and found that NLR, PLR, and LMR prior to treatment are significantly associated with OS. 41 A meta-analysis of 20 studies of 13,915 patients with resectable NSCLC showed that the preoperative NLR significantly correlated with disease-free survival and overall survival. 42 However, their value in early-stage lung cancer remains to be confirmed. The results of two previous studies43,44 suggest that preoperative NLR is a promising prognostic predictor for patients with stage I NSCLC, but other studies have shown that it is not a good predictor of prognosis in such patients.45,46 The present results are consistent with the latter findings, because the preoperative NLR, LMR, and PLR did not correlate with the CIR for patients with stage IA lung adenocarcinoma. Apart from the heterogeneity of the sample and differences in the NLR cut-off values used, one possible explanation for this inconsistency may be that stage IA lung adenocarcinoma can be associated with a relatively high prevalence of cure when radical surgery is performed, and the present findings show that <10% of patients experience tumor recurrence. Thus, the use of radical surgery may weaken the value of the preoperative measurement of markers of systemic inflammation in the prediction of the outcomes of stage IA lung adenocarcinoma. From this perspective, the serial monitoring of markers of systemic inflammation pre- and postoperatively may provide more information regarding outcomes than preoperative monitoring, but this issue requires further in-depth analysis. 47
The present study had some limitations. First, it was a retrospective study and all the data were sourced from a single center; therefore, patient selection bias was inevitable. The findings should be validated through a multicenter prospective study. Second, we assessed only the three most commonly used markers, NLR, PLR, and LMR. Therefore, in future research, the evaluation of additional markers may help improve the sensitivity and specificity of the model. Third, the concentrations of markers of systemic inflammation can be influenced by various factors, and although we used strict exclusion criteria, there may still have been unexpected factors that could have influenced these concentrations, including undiagnosed comorbidities and drug use, which may have resulted in bias.
In conclusion, we have shown that preoperative NLR is an independent indicator of the presence of a micropapillary component of stage IA lung adenocarcinoma. The use of preoperative NLR in combination with other clinicopathologic parameters should be explored in the future because if such an assessment is found to have higher sensitivity and specificity, it may help clinicians develop optimal surgical strategies. The preoperative NLR, LMR, and PLR were not found to be associated with CIR in patients with stage IA lung adenocarcinoma, but further in-depth analyses should be performed to confirm this finding.
Supplemental Material
sj-pdf-1-imr-10.1177_03000605241245016 - Supplemental material for Evaluation of the preoperative neutrophil-to-lymphocyte ratio as a predictor of the micropapillary component of stage IA lung adenocarcinoma
Supplemental material, sj-pdf-1-imr-10.1177_03000605241245016 for Evaluation of the preoperative neutrophil-to-lymphocyte ratio as a predictor of the micropapillary component of stage IA lung adenocarcinoma by Cheng Chen, Zhi-Jun Chen, Wu-Jun Li, Tao Deng, Han-Bo Le, Yong-Kui Zhang and Bin-Jie Zhang in Journal of International Medical Research
Footnotes
Author contributions
CC: Conceptualization; Data curation; Investigation; Methodology; Software; Supervision; Validation; Writing — original draft.
Z-J C: Conceptualization; Data curation; Investigation; Writing — original draft.
W-J L: Data curation; Investigation; Software.
TD: Data curation; Methodology; Validation.
H-B L: Conceptualization; Project administration; Writing — review & editing.
Y-K Z: Conceptualization; Project administration; Writing — review & editing.
B-J Z: Conceptualization; Data curation; Funding acquisition; Investigation; Methodology; Supervision; Project administration; Writing — review & editing.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This study was supported by the Zhoushan Science and Technology Bureau [grant number 2022C31016].
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
